Exploring the Health Benefits of Probiotics: Current Insights and Therapeutic Applications
Department of Microbiology, Sengamala Thayaar Educational Trust Women’s College (Autonomous), Affiliated to Bharathidasan University, Sundarakkottai, Mannargudi, Tamilnadu, India.
Corresponding Author E-mail: kannahiamf@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3485
ABSTRACT:In recent years, growing scientific attention has focused on the therapeutic potential of probiotics, particularly strains belonging to the genera Lactobacillus and Bifidobacterium, in promoting human health. They contribute to the modulation of gut microbiota and are widely used in functional foods. Emerging evidence indicates that probiotics play an important role in the prevention and management of gastrointestinal disorders, including inflammatory bowel disease, irritable bowel syndrome and ulcerative colitis. In addition, probiotics show promise in preventing obesity and in managing type 2 diabetes and cardiovascular diseases through the regulation of host metabolism and inflammatory responses. Probiotics are also implicated in the eradication of Helicobacter pylori and in reducing the risk of gastric ulcer development. This review summarizes findings from peer-reviewed articles published between 2014 and 2025, identified through major scientific databases, that investigate probiotics and their therapeutic applications. Overall, probiotics appear to be promising adjunctive therapeutic agents for gastrointestinal, metabolic and cardiovascular conditions. However, probiotic effects are highly strain-specific and are influenced by dosage, host factors and disease status. Although existing data are promising, further well-designed, large-scale clinical trials are required to establish their efficacy, safety and standardized therapeutic guidelines.
KEYWORDS:Bifidobacterium; Gut microbiota; Health benefits; Lactobacillus; Probiotics
Introduction
Overview of gut microbiota and its impact on human health
Researches have proved that the gut microbiome is crucial for human health, affecting the metabolism, nutrition, physiology and immunity. Dysbiosis, the disturbance of the gut microbiota balance, is one of the factors to provoke gastrointestinal issues like inflammatory bowel disease and irritable bowel syndrome.1-3 Additionally, dysbiosis is associated with multiple systemic diseases, such as obesity, diabetes mellitus type 2 and allergies. In recent years, the interest in rectifying the gastrointestinal health and preventing intestinal infections brought about by imbalanced diet and other lifestyle factors has been exponentially increasing.4 Administration of probiotics has been an efficient and new gateway to re-balancing of the instigated gut microbiota.5 An overview of the diverse health benefits associated with probiotic supplementation is depicted in Figure 1.
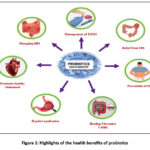 |
Figure 1: Highlights of the health benefits of probiotics
|
Evolution and definition of probiotics: Current perspectives
The definition of the term “probiotic” has evolved since it was first coined in 1965. Currently, most of the scientific and international community acknowledges the definition proposed by ISAPP in 2014. 6 Based on this definition, probiotics are “live microorganisms that when administered in sufficient quantities confer a health benefit to the host”.7 Recently various research on the human gut microbiota and evolution in analytical techniques have led to the emergence of new terms associated to the probiotic field such as next-generation probiotics, live biotherapeutic products, prebiotics, synbiotics, pharmabiotics, postbiotics and biogenics.8
Lactobacillus and Bifidobacterium: The leading probiotic strains
Among the lactic acid bacteria, the genus Lactobacillus and Bifidobacterium have also attracted much interest in the worldwide probiotic field.9 There is scientific evidence of the benefits of individual probiotic factors in various gastrointestinal diseases, including irritable bowel syndrome, the elimination of Helicobacter pylori, inflammatory bowel disease, diarrhea, gastrointestinal disorders and allergic conditions such as atopic dermatitis. Probiotics have potential uses for non-alcoholic fatty liver disease, obesity, insulin-resistant syndrome, type 2 diabetes, many types of cancer and they can also reduce the side effects of cancer treatment. Furthermore, probiotics play important roles in immune health, metabolic health, dental health and brain health. Besides, the possible role of probiotics as an adjunct therapy in the anagement of COVID-19 and its prevention is still under investigation.5,10 The beneficial effects of probiotics are strain-specific and further research is required to demonstrate the clinical benefits of each strain. Representative probiotic strains and their corresponding therapeutic targets are highlighted in Figure 2.
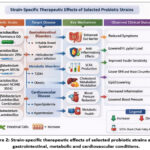 |
Figure 2: Strain-specific therapeutic effects of selected probiotic strains across gastrointestinal, metabolic and cardiovascular conditions.
|
This review has outlined the scientific foundation of using probiotics to prevent and manage several health problems. Table 1 categorizes probiotic strains according to disease indication, level of evidence and proposed mechanisms of action. The review further discussed the mechanisms of action of probiotics such as immune modulation, inflammation reduction, gut barrier enhancement and metabolic function improvement (Figure 3). Moreover, the discovery of probiotics remains pivotal in modern medical intervention.
