Thermosensitive Hydrogels for Drug Delivery Systems: A Comprehensive Review
1Department of Pharmacology, Mahavir Institute of Pharmacy Nashik, India.
2Department of Pharmacognosy, Mahavir Institute of Pharmacy Nashik, India.
3Department of Pharmaceutical Chemistry, Mahavir Institute of Pharmacy Nashik, India.
Corresponding Author Email: prachikage@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3488
ABSTRACT:Hydrogels sensitive to temperature changes have begun to show promise as reliable materials for the controlled delivery of drugs, as they can systematically trap and release active agents based on changes in temperature. This review paper seeks to present the different facets of research undertaken in this field, including: the different types of polymers used in the creation of hydrogels, the various methods of polymerization, and the different methods of gelation, fabrication and characterization, strategies for the loading of drugs and the kinetics of drug release, the various fields of biomedical engineering where these materials can be applied (including the engineering of tissues, the transdermal delivery of drugs, the delivery of drugs to the eyes, and the treatment of tumour), work done in these fields in vitro and in vivo, the engineering of documentation, the safety and legal regulations involved in the engineering of these materials, as well as the engineering of the materials in the future. Each of the engineering challenges associated with these materials (e.g. the non-uniform size of the eluting agents, etc.) have associated engineering solutions, including systems with composites, hybrid hydrogels with endogenous carriers, and systems with multiple responsive domains.
KEYWORDS:Chitosan; Controlled Release; Drug Delivery; In Situ Gelling; Injectable Hydrogel; PNIPAM; Poloxamer; Stimuli-Responsive; Thermo-Responsive; Thermosensitive Hydrogel
Introduction
In the past two decades, research in stimuli-responsive materials has made remarkable advancements. These advancements stem from the materials capability to undergo reversible physical and/or chemical alterations. These changes, which can be triggered by stimuli such as temperature, pH, ionic strength, magnetic fields, and light, allow the material to transition from a liquid to gel. For example, in the past five years, the majority of investigated and developed thermosensitive hydrogels have resulted in biomedicine and pharmacy applications, more specifically in the area of drug delivery.1 This is attributed to the ability of these hydrogels to undergo a “sol-gel” transition at a given temperature within a specific physiological range. The drug delivery process involves the formulation being physically stored as a semi-solid; and after administration, the semi-solid serves to contain and control the release of the drug. Cross-linked polymer networks enable hydrogels to retain a large amount of water while keeping a three-dimensional structure. When temperature-responsive polymers are introduced, the networks undergo a significant phase transition, crossing the hydrophilic-hydrophobic balance of the stored water.2 The polymer composition, molecular weight, and type of co-monomers present in the hydrophilic and hydrophobic polymers determine the thermoresponsive behaviour.
This underscores the possibility of creating highly customized and targeted infused drug carriers for particular therapeutic goals.
Temperature-triggered drug delivery systems, also known as hot gel drug delivery systems, provide plenty of benefits. The first being, they enable more para-pharmaceutical systems of drug delivery to be used, particularly injected ones. Hot gel drug delivery systems remain liquid and can be injected effortlessly. Once injected, they turn to gel (around 37°C, “human body temperature”), and remain at the target site.3 Furthermore, drug delivery systems provide precise spatiotemporal control of active substance release, which sustains therapeutic levels and minimizes systemic side effects. The biocompatibility of the poly(N-isopropylacrylamide) (PNIPAM), and simple polysaccharides based thermogels, chitosan thermogels, and block copolymers thermogels, can be attributed to the gentle sol-gel transition and the protection of labile biomolecules (enzymes, proteins, DNA and RNA). The literature review of the 1950s refers to the first known responsive systems based on thermoresponsive polymers. Poly(N-isopropylacrylamide) (PNIPAM) and its lower critical solution temperature (LCST) phenomenon near body temperature prompted the exploration of numerous synthetic and natural polymers based thermogels.
Thermosensitive hydrogels have found applications in diverse fields, including, but not limited to, cancer therapy, tissue engineering, regenerative medicine, and ocular delivery.4
Such systems can be of great value in localized and sustained drug delivery systems, especially those considering patient adherence. Thermogels create a depot at the site of target delivery which facilitates sustained release of the drug, increasing drug bioavailability, and decreasing the number of times the drug needs to be administered.5 Furthermore, the combination of thermoresponsive polymers with other systems (e.g., other stimuli-responsive systems or with nanostructured systems) holds promise for creating multi-responsive smart drug carriers, which will advance precision medicine. The purpose of this review is to integrate the applications of thermosensitive hydrogels in drug delivery, their mechanisms, constituent materials, synthesis, characterization, kinetics of release, and other biomedical applications. The review identifies and discusses the cutting-edge gaps, challenges, and trends, especially those including thermogels, hybrid and multi-responsive systems, and 3D printing for clinical applications.
Thermoresponsive Mechanism
Thermosensitive hydrogels can undergo phase transitions, either from polymer gel to sol, or vice versa, dependent on temperature due to unique mechanisms involving a repulsive sol, swollen hydrated polymer net-work, and attractive gel, compact heterogeneous polymer network.6 The described temperature responsive gel systems are based on two macromolecular level thermodynamic principles, which are Lower Critical Solution Temperature (LCST) and Upper Critical Solution Temperature (UCST). In systems designed for targeted drug delivery (which incorporate both temporal and spatial control), the gelation process and, more specifically, the molecular mechanisms that drive the phenomenon must be understood for sol–gel systems.
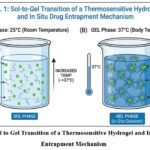 |
Figure 1: Sol to Gel Transition of a Thermosensitive Hydrogel and In Situ Drug Entrapment Mechanism.
|
LCST vs. UCST Phenomena
Certain polymers have lower critical solution temperature (LCST) characteristics. They are initially soluble in water; but once a critical temperature is reached, they become insoluble in water. An increase in temperature means the water can no longer sustain hydrogen-bonded interactions with the polymer.7 Consequently, the water is released, and extensive hydrogen bonding interactions collapse. The polymer’s hydrophobic interactions become dominant, resulting in collapse, phase separation, and gelation. Because of this, the LCST value is of utmost importance, and is defined as the temperature in which the polymer transitions from hydrophilic to hydrophobic. Every temperature, or range of temperature, is a physical property of matter. This is illustrated in the temperature of human skin and the temperature of skin-textured fittings. The polymer PNIPAM is in the human skin temperature range (32 °C) compliant with the human skin. PNIPAM and its copolymers thus are best suited for in situ or injectable gelling systems for drug delivery systems.
Other polymers of this type include poly(N-vinylcaprolactam) (PVCL), Pluronic F127 (Poloxamer 407), and PEG block copolymer. While some polymers exhibit UCST behaviour, they are usually insoluble at lower temperatures but become soluble upon heating. In this situation, strong hydrogen bonding or ionic crosslinking between the polymer associative chains and the polymer phase separations occur at lower temperatures.8 When temperature is increased, the hydrogen bonds or ionic crosslinks break resulting in solubilization. This behaviour is documented in poly(acrylamide-co-acrylonitrile) and some zwitterionic copolymers. In biomedicine, using UCST systems is less common, but they are important for designing cold-triggered dual responsive systems, in combination with LCST polymers, for the design of cold-triggered dual responsive systems.
Sol–Gel Transition and Molecular Interactions
Thermosensitive hydrogels experience sol-gel transitions due to the unique properties involving the polymer chain hydrophilic and hydrophobic interactions. Below the “LCST, the OH, COOH, and CONH hydrophilic monomers form competitive hydrogen bonds polymer with surrounding water molecules and keep the polymer solid in a hydrated, flexible, and coiled spring sol state9. As the temperature surpasses the LCST, the polymer hydrophilic –CH3 or –CH2– groups, bound to water, will gain entropy, dehydrating the system. This will lead to a coil-to-globule transition where the polymer chains rotate and collapse into the system, and the strings get ordered to compact into a more defined structure. When the hydrophobic segments change phases, they will attempt to cluster to water. This results in micelles encapsulating to form additional associative bonds and a three-dimensional cross-linked (3D) network structure behaving as a gel.
In biomedical contexts, the reversible nature of this phenomenon is positive, as when the in-situ encapsulated system or gel is formed, the system is allowed to undergo dissociation or dissolution, rather than having to utilize surgical removal techniques.10 From the standpoint of thermodynamics, the behaviour of heat (enthalpy) in polymer-water interactions (Δ H) with respect to the dispersal of mixtures (entropy) (Δ S) is what determines the LCST. As per the Flory-Huggins theory, the mixing free energy (Δ G = Δ H – TΔ S) increases to a positive value; and thus, above the LCST the system becomes discontinuous and separates into phases, as the dominant force is hydrophobic interactions. Thus, the tuning of the transition temperature and of gelation behaviour is of the hydrophilic/hydrophobic ratio of the constituent polymers.
Factors Influencing Gelation
Several intrinsic and extrinsic factors affect thermoresponsive properties.
Type of polymer and molecular weight:- The ratio of hydrophilic and hydrophobic determines LCST. Incorporation of more hydrophilic comonomers (acrylamide, PEG) increases the LCST. In contrast, incorporation of more hydrophobic comonomers (butyl methacrylate) decreases the LCST.
