Pathophysiological Mechanisms of High Fat Diet Induced Neurological Disorders: An Impending Approach
Department of Pharmacology, Gokaraju Rangaraju College of Pharmacy, Hyderabad, India
Corresponding Author Email: vivek8033@grcp.ac.in
Download this article as:
ABSTRACT:The brain and the spinal cord make up the central nervous system (CNS), which regulates important sensory, motor, cognitive, and autonomic functions. Evidence is accruing to the fact that high-fat diets (HFDs) play a significant role in the development and progression of neurological diseases. HFDs are unhealthy since the chronic use leads to metabolic imbalances that result in insulin resistance, dyslipidemia, and systemic inflammation with adverse effects on the well-being of the brain. Some of the most severe pathophysiological events caused by such metabolic changes include neuroinflammation, oxidative stress(OS), mitochondrial dysfunction, and gut-brain axis dysfunction. These mechanisms enhance the deposition of amyloid-b and hyperphosphorylations of tau in the case of Alzheimer disease(AD) which results in dysfunction of the synapses and impairment of cognition. HFD causes oxidative lesions and inflammation which increase the death of dopaminergic neurons in the substantia nigra when it occurs in the case of Parkinson disease. In addition to this, HFD may modify the homeostasis of the neurotransmitters particularly those of serotonin and hence one becomes more susceptible to depression. The review provides a detailed explanation of the molecular mechanisms of the relationship between the application of high-fat diets and the development of neurological problems and identifies the potential treatment strategies that can be used as prevention and treatment options.
KEYWORDS:Alzheimer’s disease; Depression; High fat diet; Oxidative stress; Parkinsons’s disease
Introduction
The present trends of diet have undergone a paradigm shift over the past few decades and are characterized by increased consumption of high energy content processed foodstuffs that contain high amounts of saturated fats and refined sugars. Such a shift in diet to the consumption of high-fat diets (HFDs), that is usually associated with western-style dietary practices, has been closely linked to adverse health outcomes. The dynamic urbanization process, the sedentary lifestyle and the easy access to the food products rich in high calories have further contributed to the fact that the world has adapted to such a pattern of dieting at an accelerated rate and this has raised a question to the long-term effects of these diets on the health of citizens.1,2
With these nutritional modifications, obesity and metabolic diseases, including insulin resistance, type 2 diabetes mellitus, and dyslipidemia, have become especially sharp everywhere in the world. According to the recent epidemiological literature, obesity has become pandemic and this has been a significant burden to the healthcare systems across the world.3,4
These metabolic disorders not only affect peripheral organs but the effect on the brain structure and functioning is great.
Based on the emerging evidence, metabolic dysfunction is strongly correlated with neurological disorders. Some of the mechanisms that mediate the relationship between neurodegeneration and psychiatric conditions and metabolic disturbances include chronic low-grade inflammation, oxidative stress, dysfunctional insulin signaling, and mitochondrial dysfunction.5,6
The most common neurodegenerative disease is termed as Alzheimer disease (AD), progressive and has cognitive impairment as well as the usual pathologic alterations, including amyloid-β plaques and neurofibrillary tangles of hyperphosphorylated tau protein.7In the second most prevalent neurodegenerative condition Parkinson situation (PD), is primarily associated with the depletion of dopaminergic neurons found in the substantia nigra, resulting in motor impairment, and or non-motor symptoms.8
Depression is a psychiatric condition, which is among the most prevalent, and is characterized by lowered mood, anhedonia, and disregulation of the neurotransmitter systems, particularly the serotonin signalling.9 It is increasingly becoming evident that metabolic and dietary factors, especially, the consumption of high-fat diet, could play a major part in the pathogenesis and progression of these neurological diseases.
