Review on the Use of Niosomes as a Potential Formulation for Skin Health
1Department of Pharmaceutics, Sandip Institute of Pharmaceutical Sciences, Nashik, India.
2Department of Pharmaceutics, Siddhi’s Institute of Pharmaceutical Sciences, Kalyan, India.
3Department of Pharmaceutics, Mahavir Institute of Pharmacy, Nashik, India.
Corresponding Author Email: sagar.khokale@sandippharmacy.org
DOI : http://dx.doi.org/10.13005/bbra/3486
ABSTRACT:In this study, we will review the potential of niosomal formulation. The utilization of niosomes is progressively gaining momentum owing to their eco-friendliness, cost-effectiveness in materials and production, Viability for manufacturing on a large scale, and simplified maintenance compared to other delivery systems. Niosomes play a crucial role in medication delivery systems due to their nonionic nature, enabling controlled release and precise targeting for treating skin conditions and a range of other diseases. The review methodology encompassed several key phases: formulating the research query, conducting preliminary investigations, devising search strategies, database exploration, establishing exclusion and inclusion criteria, author concurrence, selection of titles and abstracts, documenting the number of selected data, full-text retrieval, and scrutiny, supplementary manual searches across platforms like Google Scholar, Scopus, Mdpi, and ResearchGate, data extraction and quality assessment, dual verification of data, revising, and eventual submission. A comprehensive analysis revealed a noteworthy enhancement in the aesthetic appeal of cosmetic products formulated using niosomal technology.
KEYWORDS:Amphiphilic; Controlled release; Cosmetics; Encapsulation; Nanotechnology; Niosomal formulation; Niosomes; Skin penetration; Transdermal administration
Introduction
Niosomes
The term “cosmetics” consists of a diverse array of products that can be applied externally or ingested 1,2. Usually, they’re used to add a pleasant odor to the area where they’re applied or to improve its appearance3,4. Due to their numerous benefits, including enhanced skin penetration, increased bioavailability, surface adhesion, and sustained release characteristics, niosomes have garnered significant interest as a carrier system for active cosmeceutical ingredients5,6. Vast majority of cosmeceutical ingredients face challenges in passing through the skin layers and maintaining chemical or physical stability. Recent advancements in nanotechnology, particularly the utilization of niosomes, have revolutionized this scenario. These innovations now allow for the loading of various medications onto nano-sized particles, facilitating precisely targeted medication delivery while also bolstering the chemical and physical stability of cosmeceutical and pharmaceutical products. Niosomes are more stable and less expensive to produce in comparison to liposomes. Also, they are very small in size ranging from 100-200nm7. The unique structure of niosomes makes them a prime illustration of a novel drug delivery system capable of transporting amphiphilic, lipophilic, and hydrophilic molecules8. Abundant evidence highlights the use of niosomes in diverse cosmetic applications, such as anti-aging, anti-wrinkle, bleaching, anti-alopecia, and moisturizing formulations9. Niosomes have shown reduced toxicity, allowing for controlled delivery and gradual release of encapsulated active agents with beneficial properties. This facilitates skin hydration and creates a tanning effect10,11. L’Oréal pioneered the integration of niosomes into skincare products during the 1970s, marking a significant milestone in cosmetic innovation12,13. The groundbreaking skincare product incorporating niosomes was introduced to the cosmetic market by L’Oréal’s subsidiary brand, Lancôme, in 1986, under the distinguished name “Niosôme” 14. Niosomes serve as highly efficient carriers for compounds due to their unique ability to encapsulate a diverse range of active ingredients, including water-soluble, lipid-soluble, and amphiphilic substances, all without necessitating any chemical alterations15,16. Targeted drug delivery systems aim to administer medications precisely to their intended site within the body, eliciting the desired therapeutic response while minimizing interactions with non-targeted tissues. Coined “magic bullets” by Paul Ehrlich in 1909, this concept directs drug action with precision17,18. Niosomes offer unique advantages compared to other vesicular systems by acting as drug reservoirs, enabling controlled release and sustained effects19,20. Liposomes were frequently overlooked because of their composition consisting of concentric phospholipids, which made them chemically unstable and susceptible to oxidative degradation and rancidity21. Niosomes exhibit promising potential for utilization across a broad spectrum of cosmetic formulations, encompassing anti-aging properties, antioxidant and free radical scavenging effects, skin brightening, scar-reducing qualities, and loads of beneficial capabilities.
Niosome Structure
Niosome consists of a bi-layered spherical structure comprised of non-ionic surfactant and cholesterol (Fig. 1). The surfactant is arranged such that the hydrophobic end points inward (toward the lipophilic phase), while the hydrophilic end faces out (towards the aqueous phase). This arrangement creates a closed lipid bilayer enveloping solutes in aqueous phase. This bilayer resembles the outer and inner surfaces of the hydrophilic area, with the lipophilic area sandwiched in between. The distinctive structural features, composition, chemical properties, and compact size of niosomes help them to effectively stabilize and transport drug molecules to their targeted locations22.
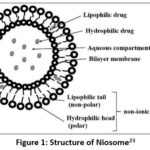 |
Figure 1: Structure of Niosome23
|
Thanks to their hydrophilic and hydrophobic characteristics, niosomes serve as excellent vesicles for encapsulating lipophilic, hydrophilic, and amphiphilic compounds24.