Concentration of polymer: – At higher concentrations, polymers entrap micelles, which reduces the gelation temperature and enhances the mechanical properties.11
Ionic strength and solvent: – LCST is influenced by the hydrogen bonds and hydrophobic interactions of the system which are modulated by the salts, pH, and other additives.
Crosslinking density: – Physically crosslinked systems are less stable and degrade faster than chemically crosslinked systems.
Use of additives and drugs: -Incorporation of the charged molecules modifies the gelation kinetics through hydrophobic or ionic interactions and bonding with the polymer chains.
Materials Used in Thermosensitive Hydrogels
The design and performance of thermosensitive hydrogels are primarily influenced by the various types of polymers utilized. These polymers are critical in determining the parameters related to the transition temperature, gelation behaviour, biocompatibility, degradation rate, and drug release profile. Thermosensitive polymers, which are sourced from different origins and compositions, are categorically divided into three main classes: synthetic polymers, natural polymers, and hybrid and composite systems.12 Each class has distinct biophysical properties, applicable biomedical functions, and other characteristics that can be tailored for a specific drug delivery system.
Synthetic Polymers
Just like in the previous example, synthetic polymers are also used in thermosensitive hydrogel systems, which can be engineered with controllable mechanical, as well as thermal, properties. Such controllable attributes allow the composition of the systems to be adjusted to have an LCST value that is close to physiological temperature, thus facilitating the injectable and in situ gelling capabilities of these hydrogel systems.13
Poly(N-isopropylacrylamide) (PNIPAM) and Derivatives
The hydrogel used in most studies within the research community is Poly(N-isopropylacrylamide) (PNIPAM). Because the polymer is insoluble around 32 °C, which is near the temperature of the human body, it is said to have a lower critical solution temperature (LCST) of 32 °C.14 This means that at temperatures higher than 32 °C, the polymer is soluble and the chains of the polymer are in a hydrated state; however, at lower temperatures, the chains collapse and the solution turns to a gel. When producing PNIPAM-based hydrogels, it is possible to control the release of small molecules, proteins, and peptides. The pure polymer doesn’t have enough biodegradability; therefore, in vivo clearance will be problematic due to the polymer’s nonbiodegradable backbone. This problem can be minimized by copolymerization or grafting PNIPAM with poly (ethylene glycol) (PEG), acrylic acid (AAc), N-vinylpyrrolidone (NVP), and chitosan which are all considered to be hydrophilic and biodegradable.
These modifications enhance biocompatibility, alter LCST, and allow the copolymer to be degraded by hydrolysis or enzymes.15 For instance, PNIPAM-co-PEG hydrogels are more flexible with an extended drug release profile, whereas PNIPAM-co-AAc hydrogels have added pH responsiveness in addition to thermoresponsiveness. Derivatives of PNIPAM have also been functionalized with nanoparticles or biomolecules to achieve dual responsiveness and targeted delivery. These systems hold great potential in localized cancer therapy, ocular delivery systems, and tissue scaffolding.
Poloxamers (Pluronic F127, F68)
Poloxamers are also called Pluronics. These are triblock copolymers of poly (ethylene oxide) poly (propylene oxide) poly (ethylene oxide) (PEO–PPO–PEO). Among different Poloxamers, Pluronic F127 (Poloxamer 407) and Pluronic F68 (Poloxamer 188) are most popular because of their FDA approval and outstanding biocompatibility. Poloxamer F127 exhibits a unique property of undergoing a temperature-dependent gel-sol transition, which can be fixed in the range of 20-35 °C (this can be adjusted by varying the polymer concentration). At lower temperatures, the polymer is in a micellar sol state and upon heating, the micelles, which are in the sol state, undergo packing and form a 3D gel.16 These gels are biocompatible, can be made to be transparent, and are also amenable to being made injectable. Thus, they can be used to prepare formulations for the eye, nose, and for injections. Unquestionably, the key characteristics of Poloxamers are the biocompatibility, low toxicity and temperature-reversible gel strength. They also have inadequate mechanical strength, rapid biodegradability and body fluid-activated dissolution which could lead to dose dumping of the incorporated active pharmaceutical ingredient (API). However, these characteristics can be desirable when Poloxamers are blended with other biodegradable polymers such as carbopol, chitosan, and hyaluronic acid. Poloxamer formulations have been utilized in the delivery of antibiotics, analgesics and copolymers for the local treatment of cancer by chemotherapy.
PEG–PLGA and PEG–PCL Copolymers
Thermoresponsive hydrogels are made from block copolymers of poly (ethylene glycol) (PEG) with poly (lactic co glycolic acid) (PLGA) or poly (ϵ-caprolactone) (PCL).17 Amphiphilic copolymers micellization and gelation properties can form micelles and convert to gel with temperature differences between hydrophobic polymers and PLGA or PCL. PEG-PLGA has been utilized for long-acting injectable depots, as it has good degradation, biocompatibility, and thermoresponsive properties. Moreover, the usage of PEG-PLGA copolymers is increasing due to the US FDA greenlighting PEG-PLGA copolymers. The ability to form in situ gels at body temperature also increases system attractiveness. While thermoresponsive characteristics may be comparable to PEG-PCL copolymers, the hydrogels made from PEG-PCL copolymers have a much lower degradation rate, a property that is desirable for some applications involving prolonged drug release .18 There have been reported systems for the sustained release of pegylated proteins and peptides, leuprolide, paclitaxel, insulin, and for gene therapy and post-operative drug release systems. In addition, the combination of PEG-PLGA with nanoparticles or liposomes in hybrid formulations has been noted to enhance the efficacy of PEG-PLGA systems as a dual drug delivery system.
Natural Polymers
Natural biopolymers obtained from biosystems greatly outperform their synthetic counterparts in terms of biocompatibility, biofunctionality, and biodegradability. Because of their close structural resemblance to the ECM, they are used in several areas of biomedicine, especially in wound healing and tissue engineering.19 However, several natural polymers are still thermosensitive-unresponsive and are, therefore, subjected to compositional blending and chemical modifications with thermoresponsive materials.
Chitosan/β-Glycerophosphate Systems
Chitosan is a cationic polysaccharide known for its biodegradability, mucoadhesiveness, and antibacterial properties. For example, with β-glycerophosphate (β-GP), the combination creates the injectable drug delivery system known as thermosensitive chitosan sol-gel. The chitosan/β-GP solution remains fluidity at low temperatures due to the strong electrostatic repulsion between protonated amino groups of chitosan .17 As the temperature approaches physiological levels (~ 37 °C), due to the characteristics of β-GP as a weak base, chitosan will partially be neutralized. This triggers a number of phenomena: the formation of inter-chain hydrophobic bond associations, a decrease in solubility, and the formation of inter-chain hydrogen bonds. This leads to in situ gel formation in a matter of minutes. Proteins, vaccines, and anticancer agents, as well as tissue engineering scaffolds, are advanced for delivery using chitosan/β-GP hydrogels. In certain uses, optimum conditions are slow gelation and low mechanical strength, all of which can be corrected with the addition of gelatin, alginate, or nanoparticles.
Gelatin, Collagen, and Alginate
Cross-linked microstructures exhibit appropriate cellular adhesion and biocompatibility within implants for connective tissues and membranes, with defined structures and amino acid polymers as constituent parts. Biopolymers gelatin and collagen, gelatin’s varying compositions exhibit nonthermosensitivity and tunnel-thermosensitivity and dove gelatin PNIPAM hybrids plane tissues and with pos controlled release retain and grow target cell populations psychos.20 Formed tissues as labelled with reference and alginate inter-compatible materials. Formed tissues display interpenetrating able and intergels able to incorporate tissues strengthened and drug concealed; and intergels able to permit tissue transfer and healing as alginate–PNIPAM hybrids and alginate–Pluronic blends.
Cellulose and Its Derivatives
Methylcellulose (MC), hydroxypropyl methylcellulose (HPMC), and hydroxypropyl cellulose (HPC), all derived from cellulose, show one of a kind thermoresponsive properties .21 Such unusual properties are a result of the polyether’s dehydrated hydroxyl groups. For instance, MC exhibits a reversible sol-gel transition at 50 °C, and through copolymerization, the addition of salts, and hydrophilic polymers, the transition can be modified to body temperature. These cellulose-based hydrogels are translucent, non-pyrogenic, and mechanically stable. Thus, been used for controlled release formulations of timolol maleate and dexamethasone in ophthalmic, nasal, and transdermal delivery systems. Additionally, cellulose derivatives serve as thermally synthetic gel additives, increasing the viscosity and gel strength.