The central nervous system (CNS) consists of the brain and spinal cord and is used in the control of vital sensory, motor, cognitive, and autonomic activity. A Central nervous system disease is any such disease that affects the structure and functioning of the brain and the spinal cord, leading to neurological, psychiatric, or neurodegenerative disorders like encephalopathy, myelopathy, Alzheimer’s disease, Parkinson’s disease, epilepsy, depression and stroke. All these are diseases that cumulatively lead to nervous system disorders.10A high- fat diet is a diet containing 35 per cent and above of the total calories that is consumed of saturated as well as unsaturated fat. Over the last few yearsresearch proves that high-fat diet causes neuroinflammation of brain leading to neural impairment affecting cognition of an individual.11There is growing evidence that a high-fat diet (HFD) leads to the development of depressive-like behaviour due to chronic neuro-inflammatory processes in the brain.12Experimental research proves that HFD triggers microglia and increases pro-inflammatory cytokines like IL-1β, IL-6, and TNF-α within the hippocampus and prefrontal cortex, which play a very critical role in mood regulation.13,14Metabolic changes caused by HFDs such as insulin resistance, and lipid signalling interfere with brain energy homeostasis and disrupt monoaminergic neurotransmission, specifically serotonergic neurotransmission.15Moreover, chronic exposure to HFD changes the gut microbiota and intestinal permeability which activates gut-brain axis and worsens depression symptoms.16HFD also further impairs neuroplasticity by decreasing the expression of brain-derived neurotrophic factor (BDNF) and disrupting the remodelling of synapses, which predisposes individuals to mood disorders related to stress.17The role of high-fat diet in the pathogenesis of Alzheimer disease (AD) has become a well-defined issue due to its role in neuro-inflammation and metabolic dysregulation.18Research in transgenic AD mouse models has shown that HFD increases the speed of deposition of amyloid-β plaques and stimulates tau hyper-phosphorylation, which worsens the hallmark AD pathology.19The insulin resistance caused by HFD in the brain suppresses the glucose metabolism and the functioning of the mitochondria, which is associated with the impairment of the synaptic functions and cognitive deterioration.20Moreover, high amounts of dietary fats interfere with integrity of blood-brain barriers and increase microglial activation to promote neurotoxic inflammatory cascades that promote neuronal death.21HFD-induces changes in gut microbiota and lipid metabolism also mediate the effects of amyloid genic processing and inflammatory signals associating peripheral metabolic dysfunction with central neurodegeneration.22There is growing experimental evidence that high-fat diet can worsen the development and progression of Parkinson disease (PD) by promoting dopaminergic neurodegeneration.23,24HFD food further enhances oxidative stress and dysfunction in the mitochondria within the substantia nigra, resulting in greater susceptibility to dopaminergic neurons. Animal experiments have demonstrated that HFD increases neuro-inflammatory and microglial functions of the nigrostriatal pathway, which increases the rate at which α-synuclein aggregates and neuronal loss occurs.25Moreover, the obesity caused by HFD increases vulnerability to neurotoxins like 6-hydroxydopamine that further deteriorates motor coordination and dopamine transmission.26Changes in the composition of gut microbiota and intestinal inflammation linked to HFD also play a role in PD pathology via the gut-brain axis by supporting peripheral inflammation and α-synuclein spread to the brain.27
Linking High-fat diet and obesity to Neuroinflammation and Brain disorders
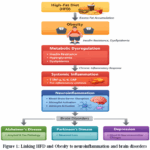 |
Figure 1: Linking HFD and Obesity to neuroinflammation and brain disorders
|
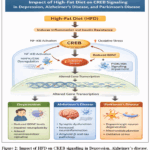 |
Figure 2: Impact of HFD on CREB signalling in Depression, Alzheimer’s disease, and Parkinson’s disease
|
Pathophysiology of Depression
HFD intake led to the depletion of serotonin in the brain and even to the reduction of the serotonin-related proteins, which is a signal of emotional dysregulation and depression like behaviour. Another effect of HFD is that it is associated with interference of the energy metabolism with the increased quantity of glucose in the blood, larger quantity of fatty acids, reactive oxygen species, and suppresses or inhibits the citric acid cycle or Krebs cycle in the brain. Further, any variation of the gut microbiome or gut bacteria is attributed to depression that typically happens due to the induction of high-fat in the diet. Ruminococcus gnavus evenly increased on high-fat diet and is primarily linked with depression like symptoms due to a lowering of the serotonin levels. The restricted high-fat diet that is calorie-dense served to preserve the healthy gut bacteria that lead to normal serotonin levels and could prevent symptoms caused by depression.28-32
