Development and HPTLC Standardization of an Ayurvedic Avleha-Based Nutraceutical Formulation of Manilkara zapota Fruit Extract with Simultaneous Estimation of Gallic Acid and Ellagic Acid
1Department of Pharmacognosy, Mahatma Gandhi Vidyamandir’s Pharmacy College, Nashik, India.
2Department of Pharmacognosy, Dr. D.Y. Patil Institute of PharmaceuticalScience & Research, Pune, India.
Corresponding Author Email: daksha511@gmail.com
Download this article as:
ABSTRACT:The growing prevalence of oxidative stress-associated disorders highlights the need for standardized herbal nutraceuticals with proven antioxidant potential. The present study aimed to develop and HPTLC standardize an Ayurvedic Avleha-based nutraceutical formulation containing extract of Manilkara zapota fruit. The extract exhibited significant phytochemical richness, with total polyphenolic content of 290 mg/g (gallic acid equivalent), total flavonoid content of 31.06 mg/g (rutin equivalent), and total flavonol content of 2.5 mg/g (rutin equivalent). Antioxidant activity assessed by DPPH assay showed an IC₅₀ value of 5180 µg/mL for the extract, compared to 4.12 µg/mL for ascorbic acid. Among the developed batches, Batch III showed optimal physicochemical characteristics and was selected for further evaluation. HPTLC analysis using a validated method with mobile phase Toluene: Ethyl acetate: Formic acid: Methanol (6:6:1.6:0.4 v/v/v/v) at 275 nm revealed well-resolved peaks with Rf values of 0.39 and 0.29 for gallic acid and ellagic acid, respectively. Simultaneous estimation showed that gallic acid and ellagic acid contents were 0.6312% w/w and 0.6918% w/w in the extract, and 0.09421% w/w and 0.04420% w/w in the formulation, respectively. The method was validated as per ICH guidelines and found to be linear, precise, accurate, and robust, supporting its suitability for routine analysis.
KEYWORDS:Avleha formulation; Ellagic acid; Gallic acid; HPTLC; Manilkara zapota; Sapodilla
Introduction
Contemporary lifestyles driven by rapid urbanization, reduced physical activity, psychological stress, and frequent consumption of nutritionally poor processed foods have contributed to widespread nutrients insufficiency and enhanced oxidative burden. Oxidative stress plays a pivotal role in the development of lifestyle-related disorders and premature aging, thereby emphasizing the need for safe, effective, and natural antioxidant-based nutraceuticalinterventions.1,2
The present investigation focuses on the development of a nutraceutical formulation enriched with vitamins and bioactive nutrients derived from the fruit of Manilkara zapota (L.) P. Royen (Sapotaceae), commonly known as Chikoo or Sapodilla. Owing to its substantial antioxidant potential, ripe fruit was selected for formulation in the traditional Ayurvedic semisolid dosage form, Avleha. The developed Avleha contains 25% ethanolic extract of the fruit to ensure adequate incorporation of therapeutically relevant phytoconstituents. Avleha is traditionally used to promote vitality and vigor and to retard age-related degenerative changes, largely due to its antioxidant attributes.3-5 The formulation was further standardized using a validated High Performance Thin Layer Chromatography (HPTLC) method for the simultaneous estimation of the polyphenolic markers gallic acid and ellagic acid.6
The formulated Avleha offers versatility in administration and can be consumed with milk or water, or used as a nutraceutical spread for convenient intake, enhancing patient compliance and acceptability.
Sapodilla, originally native to tropical America and now widely cultivated in India and the Caribbean region, is known by several vernacular names including American bully, neesberry, chikoo, sapota, sapote, and nispero. The unripe fruit and bark exude a milky latex, and the peel is rich in tannins. The mature fruit is a large berry with a brown peel, a softening skin upon ripening, and typically contains 5–6 black seeds, imparting a characteristic sweet and malty flavor. In contrast, immature fruits are firm and rich in saponins and tannins, producing a pronounced astringent sensation. Sapota is nutritionally valued as a good source of dietary fiber and calories and is commonly used in food preparations such as jams, jellies, milkshakes, and ice creams. It also serves as a useful raw material for industrial production of glucose and pectin.7
Phytochemical profiling of sapota fruit indicates the presence of proteins, carbohydrates, phenolic compounds, carotenoids, and essential minerals such as iron, copper, zinc, and calcium, along with vitamins A, E, and C. The fruit exhibits notable antioxidant activity against free radicals including 1,1-diphenyl-2-picrylhydrazyl (DPPH) and superoxide radicals. The broad-spectrum radical scavenging potential of sapota juice has been attributed to polyphenolic constituents such as gallic acid, catechin, chlorogenic acid, ellagic acid, and leucoanthocyanidins.8,9
Materials and Methods
Chemicals
Aluminium chloride, Ascorbic acid, Ferric chloride, Folin–Ciocalteu reagent,DPPH- 1,1-diphenyl-2-picrylhydrazyl,Gallic acid (Yucca enterprises, Mumbai), Ellagic acid (Yucca enterprises, Mumbai), sodium carbonate, sodium nitrite sodium hydroxide , Rutin (Yucca Enterprises,Mumbai),Analytical grade Solvents -Toluene, Ethyl acetate, Methanol, formic acid, Ethanol, Glacial acetate acid.