Types of Niosomes
Niosomes are available in three distinct variants based on particle size: large unilamellar vesicles (LUVs of size ≥ 0.05 μm), small unilamellar vesicles (SUVs of size 0.025–0.05 μm), and multilamellar vesicles (MLVs of size ≥ 0.10 μm) as shown in Figure 2. Surfactants are very important. Many research studies have looked into how nonionic surfactants are useful in creating niosomes. These niosomes are helpful for delivering molecules to specific places in the body or for making different cosmetic items. The way niosomes are formed depends a lot on the structure of the nonionic surfactants. They end up having layers made of hydrophilic (water-loving) and hydrophobic (water-repelling) parts joined together by certain kinds of chemical bonds25. Cholesterol, commonly utilized as a bilayer-inducing substance in niosome development, plays a pivotal role in stabilizing the niosome membrane. Consequently, studies have indicated that incorporating cholesterol may improve the drug-loading capacity of encapsulated drugs within niosomes. Apart from cholesterol, there are other substances called charge inducer agents that can be used when making niosomes. These materials help stop the niosome particles from clumping together and make it easier to deliver drugs to certain parts of the body. They do this by affecting something called the zeta potential26.
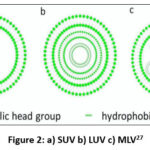 |
Figure 2: a) SUV b) LUV c) MLV27
|
Composition of Niosomes
Cholesterol: It is a steroid derivative that contributes to forming the proper shape and rigid structure of the niosome28.
Surfactant: The HLB value of a non-ionic surfactant is important when making niosomes. It’s best when the HLB value falls between 4 and 8 because it helps form stable vesicles that work well with drugs29,30. Sometimes charge inducers are also used as they induce repulsions on the surface of vesicles, resulting in increased zeta potential. This helps prevention of fusion and enhancement of stability of vesicles.
Preparation Methods31
The methods by which niosomes can be prepared are Thin film hydration, Ether injection Reverse phase evaporation, Trans-membrane pH gradient drug uptake process, Emulsion method, Lipid injection, Micelle solution and enzyme method, Bubble method, Micro fluidisation method, Formation of niosomes from pro-niosomes and Supercritical reverse phase evaporation method. The utilization of organic solvents is necessary for the dissolution of surfactants and cholesterol in thin film hydration process. Mix cholesterol and surfactant in a round-bottomed flask, followed by the organic solvent’s evaporation, leaving behind a delicate coating at the flask’s base. By adding a aqueous media and gently stirring the mixture, MLV’s are formed through the swelling of film from the wall of flask over a certain duration. These vesicles undergo further treatment to transition into unilamellar vesicles. The ether injection method involves the careful insertion of drugs and surfactants into an aqueous phase while elevating the temperature of the organic solvent above the boiling point. This meticulous process yields Large Unilamellar Vesicles (LUVs), which can subsequently undergo size reduction procedures. Conversely, in the reverse phase evaporation technique, surfactants are dispersed within an organic blend of chloroform and ether before the addition of an aqueous drug solution. Upon homogenizing the two immiscible phases and subjecting them to reduced pressure evaporation, a mixture of niosomes is anticipated to form, purportedly boasting high Encapsulation Efficiency (EE) and sizable particle dimensions. Notably, this method has shown promise in encapsulating large hydrophilic molecules more effectively than its predecessors. The transmembrane pH gradient strategy necessitates the dissolution of surfactants and cholesterol in an organic solvent to fabricate an emulsion containing niosomes. Subsequently, rotary evaporation helps the making of a thin film along the flask’s wall, which is then hydrated using a citric acid solution (of pH 4) followed by freezing, thawing, and sonication of the resulting droplets. After incorporating an aqueous drug mixture and adjusting the pH to 7, multilamellar vesicles are formed upon heating to 60 °C. Utilizing the emulsion method, a combination of surfactant and cholesterol within an organic phase is introduced to an aqueous phase housing the active ingredient, resulting in an oil-in-water emulsion. Upon evaporation of the organic solvent, a niosomal suspension emerges without the necessity of organic solvents. The lipid injection technique deviates from the reliance on organic solvents. In this process, a hot and vigorously stirred water-based solution with dissolved drug molecules is mixed with a non-ionic surfactant and a substance that helps form layers (like cholesterol). This mixture creates a suspension of niosomes. Additionally, enzymes can help make niosomes from a solution containing mixed micelles. For instance, esterase can break apart certain bonds in chemicals like polyethylene stearyl derivatives when mixed with dicetyl phosphate and other lipids, yielding breakdown products like cholesterol and polyoxyethylene, which can then be employed to produce multilamellar niosomes. The groundbreaking microfluidization technique yields smaller unilamellar particles with a constrained size distribution. By subjecting a blend of surfactant and drug to high pressure within a chamber, typically at a flow rate of around 100 mL/min, niosomes are formed. Subsequently, the fluid undergoes a cooling process to dissipate the heat generated during microfluidization, facilitating the creation of niosomes. In contrast to methods reliant on organic solvents, the reverse phase evaporation technique offers versatility without the need for potentially hazardous solvent removal. This method is particularly adept at generating substantial quantities of niosomes. Nevertheless, it tends to produce LUVs with sizes spanning from 100 to 500 nm. To mitigate this, techniques like sonication or extrusion can be used to reduce the niosomes into smaller dimensions. Lastly, the bubble approach presents a streamlined, solvent-free process. By combining a nonionic surfactant and a bilayer-inducing agent (cholesterol) in a buffer solution at 70 °C, followed by a brief 15-second mixing period using a high-shear homogenizer, LUVs are generated by cycling nitrogen gas through the mixture32.