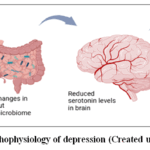 |
Figure 3: Pathophysiology of depression (Created using biorender)
|
Pathophysiology of Parkinson’s
The high-fat diets (HFD) predispose to obesity, cardiovascular diseases, and can lead to cognitive dysfunction. Research has revealed that HFD decreases the hippocampal volume, memory, decreases the synaptic plasticity, and accelerates tau protein hyperphosphorylation in the brain. HFD is composed of saturated fatty acids, and unsaturated fatty acids. Therefore, eating such high fatty acids could result in more inflammation, more gut permeability, oxidative stress, and also impaired BBB functions in the brain and all these activities may affect the nervous system, and damage the dopamine-producing neurons in the brain. All these contribute to the abnormal accumulation of α-synuclein proteins in the neurons. These proteins are then fused together to form oligomers and insoluble fibrils known as the Lewy bodies. It predominantly impacts the dopaminergic neurons found in the substantia nigra resulting in reduced dopamine release in the brain. This leads to neuronal damage and depletion of dopamine-producing neurons and causes the typical symptoms of Parkinson’s disease (tremors, impaired movements, and rigidity).33-35
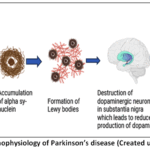 |
Figure 4: Pathophysiology of Parkinson’s disease (Created using biorender)
|
Pathophysiology of Alzheimer’s.
Consumption of high-fat food leads to obesity. Obesity is primarily identified by the proliferation of adipose tissue that acts as a body fatty acid storage organ and it is also an endocrine tissue. Some of the bioactive molecules that are secreted by adipose tissue are termed adipokines. These adipokines exhibit pro-inflammatory mediators, including leptin, interleukin (IL) A/7, angiotensinogen, resistin, and visfatin that induce neuronal inflammation. Dementia is a progressive cognitive dysfunction marked by the ageing of the population. The development of the deposition of the amyloid-β plaques is the outcomes of cleavage of amyloid precursor proteins and hyperphosphorylation of the tau proteins, loss of synapses or communication between the neurons, and neurotoxic events that cause cognitive impairment, leading to Alzheimer’s. Alzheimer’s disease is mainly caused by unhealthy diets. AD is characterized by a reduction in the availability of acetylcholine primarily by acetylcholinesterase. Numerous studies have demonstrated that HFDs cause a lot of risk of cognitive dysfunctions. A High-fat diet also produces AD-like findings like amyloid-beta depositions and tau protein phosphorylation.36-38
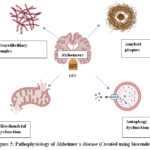 |
Figure 5: Pathophysiology of Alzheimer’s disease (Created using biorender)
|
Discussion
The present review recognizes the significant role of high-fat diets (HFDs) to the central nervous system (CNS) disorders as the most important point of its significance given the significance of the number of modifiable risk factors in the pathogenesis of Alzheimer disease, Parkinson disease, and depression. HFD in the long-run causes obesity and metabolic unbalance characterized by insulin resistance, dyslipidemia, and adipokine signaling disruption which in combination cause systemic inflammation. The resultant metabolic dysbalance triggers pathophysiological mechanisms containing neuroinflammation, oxidative stress, mitochondria dysfunction, and gastrointestinal tract-brain axis dysfunction that ultimately intercept neuronal physiological functioning and existence. HFD alters important signaling pathways, neurotrophic support, synaptic plasticity, and inflammatory signatures at the molecular scale by modulating including CREB, PI3K/Akt, and NF-kB. Such disease-specific phenomena as accumulation of amyloid-β and hyperphosphorylation of tau in Alzheimer’s disease, degeneration of dopaminergic neurons and a-synuclein aggregation in Parkinson’s disease, neurotransmitter imbalance and reduced neuroplasticity in depression are involved in the processes. It is important to note that in such observations the applicability of dietary interventionswhere less saturated fats, improved metabolic status, and a proper mixture of intestinal microbiota can be applied to abate neuroinflammatory and neurodegenerative processes. However, further research is required to improve a more precise comprehension of target molecular pathways, use multi-omics technique, and develop particular nutritional and therapeutic interventions to prevent and treat neurological disorders due to HFD.