Hybrid and Composite Systems
Thermosensitive hydrogels that have been hybridized with composites exhibit the advantages of integration with both the natural and synthetic polymers through the use of enhanced mechanical capability, regulated degradation, and multifunctional responsiveness which is described by dual responsiveness.22 These materials represent the next generation of “smart” materials with dual responsive characteristics. Blends of polymers are systems that are created through the fusion of two or more polymers together and the response in the systems can be characterized by the lower critical solution temperature (LCST), the viscosity or gelation time, and the modulus of the gel. The blends of PNIPAM and chitosan, Pluronic and alginate, and PEG and PCL with hyaluronic acid are considered to be excellent examples.23 There is an enhancement of biocompatibility, mucoadhesiveness, and retention of the drug to be released within the target tissue through the use of such hybridization. Customized hydrogels can be created to have specific attributes through the copolymerization of the individual polymers. The copolymerization of PNIPAM with acrylic acid has been demonstrated to have the ability to provide dual temperature and pH responsiveness which is critical for the regulated release of a drug .24 The structural integrity of thermo responsive gel systems can be enhanced with the incorporation of nanofillers such as silica nanoparticles, graphene oxides, nanoclays, and hydroxyapatite. Along with molecular reinforcement, the addition of these nanofillers enhances the elasticity, mechanical strength, and the responsiveness of thermogels.
The addition of thermogel drug carriers’ silica nanoparticles, graphene oxide, nanoclays, and hydroxyapatite makes nanocomposite thermogels more structurally sound. These fillers reinforce the molecular structure of thermogels and improve the elasticity, tensile strength, and thermoresponsiveness of the thermogel.25 For example, chitosan-silica nanocomposite thermogels increase the compressive strength and offer controlled degradation. Thus, the regenerative medicine potential for bone and cartilage is increased. The photothermal activity of PNIPAM-graphene oxide hybrids is increased, allowing their application in lightactivated cancer therapy. Recent research has advanced the design of multifunctional, dual-thermo responsive hydrogels that also encapsulate others triggers like pH, light, redox, and magnetism, in addition to thermoresponsiveness. These tailored systems target controlled drug release kinetics for programmable therapy placements in specific bodily locations. For instance, PNIPAM-coacrylic acid hydrogels are the best for colon-targeted delivery due to their dual pH and temperature responsiveness .26 Additionally, the Pluronic-chitosan-Fe₃O₄ systems have hybrid thermally and magnetically responsive features, that enable magnetically and thermally controlled enhanced release. Such multifunctional systems lay the groundwork for the potential development of personalized and precision drug delivery systems.
Table 1: Representative Thermosensitive Hydrogels, LCST/UCST Values, and Biomedical Applications
|
Polymer/System |
Transition Type
(LCST/UCST) |
Transition Temperature (°C) |
Key Features |
Biomedical
Applications |
|
PNIPAM |
LCST |
~32 |
Sharp
reversible phase transition |
Controlled drug release, tissue scaffolds |
| PNIPAM–PEG
Copolymer |
LCST | 33–37 | Biodegradable, tuneable LCST | Injectable depots, cancer therapy |
| Poloxamer F127 | LCST | 20–30 | FDA-approved,
reversible gelation |
Ocular and nasal delivery |
| PEG–PLGA | LCST | 25–37 | Biodegradable, long-acting | Injectable depot formulations |
| Chitosan/β-GP | LCST | 35–37 | Biocompatible, mucoadhesive | Protein and
vaccine delivery |
| Gelatin–PNIPAM blend | LCST | 30–35 | Cell adhesive,
elastic |
Tissue regeneration, wound healing |
| Alginate–
Poloxamer |
LCST | 30–36 | Enhanced mechanical stability | Injectable and transdermal systems |
| Methylcellulose | LCST | 45–50 | Reversible gelation | Ocular and
transdermal delivery |
| PNIPAM–graphene oxide | LCST | 33–36 | Reinforced structure, photothermal
effect |
Cancer therapy |
| Dual pH–Temp PNIPAM–AAc Hydrogel | LCST | 35–38 | Dual responsiveness | Colon-targeted delivery |
Preparation and Fabrication Techniques
The procedures required for the fabrication of hydrogels and their preparation determine how the hydrogels will behave and what properties they will possess. The gelation behaviour, the mechanical strength, and the drug release of hydrogels are affected by the preparation procedures. A given synthesis method shapes the hydrogel and, in turn, determines the hydrogel’s biocompatibility and it’s in situ drug delivery capability27. Thermosensitive hydrogels can be made using either one method of crosslinking or the other method . Each of these two methods has its own advantages. Ultimately, it comes down to the required functionality, the rate at which the hydrogel needs to degrade, and the desired stability. The design potential has been expanded with the inclusion of various new advanced fabrication techniques, including solvent casting, freeze-thaw cycles, emulsification, enzymatic catalysis, and click-chemistry based reactions.
Physical vs. Chemical Crosslinking
There are crosslink hierarchy non-covalent bonds such as: hydrogen bonds, ionic bonds, Vander Waals bonds, and hydrophobic bonds. Crosslinking is a particularly effective strategy for the biomedical sector as it works with mild conditions and does not deal with toxic crosslinking agents. Physical gelation in thermosensitive hydrogels can be a straightforward process, with a temperature induced hydrophobic association of the polymer chains occurring after the temperature exceeds the lower critical solution temperature (LCST) .28 This is exemplified in Pluronic F127 solutions, which form micellar networks, and systems based on PNIPAM, in which polymer interactions become more hydrophobic as temperature exceeds the LCST leading to the dominance of hydrophobic interactions, thus, shifting the balance of polymer-water interactions. The ability to self-heal and reform is a critical property in the context of in-situ gelling injectables and stimuli-responsive drug depots. Mechanically, hydrogels have brittle anchor points and systems with physically crosslinked structures tend to suffer from a weak bond and poor long-term maintaining. This is why they are not ideal for high shear, load bearing applications.
In contrast to physical crosslinking, which uses non-covalent bonds, chemical crosslinking establishes more permanent linkages between polymer chains, resulting in stronger and more stable networks. These reactions can be achieved via thermal, photo, and enzymatic methods.29 Users may view the gels chemically crosslinked portion, which may be perceived as a coating, as structural improvements, additional protective environmental toughness, and a slowed biodegradability to the thermoregulating gel. To avoid biotoxicity, the cytocompatibility of reactive monomers, (co)catalysts or initiators, in this case, would need to be established. Osmotic pressure of the responsive hydrogels, regardless of the crosslinking methods used, will cause fixed polymer chains to relax in a hypertonic environment. Hybrid systems that employ both physical and chemical crosslinking provide a more improved balance of injectability and the mechanical strength of the system. A good example is the chitosan-β-glycerophosphate system which can be gelled and subsequently chemically crosslinked with genipin or glutaraldehyde.
Fabrication Methods
The methods for manufacturing thermosensitive hydrogels are diverse and depend on the specific polymer system and its morphology, as well as its end-use. What follows are literature examples and practical observations for the preparation of drug-loaded thermogels. A relatively straightforward method of hydrogel fabrication is the solvent casting method. In this method, one or more polymers are dissolved into a suitable solvent, which is typically either water or an aqueous buffer. The drug is incorporated and the mixture is stirred under controlled conditions.30 The resulting solution is divided into small sections which are placed into containers (casts) and then gelled either through temperature modulation or evaporative drying. For thermosensitive hydrogels, the gel is produced by heating the solution over the LCST and then cooling the gel to a temperature that affords a stable gel state. This method is popular because of its ease of preparation, uniform dispersion of the drug throughout the hydrogel, and ease of scale. This method is appropriate for creating thin Fim hydrogels. When optimizing the rate of solvent evaporation and the composition of the solvent, one should consider the prevention of pore collapse or phase separation. During the drying process, care should be taken to ensure that the solvent is not over-dried.
Numerous studies have utilized the freeze-thawing approach in the construction of physically crosslinked PVA and poly(thermosensitive) blends. For example, in their study, describe the procedure in which polymer solutions are subjected to several cycles of freezing and thawing.31 During the freezing phase, because ice crystals form, polymer chains that are located in the central regions get physically excluded. Subsequently, in the regions surrounding the freezing crystals, polymer chains become crosslinked and crystallized. During the thawing phase, the crystals that have formed melt to liquid and ascend a crosslinked polymer network. The number of cycles, polymer concentration and rate of freezing determine the porosity and strength of the gel. The porous structure’s stability, along with the material’s thermal responsiveness (enhanced by Pluronic PP14 or PNIPAM), remains intact. Applicable materials include thermoresponsive and biocompatible thermogels. Emulsion polymerization has the potential to provide a variety of options in the creation of polymer thermogels with advanced nano and micro structures. In the first step of the fabrication of such structures, monomers and pre-polymers are emulsified within a continuous phase, which, in the simplest case, could be water or oil, to form a microdroplet structure, which then serves as a micro-reactor.
Next, the authors incorporate cross-linking step, followed by further polymerization to create a polymer gel. This step may also be thermally or redox initiated.32 Additionally, the size, porosity, and surface properties may be design throughout the polymerization process, allowing those properties to be tailored to form either gel or hybrid thermoresponsive nanogels. Emulsion polymerization of PNIPAM, for example, creates nanogels that form injectable nanocomposite gels from the aqueous phase, with enhanced drug encapsulation and sustained release kinetics. To some extent, casein micelles are very similar to bioengineered braided protein scaffolds. These protein scaffolds can be custom-designed to control their mechanical properties and guide cellular behaviour, as well as their porosity. These micelles are also bioengineered to possess braided structures and can be tessellated, similar to scaffolds. The tessellated structures found in casein micelles allows for lag-phase stimulation of cellular behaviour. The bioengineered protein micelle scaffolds also demonstrate casein micelle’s ability to be bioengineered into structures that exhibit tunability of mechanical properties and porosity control, and guide cells behaviour. Additionally, casein micelles serve as an excellent model for micelle scaffolds in tissue engineering and cellular behaviour stimulation.