Niosomes in Cosmetics: Their Mechanism on Skin Enhancement
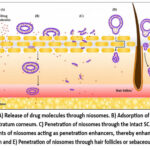
An essential criterion for transdermal delivery is the ability of the drug, carried by a carrier, to effectively reach the skin surface at a sufficient rate and in adequate quantities. A wide array of applications and the discovery of numerous mechanisms that enhance the penetration of drugs into deeper skin layers have been facilitated by the versatility of novel drug delivery methods. Generally, three potential routes through which the epidermis can be traversed by a permeant applied to the skin are available. The transcellular route, involving passage through the lipid domain associated with proteins within corneocytes, the intercellular route, and the appendageal route, which includes penetration through hair follicles, associated sebaceous glands, and sweat ducts, are included in these routes. The mechanism of drug transport across these routes may vary depending on the nature of the drug itself33. Various strategies are encompassed by mechanisms for enhancing the transdermal delivery of hydrophilic drugs. Increasing the thermodynamic activity of the drug is involved as one such method. The achievement of this involves the adsorption and fusion of drug-containing vesicles onto the skin surface. Subsequently, a thermodynamic activity gradient is established, resulting in heightened diffusion pressure for drug permeation at the skin’s surface. The penetration of the drug across the stratum corneum (SC) is facilitated by this gradient, which acts as a driving force34,35. Various mechanisms can be utilized for the transdermal enhancement of hydrophilic drugs like transdermal delivery enhancement of ionic drugs can be achieved by modifying their surface electrical charge. By altering the charge, the drugs can interact more favorably with the skin’s surface, thereby promoting their penetration through the stratum corneum. Solubilization of sebum by vesicles is facilitated. Vesicles have the capability to solubilize sebum, the oily substance naturally present on the skin. The delivery of drugs into hair follicles is facilitated by this property, as the solubilized sebum acts as a carrier for the drug, allowing it to reach deeper layers of the skin36. The pore pathway can be utilized by large water-soluble molecules loaded in niosomes. Such molecules can be encapsulated by niosomes, which are lipid-based vesicles. When applied to the skin, interaction with the skin’s pores can allow the encapsulated molecules to pass through and penetrate into the deeper layers of the skin with these niosomes. The transdermal delivery of hydrophilic drugs loaded in niosomes is enhanced by this pore pathway mechanism. Several strategies encompass mechanisms for enhancing the transdermal delivery of hydrophobic drugs which includes disruption of the lipid bilayer of the Stratum Corneum (SC) can lead to structural modifications, resulting in the integrity being disrupted, extracellular spaces being filled, and permeation rates being enhanced. The passage of hydrophobic drugs through the SC is facilitated by this disruption. Transdermal permeation can be enhanced through nano-sizing, which increases the surface area of hydrophobic drug particles, facilitating their interaction with the skin and enhancing transdermal permeation37. Drug partitioning into skin layers can be altered: The transdermal delivery of hydrophobic drugs can be enhanced by modifying their partitioning behavior into different skin layers. Various formulation techniques can be employed to achieve this. The skin can be hydrated, and the dilation of intercellular channels in the stratum corneum (SC) can occur: The barrier properties of the stratum corneum (SC) can be altered by niosomes, which are lipid-based vesicles. This alteration leads to enhanced skin hydration by reducing trans-epidermal water loss38. The tightly packed structure of the SC can be loosened by this hydration, which may result in membrane lysis by lysozyme, Hydrophobic drugs entrapped within the system can be released39,40. The permeation pathway can be changed to facilitate follicular delivery: The delivery of hydrophobic drugs to the skin’s follicles can be facilitated by altering the permeation pathway. Penetration enhancers as non-ionic surfactants enter into intercellular lipids through endocytosis and hence facilitating the delivery of hydrophobic drugs to the follicular region41. Non-ionic surfactants serve as pivotal penetration enhancers in transdermal delivery by permeating into intercellular lipids through endocytosis42,43. To comprehend the impact of niosomes on the skin, their interactions with the outermost layer, the stratum corneum (SC), have been investigated by researchers, employing microscopic techniques. Evidence suggests a prevalence of vesicular constituents on or near the SC surface, with their concentration gradually tapering off towards the inner SC region. At this juncture, it is posited that niosomes are likely to be merged and intermingled with the native SC lipids. While some images depict vesicular components even in deeper SC layers, there are instances where vesicular components have been observed in deeper SC layers, it remains ambiguous whether these represent intact niosomes traversing from the skin surface or if there’s uncontrolled vesicle turnover as the SC becomes more hydrated. The enhanced permeability of the vesicles in the skin may be attributed to a modified thermodynamic activity or the influence of “released” particles on the Stratum Corneum’s barrier function. Remarkable efficacy in delivering substances like enoxacin has been demonstrated by niosomes compared to liposomes or conventional active ingredient solutions. Larger molecules can penetrate the skin using niosomes, if the barrier function is compromised compared to intact skin44. The mechanisms of action to enhance skin penetration.