Plant material
The fruiting branch of Sapota was collected from Deola village, Nashik district, Maharashtra, for authentication. A herbarium specimen was prepared and deposited at the Botanical Survey of India, Pune. The plant material was authenticated and a certificate (BSI/WRC/100-1/Tech./2022 dated 18/04/2022) was issued for Manilkara zapota (L.) P. Royen (family Sapotaceae). Subsequently, bulk quantities of the fruits were collected, cut into small pieces, shade-dried, coarsely powdered, passed through sieve no. 12, and stored in airtight polybags for further use.
Equipments
HPTLC CAMAG Switzerland- Twin through chamber, Win CATS Software,TLC plate Visualizer, Linomat Syringe V, TLC Scanner 3,Digi store – Reprostar 3, Pre-coated silica gel 60 F 254 aluminum plates (0.2mm thick, Merck, Germany)
Brookfield viscometer LV DV-II +PRO,LABINDIA Digital pH conductivity meter.
Extraction
Dried fruit powder was extracted with ethanol using Soxhlet assembly at temperature not more than 60 ͦC. Alcoholic extract was filtered, concentrated by rotary evaporation,yield was calculated and the extract was designated as MZE (Manilkara zapota extract).
The obtained extract (MZE) was subsequently subjected to phytochemical screening, quantitative estimation, and antioxidant evaluation prior to formulation development.
Phytochemical Screening of MZE
One hundred mg of MZE was dissolved in 100 mL of alcohol and filtered. The resultant extract subjected for phytochemical tests for carbohydrates, proteins, amino acids, flavonoids, alkaloids, and tannins.
Quantification of Total Polyphenols, Flavonoids, and Flavonols in MZE10-12
Total Phenolic Content (TPC) in MZE
The total phenolic content of MZE was estimated using the Folin–Ciocalteu colorimetric approach and expressed as gallic acid equivalents. A calibration curve was prepared using gallic acid (10–100 µg/mL). The extract solution (1000 ppm) was treated with diluted Folin–Ciocalteu reagent, followed by sodium carbonate solution, and allowed to react under dark conditions for 2 h. The absorbance was recorded at 760 nm against a reagent blank. Measurements were carried out in triplicate, and phenolic content in MZEwas calculated from the regression equation derived from the standard curve.
Total Flavonoid Content (TFC) in MZE:
Flavonoid content was quantified by the Aluminium Chloride method using Rutin as the reference standard (100–500 µg/mL). The reaction involved sequential addition of sodium nitrite, aluminium chloride, and sodium hydroxide to the extract (1000 ppm) and standard solutions, followed by dilution to volume. The resulting chromophore intensity was measured at 510 nm. All measurements were performed in triplicate, and the flavonoid content in MZE was determined using the corresponding calibration curve.
Total Flavonol Contentin MZE:
Flavonol estimation was carried out using a modified Aluminium Chloride assay with Rutin as standard (100–500 µg/mL). The extract (1000 ppm) was reacted with aluminium trichloride and sodium acetate solution and incubated at ambient temperature for 2.5 h. The absorbance was measured at 440 nm. Triplicate readings were taken, and flavonol content in MZE was calculated using the regression equation obtained from the standard curve.
Antioxidant Activity by DPPH Radical Scavenging Assay for MZE13,14
The antioxidant potential of MZE was assessed using the DPPH free radical scavenging method, with ascorbic acid as the reference compound. Stock solutions of ascorbic acid, DPPH, and MZE were prepared in methanol and further diluted to obtain the required concentration ranges. Each test solution was mixed with DPPH reagent and incubated for 15 min in the dark. Absorbance was then recorded at 517 nm against a control.
The percentage radical scavenging activity was calculated using the equation:
% Inhibition =(Absorbance control – Absorbance solution) / Absorbance control × 100. All experiments were conducted in triplicate. A plot of percentage inhibition versus concentration was constructed, and the IC50 values for both ascorbic acid and MZE were determined.
Preparation of ‘Avleha’ based formulation15-17
For formulation development, different batches of Avleha were designed (Table 1).The composition of batches was varied primarily to optimize viscosity and palatability while maintaining therapeutic potential. The general procedure for Avleha preparation was followed as described in the Ayurvedic Formulary of India.
A weighed quantity of jaggery was dissolved in water (Q.S.) and filtered to remove foreign particles. The solution was boiled over moderate heat with continuous stirring. During the heating process, the stage of paka was monitored by pressing a small mass between two fingers. When the syrup attained a thread-like consistency (tantuvatva) or when a small portion sank in water without dissolving readily, heating was stopped. At this stage, the weighed quantity of MZE extract was incorporated with continuous stirring to obtain a homogeneous mass. Subsequently, the weighed quantity of ghee was added with stirring until uniformity was achieved. The mixture was then allowed to cool, after which honey was added with constant stirring. The final formulations were designated as Batch I to Batch V.