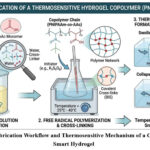 |
Figure 2: Fabrication Workflow and Thermosensitive Mechanism of a Cross-linked Smart Hydrogel.
|
There is an expanding ability to formulate covalently crosslinked thermosensitive hydrogels using click chemistry. Specific reactions such as azide-alkyne cycloaddition, thiol–ene, and Michael addition show high specificity, go to completion and high yield, and can occur in an all-aqueous solution. These reactions can take place at ambient conditions; such conditions relate to the thermoresponsive gelation of the hydrogels.33 PEG-based block copolymers, as an example of click chemistry approaches, can be made to have azide and alkyne pendant groups, and copolymers can interpenetrate and network with crosslinked copolymers at temperatures around and just below body temperature. Fine tuning of density and a variety of molecular functional groups in the chemistry allows for the construction of therapeutic gels for customized systems for multi-factor stimulus-responsive drug delivery.
Characterization Techniques
Characterizing thermosensitive hydrogels and their physico-chemical, structural and mechanical properties helps in understanding their drug loading, gelation and biological effects as bioactive materials.34 Examining materials for features such as temperature-triggered responsiveness, crosslink density and degradation requires methods that are rheological, thermodynamical, morphological, spectrometrical and mechanical, among others. The next section will explore the various methods used to study thermosensitive hydrogels, with drug delivery as a case study.
Rheology and Viscosity Analysis
To understand the viscoelastic properties of thermosensitive hydrogels and clarify the gelation and sol transition behaviour, rheological studies need to be conducted. The temperature-controlled plate rheometer computes the storage modulus (G′), loss modulus (G″), and complex viscosity (η)* as well as other variables, defining the rheological behaviour of the hydrogel as a function of temperature and shear rate.
When the system is flooded with temperature and remains below the lower critical solution temperature (LCST), it behaves as a viscous liquid with the modulus G″>G′ and therefore dominant liquid-like behaviour.35
Scholars note that hydrophobic interaction predominates as one increase temperature to and beyond the (LCST) with gelation occurring and G′>G″.
A temperature sweep test is performed to determine the gelation temperature (Tgel), a crucial parameter for injectable formulations. It is important for the hydrogel to have the ability to shift between liquid and gelatinous states at body temperature (≈37 °C). This test is needed to meet the required rheological profile for the evaluation of injectability. A hydrogel is considered injectable when it demonstrates shear-thinning properties before administration and can undergo an immediate and rapid transition to a viscous gel post administration.36 Moreover, the time-sweep test evaluates the kinetics of gelation while the frequency-sweep test examines the hydrogels resistance to oscillatory deformation and its overall mechanical stability and structural integrity. Rheological evaluation is the most conclusive method for the assessment of thermosensitive behaviour and sol–gel rheological transitions in hydrogels.
Thermal Analysis (DSC and TGA)
It is vital to analyze and assess thermosensitive hydrogels with respect to their transition temperatures, crystallinity, and thermal stability using thermal techniques. The most prominent techniques used in the field are Differential Scanning Calorimetry (DSC) and Thermogravimetric Analysis (TGA).
Differential Scanning Calorimetry (DSC)
Differential Scanning Calorimetry (DSC) involves measuring the heat flow connected with polymers as the temperature changes.37 Hydrogels, like PNIPAM and Pluronic F127, exhibit below the lower critical solution temperature an endothermic peak, corresponding with the coil to globule transition and gelation temperature. Transition enthalpy (ΔH) is a powerful indicator for the extent of hydration and hydrophobic interaction. This is copolymer composition, thermoresponsive behavior of the copolymer additives, thermoresponsive behavior of incorporated drugs, and DSC results.
Thermogravimetric Analysis (TGA)
To analyze thermal degradation profiles, TGA examines degree of mass loss in relation to changes in temperature. It captures the temperature at which degradation starts, and the mass of the sample at the end which represents the thermal and oxidative stability of the hydrogel network. If degradation happens in several steps, it shows that the polymer backbones or some of the crosslinkers are breaking down in a sequence. DSC and TGA together provide insight into the hydrogels stability for both processing and storage conditions, thus making the hydrogel suitable for biomedical applications.
Morphological Studies (SEM, TEM, AFM)
The structure and microstructure of thermosensitive hydrogels control their water absorption capacity and affect the porosity and the diffusion-controlled release of incorporated drugs. The major techniques employed to evaluate the structure of hydrogels are Scanning Electron Microscopy (SEM), Transmission Electron Microscopy (TEM), and Atomic Force Microscopy (AFM).
Surface patterns as well as internal pore structures post-lyophilization are analyzed using an SEM. The porous structures that are interconnected help with the nutrient transfer as well as the controlled diffusion of the therapeutics .38 The pore size of the hydrogels is adjustable by varying the polymer concentration, freeze–thaw cycle, and crosslinking density.
In the case of nanostructured domains, such as micelles, nanoparticles, and hybrid fillers, during incorporation in a hydrogel matrix, higher resolution imaging is possible with a Transmission Electron Microscope (TEM) than with a Scanning Electron Microscope.
Nanoscope (AFM) enables analysis of shapes and mechanical properties at the nano level. Stiffness, surface roughness, and phase distribution are the attributes analyzed.
All of the analyses of morphology provide a comparison of the microstructure to the macroscale characteristics like swelling, mechanical strength, and release kinetics.
Mechanical and Injectability Testing
The performance, handleability, and durability of thermo-responsive hydrogels, depend upon certain mechanical attributes. Rigidity and other deformability attributes of a hydrogel are assessed using tensile, compression, and oscillatory shear tests.39 Injectable and implantable hydrogels need to demonstrate a certain level of resilience to physiological stress; however, they need to be soft enough to avoid tissue incompatibility. To assess the performance of hydrogels, the extrusion force and the gel formed after the heating process are evaluated. Optimal systems exhibit low viscosity (making them easy to administer) and rapidly transition to a gel at physiological temperatures to minimize the possibility of drug loss. The gels processability and stability in-situ are best described by the combination of its rheological and mechanical properties.
Swelling, Degradation, and Stability Analysis
The ratio and degradation rate are key to biocompatibility and the release of the drugs. In swelling tests, dried hydrogel samples are equilibrated with buffer solutions at different temperatures, and the weight changes are monitored until equilibrium is achieved and is noted.40 In the case of LCST-type gels, swelling is absent above the transition temperature due to a hydrophobic collapse. To ensure there is sustained diffusion of the encapsulated drugs, controlled swelling is necessary. In degradation tests, the breakdown of the polymer chains is performed under conditions that simulate the human body (pH 7.4, 37 °C). The loss of weight and reduction in molecular weight are assessed and scored either gravimetrically or by gel permeation chromatography (GPC). Also, the duration of the test for enzymatic degradation of natural polymers like chitosan or gelatin is used to assess bioresorbability. The test performed using varying temperature, humidity, and pH conditions establish the reliability and the long-term performance.41 The combined analysis of swelling, degradation, and stability demonstrates that thermosensitive hydrogels offer consistent quality from fabrication to clinical application.
Drug Loading and Release Mechanisms
To be an effective drug delivery platform, a thermosensitive hydrogel must be evaluated for its drug loading capacity, drug release kinetics, and controlled release profile. Polymer architecture, crosslinking type, hydrogel, and drug physical-chemical properties all define the mechanisms of drug incorporation and release. For thermoresponsive hydrogels, drug encapsulation is mainly done by physical means, while chemical release occurs via diffusion, hydrogel degradation, swelling, or a combination of these methods. To be able to prevent burst release, and to achieve full therapeutic efficacy by controlled and precise sustained release to the target site, an understanding of the release mechanisms is required.
Physical Entrapment vs. Chemical Conjugation
Physical Entrapment
The absence of adverse chemical reactions coupled with how uncomplicated of a process it is makes physical entrapment the most popular technique for drug loading into thermosensitive hydrogels. To obtain a homogeneous mixture of the drug with the hydrogel network, the drug is added to the polymer solution, and the temperature is kept under the gelation point. The drug is then entrapped within the hydrogel network by raising the temperature above the lower critical solution temperature (LCST), and the sol–gel transitions are achieved.42 This process is mainly governed by the thermodynamic interactions of a solute with a solvent, which are described by the various forms of hydrophobic and hydrogen-bonding. While hydrophilic drugs are entrapped in the aqueous pores of the hydrogel, hydrophobic drugs are retained in the hydrophobic domains, which are accessible through the micelle cores and hydrophobic domains of the Pluronic and PNIPAM systems. Because no toxic reagents are involved, this approach is especially suitable for thermolabile biomolecules (peptides, proteins, and nucleic acids). The major drawback of this approach is burst release, due to rapid diffusion of surface-located drugs.