Potential mechanisms of action of niosomes for enhancing skin penetration in topical and transdermal drug delivery can be outlined as shown in Figure 3:
Advantages/Disadvantages of Niosomes
Advantages of niosomes
Niosomes possess an amphiphilic structure, providing a broad spectrum of solubility for drug molecules46. Their aqueous dispersion form enhances patient compliance compared to oily preparations47. Niosomes facilitate increased skin penetration of medications and enhance the oral bioavailability of poorly soluble drugs48. They can be administered via various routes including oral, parenteral, topical, and transdermal49. Composed of biocompatible, biodegradable, and non-toxic materials, niosomes are considered safe for use50. Characteristics of niosome formulations are adjustable by modifying parameters such as composition, size, lamellarity, surface charge, and concentration51. Niosomes can serve as drug depots, allowing controlled release of the drug to the target site from the closed bilayer structure52. They exhibit chemical stability, thus requiring no special handling or storage conditions. Niosomes are osmotically active, stable, and possess a longer shelf life. They shield the drug from enzymatic degradation53. Niosomes are highly cost-effective for large-scale production.
Disadvantages of niosomes
The dispersion form of niosomes may lead to sedimentation, accumulation, fusion, or leakage of the entrapped drug during storage54. Certain formulation techniques for niosomes can be time-consuming and necessitate specialized equipment. There is a risk of incomplete hydration of surfactants during the hydration process55.
Future of Niosomes in Cosmetics
The future looks bright for niosomes in cosmetics. Here’s why: They stay fresh longer: Niosomes keep the drug safe from breaking down, thus extending its duration in beauty products. Goes deeper: These niosomes assist in facilitating deeper penetration of the drug into the skin, thereby enhancing its efficacy. The target is reached by them: Niosomes can be tweaked to aim at specific skin issues, like dark spots or uneven tone, delivering the drug right where it is needed. Bye-bye irritation: Putting the drug into niosomes can calm down any potential skin irritation if present, making it nicer to use and safer too. Synergistic: Mix niosomes with other good-for-skin stuff, and a powerful combination is formed that can yield numerous benefits for your skin, like fighting aging or keeping it hydrated. Consumer preference: More and more folks want skincare products that are kind to the planet and their skin. By strategically blending drug with other active ingredients, its potential can be greatly magnified. This synergistic approach unlocks a range of benefits, comprehensively addressing various aspects of skin health: Enhanced Skin Brightening: preparing niosomes with potent skin-brightening agents like arbutin or licorice extract may yield powerful results56,57. Antioxidant Protection: Incorporating antioxidants such as vitamin C or green tea extract not only inhibits melanin production but also guards against oxidative stress, promoting overall skin health58,59. Moisture Retention and Hydration: Formulating with hyaluronic acid or glycerin enhances moisturization, ensuring optimal hydration and a radiant complexion60,61. Anti-Inflammatory Support: Partnering with anti-inflammatory agents like chamomile extract or aloe vera soothes sensitive skin, reducing irritation and redness62,63. Collagen Boosting: Introducing collagen-boosting ingredients such as peptides or retinol supports skin elasticity and firmness, addressing pigmentation concerns while promoting a youthful appearance64,65. Sunscreen Integration: Combining with broad-spectrum sunscreen ingredients like zinc oxide or titanium dioxide provides comprehensive protection against UV-induced pigmentation and other sun-related issues. Customizable Formulations: Tailoring niosomal formulation to individual skin needs, incorporating acne-fighting agents or anti-aging compounds, allows for personalized skincare solutions66. Niosomes fit the bill, ticking all the boxes for safer, eco-friendly beauty options. So, in short, niosomes are set to make a big splash in cosmetics, offering clever ways to tackle different skin issues and keeping everyone happy with healthier, happier skin.
Conclusion
The future outlook for niosomes in skincare appears promising. In conclusion, the future potential of niosomes lies in its ability to synergize with a spectrum of active ingredients. In simpler terms, the mentioned combinations provide advanced skincare formulations that not only address pigmentation concerns but also boost overall skin health, meeting diverse needs and preferences in the ever-changing skincare landscape. The data suggests that niosomes offer a promising way to improve skin health by delivering drugs through the skin. Traditional forms of medication often struggle with issues like low effectiveness and imprecise targeting. However, the tough outer layer of skin, the stratum corneum, limits the absorption of certain drugs and large molecules. Niosomes, a new type of vesicle made from non-ionic surfactants, are a promising solution to these problems. They offer benefits like controlled and sustained drug release without needing special storage. Recent studies show they can improve drug absorption through the skin and are useful in cosmetics for better aesthetic results. Niosome-based cosmetic products show improvements over traditional methods, especially in treating various skin conditions. But there’s still more to learn about using niosomes in cosmetics. More research and commercial products could make a big difference in skincare.
Acknowledgement
The author would like to thank Sandip Institute of Pharmaceutical Sciences, Mahiravani, Nashik for granting the research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Sagar Kashinath Khokale: Conceptualization, Methodology, Writing – Original Draft.