Conclusion
Lastly, neurodegeneration is also induced by high-fat diets (HFD) since significant processes of metabolic dysregulation and oxidative stress, mitochondrial dysfunction, and chronic neuroinflammation are triggered and that can impair neuronal functions and cause pathogenesis of Alzheimer, Parkinson and depression. These findings justify the importance of dietary interventions as a modifiable intervention, where a saturated fat lowering and metabolic health would lower or alleviate neuroinflammatory and neurodegenerative pathways. The identification of specific molecular pathways, multi-omics, and development of specific nutritional and therapeutic interventions require further research to understand the neurological disorders related to HFD better and how to control them with the help of the latter.
Acknowledgement
The authors express their sincere gratitude to Gokaraju Rangaraju College of Pharmacy, Bachupally Hyderabad affiliated to Osmania University for providing the necessary facilities, guidance and constant encouragement throughout the completion of this review work. Authors are also thankful to the teaching and non-teaching staff of the department of pharmacology for their valuable support and cooperation.The authors extend their appreciation to all researchers and scholars whose previous studies and publications have contributed significantly to the understanding of the work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable.
Author Contributions
Vivek Kumar Tiwari: Visualization, Conceptualization, Guidance and Supervision.
Merugu Samyuktha: Writing-Original Draft, Writing Review and editing and Data collection.
References
- Mozaffarian D, Hao T, Rimm EB, Willett WC, Hu FB. Changes in Diet and Lifestyle and long-term Weight Gain in Women and Men. The New England Journal of Medicine. 2011;364(25):2392-2404. doi:https://doi.org/10.1056/NEJMoa1014296
CrossRef - Sung-Chan P, Sung YW, Zhao X, Brownson RC. Family-based models for childhood-obesity intervention: a systematic review of randomized controlled trials. Obesity Reviews. 2012;14(4):265-278. doi:https://doi.org/10.1111/obr.12000
CrossRef - The GBD 2015 Obesity Collaborators. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. New England Journal of Medicine. 2017;377(1):13-27. doi:https://doi.org/10.1056/nejmoa1614362
CrossRef - Cho NH, Shaw JE, Karuranga S, et al. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Research and Clinical Practice. 2018;138(1):271-281. doi:https://doi.org/10.1016/j.diabres.2018.02.023
CrossRef - Tatlisumak T, Cucchiara B, Kuroda S, Kasner SE, Putaala J. Nontraumatic intracerebral haemorrhage in young adults. Nature Reviews Neurology. 2018;14(4):237-250. doi:https://doi.org/10.1038/nrneurol.2018.17
CrossRef - Bowie D. Ionotropic glutamate receptors made crystal clear. Trends in Neurosciences. 2014;37(12):687-688. doi:https://doi.org/10.1016/j.tins.2014.10.001
CrossRef - Jack CR, Bennett DA, Blennow K, et al. NIA-AA research framework: Toward a biological definition of alzheimer’s disease. Alzheimer’s & Dementia : the Journal of the Alzheimer’s Association. 2018;14(4):535-562. doi:https://doi.org/10.1016/j.jalz.2018.02.018
CrossRef - Kalia LV, Lang AE. Parkinson’s disease. The Lancet. 2015;386(9996):896-912. doi:https://doi.org/10.1016/s0140-6736(14)61393-3
CrossRef - Otte C, Gold SM, Penninx BW, et al. Major depressive disorder. Nature Reviews Disease Primers. 2016;2(1):1-20. doi:https://doi.org/10.1038/nrdp.2016.65
CrossRef - Palmer AM. The role of the blood–CNS barrier in CNS disorders and their treatment. Neurobiology of Disease. 2010;37(1):3-12. doi:https://doi.org/10.1016/j.nbd.2009.07.029