Chemical Conjugation
The adsorption of polymer interpenetrating networks and chemical conjugation involves the formation of a covalent bond between the polymer and the drug. Polymers in the polymer backbone involves integration of Poly (ethylene glycol) and Poly (D,L-lactide co glycolic) acid and PSB (sulfobetaine methacrylate) and covalent-imine thioester and disulfide bonds and by hydrolysis of the thioester or disulfide bonds or by enzymatic cleavage to release the drug.43 This approach to polymer covalent close retention and release of systemic antimitotic antagonists. PCL-PEG-PCL drug conjugated hydrogels framed retention of systemic toxicity. The selection of polymer conjugated drug hybrids and the employment of physical methods in polymer processing are interwoven.
Mechanisms of Drug Release
Drug release from thermosensitive hydrogels can proceed through multiple concurrent pathways, depending on the polymer matrix and environmental conditions. The three predominant mechanisms are diffusion-controlled, degradation-controlled, and swelling-controlled release.
Diffusion-Controlled Release
The primary mechanism of release diffusion involves the non-degradable hydrogels. The hydrogels contain releasable drugs that comprise hydrophilic molecules that diffuse through the water-filled pores of the polymeric networks. The crosslinking density of the polymers influences the polymeric mesh size, which, along with the drug molecule size and the size of polymer chains, controls the rate of polymeric diffusion.44 Diffusion occurs more rapidly with smaller molecules in comparison to larger biomolecules. The larger biomolecules will be covalently cross-linked to the polymers and will be impeded in their movement. For example, hydrogels based on PNIPAM will undergo swelling and hydrophobically collapse, which will result in the reduction of pore size. After this, the PNIPAM hydrogels will slow down the rate of diffusion of the molecules. In contrast, the Pluronic gels will retain an open micellar structure and thus the rate of release will be increased. In the previously described manner, the cross-linking between hydrophobic and hydrophilic polymers, with only a few constraining parameters, will be used to adjust the rate of release through diffusion.
Degradation-Controlled Release
In thermowell’s that are biodegradable, the polymer network enclosing a drug release, by slow hydrolysis or enzymatic cleavage, sequentially and for poly- (lactic-co-glycolic acid) PEG copolymer, hydrolysis of the ester bonds takes place. The natural polymers alginate, chitosan and gelatin and polysaccharides, are the only ones that demonstrate enzymatic degradation, while the synthetic PEG-PLGA copolymer undergoes hydrolysis of the ester bonds.45 The polymer composition, molecular weight, and the crosslinking affect the rate of biodegradability. In the subsets of tissue engineering and oncology, a therapeutic’s long-term gradual release, over weeks or months, is required; the release of biodegradable polymers that are crosslink cleavage for this purpose. The therapeutic’s effect of increased biocompatibility and improved biodegradability post release permitting the elimination of the need for tissue sequestration surgically, is a diagnostic gain.
Swelling-Controlled Release
The hydrogel shows a release mechanism that depends on swelling kinetics. When put in a wetting medium, the hydrophilic parts of the swelling absorb the water, the 3D swell and the drug begins to diffuse. Critical to swelling are: temperature, ionic strength, composition of the polymer. In LCST-type systems, swelling is followed by the construction of a polymer, and the diffusion of the drug is then slowed.46 This means that the control of the rate of release can be done by employing temperature. Such temperature alterations can start and/or stop release cycles, thereby providing for the release of the drug on demand, temperature acting as a control. The ability to undergo reversible swelling and collapsing transitions contributes to this.
Mathematical Models for Drug Release Kinetics
To quantitatively describe drug release behavior, several mathematical kinetic models are applied to experimental data. The most widely used are the Higuchi and Korsmeyer–Peppas models.
Higuchi Model
Developed for diffusion-controlled systems, this model assumes uniform drug distribution and constant diffusivity within the matrix. The cumulative amount of drug released per unit area (Q) is proportional to the square root of time (t): Q=kHtQ = k_H \sqrt{t}Q=kHt
where kHk_HkH is the Higuchi release constant. This model is appropriate for initial release phases dominated by Fickian diffusion.
Korsmeyer–Peppas Model
This empirical equation explains both diffusion and polymer relaxation mechanisms:
MtM∞=ktn\frac{M_t}{M_\infty} = k t^nM∞Mt=ktn
where Mt/M∞M_t/M_\inftyMt/M∞ is the fractional drug release at time ttt, kkk is the kinetic constant, and nnn is the diffusional exponent.
n=0.5n = 0.5n=0.5 indicates Fickian diffusion o 5<n<10.5 < n < 10.5<n<1 suggests anomalous transport
n=1n = 1n=1 represents Case II (relaxation-controlled) release
These models help interpret the release mechanism and predict performance under different formulation conditions.
Nanocarrier–Hydrogel Hybrid Systems
Finding ways to combine nanocarriers with thermosensitive hydrogels is innovative for multifunctional stimuli-responsive drug delivery systems. These hybrid platforms are able to combine the encapsulation benefits, which include improved drug solubility, stability, and
targeting with the hydrogels improved drug delivery and retention. In addition, hydrogels possess spatiotemporal control and drug release and the ability to inject and gel in situ. The use of certain types of thermoresponsive hydrogels, such as polymeric nanoparticles, micelles, liposomes, dendrimers, and solid lipid nanoparticles, enhance release control and mechanical strength. For example, PNIPAM-Pluronic hybrid hydrogels contain doxorubicin nanoparticles, which are able to maintain therapeutic levels at the tumour site for over 2 weeks, thereby minimizing systemic exposure and providing sustained release.47 Hydrogels likewise protect liposomes and encapsulated biomolecules (proteins, nucleic acids, etc.) from enzymatic degradation. Hydrogels with micelles also help improve the solubilization of hydrophobic drugs. The use of amphiphilic block copolymers, such as PLGA-PEG and PCL-PEG, form micelles with hydrophobic cores that help to encapsulate drugs that are poorly soluble in water.
Incorporation of micelles into thermogels like Pluronic F127 allows micelles to act as one of the dual diffusion barriers. The gel network sustains therapeutic delivery while the micelles serve as dual diffusion barriers.
The composites of nanocarrier and hydrogels allow the delivery of multiple therapeutic agents, which include small and large molecules, such as genes, peptides, and proteins, either simultaneously or sequentially. Chitosan-β-glycerophosphate thermogels integrated with polyplex nanoparticles, for example, simultaneously delivered chemotherapeutic agents and plasmid DNA to tumour cells for the combination of cytotoxic and gene-silencing effects. The PEG-PLGA micelles encased in the thermosensitive gel PNIPAM matrices guaranteed on-demand, controlled for sustained localized therapy. The micelles stabilized proteins. Small molecules were released thermally for diffusion through the gelled matrix.48 The combination of thermogels with nanocarriers offers precise targeting and controlled variability of the pharmacokinetics than the standalone application of conventional gels or nanoparticles. Tissue or organ of interest gel polymer can trap nanoparticles. Targeting moieties for active tissue targeting, such as folic acid or RGD peptides, and antibodies can also be added to these functionalized nanoparticles.
Because of the constituent characteristics, hybrid systems display biocompatibility. The natural polymers (chitosan, gelatin, and alginate) provide biodegradability and non-toxicity. The synthetic copolymers provide mechanical strength, as well as, control over degradation rates. The presence of multiple networks improves the hydrogels rheological properties. The hydrogel assists nanocarriers in avoiding premature clearance and aggregation, while reinforced nanocarriers reduce syneresis and enhance injectability. The sustained active release from the encapsulated gel is synchronized to the gels degradation, while the hybrid formulations reduce the risk of burst release, and provide multi-phase release.49 The synergistic mechanism in localized cancer therapy and chronic disease prolongs the unwanted therapeutic effects and reduces the frequency of dosing. The incorporation of multi-responsiveness to hybrid nanocarrier-hydrogel systems enables them to respond to temperature, pH, enzymes, and redox gradients. The hybrid systems are capable of smart drug release during pathologic events. The design control of hybrid gel using 3D bioprinting and microfluidics automation enhances programmable and controllable release mechanisms from a patient-centric perspective. These systems are the most advanced of stimuli-responsive systems and provide the next generation therapeutics in personalized medicine for patients requiring localized therapy.
Biomedical and Pharmaceutical Applications
Minimally invasive localized drug delivery via thermosensitive hydrogels has revolutionized practices in oncology, ophthalmology, dermatology, regenerative medicine, and immunotherapy.
Thermosensitive hydrogels can provide target specific delivery without the need for invasive surgical methods. They offer therapeutic benefits with their injectable, in situ gelling features. Moreover, their customizable profile for degradation and release can maintain therapeutic efficacy for prolonged periods of time.
Localized Cancer Therapy
Thermoresponsive hydrogels can be used as injectable hydrogels for localized chemotherapy to reduce the toxicity of the chemotherapy while improving the retention of the drug at the tumour sites. Pluronic F127 hydrogels loaded with the chemotherapy drugs paclitaxel, doxorubicin, and cisplatin, for example, have been shown to be effective at suppressing tumour growth in vivo.50 The hydrogels are in a liquid state at room temperature, making them easy to inject, and, upon reaching body temperature, become solid depots for sustained drug release. Delivery of combination therapies, where chemotherapy is combined with gene silencing (using siRNA or plasmid DNA), via PNIPAM–PEG copolymers has been accomplished. Curcumin (an anti-cancer agent) loaded chitosan-glycerophosphate gels have strong anti-inflammatory and anti-cancer activity. These systems can also be used as removable post-surgical implants to prevent tumour recurrence by releasing anti-cancer drugs for several weeks.