Table 1: Formulation design for different batches of Avleha
| Batches | ||||||
| Ingredient in % | I | II | III | IV | V | Use |
| Extract (MZE* ) | 25 | 25 | 25 | 25 | 25 | Antioxidant Nutraceutical |
| Jaggary | 35 | 40 | 45 | 50 | 55 | Sweetener & viscosity builder |
| Ghee | 25 | 20 | 15 | 10 | 5 | Digestive, Nutritive |
| Honey | 15 | 15 | 15 | 15 | 15 | Antibacterial |
| Water | QS | QS | QS | QS | QS | Vehicle |
MZE *- Manilkara zapota extract
Evaluation of formulation for pH, viscosity and organoleptic parameters18,19
The prepared formulations (Batch I–V) were evaluated for pH, viscosity, and organoleptic characteristics. Viscosity was measured using a Brookfield viscometer under the following conditions: temperature 25.6 °C, spindle no. 64 (LV-4), speed 20 rpm, and sample weight 100 g. The viscosity values were recorded in centipoise (cP).
The pH was determined using a calibrated digital pH meter for a 10% w/v aqueous solution of each batch (Batch I–V). Organoleptic evaluation, including color, odor, taste, and consistency, was carried out by visual and sensory assessment.
Based on the evaluation results, Batch III was selected for further HPTLC quantification and validation studies.
Sample preparation for HPTLC
Treatment to formulation MZF
Two grams of the selected Batch III Avleha were mixed with water and extracted with ethyl acetate. The ethyl acetate layer was collected, concentrated, and reconstituted in 1 mL of methanol. The resulting solution was designated as MZF (Manilkara zapota formulation).
Sample for extract (MZE)
Two hundred milligrams of the alcoholic extract of Manilkara zapotafruits were dissolved in 1 mL of methanol and designated as MZE.
Standard Gallic acid solution (SG)
Five milligrams of standard gallic acid were dissolved in 5 mL of methanol in a volumetric flask to obtain a stock solution of 1000 ppm. From this stock, a working solution of 100 ppm was prepared in methanol and designated as SG.
Standard Ellagic acid solution (SE)
Five milligrams of standard ellagic acid were dissolved in 5 mL of methanol in a volumetric flask to obtain a stock solution of 1000 ppm. From this stock, a working solution of 100 ppm was prepared in methanol and designated as SE.
All sample and standard solutions were filtered through Whatman No. 1 filter paper prior to application.
HPTLC
Chromatographic condition
Stationary phase:Pre-coated silica gel G60 F254 HPTLC plates (0.2mm thick),
Mobile phase: Toluene: Ethyl acetate: Formic acid: Methanol (6:6:1.6:0.4 v/v/v/v)20,
Spraying reagent – Ferric chloride (5% alcoholic),
Saturation time – 15 min,
Wavelength – 275 nm (Isoabsorbtive between Gallic acid and Ellagic acid spectra),
Scanning silt width – 6×0.45 mm,
Scanning speed – 20mm/s, Sample band length -8 mm,Solvent run – 80 mm ,
Room temperature -16 ͦ C± 2 °C. , Syringe volume -100 µL,
Spray gas – nitrogen gas,
Sample application speed – 0.2 µL/s,
Sample Applicator –Linomat 5,
Scanner 4 – CAMAG TLC Scanner 3,
Photo documentation – CAMAG REPROSTAR 3
Prevalidation21-23
Simultaneous Wavelength selection , Rf values, and fingerprinting of standard gallic acid (SG) and standard ellagic acid (SE) in the plant extract (MZE) and in the formulation (MZF)
The chromatographic conditions described above were employed using 20 × 20 cm HPTLC plates. Sample application was performed as follows: SG on tracks 1 and 2 (1 and 2 µL, respectively); MZE on tracks 3, 4, and 5 (2, 3, and 4 µL, respectively); MZF on tracks 6, 7, and 8 (2, 3, and 4 µL, respectively); and SE on tracks 9 and 10 (1 and 2 µL, respectively).
Initial scanning was carried out at 254 nm in absorbance mode. Spectral scanning of selected spots was then performed over the range of 200–400 nm. The overlaid spectra for the spots of SG and SE were used to identify the isoabsorptive wavelength suitable for simultaneous densitometric estimation of both biomarkers. The Rf values of both standards were recorded, and three-dimensional Rf overlays of SG, SE,MZF and MZE were compared to confirm peak identity. The selected isoabsorptive wavelength was subsequently used for further densitometric quantification and method validation.This ensured reliable simultaneous estimation of both biomarkers at a single wavelength.