Harshali Raosaheb Ahire: Funding Acquisition, Resources, Supervision.
Mohit Madhukar Jadhav: Data Collection, Analysis, Writing – Review & Editing.
Chetana Krushnarao Shinde: Funding Acquisition, Resources, Supervision.
Sakshi Rajendra Shinde: Visualization, Supervision, Project Administration.
References
- Chen S, Hanning S, Falconer J, Locke M, Wen J. Recent advances in non-ionic surfactant vesicles (niosomes): Fabrication, characterization, pharmaceutical and cosmetic applications. .European Journal of Pharmaceutics and Biopharmaceutics.. 2019;144:18–39. [https://doi.org/10.1016/j.ejpb.2019.08.015](https://doi.org/10.1016/j.ejpb.2019.08.015).
CrossRef - Cerqueira-Coutinho C, dos Santos EP, Mansur CRE. Niosomes as nano-delivery systems in the pharmaceutical field. Critical Reviews in Therapeutic Drug Carrier Systems.. 2016;33(2):195–212. [https://doi.org/10.1615/CritRevTherDrugCarrierSyst.2016016167](https://doi.org/10.1615/CritRevTherDrugCarrierSyst.2016016167)
CrossRef - Umbarkar MG. Niosome as a novel pharmaceutical drug delivery: A brief review highlighting formulation, types, composition, and application. .Indian Journal of Pharmaceutical Education and Research.. 2021;55(1):11–28.
CrossRef - Hatem S, El Hoffy NM, Elezaby RS, Nasr M, Kamel AO, Elkheshen AS. Background and different treatment modalities for melasma: Conventional and nanotechnology-based approaches. .Journal of Drug Delivery Science and Technology.. 2020;60:101984. [https://doi.org/10.1016/j.jddst.2020.101984](https://doi.org/10.1016/j.jddst.2020.101984).
CrossRef - Bilal M, Iqbal HMN. New insights on unique features and role of nanostructured materials in cosmetics. .Cosmetics.. 2020;7(2):24. [https://doi.org/10.3390/cosmetics7020024](https://doi.org/10.3390/cosmetics7020024).
CrossRef - Wu X, Guy RH. Applications of nanoparticles in topical drug delivery and cosmetics. .Journal of Drug Delivery Science and Technology.. 2009;19(6):371–384. [https://doi.org/10.1016/S1773-2247(09)50061-8](https://doi.org/10.1016/S1773-2247%2809%2950061-8).
CrossRef - Mohanty D, Jhansi M, Bakshi AK. Niosomes: A novel trend in drug delivery. .Research Journal of Pharmacy and Technology.. 2018;11(11):5205–5211. [https://doi.org/10.5958/0974-360X.2018.00952.2](https://doi.org/10.5958/0974-360X.2018.00952.2)
CrossRef - Kheilnezhad B, Hadjizadeh A. Factors affecting the penetration of niosome into the skin, their laboratory measurements and dependency to the niosome composition: A review. .Current Drug Delivery.. 2021;18(5):555–569.[https://doi.org/10.2174/1567201 817999200916103907] (https://doi.org/10.2174/1567201817999200916103907)
CrossRef - Zhou H, Luo D, Chen DW. Current advances of nanocarrier technology-based active cosmetic ingredients for beauty applications. .Clinical, Cosmetic and Investigational Dermatology.. 2021;14:867–887. [https://doi.org/10.2147/CCID.S303334](https://doi.org/10.2147/CCID.S303334)
CrossRef - Chen S, Hanning S, Falconer J, Locke M, Wen J. Recent advances in non-ionic surfactant vesicles (niosomes): Fabrication, characterization, pharmaceutical and cosmetic applications. .European Journal of Pharmaceutics and Biopharmaceutics.. 2019;144:18–38. [https://doi.org/10.1016/j.ejpb.2019.08.015](https://doi.org/10.1016/j.ejpb.2019.08.015)
CrossRef - Bilal M, Iqbal HMN. New insights on unique features and role of nanostructured materials. .Cosmetics.. 2020;7(2):24. [https://doi.org/10.3390/cosmetics7020024](https://doi.org/10.3390/cosmetics7020024)
CrossRef - Cerqueira-Coutinho C, dos Santos EP, Mansur CRE. Niosomes as nano-delivery systems in the pharmaceutical field. .Critical Reviews in Therapeutic Drug Carrier Systems.. 2016;33(2):195–212. [https://doi.org/10.1615/CritRevTherDrugCarrierSyst.2016016167](https://doi.org/10.1615/CritRevTherDrugCarrierSyst.2016016167)
CrossRef - Bilal M, Iqbal HMN. New insights on unique features and role of nanostructured materials. .Cosmetics.. 2020;7(2):24. [https://doi.org/10.3390/cosmetics7020024](https://doi.org/10.3390/cosmetics7020024)
CrossRef - Saraf S, Kaur CD. Skin targeting approaches in cosmetics. .Indian Journal of Pharmaceutical Education and Research.. 2019;53(4):577–594. [https://doi.org/10.5530/ijper.53.4.114](https://doi.org/10.5530/ijper.53.4.114)
CrossRef - Manosroi A, Chutoprapat R, Abe M, Manosroi W, Manosroi J. Anti-aging efficacy of topical formulations containing niosomes entrapped with rice bran bioactive compounds. .Pharmaceutical Biology.. 2012;50(2):208–224. [https://doi.org/10.3109/13880209. 2011.596206](https://doi.org/10.3109/13880209.2011.596206)
CrossRef - Santos AC, Rodrigues D, Sequeira JAD, Pereira I, Simões A, Costa D, Peixoto D, Costa G, Veiga F. Nanotechnological breakthroughs in the development of topical phytocompounds-based formulations. .International Journal of Pharmaceutics.. 2019;572:118787. [https://doi.org/10.1016/j.ijpharm.2019.118787](https://doi.org/10.1016/j.ijpharm.2019.118787)
CrossRef - Kaur D, Kumar S. Niosomes: Present scenario and future aspects. .Journal of Drug Delivery and Therapeutics.. 2018;8(5):35–43. [https://doi.org/10.22270/jddt.v8i5.1923](https://doi.org/10.22270/jddt.v8i5.1923)
CrossRef - Madhav NV, Saini A. Niosomes: A novel drug delivery system. .International Journal of Research in Pharmacy and Chemistry.. 2011;1(3):498–511.