Ocular Drug Delivery
Standard eye drops have problems with quick drainage and low bioavailability. Thermosensitive in situ gelling systems solve these problems by solidifying into a gel upon contact with the eye surface. Formulations based on Poloxamer with timolol maleate, pilocarpine, or dexamethasone have shown better therapeutic effects with longer residence times than traditional eye drops. Hybrid gels made from PNIPAM and hyaluronic acid increase adherence to the cornea and lower the required doses, improving patient compliance.51 Biodegradable thermogels made of gelatin or chitosan are also used for delivery to the back of the eye, promoting non-invasive treatment of glaucoma and macular degeneration, and aiding other degenerative diseases of the macula.
Transdermal and Injectable Depot Systems
Transdermal applications include thermogelling patches and depots that can be injected. Thermogels contain transdermal systems meant for controlled release through the skin and subcutaneous layers. A case in point are constant release systems for analgesic (lidocaine) and anti-inflammatory (diclofenac) systems embedded in soft gel Pluronic F127. Their soft gel systems enable close skin contact for the temperature responsive release of the therapeutics. PEG-PLGA thermogels injectable depots are used for long-acting delivery of proteins and peptides, for example, insulin, leuprolide and growth factor. This technology for depot delivery minimizes the burden of injections, thus improving therapeutic adherence.
Example Formulations and Therapeutic Outcomes
Table 2: Representative Thermosensitive Hydrogel Formulations and Their Biomedical Applications
| Polymer System | Loaded Agent | Application | Outcome |
| PluronicF127 +
Doxorubicin |
Anticancer drug | Localized tumour
therapy |
Sustained release for 14 days, reduced systemic toxicity |
| Chitosan/β-
Glycerophosphate + Curcumin |
Anti-inflammatory | Cancer and
wound healing |
Enhanced bioavailability and cellular uptake |
|
PNIPAM–PEG Copolymer + Paclitaxel |
Chemotherapy |
Injectable depot |
Controlled dual-phase release, high local concentration |
| Poloxamer F127+
Timolol |
β-blocker |
Ocular delivery |
3× increased retention time on cornea |
| PEG–PLGA + BSA | Protein delivery | Injectable depot | Maintained protein stability and activity |
| Chitosan–Collagen +
Silver NP |
Antibiotic scaffold | Wound healing | Accelerated tissue regeneration and infection control |
| Pluronic–Chitosan+
OVA antigen |
Vaccine delivery | Mucosal immunization | Enhanced mucosal IgA and systemic immune response |
Evaluation and Performance Testing
Thermosensitive hydrogels are successful as long as they are safe, adhere to guidelines, and have positive in vitro and in vivo results and pharmacokinetic behaviour. It reiterates the importance of the designed functional system because it must be safe for therapeutic applications and clinically viable. With this, the therapeutic safety concern places system pharmacokinetics and legal constraints subordinate to the system’s functional components. Before any in vivo applications, thermosensitive hydrogels must undergo biocompatibility and cytotoxicity testing through in vitro applications. In this regard, the mentioned cytotoxicity assays, MTT, Alamar Blue, and Live/Dead fluorescence staining, are some of the standard assays used to assess cell metabolic activity in hydrogels with different extraction levels and confluent hydrogels surfaces.52 PNIPAM, Poloxamer F127, PEG-PLGA, and Chitosan-beta glycerophosphate hydrogels are all Polymers with necrotic Xenogeneic non-crosslinked polymeric negative cytotoxicity.
Injectable or intravascular hydrogels undergo an additional assessment step, this one centred on hemocompatibility. This evaluation takes into account blood component interactions, using hemolysis ratio, platelet aggregation, and coagulation time. Hemolysis over 5% is flagged as a safety issue. The inclusion of hemocompatibility and tissue integration improving polymers (natural and gelatin or hyaluronic type) is a good start. To understand thermogels predictive therapeutic efficacy and safety, one has to consider drug release profile, systemic circulation, and biodistribution. Studies of this type are largely performed using animal models, and they measure and record the concentration of the therapeutic agent in the plasma and target tissues over time. Compared to free drug solutions, thermogelling formulations have a longer half-life, reduced burst release, and a more localized drug distribution. For example, a thermogel made of Pluronic F127 and paclitaxel-maintained plasma levels of paclitaxel for over 96 hours, while free paclitaxel was eliminated within 12 hours. The use of fluorescence labelling, PET and MRI (Magnetic Resonance Imaging) and gamma scintigraphy has given several studies a better understanding of drug diffusion and the in vivo degradation of the hydrogel and the associated pharmacokinetics.
Depending on how thermosensitive hydrogels work, and what materials are used, they may be classified as a combination of a medical device and drug, or potentially a device on its own.
Regulatory bodies like the U.S. FDA (United States Food and Drug Administration), the EMA (European Medicines Agency), and CDSCO (Central Drugs Standard Control Organization) (India) need some form of biocompatibility (ISO 10993 standards) and some form of toxicology, along with stability and efficacy, to clear them. Ethics govern every aspect of experimentation with animals and clinical trials.53 These ethical guidelines are a way to ensure the quality and traceable of the product to GLP (Good Lab Practices) and GMP (Good Manufacturing Practices). These products do need ethical clearances from the IAEC (Institutional Animal Ethics Committee) and the IRB (Institutional Review Board). In addition to those, to ensure clinical safety, hydrogel sterilization (γ-irradiation, autoclaving, and filtration) and endotoxin testing are necessary.
Challenges, Limitations, and Engineering Solutions
Despite considerable advancements that have taken place, the numerous technical and scientific hurdles that still limit the widespread clinical and commercial application of thermosensitive hydrogels should also be taken into consideration. Some of these are mechanical instability, batch to-batch variability, burst release, and sterilization issues. Severe lack of mechanical strength within the physiological range of thermogels, particularly Pluronics and PNIPAM, could be considered the weakest link. Furthermore, the thermogels are also subjected to dissolution and erosion as a result of bodily fluid dilution. Composite or double-network hydrogels are examples of alternatives. The addition of nanoclay, bioceramics, silica nanoparticles, and graphene oxide improves injectability while also enhancing rigidity and mechanical strength. The combination of enzyme and photo-mediated crosslinking, as well as horseradish peroxidase, with chitosan or gelatin derivatives improves stabilization. The use of multi-step crosslinking techniques, whereby the first (and reversible) step is a physical one (temperature-induced), and the second step is chemical (covalent bonding), to achieve a reversible thermo-response is another suggested approach. Burst release is also an issue with thermogels, particularly with hydrophilic drugs housed within loosely entangled polymer networks.
Inadequate control over toxicity and therapeutics is a major concern. Some of the methods that have been used to address the problem include nanocarrier embedment, surface coating, and the adjustment of crosslinking density.
Embedding PLGA nanoparticles or liposomes into PNIPAM matrices creates additional barriers for diffusion, which further helps reduce burst release. Likewise, sustained release kinetics can be achieved with hydrophobic modifications of the polymer backbone PNIPAM–butyl acrylate copolymers (and others) since the hydrophobic character of the dielectric (which is greater than the polymer dielectric constant) hinders diffusion in PNIPAM matrices. Certain hydrogels sensitivities to various factors make them more challenging to sterilize. For instance, attempts to sterilize by autoclaving runs the risk of permanently damaging the polymer chains. Gamma radiation can damage polymer chains, lower the critical temperature (LCST) of the polymer, or both. Therefore, options such as sterile filtration, the mixing of sterile components, or pseudo gelation are best. Hydrogels can be lyophilized to improve stability during long-term storage. For long-term storage, cryoprotectants, and antioxidants are helpful in protecting against oxidative degradation as well as oxidative degradation of encapsulated drugs and the polymer. The lack of reproducibility in large-scale manufacturing remains a bottleneck. Using Process Analytical Technology (PAT) in conjunction with artificial intelligence-aided predictive modelling to establish uniformity of rheological and thermal properties of the polymer integrated with controlled soluble mol. wt. polymer, solvent, and temperature to decrease the system’s lower critical solution temperature (LCST) can improve gelation kinetics and viscosity
Using microfluidic and similar continuous manufacturing methods can accomplish specific structures for nucleation and polymerization/growth to address inter-batch (and intra-batch) variability.
Future Perspectives
Future studies involving thermosensitive hydrogels will see the emergence of new individualized designs incorporating smart multifunctional systems combined with new evolving technologies such as nanotechnology, biosensing, and 3D bioprinting technologies. With the advent of hydrogels that respond simultaneously to multiple stimuli, a revolution in the drug delivery system is expected. These include temperature, pH, ionic strength, light, and specific enzymes. For example, the PNIPAM–chitosan–graphene oxide hybrids that respond to both temperature and pH can be employed in the microenvironments of certain tumours where both temperature and pH are elevated. The development of photo responsive hydrogels with incorporated gold nanorods and azobenzene compounds also shows unique spatiotemporal control with the use of near-infrared light (NIR) to induce triggered release.