Specificity: Spectral Overlay, Spiking, and Derivatization Study
Specificity of the method was evaluated by spectral overlay and spiking experiments under the optimized chromatographic conditions using 20 × 20 cm HPTLC plates. Samples were applied as bands in the following track positions and volumes: MZE on tracks 1 and 2 (2 and 3 µL, respectively); SG on tracks 3 and 4 (1 and 2 µL, respectively); SE on tracks 5 and 6 (1 and 2 µL, respectively); SG + MZE on tracks 7 and 8 (1 and 2 µL of SG spiked into 2 and 3 µL of MZE, respectively); and SE + MZE on tracks 9 and 10 (1 and 2 µL of SE spiked into 2 and 3 µL of MZE, respectively).
After development, the plate was scanned densitometrically at 275 nm. The spots observed in MZE and in the spiked tracks (tracks 7–10) that exhibited Rf values corresponding to SG and SE were subjected to spectral scanning, and the resulting spectra were overlaid to confirm peak purity and specificity as shown in Fig. 1.The plate was then derivatized with 5% ethanolic Ferric chloride solution, and the same spots were visualized to further confirm identity.
Simultaneous Quantification of Gallic acid and Ellagic acid by Linearity in MZE and MZF24-27
Simultaneous densitometric quantification of gallic acid and ellagic acid in the plant extract (MZE) and formulation extract (MZF) were performed using the optimized chromatographic conditions on 20 × 20 cm HPTLC plates. Sample application was carried out as follows: SG was applied on tracks 1–6 at volumes of 2, 3, 4, 5, and 6 µL; MZE was applied on tracks 7 and 8 at 1.0 and 1.5 µL, respectively; MZF was applied on tracks 9 and 10 at 1.0 and 1.5 µL,respectively; and SE was applied on tracks 11–16 at volumes of 2, 3, 4, 5, and 6 µL.
The plates were developed under the chromatographic conditions described above and densitometrically scanned at 275 nm. The procedure was repeated in triplicate on three different plates. The average area under the curve (AUC) and percentage relative standard deviation (% RSD) were calculated. Calibration curves were constructed in Figs. 2 a and 2 b by plotting average AUC versus concentration (ng/spot) for SG and SE. The contents of gallic acid and ellagic acid in MZE and MZF were calculated using the respective regression equations.
Method Validation (as per ICH Q2 (R1) Guidelines)28-33
Method validation was performed in accordance with International Council for Harmonisation guidelines Q2 (R1) for linearity, precision, accuracy, LOD, LOQ, and robustness.
LOD, and LOQ
The limit of detection (LOD) and limit of quantification (LOQ) were determined using the following equations, LOD = 3.3 × (σ/S)and LOQ = 10 × (σ/S) , where σ is the standard deviation of the response and S is the slope of the calibration curve. The values are reported in Table 3.
Precision
Intermediate Precision (Intra-day and Inter-day)
Precision was evaluated for SG and SE over three consecutive days at three different time intervals (morning, afternoon, and evening). Three concentrations (2, 4, and 6 µL) were analyzed in triplicate under the optimized chromatographic conditions using 20 × 20 cm plates. The samples were scanned at 275 nm, and the average AUC was recorded. The corresponding amounts were calculated using the regression equation, and %RSD values were determined.
Repeatability
Repeatability was assessed by applying the mid-level concentration (4 µL) of SG and SE separately, nine times on the same plate. The plates were developed under the same chromatographic conditions and scanned at 275 nm. The %RSD of AUC was calculated.
Accuracy (% Recovery)
Accuracy was determined by the recovery study at three levels (80%, 100%, and 120%). Known amounts of SG and SE were spiked into MZE and analyzed under the optimized conditions. The plates were scanned at 275 nm, and the average AUC was recorded. The percentage recovery and %RSD were calculated. Results are presented in Table 4.
Robustness
Robustness was evaluated by introducing small deliberate variations in chromatographic conditions. The mid-level concentration (4 µL) of SG and SE was applied in triplicate. Chromatographic conditions are challenged as: Wavelength – 280 nm, Slit width – 6 × 0.3. mm, scan speed – 40 mm/s, Mobile phase – Toluene: Ethyl acetate: Formic acid: Methanol (4:4:2:0.5), Saturation time – 20 min. The average AUC and %RSD were calculated to assess method robustness.
Results
The ethanolic extract of M. zapota (MZE) exhibited a yield of 14.68% w/w and appeared brown in color with a characteristic odor. Phytochemical screening of MZE showed positive results for carbohydrates, proteins, amino acids, flavonoids, and tannins. The extract was further subjected to quantitative estimation of total phenolic, flavonoid, and flavonol contents, and the results are presented in Table 2. MZE also demonstrated significant antioxidant activity as determined by the DPPH assay, with the corresponding results included in Table 2.