- Mittal S, Chaudhary A, Kumar A. Proniosomes: The effective and efficient drug-carrier system. .Therapeutic Delivery.. 2020;11(2):125–137. [https://doi.org/10.4155/tde-2019-0082](https://doi.org/10.4155/tde-2019-0082)
CrossRef - Lakshmi Radhika K. Proniosomal gel: A novel approach for drug delivery. .Indo-American Journal of Pharmaceutical Research.. 2017;7(3):7854–7860.
- Bachhav AA. Proniosome: A novel non-ionic provesicules as potential drug carrier. .Asian Journal of Pharmaceutics.. 2016;10(3).
CrossRef - Yeo PL. Niosomes: A review of their structure, properties, methods of preparation, and medical applications. .Asian Biomedicine.. 2018;11(4):301–314. [https://doi.org/10.1515/abm-2018-0037](https://doi.org/10.1515/abm-2018-0037)
CrossRef - Katrolia A, Chauhan SB. Formulation and evaluation of metformin hydrochloride-loaded curcumin–lycopene niosomes. .SN Applied Sciences.. 2019;1:1703. [https://doi.org/10.1007/s42452-019-1733-6](https://doi.org/10.1007/s42452-019-1733-6)
CrossRef - Saraswathi MK. Niosomes as an emerging formulation tool for drug delivery—A review. .International Journal of Applied Pharmaceutics.. 2019;11:7–15. [https://doi.org/10.22159/ijap.2019v11i4.32075](https://doi.org/10.22159/ijap.2019v11i4.32075)
CrossRef - Sudheer P. Review on niosomes—A novel approach for drug targeting. .Journal of Pharmaceutical Research.. 2015;14(1):20–25.
CrossRef - Saraswathi MK. Niosomes as an emerging formulation tool for drug delivery—A review. .International Journal of Applied
Pharmaceutics.. 2019;11:10–14. [https://doi.org/10.22159/ijap.2019v11i4.32075](https://doi.org/10.22159/ijap.2019v11i4.32075) - Ge X, He S. Advances of non-ionic surfactant vesicles (niosomes) and their application in drug delivery. .Pharmaceutics.. 2019;11(2):55. [https://doi.org/10.3390/pharmaceutics11020055](https://doi.org/10.3390/pharmaceutics11020055)
CrossRef - Lohumi A. A novel drug delivery system: Niosomes review. .Journal of Drug Delivery and Therapeutics.. 2012;2(5):274–281.
CrossRef - Gharbavi M, Amani J, Kheiri-Manjili H, Danafar H, Sharafi A. Niosome: A promising nanocarrier for natural drug delivery through the blood–brain barrier. .Advances in Pharmacological and Pharmaceutical Sciences.. 2018;2018:6847971. [https://doi.org/10.1155/2018/ 6847971](https://doi.org/10.1155/2018/6847971)
CrossRef - Seleci DA, Seleci M, Walter JG, Stahl F, Scheper T. Niosomes as nanoparticular drug carriers: Fundamentals and recent applications. .Journal of Nanomaterials.. 2016;2016:7372306. [https://doi.org/10.1155/2016/7372306](https://doi.org/10.1155/2016/7372306)
CrossRef - Chen S, Hanning S, Falconer J, Locke M, Wen J. Recent advances in non-ionic surfactant vesicles (niosomes): Fabrication, characterization, pharmaceutical and cosmetic applications. .European Journal of Pharmaceutics and Biopharmaceutics.. 2019;144:18–39. [https://doi.org/10.1016/j.ejpb.2019.08.015](https://doi.org/10.1016/j.ejpb.2019.08.015)
CrossRef - Chen S, Hanning S. Recent advances in non-ionic surfactant vesicles (niosomes): Fabrication, characterization, pharmaceutical and cosmetic applications. .European Journal of Pharmaceutics and Biopharmaceutics.. 2019;144:20–36.