Table 1: Strain-Specific Probiotic Evidence Summary
| Conditions |
Key strains
|
Evidence level | Primary mechanism |
| Ulcerative colitis | VSL#3, E. coli Nissle 1917 | Strong (RCTs = mesalazine) | Barrier repair, ↓NF-κB.52-60 |
| IBS | B. infantis,
L. plantarum 299v |
Moderate (symptom relief) | ↓Visceral hypersensitivity.70-78 |
| H. pylori | L. reuteri,
S. boulardii |
Strong (↑eradication 10-15%) | Urease inhibition, adhesion block.61-69 |
| T2DM | L. rhamnosus, B. longum | Emerging (↓HbA1c, ↑insulin sensitivity) | SCFA production, ↓inflammation.23-38
|
| Obesity | L. gasseri SBT2055 | Preliminary (↓BMI 0.5-1kg/m²) | Leptin/adiponectin modulation.39-51
|
| CVD | L. plantarum 299v | Preliminary (↓LDL, ↑HDL) | Bile salt hydrolase.11-22 |
| IBD | S. boulardii,
L. rhamnosus GG, E. coli Nissle |
Moderate-Strong (adjunct remission)
|
↓TNF-α/IL-6/IL-1β, tight junctions.79-91
|
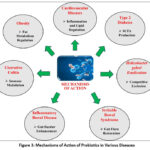 |
Figure 3: Mechanisms of Action of Probiotics in Various Diseases
|
Materials and Methods
This narrative review synthesized evidence on probiotic health benefits through a systematic literature search conducted in February 2026, covering peer-reviewed articles from January 2014 to February 2026 in PubMed/MEDLINE, Google Scholar, Web of Science, Scopus, ScienceDirect, Elsevier, MDPI, SciFinder and Scirus (with hand-searching of references); keywords included (“probiotic*” OR “Lactobacillus*” OR “Bifidobacterium*” OR “gut microbiota”) AND (“health benefit*” OR “therapeutic*” OR “gastrointestinal” OR “inflammatory bowel disease” OR “irritable bowel syndrome” OR “ulcerative colitis” OR “obesity” OR “type 2 diabetes” OR “cardiovascular” OR “Helicobacter pylori” OR “mechanism*”) using Boolean operators, no language limits. Inclusion focused on original research (in vitro/animal/human trials) or reviews demonstrating strain-specific effects on GI/metabolic/cardiovascular conditions or mechanisms (e.g., immune modulation, gut barrier); exclusions were pre-2014 studies (except seminal mechanism papers), non-peer-reviewed items and irrelevant topics. Two authors independently screened approximately 500 titles/abstracts, reviewed full texts, resolved discrepancies by consensus, and narratively extracted data from 82 high-quality sources prioritizing RCTs and recent high-impact publications, without formal PRISMA or quality scoring as per narrative review standards.
Health Attributes of Probiotics
Probiotics have demonstrated potential to support the management of and reduce risk factors for various conditions including cardiovascular disease (CVD), type 2 diabetes mellitus (T2DM), obesity, ulcerative colitis (UC), Helicobacter pylori infections, inflammatory bowel disease (IBD) and irritable bowel syndrome (IBS).