Table 2: Quantification of Total Polyphenolic, Total Flavonoid and Total Flavonol content in MZE ; Antioxidant activity by DPPH for MZE.
| a) Quantification of Total Polyphenolic, Total Flavonoid and Total Flavonol content in MZE | ||||
| Parameters | Total Polyphenolic | Total flavonoid | Total flavonol | |
| Estimation | As Gallic Acid equivalence
= 290 mg/gm |
as Rutin equivalence
= 31.06 mg/gm |
As Rutin equivalence
=2.5 mg/gm |
|
| Linearity Range | 10 to 100 μg/ml | 100 to 500 μg/ml | 100 to to 500 μg/ml | |
| Regression Equation, Corelation Coefficient | Y= 0.2994x – 0.1783,
R2= 0.9438
|
Y= 0.0003X -0.0132,
R2 = 0.9753
|
Y= 0.003X +0.3305,
R2 = 0.967 |
|
| Absorbance of MZE ± S.D. (n=3), | 0.692 ± 0.0005
|
0.080 ± 0.0179, | 0.338 ± 0.0010 | |
| b) Antioxidant activity By DPPH method in MZE | ||||
| IC 50 Std Ascorbic Acid ,
Average Absorbance ± S.D. (n=3) Control, Conc. 2 to 10 μg /ml |
4.12 μg /ml
0.9836 ± 0.0011 , 0.662 ± 0.0251, to 0.1153 ±0.0011 |
|||
| IC 50 MZE
Average Absorbance ± S.D. (n=3) Control, Conc. 2000 to 10,000 μg /ml
|
5180 μg /ml
0.9936 ± 0.011 , 0.6353 ± 0.0011 to 0.293 ± 0.001
|
|||
MZE- Manilkara zapota extract, IC 50 – 50 % Inhibition concentration ,S.D.-Standard deviation
For formulation development, different batches of Avleha (Batch I–V) were prepared and evaluated for pH, viscosity, and organoleptic characteristics. All batches exhibited semisolid consistency with brown color, sweet taste, and characteristic odor. The viscosity of the formulations increased progressively from Batch I (10525 cP) to Batch V (64765 cP), indicating the effect of varying composition on rheological properties. The pH of all batches was found to be near neutral (6.4–7.09), suggesting suitability for oral administration. Among all, Batch III showed optimum characteristics with a viscosity of 18036 cP, appropriate semisolid consistency, non-sticky nature, and acceptable organoleptic properties, and was therefore selected for further analysis.
In the fingerprinting study, using the optimized chromatographic conditions, the mobile phase system of Toluene: Ethyl acetate: Formic acid: Methanol (6:6:1.8:0.4 v/v/v/v) provided well-resolved peaks with Rf values of 0.39 and 0.29 for SG and SE, respectively. Corresponding spots with identical Rf values were observed in MZE, indicating the presence of these biomarkers in the extract. Spectral scanning of the selected spots showed maximum absorbance at 280 nm for SG and 276 nm for SE. Overlay of the spectra revealed an isoabsorptive point at 275 nm, which was selected for simultaneous densitometric estimation.
In the specificity study, spectral overlay of SG and SE with the corresponding spots in MZE and spiked samples exhibited similar spectral patterns at matching Rf values, shown in Fig. 1. Further confirmation was achieved through derivatization with Ferric chloride reagent, which produced characteristic blue coloration for SG and SE, also observed in MZE and spiked tracks, confirming their presence in the extract.
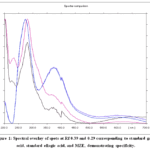 |
Figure 1: Spectral overlay of spots at Rf 0.39 and 0.29 corresponding to standard gallic acid, standard ellagic acid, and MZE, demonstrating specificity.
|
The calibration curves for SG and SE exhibited good linearity over the studied concentration range. The quantified amounts of gallic acid and ellagic acid in MZE and MZF are presented in Table 3. The chromatograms obtained showed well-resolved and distinct peaks for both biomarkers (Figs. 3 and 4 a to d).
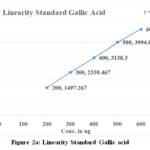 |
Figure 2a: Linearity Standard Gallic acid
|
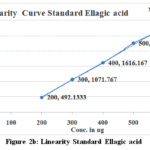 |
Figure 2b: Linearity Standard Ellagic acid
|
 |
Figure 3: Photodocumentation of Linearity plate at 254 nm
|
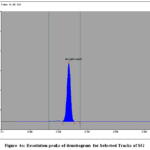 |
Figure 4a: Resolution peaks of densitogram for Selected Tracks of SG
|
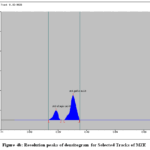 |
Figure 4b: Resolution peaks of densitogram for Selected Tracks of MZE
|
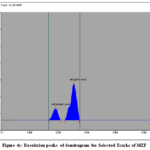 |
Figure 4c: Resolution peaks of densitogram for Selected Tracks of MZF
|
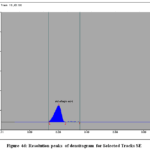 |
Figure 4d: Resolution peaks of densitogram for Selected Tracks SE
|
Table 3: Validation parameters for simultaneous estimation Gallic acid and Ellagic acid
| Parameter | Gallic Acid | Ellagic Acid |
| Wavelength nm | 280 | 276 |
| Isoabsorptive Wavelength | 275 nm | 275 nm |
| Rf Value | 0.39 | 0.29 |
| Linearity Range ng /band | 200-600 | 200 -600 |
| Regression Equation | Y=8.0709x -93.459 | Y= 5.6407x -625.18 |
| Correlation Coefficient | 0.9992 | 0.9983 |
| Limit of Detection ng/band LOD | 6.23 | 9.09 |
| Limit of Quantification ng/band LOQ | 18.89 | 27.56 |
| Specificity | Retention factor, overlay spectra, derivatization. | Retention factor, overlay spectra , derivatization |
| Simultaneous estimation in MZE | 0.6312 % w/w | 0.6918 % w/w |
| Simultaneous estimation in MZF | 0.09421 % w/w | 0.04420 % w/w |
The precision of the method, including repeatability and intermediate precision, showed low %RSD values, indicating good reproducibility (Table 4).