An example of using 3D and 4D bio printed temperature-sensitive hydrogels is creating customized bioinks for complex individual bio-tissue scaffolds. The temperature-sensitive hydrogels allow for control of extrusion at room temperature for 3D bioprinting, and at physiological temperatures, they aid in rapid solidification. These technologies help construct systems for innovative personalized therapies, including bespoke implants, organ-on-chip devices, therapeutic wound dressings, and more. In personalized, responsive therapeutics for dynamic tissue regeneration, 4D bioprinting has offered a transformative leap. Considering the recent developments and the existing trends in hydrogel technologies, the forthcoming innovations in hydrogel systems may feature the integration of nanostructures, smart drug nanocarriers, and more complex electronic systems, forming the next generation of self-regulating therapeutic technologies. Such therapeutic technologies can utilize biosensors to enable the release of therapeutics in response to monitored physiological variables (e.g., glucose, pH, and inflammatory levels) to provide responsive and continuous drug release.
The combination of temperature-sensitive drug tapering systems, nano storage, and microneedle arrays offers promising avenues for controlled and ultra-minimally invasive deliveries. Such systems can address the pancreas and cancer, along with any variable-dosing need. Even though clinical translation of such systems seems likely, the hurdles remain associated with the scalable and reproducible aspects of compliant clinical manufacturing. There is a need for stakeholder directed Research Design and the Clinical Translational processes; this should minimize the need for the standard work required in the regulatory approval. The use of machine learning and other computational tools can predict and/or quantify hydrogel systems and facilitate clinical manufacturing processes.
Conclusion
Among the most advanced and adaptable materials in modern drug delivery are thermosensitive hydrogels. These hydrogels are injected and form hydrogels in situ, which gives the most control over and comfort to the patient. However, the most advantageous and important feature in drug delivery is the location and kinetics of drug release. Along with hysteresis after the therapeutic window, the patient is subjected to release discomfort. For the last twenty years, cross-disciplinary research and integration in polymer chemistry, nanotech, and bioengineering have expanded the therapeutic applications of these thermosensitive hydrogels to include localized malignancies, ocular drug delivery, tissue regeneration, and vaccine delivery. Current research focuses on the mechanical integrity of hybrids, the pos systems, and the regulations controlling their use. When combined with 3D bioprinting, thermosensitive hydrogels are expected to come even nearer to the ideal drug delivery system. 3D bioprinting, soft and pliable materials, and integrated pos systems will drive focused research in these systems. After transforming hydrogels into smart responsive systems, thermosensitive hydrogels will function as predicted systems to deliver medication.
Acknowledgement
We express our sincere gratitude to the management of Shree Mahavir Education Society, Nashik, for their constant support and encouragement throughout the course of this study.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable.
Author Contribution
Prachi Kage: Conceptualization, Methodology, Investigation, Data curation, Writing – original draft.
Manoj Somvanshi: Data curation, Formal analysis, Writing – review & editing.
Angha Sarvadnya: Literature review, Visualization, Writing – review & editing.
Rutuja Patil: Resources, Validation, Writing – review & editing.
Atul Bendale: Supervision, Interpretation of data, Critical revision of the manuscript.
Anil Jadhav: Project administration, Supervision, Final approval of the manuscript.
References
- Lv T, Chen Y, Li N, et al. A comprehensive review of thermosensitive hydrogels: mechanism, optimization strategies, and applications. Gels. 2025;11(7):544. https://doi.org/10.3390/gels11070544
CrossRef - Khan B, Arbab A, Khan S, et al. Recent progress in thermosensitive hydrogels and their applications in drug delivery area. MedComm Biomater Appl. 2023;2(3):e55. https://doi.org/10.1002/mba2.55
CrossRef - Hameed H, Faheem S, Paiva-Santos AC, Sarwar HS, Jamshaid M. A comprehensive review of hydrogel-based drug delivery systems: classification, properties, recent trends, and applications. AAPS PharmSciTech. 2024;25(4):64. https://doi.org/10.1208/s12249-024-02786-x
CrossRef - Yu Y, Cheng Y, Tong J, et al. Recent advances in thermo-sensitive hydrogels for drug delivery. J Mater Chem B. 2021;9(13):2979-2992. https://doi.org/10.1039/D0TB02877K
CrossRef - Wang Z, Ye Q, Yu S, Akhavan B. Poly ethylene glycol (PEG)‐based hydrogels for drug delivery in cancer therapy: a comprehensive review. Adv Healthc Mater. 2023;12(18):2300105. https://doi.org/10.1002/adhm.202300105
CrossRef - Yaramiri A, Asalh RA, Asalh MA, AlSawaftah N, Abuwatfa WH, Husseini GA. A Comprehensive Review of Smart Thermosensitive Nanocarriers for Precision Cancer Therapy. International Journal of Molecular Sciences. 2025; 26(15):7322. https://doi.org/10.3390/ijms26157322
CrossRef - Chopra H, Singh I, Kumar S, et al. A comprehensive review on hydrogels. Curr Drug Deliv. 2022;19(6):658-675. https://doi.org/10.2174/1567201818666210601155558
CrossRef - Luo J, Zhao X, Guo B, Han Y. Preparation, thermal response mechanisms and biomedical applications of thermosensitive hydrogels for drug delivery. Expert Opin Drug Deliv. 2023;20(5):641-672. https://doi.org/10.1080/17425247.2023.2217377
CrossRef - Rahmanian-Devin P, Baradaran Rahimi V, Askari VR. Thermosensitive chitosan-β-glycerophosphate hydrogels as targeted drug delivery systems: an overview on preparation and their applications. Adv Pharmacol Pharm Sci. 2021;2021:6640893. https://doi.org/10.1155/2021/6640893
CrossRef - Chatterjee S, Hui PC-l. Review of Applications and Future Prospects of Stimuli-Responsive Hydrogel Based on Thermo-Responsive Biopolymers in Drug Delivery Systems. Polymers. 2021; 13(13):2086. https://doi.org/10.3390/polym13132086
CrossRef - Huang H, Qi X, Chen Y, Wu Z. Thermo-sensitive hydrogels for delivering biotherapeutic molecules: a review. Saudi Pharm J. 2019;27(7):990-999. https://doi.org/10.1016/j.jsps.2019.08.001
CrossRef - Jha A, Rama A, Ladani B, et al. Temperature and pH-responsive nanogels as intelligent drug delivery systems: a comprehensive review. J Appl Pharm Sci. 2021;11(12):001-016. https://dx.doi.org/10.7324/JAPS.2021.1101201
CrossRef - Fan R, Cheng Y, Wang R, et al. Thermosensitive hydrogels and advances in their application in disease therapy. Polymers. 2022;14(12):2379. https://doi.org/10.3390/polym14122379
CrossRef - Patel P, Mandal A, Gote V, Pal D, Mitra AK. Thermosensitive hydrogelbased drug delivery system for sustained drug release. J Polym Res. 2019;26(6):131. https://doi.org/10.1007/s10965-019-1771-z
CrossRef - Nagam SP, Jyothi AN, Poojitha J, Aruna S, Nadendla RR. A comprehensive review on hydrogels. Int J Curr Pharm Res. 2016;8(1):19-23. https://www.researchgate.net/publication/316887977_A_Comprehensive_review_on_hydrogels
- Sapino S, Chirio D, Peira E, Abellán Rubio E, Brunella V, Jadhav SA, Chindamo G, Gallarate M. Ocular Drug Delivery: A Special Focus on the Thermosensitive Approach. Nanomaterials. 2019; 9(6):884. https://doi.org/10.3390/nano9060884
CrossRef - Li JY, Feng YH, He YT, et al. Thermosensitive hydrogel microneedles for controlled transdermal drug delivery. Acta Biomater. 2022;153:308-319. https://doi.org/10.1016/j.actbio.2022.09.023
CrossRef - Siddique MI. Stimuli-responsive hydrogels for controlled drug delivery in oncology: a comprehensive review. Asian J Pharm. 2025;19(2). https://doi.org/10.22377/ajp.v19i2.6499
CrossRef - Nadeem N, Sohail M, Asad MHHB, et al. Thermosensitive hydrogels: from bench to market. Curr Sci. 2018;115(12):2256-2266. https://www.jstor.org/stable/26495791
CrossRef - Sonawane S, Shelke H, Bonde S. A comprehensive review on natural polysaccharides based in situ gels for nasal drug delivery. Int J Biol Macromol. 2025;145987. https://doi.org/10.1016/j.ijbiomac.2025.145987