Probiotics in cardiovascular disease prevention
Probiotics have been investigated for their potential role in modulating certain cardiovascular risk factors; however, clinical evidence remains strain-specific and variable.11 In hypertension, Lactobacillus helveticus lowers blood pressure by producing bioactive peptides that inhibit angiotensin-converting enzyme (ACE), thereby reducing vasoconstriction and improving vascular tone.12 In atherosclerosis, Lactobacillus plantarum and Bifidobacterium longum promote bile acid deconjugation, enhance cholesterol excretion, improve lipid metabolism, and attenuate plaque development while suppressing oxidative stress–mediated vascular injury.13,14 In patients with stable coronary artery disease, supplementation with Lactobacillus plantarum 299v (1 × 10¹⁰ CFU/day) for 12 weeks improved endothelial function and reduced IL-6 and C-reactive protein levels.15 Similarly, in individuals undergoing coronary angiography, administration of L. plantarum 299v (2 × 10¹⁰ CFU/day) for six weeks significantly reduced systolic blood pressure, IL-8, IL-12, and leptin, supporting its potential role in secondary prevention.11 Lactobacillus acidophilus improves dyslipidemia by lowering LDL-cholesterol and triglycerides while increasing HDL-cholesterol.16,17 Emerging evidence suggests that probiotics may influence stroke outcomes through modulation of the gut–brain axis and neuroinflammation.18,19 In heart failure and myocardial infarction, probiotics may attenuate systemic inflammation, oxidative stress, trimethylamine-N-oxide (TMAO) production, and adverse cardiac remodeling.20-22
The role of probiotics in managing type 2 diabetes mellitus
Probiotics demonstrate therapeutic potential in type 2 diabetes mellitus (T2DM) by improving insulin sensitivity, lowering blood glucose, and reducing inflammation.23,24 Gut microbiota dysbiosis disrupts carbohydrate and lipid metabolism, contributing to insulin resistance and chronic inflammation in T2DM patients.25,26 Lactobacillus rhamnosus and Lactobacillus paracasei attenuate fasting blood glucose and enhance insulin sensitivity through gut microbiota remodulation and downregulation of pro-inflammatory cytokines (TNF-α, IL-6). 27,28 Bifidobacterium longum and Bifidobacterium bifidum improve glucose metabolism by increasing short-chain fatty acid (SCFA) production, particularly butyrate, thereby enhancing gut barrier integrity and reducing gut permeability and inflammation.29,30 Lactobacillus plantarum and Lactobacillus acidophilus decrease oxidative stress via antioxidant activity in the pancreas and liver, while Bifidobacterium animalis limits lipopolysaccharide (LPS)-induced endotoxemia.31-33 Probiotics also influence incretin hormones, gut–brain axis signaling, and glycemic regulation.34,35 Clinical trials support these mechanisms. A 12-week randomized study involving 150 patients showed that probiotics combined with metformin significantly reduced fasting blood glucose, postprandial glucose, and HbA1c, while lowering gastrointestinal side effects. Another 12-week trial in 58 patients demonstrated enhanced hypoglycemic response with multi-strain probiotics through modulation of gut microbiota, bile acid metabolism, and SCFA production.36 Meta-analyses of 15–30 randomized controlled trials (902–1827 patients) confirmed significant reductions in HbA1c, fasting glucose, insulin levels, and HOMA-IR.37 Collectively, probiotics represent a promising adjunct therapy that may also reduce complications such as diabetic retinopathy.38
Probiotics in obesity management
Probiotics play a significant role in obesity management by modulating gut microbiota composition, improving metabolic function, and suppressing chronic low-grade inflammation.39,40 Obesity-associated dysbiosis, characterized by an increased Firmicutes-to-Bacteroidetes ratio, has been linked to enhanced energy harvest and fat accumulation.41,42 Probiotic strains such as Lactobacillus gasseri, Lactobacillus plantarum, and Bifidobacterium longum contribute to body fat reduction through increased production of short-chain fatty acids, particularly butyrate, which promotes fatty acid oxidation, improves insulin sensitivity, and attenuates inflammation.43-45 Lactobacillus rhamnosus and Lactobacillus acidophilus further regulate lipid metabolism, reducing hepatic fat deposition.46,47 Probiotics also strengthen gut barrier integrity, limiting translocation of lipopolysaccharides into circulation and thereby decreasing metabolic endotoxemia and insulin resistance.48,49 Additionally, modulation of appetite-regulating hormones such as leptin and adiponectin supports improved satiety and adipose tissue regulation.50 Clinical trials and meta-analyses report modest but significant reductions in body weight and body mass index, particularly with multi-strain formulations administered for more than eight weeks in overweight and obese adults. Although findings remain strain-specific and duration-dependent, probiotics represent a promising adjunct strategy for improving metabolic health and weight management.51
Probiotics in the management of ulcerative colitis
Probiotics such as Escherichia coli Nissle 1917 (EcN), Lactobacillus spp. (Lactobacillus acidophilus, Lacticaseibacillus casei), and Bifidobacterium spp. (Bifidobacterium infantis) demonstrate therapeutic potential in ulcerative colitis (UC) by restoring gut microbiota balance.52 Patients with UC-associated dysbiosis promotes intestinal inflammation and epithelial barrier dysfunction.53 Probiotics strengthen barrier integrity by enhancing mucin secretion and tight junction protein expression, thereby limiting pathogen translocation.54,55 They modulate immune responses through inhibition of the NF-κB pathway, reducing pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) and increasing anti-inflammatory cytokines (IL-10, TGF-β), thus attenuating chronic inflammation.56 Additionally, probiotics increase short-chain fatty acid production, particularly butyrate, which nourishes colonocytes and promotes mucosal healing.57 Clinical trials support these effects. A pediatric study demonstrated that EcN effectively maintains remission in young UC patients.58 Evidence indicates that active UC responds favorably to EcN (Mutaflor) and the multi-strain formulation VSL#3, comprising Lacticaseibacillus casei, Lactobacillus plantarum, Lactobacillus acidophilus, Lactobacillus delbrueckii subsp. bulgaricus, Bifidobacterium longum, Bifidobacterium breve, Bifidobacterium infantis, and Streptococcus salivarius subsp. thermophilus. VSL#3 induces clinical remission and symptom improvement without increased adverse effects; notably, over 50% of mild-to-moderate UC patients unresponsive to conventional therapy achieved remission after six weeks. Similarly, EcN is as effective and safe as mesalazine for maintaining remission and is endorsed in ECCO guidelines as an evidence-based probiotic for adults and children.59 Although probiotics alleviate symptoms and maintain remission, they are most effective as adjuncts within a comprehensive treatment strategy rather than as monotherapy.60 Further long-term studies are needed to confirm sustained efficacy and safety.