The accuracy of the method, evaluated by recovery studies at 80%, 100%, and 120% levels, demonstrated acceptable recovery for both SG and SE (Table 5).
The robustness study indicated that small deliberate variations in chromatographic conditions did not significantly affect the results, confirming the reliability of the method.
Table 4: Intermediate precision study for standard Gallic acid and Ellagic acid.
| Concentration (ng/band) | Average Concentration (n=3) Intraday
(ng /band ) |
Intra day
%RSD |
Average Concentration (n=3) Inter day (ng / band ) |
Inter day
%RSD |
|
| 200 | 186.76 | 0.3386 | 182.23 | 3.4321 | |
| SG | 400 | 393.96 | 1.5115 | 397.61 | 0.9749 |
| 600 | 589.1 | 0.1149 | 590.09 | 0.1711 | |
| 200 | 207.56 | 1.1279 | 207.39 | 1.1886 | |
| SE | 400 | 389.28 | 1.8520 | 399.65 | 3.7829 |
| 600 | 609.29 | 0.4771 | 609.29 | 0.7574 |
Table 5: Accuracy by % recovery for Gallic acid and Ellagic acid in MZE (80, 100, 120%)
| In MZE | Level of % Recovery | Amount in MZE (ng) | Amount of standard Added (ng ) | Total amount of standard taken (ng) | Total amount of standard obtained (ng) | %Recovery ±SD (n=3) |
| SG | 80 | 366.73 | 293.38 | 660.11 | 658 | 99.68 |
| 100 | 366.73 | 366.73 | 733.46 | 723.87 | 98.69 | |
| 120 | 366.73 | 440.07 | 806.8 | 743.18 | 92.11 | |
| SE | 80 | 336.85 | 269.48 | 606.33 | 647.44 | 108.78 |
| 100 | 336.85 | 336.85 | 673.7 | 681.84 | 101.2 | |
| 120 | 336.85 | 404.22 | 741.07 | 723.87 | 97.67 |
Discussion
The ethanolic extract of M. zapota (MZE) exhibited a satisfactory yield, indicating efficient extraction of bioactive constituents. Phytochemical screening confirmed the presence of key secondary metabolites such as flavonoids and tannins, which are well known for their antioxidant potential.
The quantitative estimation revealed appreciable levels of total phenolic, flavonoid, and flavonol content in MZE. These phytoconstituents are widely reported to contribute significantly to free radical scavenging activity, which is supported by the observed antioxidant potential of the extract in the DPPH assay. The results indicate a direct correlation between phenolic content and antioxidant activity, justifying the selection of MZE for formulation development.
The Avleha formulations were successfully prepared and evaluated, where variation in composition influenced the rheological properties. An increase in viscosity was observed with higher jaggery content, indicating its role as a viscosity-enhancing agent. Among the batches, Batch III demonstrated optimum characteristics in terms of viscosity, consistency, pH, and organoleptic properties, making it suitable for further standardization.
HPTLC fingerprinting provided clear resolution of gallic acid and ellagic acid with distinct Rf values, confirming their presence in the extract. The selection of an isoabsorptive wavelength enabled simultaneous estimation of both biomarkers with improved accuracy. Specificity studies, including spectral overlay and derivatization, further confirmed the identity and purity of the peaks.
The developed method showed good linearity, precision, and accuracy, indicating its suitability for quantitative analysis. The low %RSD values observed in precision studies reflect the reproducibility of the method, while acceptable recovery values confirm its accuracy. Robustness results suggest that minor variations in chromatographic conditions do not significantly affect the analysis, demonstrating the reliability of the method.
Overall, the presence of bioactive phenolic compounds along with significant antioxidant activity supports the potential of M. zapota extract as a functional ingredient in nutraceutical formulations. The validated HPTLC method ensures reliable standardization of the developed Avleha formulation.
Conclusion
The present study demonstrates the successful development and standardization of an Avleha formulation using M. zapota extract as a promising antioxidant nutraceutical. The significant phytochemical content and antioxidant activity of the extract justify its therapeutic potential. The validated HPTLC method provides a reliable tool for simultaneous estimation of gallic acid and ellagic acid, ensuring quality control of the formulation. This work supports the utilization of M. zapota in the development of scientifically validated herbal formulations.