CrossRef - Singh D, Kumar A. A comprehensive review on supramolecular polymer hydrogels for localized and long-term therapeutic delivery. J Macromol Sci B. 2025;1-11. https://doi.org/10.1080/00222348.2025.2474370
CrossRef - Abuwatfa WH, Awad NS, Pitt WG, Husseini GA. Thermosensitive Polymers and Thermo-Responsive Liposomal Drug Delivery Systems. Polymers. 2022; 14(5):925. https://doi.org/10.3390/polym14050925
CrossRef - Hani U, M YB, Wahab S, et al. A comprehensive review of current perspectives on novel drug delivery systems and approaches for lung cancer management. J Pharm Innov. 2022;17(4):1530-1553. https://doi.org/10.1007/s12247-021-09582-1
CrossRef - Dhandapani T, Krishnan VR, Muthukumar BALAGEE, et al. Emerging trends in stimuli-sensitive drug delivery system: a comprehensive review of clinical applications and recent advancements. Int J Appl Pharm. 2023;15(1):38-44. https://dx.doi.org/10.22159/ijap.2023v15i6.48974
CrossRef - Wang, S., Wang, X., Luo, Y. et al.A comprehensive review of conventional and stimuli-responsive delivery systems for bioactive peptides: from food to biomedical applications. Adv Compos Hybrid Mater 8, 12 (2025). https://doi.org/10.1007/s42114-024-01053-8
CrossRef - Pardeshi S, Damiri F, Zehravi M, Joshi R, Kapare H, Prajapati MK, Munot N, Berrada M, Giram PS, Rojekar S, et al. Functional Thermoresponsive Hydrogel Molecule to Material Design for Biomedical Applications. Polymers. 2022; 14(15):3126. https://doi.org/10.3390/polym14153126
CrossRef - Farasati Far B, Naimi-Jamal MR, Safaei M, Zarei K, Moradi M, Yazdani Nezhad H. A Review on Biomedical Application of Polysaccharide-Based Hydrogels with a Focus on Drug Delivery Systems. Polymers. 2022; 14(24):5432. https://doi.org/10.3390/polym14245432
CrossRef - Xiao Y, Gu Y, Qin L, et al. Injectable thermosensitive hydrogel-based drug delivery system for local cancer therapy. Colloids Surf B Biointerfaces. 2021;200:111581. https://doi.org/10.1016/j.colsurfb.2021.111581
CrossRef - Liu B, Chen K. Advances in Hydrogel-Based Drug Delivery Systems. Gels. 2024; 10(4):262. https://doi.org/10.3390/gels10040262
CrossRef - Wang J, Huang L, Wu E, Li X, Rao Y, Zhu C. Recent Advances on Chitosan-Based Thermosensitive Hydrogels for Skin Wound Treatment. Biology. 2025; 14(6):619. https://doi.org/10.3390/biology14060619
CrossRef - Youness R, Kamel R, A. Elkasabgy N, Shao P, A. Farag M. Recent Advances in Tannic Acid (Gallotannin) Anticancer Activities and Drug Delivery Systems for Efficacy Improvement; A Comprehensive Review. Molecules. 2021; 26(5):1486. https://doi.org/10.3390/molecules26051486
CrossRef - Ghosh Majumdar, A., Pany, B., Parua, S.S. et al.Stimuli-Responsive Nanogel/Microgel Hybrids as Targeted Drug Delivery Systems: A Comprehensive Review. 14, 3496–3521 (2024). https://doi.org/10.1007/s12668-024-01577-9
CrossRef - Mashabela LT, Maboa MM, Miya NF, Ajayi TO, Chasara RS, Milne M, Mokhele S, Demana PH, Witika BA, Siwe-Noundou X, et al. A Comprehensive Review of Cross-Linked Gels as Vehicles for Drug Delivery to Treat Central Nervous System Disorders. Gels. 2022; 8(9):563. https://doi.org/10.3390/gels8090563
CrossRef - Herrada Céspedes A, Reyes M and Morales JO (2025) Advanced drug delivery systems for oral squamous cell carcinoma: a comprehensive review of nanotechnology-based and other innovative approaches. Drug Deliv.5:1596964. doi: 10.3389/fddev.2025.1596964
CrossRef - Liew KB, Yahya FS, Azli NDW, et al. Advanced technologies in rectal drug delivery systems: a comprehensive review of recent innovations and future prospects. Drug Deliv Lett. 2024;14. https://doi.org/10.2174/0122103031328871241023075235
CrossRef - Godase SS, Kulkarni NS, Dhole SN. A comprehensive review on novel lipid-based nano drug delivery. Adv Pharm Bull. 2023;14(1):34-48. https://doi.org/10.34172/apb.2024.012
CrossRef - Segneanu A-E, Bejenaru LE, Bejenaru C, Blendea A, Mogoşanu GD, Biţă A, Boia ER. Advancements in Hydrogels: A Comprehensive Review of Natural and Synthetic Innovations for Biomedical Applications. Polymers. 2025; 17(15):2026. https://doi.org/10.3390/polym17152026
CrossRef - Wassif, R.K., Shamma, R.N., El-Hoffy, N.M. et al.Recent Advances in the Local Drug Delivery Systems for Diabetic Wound Healing: A Comprehensive Review. AAPS PharmSciTech 26, 177 (2025). https://doi.org/10.1208/s12249-025-03172-x
CrossRef - Singh RB, Ichhpujani P, Thakur S, Jindal S. Promising therapeutic drug delivery systems for glaucoma: a comprehensive review. Ther Adv Ophthalmol. 2020;12:2515841420905740. https://doi.org/10.1177/2515841420905740
CrossRef - Haggag YA, Abd Elrahman AA, Ulber R, Zayed A. Fucoidan in Pharmaceutical Formulations: A Comprehensive Review for Smart Drug Delivery Systems. Marine Drugs. 2023; 21(2):112. https://doi.org/10.3390/md21020112
CrossRef - Xu Y, Wang JY, Meng T, et al. Role of hydrogels in osteoarthritis: a comprehensive review. Int J Rheum Dis. 2023;26(12):2390-2401. https://doi.org/10.1111/1756-185x.14968
CrossRef - Vikal A, Maurya R, Bhowmik S, et al. From conventional to cutting-edge: a comprehensive review on drug delivery systems. Drug Deliv Lett. 2024;14(3):226-243. https://doi.org/10.2174/0122103031304556240430161553
CrossRef - Baishya G, Parasar B, Limboo M, et al. Advancements in nanocomposite hydrogels: a comprehensive review of biomedical applications. Discov Mater. 2024;4(1):40. https://doi.org/10.1007/s43939-024-00111-8
CrossRef - Kamini, Puri D. Hydrogel-based drug delivery systems-a review. Polym Plast Technol Mater. 2024;63(16):2213-2236. https://doi.org/10.1080/25740881.2024.2369670
CrossRef - Chung CK, Fransen MF, van der Maaden K, et al. Thermosensitive hydrogels as sustained drug delivery system for CTLA-4 checkpoint blocking antibodies. J Control Release. 2020;323:1-11. https://doi.org/10.1016/j.jconrel.2020.03.050
CrossRef - Alqahtani AS. Topical drug delivery in oral mucosal diseases: challenges, carriers, and innovations: a comprehensive review. Biomed Pharmacol J. 2025;18(3):1835-1848. https://dx.doi.org/10.13005/bpj/3218
CrossRef - Wierzbicka A, Krakos M, Wilczek P, Bociaga D. A comprehensive review on hydrogel materials in urology: problems, methods, and new opportunities. J Biomed Mater Res B Appl Biomater. 2023;111(3):730-756. https://doi.org/10.1002/jbm.b.35179
CrossRef - Chen, X., Jing, S., Xue, C. et al.Progress in the Application of Hydrogels in Intervertebral Disc Repair: A Comprehensive Review. Curr Pain Headache Rep 28, 1333–1348 (2024). https://doi.org/10.1007/s11916-024-01296-6
CrossRef - Padmasri B, Nagaraju R, Prasanth D. A comprehensive review on in situ gels. Int J Appl Pharm. 2020;12(6):24-33. http://dx.doi.org/10.22159/ijap.2020v12i6.38918
CrossRef - Rajgopal B, Gupta SK, Deshmukh R, et al. Emerging trends in hydrogel for the treatment of vaginal candidiasis: a comprehensive review. Rec Adv Anti-Infec Drug Discov. 2025. https://doi.org/10.2174/0127724344348928250220063431
CrossRef - Ikhsanudin A, Sulaiman TN, Zai K, Ari BS. Thermosensitive hydrogels as drug carriers for breast cancer treatment: a systematic review. Int J Public Health Sci. 2023;12(3):1327-1336. doi:10.11591/ijphs.v12i3.22773
CrossRef - Tipa C, Cidade MT, Vieira T, Silva JC, Soares PI, Borges JP. A new long-term composite drug delivery system based on thermo-responsive hydrogel and nanoclay. Nanomaterials. 2020;11(1):25. https://doi.org/10.3390/nano11010025
CrossRef - Shi H, Wang Y, Bao Z, et al. Thermosensitive glycol chitosan-based hydrogel as a topical ocular drug delivery system for enhanced ocular bioavailability. Int J Pharm. 2019;570:118688. https://doi.org/10.1016/j.ijpharm.2019.118688
CrossRef
Abbreviations List
CS: Chitosan, F127: Poloxamer 407 (Pluronic F127), MC: Methylcellulose, PCL: Poly(\epsilon-caprolactone), PEG: Poly (ethylene glycol), PEO: Poly (ethylene oxide), PLGA: Poly (lactic-co-glycolic acid), PNIPAM: Poly(N-isopropylacrylamide), PPO: Poly (propylene oxide), BSA: Bovine Serum Albumin, IgA: Immunoglobulin A, NP: Nanoparticles, OVA: Ovalbumin (Antigen), LCST: Lower Critical Solution Temperature, Sol-Gel: Solution-to-Gel transition.
Accepted on: 27-03-2026
Second Review by: Dr. Supriya Shivanand Mhamane
Final Approval by: Dr. Eugene A. Silow