Helicobacter pylori eradication via probiotic modulation of gastric gut microbiota
Probiotics, including Lactobacillus acidophilus, Lactobacillus reuteri, Bifidobacterium lactis, Saccharomyces boulardii, and Lactiplantibacillus plantarum, enhance Helicobacter pylori eradication rates and reduce treatment side effects when used as adjuncts to standard antibiotic therapy through improved gut health and immune modulation.61,62 They compete with H. pylori for gastric mucosal adhesion sites, produce antimicrobial metabolites such as lactic acid, short-chain fatty acids, and hydrogen peroxide, and suppress urease activity, thereby impairing bacterial survival in the acidic gastric environment.63-65 Probiotics also enhance mucosal immunity by increasing IgA production, reinforcing barrier integrity, and reducing pro-inflammatory cytokines including IL-6 and TNF-α.66,67 Furthermore, they restore microbiota balance disrupted by infection or antibiotic therapy.68 Clinical studies in children and adults demonstrate that multi-strain formulations such as Lacidofil® significantly increase eradication rates, frequently achieving ≥90% compared with 70–86% for standard therapy alone, while reducing gastroduodenal inflammation and antibiotic-associated adverse effects. Adjunctive probiotic supplementation is also associated with improved gastrointestinal tolerance and faster symptom resolution, supporting its role as an effective complementary strategy in H. pylori management and long-term gastric health maintenance.69
Probiotics in treating irritable bowel syndrome (IBS)
Probiotics are increasingly recognized for their therapeutic potential in irritable bowel syndrome (IBS), a condition associated with gut microbiota dysbiosis.70 Restoration of microbial balance through strains such as Bifidobacterium infantis, Lactobacillus plantarum, Lactobacillus rhamnosus, and Escherichia coli Nissle 1917 has shown clinical benefit.71 Probiotics reduce pathogenic bacteria, enhance microbial diversity, and strengthen intestinal barrier integrity by improving epithelial function and lowering gut permeability. They promote short-chain fatty acid production, particularly butyrate, supporting mucosal integrity.72,73 Additionally, probiotics exert immunomodulatory effects by reducing pro-inflammatory cytokines, modulating gastrointestinal motility, and decreasing visceral hypersensitivity through actions on opioid and cannabinoid receptors.74-76 Clinical evidence supports these mechanisms. A 4-week randomized, double-blind, placebo-controlled trial in Vietnamese patients with unconstipated IBS reported significantly higher overall symptom improvement (80.8% vs. 45.8%, p = 0.009) and abdominal pain reduction (69.2% vs. 41.7%, p = 0.048) with multi-strain Lactobacillus (1 × 10⁹ CFU/day) compared to placebo, without adverse events.77 Similarly, a 16-week trial in IBS-D patients demonstrated significant reductions in abdominal pain (69% vs. 47%, p < 0.001) and improved IBS severity and quality of life with a 14-strain probiotic (8 × 10⁹ CFU, twice daily).78
Probiotics in treating inflammatory bowel disease (IBD)
Probiotics show therapeutic promise in the management of IBD, including Crohn’s disease (CD) and ulcerative colitis (UC).79 IBD is characterized by chronic gastrointestinal inflammation associated with dysbiosis, impaired gut barrier integrity, and increased permeability.80 Specific strains such as Saccharomyces boulardii, Escherichia coli Nissle 1917, Lactobacillus rhamnosus, and Faecalibacterium prausnitzii help restore microbial balance, suppress inflammation, and support epithelial integrity.81,82 S. boulardii reduces recurrent inflammation following surgical diversion, while E. coli Nissle 1917 and L. rhamnosus upregulate tight junction proteins, strengthening barrier function.83-86 Probiotics inhibit pro-inflammatory cytokines (TNF-α, IL-6, IL-1β), produce antimicrobial substances such as lactic acid and hydrogen peroxide, and stimulate aryl hydrocarbon receptors (AhRs), thereby promoting anti-inflammatory immune modulation.87-89 Clinical evidence supports these mechanisms. A 2019 UK study reported that a multi-strain probiotic significantly reduced intestinal inflammation