Acknowledgement
The authors are thankful to Mahatma Gandhi Vidyamandir’s Pharmacy College, Nashik, for providing laboratory facilities.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable.
Author Contributions:
Daksha Lalit Attarde: Conceptualization, Methodology, Data Collection, Investigation,Writing, Review ,Editing and Supervision.
Sarika Tanaji Nikam: Conceptualization, Literature review, Methodology, Formal analysis, Investigation, Data Collection, Writing.
Komal Bhausaheb Dawange: Literature review, Data Collection, Writing, Review and Editing.
References
- Pandey P, Pal R, Koli M, Malakar RK, Verma S, Kumar N, Kumar P. A traditional review: The utilization of nutraceutical as a traditional cure for the modern world at current prospectus for multiple health conditions. Drug Deliv. Ther. 2024;14(3):154–163. doi:10.22270/jddt.v14i3.6445.
CrossRef - Taylor CL. Regulatory frameworks for functional foods and dietary supplements. Rev. 2004;62(2):55–59.doi:10.1111/j.1753-4887.2004.tb00024.x.
CrossRef - Ahmed T, Burhanuddin M, Haque MA, Hossain MA. Preparation of jam from Sapota (Achras zapota). 2011;9(1–2):1-7.doi:10.3329/agric.v9i12.9473.
CrossRef - Ujjaliya N, Dash S, Jain SK. A review on nutraceuticals in Ayurveda. World J. Pharm. Pharm. Sci. 2018;7(5):277–281. doi:10.20959/wjpps20185-11447.
CrossRef - Singh S, Bhatt AK, Manohar AK. Nutraceuticals in Ayurveda: Bridging ancient wisdom with modern nutrition. World J. Pharm. Res. 2025;14(9):933–938. doi:10.20959/wjpr20259-36324.
CrossRef - Ma J, Luo X, Protiva P, Yang H, Ma C, Basile MJ, Weinstein IB, Kennelly EJ. Bioactive novel polyphenols from the fruit of Manilkara zapota (Sapodilla). Nat. Prod. 2003;66:983–986. doi:10.1021/np020576x.
CrossRef - Kritikar KR, Basu BD. Indian medicinal plants. 2nd ed. Periodical Experts Book Agency; 1984. pp 1486–1487
- Gomathy K, Baskar R, Kumaresan K. Comparison of antioxidant potential in pulp and peel extracts of Manilkara zapota (L.) P. Royen. J. Biotechnol. 2013;12(31):4936–4943. doi:10.5897/AJB2012.2961.
CrossRef - Kulkarni AP, Policegoudra RS, Aradhya SM. Chemical composition and antioxidant activity of sapota (Achras sapota Linn.) fruit. Food Biochem. 2007;31(3):399–414. doi:10.1111/j.1745-4514.2007.00122.x.
CrossRef - Nortjie E, Basitere M, Moyo D, Nyamukamba P. Extraction methods, quantitative and qualitative phytochemical screening of medicinal plants for antimicrobial textiles: A review. 2022;11(15):2011. doi:10.3390/plants11152011.
CrossRef - Singleton VL, Orthofer R, Lamuela-Raventós RM. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteu reagent. Methods Enzymol. 1999;299:152–178. doi:10.1016/S0076-6879(99)99017-1.
CrossRef - Chang CC, Yang MH, Wen HM, Chern JC. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. Food Drug Anal. 2002;10(3):178–182.doi: 10.38212/2224-6614.2748
CrossRef - Kumaran A, Karunakaran RJ. Antioxidant and free radical scavenging activity of an aqueous extract of Coleus aromaticus. Food Chem. 2007;100(1):356–361. doi:10.1016/j.foodchem.2005.09.040.
CrossRef - Roman Cortes NR, De Jesus Ariza Ortega T, Cruz Munoz R. Phytochemical content and antioxidant potential of tropical sapodilla fruit (Manilkara zapota). 2022;77(3). doi:10.17660/th2022/013.
CrossRef - Government of India, Ministry of Health & Family Welfare. The Ayurvedic formulary of India. 2nd revised English ed. Part I. 2003;31.
- Pandey MM, Rastogi S, Rawat AKS. Indian traditional Ayurvedic system of medicine and nutritional supplementation. -Based Complementary Altern. Med. 2013.doi: 10.1155/2013/376327.
CrossRef - Patel JR, Tripathi P, Sharma V, Chauhan NS, Dixit VK. Phyllanthus amarus: ethnomedicinal uses, phytochemistry and pharmacology. Ethnopharmacol. 2011.doi: 10.1016/j.jep.2011.01.040.
CrossRef - Mishra K, Shukla K, Pal P, Mahajan SC. Comparative standardization of a polyherbal Ayurvedic formulation: Talisadi churna. J. Pharm. Technol. 2015;7(1):34–37. doi:10.5958/0975-4385.2015.00008.4
CrossRef - Royal Society of Chemistry. The Merck Index: An encyclopedia of chemicals, drugs and biologicals. 12th ed. 1996;4364.