CrossRef - Mittal S, Chaudhary A, Chaudhary A, Kumar A. Proniosomes: The effective and efficient drug-carrier system. .Therapeutic Delivery.. 2020;11(2):125–137. [https://doi.org/10.4155/tde-2019-0082](https://doi.org/10.4155/tde-2019-0082)
CrossRef - Javadzadeh Y, Hamishehkar H. Enhancing percutaneous delivery of methotrexate using different types of surfactants. .Colloids and Surfaces B: Biointerfaces.. 2011;82(2):422–426. [https://doi.org/10.1016/j.colsurfb.2010.10.005](https://doi.org/10.1016/ j.colsurfb. 2010.10.005)
CrossRef - Fang JY, Hong CT, Chiu WT, Wang YY. In vitro skin permeation of estradiol from various proniosome formulations. .International Journal of Pharmaceutics.. 2001;215(1-2):91–99. [https://doi.org/10.1016/S0378-5173(00)00669-1](https://doi.org/10.1016/S0378-517328002900669-1)
CrossRef - Junyaprasert VB, Teeranachaideekul V, Supaperm T. Influence of chemical penetration enhancers on skin permeability of ellagic acid-loaded niosomes. .Asian Journal of Pharmaceutical Sciences.. 2013;8(2):110–117. [https://doi.org/10.1016/j.ajps.2013.07.005](https://doi.org/10.1016/j.ajps.2013.07.005).
CrossRef - Gupta PN, Mishra V, Rawat A, Dubey P, Mahor S, Jain S, Chatterji DP, Vyas SP. Non-invasive vaccine delivery in transfersomes, niosomes and liposomes: A comparative study. International Journal of Pharmaceutics. 2005;293(1-2):73–82. [https://doi.org/10.1016/j.ijpharm.2004.12.022](https://doi.org/10.1016/j.ijpharm.2004.12.022)
CrossRef - Choi MJ, Maibach HI. Liposomes and niosomes as topical drug delivery systems. .Skin Pharmacology and Physiology.. 2005;18(5):209–219. [https://doi.org/10.1159/000086389](https://doi.org/10.1159/000086389)
CrossRef - Sambhakar S, Paliwal S. Formulation of risperidone loaded proniosomes for effective transdermal delivery: An in-vitro and in-vivo study. .Bulletin of Faculty of Pharmacy, Cairo University.. 2017;55(2):239–247. [https://doi.org/10.1016/j.bfopcu.2017.06.001](https://doi.org/10.1016/j.bfopcu.2017.06.001)
CrossRef - Maheshwari C, Dixit VK. Non-ionic surfactant vesicles mediated transcutaneous immunization against hepatitis B. .International Immunopharmacology.. 2011;11(10):1516–1522. [https://doi.org/10.1016/j.intimp.2011.05.004](https://doi.org/10.1016/ j.intimp.2011.05.004)
CrossRef - Kassem AA, Abd El-Alim SH, Asfour MH, Ahmed MB. Enhancement of 8-methoxypsoralen topical delivery via nanosized niosomal vesicles: Formulation development, in vitro and in vivo evaluation of skin deposition. .International Journal of Pharmaceutics.. 2017;517(1-2):256–268. [https://doi.org/10.1016/j.ijpharm.2016.12.005](https://doi.org/10.1016/j.ijpharm.2016.12.005)
CrossRef - Fang JY, Yu SY, Wu PC, Huang YB, Tsai YH. Effect of liposomes and niosomes on skin permeation of enoxacin. .International Journal of Pharmaceutics.. 2001;219(1-2):61–72. [https://doi.org/10.1016/S0378-5173(01)00616-8](https://doi.org/10.1016/S0378-5173%2801%2900616-8)
CrossRef - Shaker DS, Shaker MA, Hanafy MS. Nanoemulsion: A review on mechanisms for the transdermal delivery of hydrophobic and hydrophilic drugs. .Scientia Pharmaceutica.. 2019;87(3):17. [https://doi.org/10.3390/scipharm87030017](https://doi.org/10.3390/scipharm87030017)
CrossRef - El Maghraby GM, Williams AC, Barry BW. Can drug-bearing liposomes penetrate intact skin? .Journal of Pharmacy and Pharmacology.. 2006;58(4):415–429. [https://doi.org/10.1211/jpp.58.4.0001](https://doi.org/10.1211/jpp.58.4.0001)
CrossRef - Seleci DA, Seleci M, Walter JG, Stahl F, Scheper T. Niosomes as nanoparticular drug carriers: Fundamentals and recent applications. .Journal of Nanomaterials.. 2016;2016:7372306. [https://doi.org/10.1155/2016/7372306](https://doi.org/10.1155/2016/7372306)
CrossRef - Madhav NV, Saini A. Niosomes: A novel drug delivery system. .International Journal of Research in Pharmacy and Chemistry.. 2011;1(3):498–511.
- Jaydeep DY. Niosomes: A review. .Journal of Pharmaceutical Research.. 2011;4:632.
- Kalra N, Jain NK. Non-ionic surfactant vesicles and their therapeutic potentials. .Journal of Pharmaceutical Sciences.. 2016;3(2):193–201.