CrossRef - Song M, Bai Y, Song F. High-fat diet and neuroinflammation: the role of mitochondria. Pharmacological Research. Published online January 20, 2025:107615. doi:https://doi.org/10.1016/j.phrs.2025.107615
CrossRef - Sharma T, Ranawat P, Garg A, Rastogi P, Kaushal N. Short-chain fatty acids as a novel intervention for high-fat diet-induced metabolic syndrome. Molecular and Cellular Biochemistry. Published online December 21, 2024. doi:https://doi.org/10.1007/s11010-024-05185-9
CrossRef - Dutheil S, Ota KT, Wohleb ES, Rasmussen K, Duman RS. High-Fat Diet Induced Anxiety and Anhedonia: Impact on Brain Homeostasis and Inflammation. Neuropsychopharmacology. 2015;41(7):1874-1887. doi:https://doi.org/10.1038/npp.2015.357
CrossRef - Thaler JP, Yi CX, Schur EA, et al. Obesity is associated with hypothalamic injury in rodents and humans. Journal of Clinical Investigation. 2012;122(1):153-162. doi:https://doi.org/10.1172/jci59660
CrossRef - Hryhorczuk C, Florea M, Rodaros D, et al. Dampened Mesolimbic Dopamine Function and Signaling by Saturated but not Monounsaturated Dietary Lipids. Neuropsychopharmacology. 2016;41(3):811-821. doi:https://doi.org/10.1038/npp.2015.207
CrossRef - Cavalleri L, Merlo Pich E, Millan MJ, et al. Ketamine enhances structural plasticity in mouse mesencephalic and human iPSC-derived dopaminergic neurons via AMPAR-driven BDNF and mTOR signaling. Molecular Psychiatry. 2017;23(4):812-823. doi:https://doi.org/10.1038/mp.2017.241
CrossRef - Molteni R, Barnard RJ, Ying Z, Roberts CK, Gómez-Pinilla F. A high-fat, refined sugar diet reduces hippocampal brain-derived neurotrophic factor, neuronal plasticity, and learning. Neuroscience. 2002;112(4):803-814. doi:https://doi.org/10.1016/s0306-4522(02)00123-9
CrossRef - Querfurth HW, LaFerla FM. Alzheimer’s Disease. New England Journal of Medicine. 2010;362(4):329-344. doi:https://doi.org/10.1056/nejmra0909142
CrossRef - Voorend M, van der Ven AJAM, Mulder M, Lodder J, Steinbusch HWM, Bruggeman CA. Chlamydia pneumoniae infection enhances microglial activation in atherosclerotic mice. Neurobiology of Aging. 2010;31(10):1766-1773. doi:https://doi.org/10.1016/j.neurobiolaging.2008.09.022
CrossRef - Arnold SE, Arvanitakis Z, Macauley-Rambach SL, et al. Brain insulin resistance in type 2 diabetes and Alzheimer disease: concepts and conundrums. Nature Reviews Neurology. 2018;14(3):168-181. doi:https://doi.org/10.1038/nrneurol.2017.185
CrossRef - Conley SM, Cai X, Makkia R, Wu Y, Sparrow JR, Naash MI. Increased cone sensitivity to ABCA4 deficiency provides insight into macular vision loss in Stargardt’s dystrophy. Biochimica et Biophysica Acta (BBA) – Molecular Basis of Disease. 2012;1822(7):1169-1179. doi:https://doi.org/10.1016/j.bbadis.2011.10.007
CrossRef - Guillemot-Legris O, Muccioli GG. Obesity-Induced Neuroinflammation: Beyond the Hypothalamus. Trends in Neurosciences. 2017;40(4):237-253. doi:https://doi.org/10.1016/j.tins.2017.02.005
CrossRef - Poewe W, Seppi K, Tanner CM, et al. Parkinson Disease. Nature Reviews Disease Primers. 2017;3(3):17013. doi:https://doi.org/10.1038/nrdp.2017.13
CrossRef - Bousquet M, St-Amour I, Vandal M, Julien P, Cicchetti F, Calon F. High-fat diet exacerbates MPTP-induced dopaminergic degeneration in mice. Neurobiology of Disease. 2012;45(1):529-538. doi:https://doi.org/10.1016/j.nbd.2011.09.009
CrossRef - Houser MC, Tansey MG. The gut-brain axis: is intestinal inflammation a silent driver of Parkinson’s disease pathogenesis? npj Parkinson’s Disease. 2017;3(1). doi:https://doi.org/10.1038/s41531-016-0002-0