- Wagner H, Bladt S. Plant drug analysis: A thin layer chromatography atlas. 2nd ed. Springer; 1996; 360–361.
- Sima IA, Sârbu C. Evaluation of polyphenolic fingerprints and antioxidant profiles of wild fruits. J. Food Sci. Technol. 2016;51(6):1433–1440. doi:10.1111/ijfs.13111.
CrossRef - Bagul M, Srinivasa H, Padh H, Rajani M. A rapid densitometric method for simultaneous quantification of gallic acid and ellagic acid in herbal raw materials using HPTLC. Sep. Sci. 2005;28(6):581–584. doi: 10.1002/jssc.200301695
CrossRef - Shulammithi R, Sharanya M, Tejaswini R, Kiranmai M. Standardization and quality evaluation of herbal drugs. IOSR J. Pharm. Biol. Sci. 2016;11(5):89–100. doi:10.9790/3008-11050189100.
CrossRef - Mohamed ITA, Khan W, Chester K, Mohamed AH, Ahmad S, Ayoub SMH. Simultaneous quantitative estimation of ellagic acid and gallic acid in Sudanese Solanum dubium seed by high performance thin layer chromatography (HPTLC). GSC Biol. Pharm. Sci. 2020;13(1):054–061.doi:10.30574/gscbps.2020.13.1.0311.
CrossRef - Shrivastava S, Dubey PK, Shrivastava B, Sharma P. Development and validation of HPTLC method for estimation of ellagic acid in antidiabetic herbal formulation. Drug Deliv. Ther. 2017;7(7):180–182. doi: 10.22270/jddt.v7i7.1628
CrossRef - Cimpoiu C. Analysis of some natural antioxidants by thin-layer chromatography and high performance thin-layer chromatography. Liq. Chromatogr. Relat. Technol. 2006;29(7–8):1125–1142. doi:10.1080/1082607060057491.
CrossRef - Attimarad M, Mueen Ahmed KK, Aldhubaib BE, Harsha S. High-performance thin layer chromatography: A powerful analytical technique in pharmaceuticaldrug discovery. Pharm. Methods. 2011;2(2):71–75. doi:10.4103/2229-4708.84436.
CrossRef - Patel KG, Patel VG, Patel KV, Gandhi TR. Validated HPTLC method for quantitative determination of gallic acid in stem bark of Myrica esculenta. AOAC Int.2010;93(5):1422–1427.doi: 10.1093/jaoac/93.5.1422
CrossRef - Godiyal S, Laddha K. Validated high-performance thin-layer chromatographic method for quantification of gallic acid and ellagic acid in fruits. Sep. Sci. 2023.doi: 10.1002/jssc.202200991
CrossRef - Meena AK, Narasimhaji CV, Velvizhi D, Singh A, Rekha P, Kumar V, Ilavarasan R, Srikanth N, Dhiman KS. Determination of gallic acid in Ayurvedic polyherbal formulation Triphala churna and its ingredients by HPLC and HPTLC. J. Pharm. Technol. 2018;11(8):3243–3249. doi:10.5958/0974-360X.2018.00596.6.
CrossRef - Patel BC, Patel NM. Standardisation of Bhumyamalaki Churna: An Ayurvedic polyherbal formulation. J. Pharm. Sci. Res. 2013;4(10):4010–4015. doi:10.13040/IJPSR.0975-8232.4(10).4010-15.
CrossRef - Singh DP, Govindarajan R, Rawat AKS. High-performance liquid chromatography as a tool for the chemical standardisation of Triphala—an Ayurvedic formulation. Anal. 2007;19(2):164–168. doi:10.1002/pca.10.
CrossRef - Mukherjee PK, Ponnusankar S, Pal BC. Marker profiling: An approach for quality evaluation of Indian medicinal plants. Ther Innov Regul Sci. 2011;45(1):1–14. doi:10.1177/009286151104500101.
CrossRef
Abbreviations
AUC – Area Under Curve
cP – centipoise
DPPH– 1,1-Diphenyl-2-picrylhydrazyl
g– gram
HPTLC – High Performance Thin Layer Chromatography
ICH – International Council for Harmonisation
IC50 – half maximal inhibitory concentration
LOD – Limit of Detection
LOQ – Limit of Quantification
mm– millimeter
min– minutes
MZE – Manilkara zapota Extract
MZF – Manilkara zapota Formulation
nm – nanometer
ppm – parts per million
Q.S– Quantity sufficient
Rf – Retention factor
RSD – Relative Standard Deviation
SD – Standard Deviation
SE – Standard Ellagic Acid
SG – Standard Gallic Acid
TFC – Total Flavonoid Content
TPC – Total Phenolic Content
°C – degree celsius
μg – microgram
mL –milliliter
μL – Microliter
rpm– revolutions per minute
v/v: volume per volume
w/w: weight per weight
%: Percentage
σ: sigma
S: slope
Accepted on: 19-04-2026
Second Review by: Dr. Anjaneyulu Vinukonda
Final Approval by: Dr. Wagih Ghannam