- Muzzalupo R, Tavano L. Niosomal drug delivery for transdermal targeting: Recent advances. .Research and Reports in Transdermal Drug Delivery.. 2015;4:23–33. [https://doi.org/10.2147/RRTD.S64773](https://doi.org/10.2147/RRTD.S64773)
CrossRef - Kumari R, Kumar S. Proniosomes: A key to improved drug delivery. .Journal of Drug Delivery and Therapeutics.. 2014;4(3):56–65.
CrossRef - Azeem A, Anwer MK, Talari MK, Jain GK, Talegaonkar S. Niosomes in sustained and targeted drug delivery: Recent advances. .Journal of Drug Targeting.. 2009;17(9):671–689. [https://doi.org/10.3109/10611860903048369](https://doi.org/10.3109/106118609 03048369)
CrossRef - Jothy MA, Shanmuganathan S, Nagalakshmi G. An overview on niosome as carrier in dermal drug delivery. .Journal of Pharmaceutical Sciences and Research.. 2015;7(11):923–927.
- Shirsand SB, Para MS, Nagendrakumar D, Kanani KM, Keerthy D. Recent advances in niosomal drug delivery—A review. .Research Journal of Life Sciences, Bioinformatics, Pharmaceutical and Chemical Sciences.. 2019;5(3):516–528.
- Arunachalam A, Karthikeyan M, Vinaykumar D, Prathap M, Sethuraman S, Ashutoshkumar S. Niosomes: A novel drug delivery system. .International Journal of Novel Trends in Pharmaceutical Sciences.. 2012;2(1):25–31.
CrossRef - Sanklecha VM, Pandey A. Review on niosomes. .Austin Pharmacology and Pharmaceutics.. 2018;3(2):1–7.
- Eimpunth S, Wanitphakdeedecha R, Manuskiatti W. A focused review on acne-induced and aesthetic procedure-related postinflammatory hyperpigmentation in Asians. .Journal of the European Academy of Dermatology and Venereology.. 2013;27:7–18. [https://doi.org/10.1111/j.1468-3083.2012.04594.x](https://doi.org/10.1111/j.1468-3083.2012.04594.x)
CrossRef - Searle T, Al-Niaimi F, Ali FR. The top 10 cosmeceuticals for facial hyperpigmentation. .Dermatologic Therapy.. 2020;33:e14095. [https://doi.org/10.1111/dth.14095](https://doi.org/10.1111/dth.14095).
CrossRef - Rajaratnam R, Halpern J, Salim A, Emmett C. Interventions for melasma. .Cochrane Database of Systematic Reviews.. 2010;(7):CD003583. [https://doi.org/10.1002/14651858.CD003583.pub2](https://doi.org/10.1002/14651858.CD003583.pub2)
CrossRef - Wang F, Li Y, Wang J. Review on oral plant extracts in skin whitening. .Food Science and Technology.. 2022;42:e83922. [https://doi.org/10.1590/fst.83922](https://doi.org/10.1590/fst.83922)
CrossRef - Souto EB, Ribeiro AF, Ferreira MI, Teixeira MC, Shimojo AAM, Lopes PS. Nanomaterials for skin delivery of cosmeceuticals and pharmaceuticals. .Applied Sciences.. 2020;10:1594. [https://doi.org/10.3390/app10051594](https://doi.org/10.3390/app10051594)
CrossRef - Kong M, Chen XG, Kweon DK, Park HJ. Investigations on skin permeation of hyaluronic acid-based nanoemulsion as transdermal carrier. .Carbohydrate Polymers.. 2011;86:837–843. [https://doi.org/10.1016/j.carbpol.2011.05.029](https://doi.org/10.1016/j.carbpol.2011.05.029)
CrossRef - Lieu AG, Fischer AH, Goldberg DJ. Melasma quality of life measures. .Dermatologic Clinics.. 2012;30:269–280. [https://doi.org/10.1016/j.det.2011.11.005](https://doi.org/10.1016/j.det.2011.11.005)
CrossRef - Izzo AA, Ernst E. Interactions between herbal medicines and prescribed drugs: An updated systematic review. .Phytotherapy Research.. 2016;30:691–700. [https://doi.org/10.1002/ptr.5591](https://doi.org/10.1002/ptr.5591)
CrossRef - Searle T, Al-Niaimi F, Ali FR. The top 10 cosmeceuticals for facial hyperpigmentation. .Dermatologic Therapy.. 2020;33(6):e14095. [https://doi.org/10.1111/dth.14095](https://doi.org/10.1111/dth.14095)
CrossRef - Bissett DL, Oblong JE, Berge CA. Niacinamide: A B vitamin that improves aging facial skin appearance. .Dermatologic Surgery.. 2005;31(7):860–865. [https://doi.org/10.1111/j.1524-4725.2005.31732](https://doi.org/10.1111/j.1524-4725.2005.31732).
CrossRef - Joshi SS, Boone SL, Alam M. Effectiveness, safety, and effect on quality of life of topical salicylic acid peels for treatment of postinflammatory hyperpigmentation in dark skin. .Dermatologic Surgery.. 2009;35:638–644. [https://doi.org/10.1111/j.1524-4725.2009.01107.x](https://doi.org/10.1111/j.1524-4725.2009.01107.x).
CrossRef
Accepted on: 06-03-2026
Second Review by: Dr. Shankar Yelmame
Final Approval by: Dr. Wagih Ghannam