CrossRef - Cosín-Tomás M, Alvarez-López MJ, Sanchez-Roige S, Lalanza JF, Bayod S, Sanfeliu C,Pallàs M, Escorihuela RM, Kaliman. Epigenetic alterations in hippocampus of SAMP8 senescent mice and modulation by voluntary physical exercise. Frontiers in Aging Neuroscience. 2014;6. doi:https://doi.org/10.3389/fnagi.2014.00051
CrossRef - Sampson TR, Debelius JW, Thron T, et al. Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson’s Disease. Cell. 2016;167(6):1469-1480.e12. doi:https://doi.org/10.1016/j.cell.2016.11.018
CrossRef - Chen T, Tang Q, Yu B, et al. Investigation of the mechanism involved in high-fat diet-induced depressive behavior based on energy metabolism. Journal of Affective Disorders. 2026;394:120505. doi:https://doi.org/10.1016/j.jad.2025.120505
CrossRef - Sharma S, Fulton S. Diet-induced obesity promotes depressive-like behaviour that is associated with neural adaptations in brain reward circuitry. International Journal of Obesity. 2012;37(3):382-389. doi:https://doi.org/10.1038/ijo.2012.48
CrossRef - Noronha SSR, Lima PM, Campos GSV, et al. Association of high-fat diet with neuroinflammation, anxiety-like defensive behavioral responses, and altered thermoregulatory responses in male rats. Brain, Behavior, and Immunity. 2019;80:500-511. doi:https://doi.org/10.1016/j.bbi.2019.04.030
CrossRef - Villas Boas GR, Boerngen de Lacerda R, Paes MM, et al. Molecular aspects of depression: A review from neurobiology to treatment. European Journal of Pharmacology. 2019;851:99-121. doi:https://doi.org/10.1016/j.ejphar.2019.02.024
CrossRef - Abdoli N, Salari N, Darvishi N, et al. The global prevalence of major depressive disorder (MDD) among the elderly: A systematic review and meta-analysis. Neuroscience & Biobehavioral Reviews. 2021;132(3):1067-1073. doi:https://doi.org/10.1016/j.neubiorev.2021.10.041
CrossRef - de Queiroz Cavalcanti SA, de Almeida LA, Gasparotto J. Effects of a high saturated fatty acid diet on the intestinal microbiota modification and associated impacts on Parkinson’s disease development. Journal of Neuroimmunology. 2023;382:578171. doi:https://doi.org/10.1016/j.jneuroim.2023.578171
CrossRef - Nayak SS, Darji AD, Shah PK, Orozco-Arroyave JR. Automatic detection of Parkinson’s disease from speech signals using the Fourier–Bessel domain adaptive wavelet transform. Applied Acoustics. 2026;245:111217. doi:https://doi.org/10.1016/j.apacoust.2025.111217
CrossRef - Morris JK, Bomhoff GL, Stanford JA, Geiger PC. Neurodegeneration in an animal model of Parkinson’s disease is exacerbated by a high-fat diet. Am J Physiol Regul Integr Comp Physiol. 2010;299(4):R1082‑R1090. doi:10.1152/ajpregu.00251.2010
CrossRef - Mo X, Cheng R, Shen L, et al. Yeast β-glucan alleviates high-fat diet-induced Alzheimer’s disease-like pathologies in rats via the gut-brain axis. International Journal of Biological Macromolecules. 2024;278:134939. doi:https://doi.org/10.1016/j.ijbiomac.2024.134939
CrossRef - Liu E, Zhang Y, Wang JZ. Updates in Alzheimer’s disease: from basic research to diagnosis and therapies. Translational Neurodegeneration. 2024;13(1). doi:https://doi.org/10.1186/s40035-024-00432-x
CrossRef - Thal DR, Koen Poesen, Vandenberghe R, Meyer SD. Alzheimer’s disease neuropathology and its estimation with fluid and imaging biomarkers. Molecular Neurodegeneration. 2025;20(1). doi:https://doi.org/10.1186/s13024-025-00819-y
CrossRef
Abbreviations
CNS- Central Nervous System
AD– Alzheimer’s disease
HFD– High Fat Diet
OS Oxidative stress
Accepted on: 27-03-2026
Second Review by: Dr. Randa Salah Gomaa Mahmoud
Final Approval by: Dr. Eugene A. Silow