in ulcerative colitis but showed no comparable benefit in Crohn’s disease, indicating strain- and disease-specific effects. Similarly, a 2019 study from China found that adjunct probiotic therapy alongside standard treatment improved gut microbiota composition and reduced pro-inflammatory cytokines compared with conventional therapy alone.90 In addition, findings from other clinical trials indicate that probiotics have been investigated as adjunctive therapy in inflammatory bowel disease to prevent treatment-associated dysbiosis during prolonged antibiotic or immunosuppressive therapy and to help restore microbial balance in newly diagnosed patients or during disease exacerbations.91
Conclusion
The health benefits of probiotics have been widely investigated and accumulating evidence suggests that certain strains may contribute to the management of digestive conditions such as inflammatory bowel disease (IBD), irritable bowel syndrome (IBS), ulcerative colitis and Helicobacter pylori infection. Emerging studies have also explored their potential roles in obesity, type 2 diabetes and cardiovascular diseases, although the strength of evidence varies across conditions. Probiotics exert their effects through mechanisms including competitive exclusion of pathogens, enhancement of mucosal immunity, production of antimicrobial compounds and modulation of inflammatory responses. However, probiotic efficacy remains highly strain-specific and influenced by dosage, host characteristics and disease status. Further well-designed, large-scale human clinical trials are required to determine optimal strains, dosages, long-term safety and standardized therapeutic recommendations. In addition, clearer regulatory frameworks are necessary to support evidence-based health claims and ensure safe application in medical and food industries.
Acknowledgement
The authors acknowledge Sengamala Thayaar Educational Trust Women’s College (Autonomous), Sundarakkottai, Mannargudi, Tamilnadu, India.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from other Sources
Not Applicable
Author Contributions
Sarumathi Chandrasekaran: developed the review outline, conducted the literature search and drafted the manuscript.
Kannahi Manoharan: contributed to the literature search, critically revised the manuscript and performed the final editing.
References
- Paul J K, Azmal M, Haque A S N B, Meem M, Talukder O F, Ghosh A. Unlocking the secrets of the human gut microbiota: comprehensive review on its role in different diseases. World J Gastroenterol. 2025; 31(5): 99913. https://doi.org/10.3748/wjg.v31.i5.99913
CrossRef - Renukadevi J, Helinto J S, Prena D. Immunotherapeutic potential of lactobacillus species as immune checkpoint inhibitors in cancer immunotherapy. J Bio-X Res. 2025; 8(01): 1-7. https://doi. org/10.34133/jbioxresearch.0028
CrossRef - Renukadevi J, Ranjani M, Sneha P, Hussain J S, Shakthi H. Gut‐targeted nutraceutical delivery: engineering microbiome‐responsive nutraceutical interfaces. Food Bioeng. 2025; 4(3): 248-61. https://doi.org/10.1002/fbe2.70024
CrossRef - Shen Y, Fan N, Ma S, Cheng X, Yang X, Wang G. Gut microbiota dysbiosis: pathogenesis, diseases, prevention, and therapy. 2025; 6(5): e70168. https://doi.org/10.1002/mco2.70168
CrossRef - Manan M A. The role of probiotics in personalized therapeutics: Advances in gut microbe-driven interventions. The Microbe. 2025; 8: 100497. https://doi.org/10.1016/j.microb.2025.100497
CrossRef - Lau L Y, Quek S Y. Probiotics: Health benefits, food application, and colonization in the human gastrointestinal tract. Food Bioeng. 2024; 3(1): 41-64.https://doi.org/10.1002/fbe2.12078
CrossRef - Latif A, Shehzad A, Niazi S, et al. Probiotics: mechanism of action, health benefits and their application in food industries. Microbiol. 2023; 14. https://doi.org/10.3389/fmicb.2023.1216674
CrossRef - Abouelela M E, Helmy Y A. Next-Generation Probiotics as Novel Therapeutics for Improving Human Health: Current Trends and Future Perspectives. Microorganisms. 2024; 12(3): 430. https://doi.org/10.3390/microorganisms12030430
CrossRef - Linares D M, Gómez C, Renes E, et al. Lactic Acid Bacteria and Bifidobacteria with Potential to Design Natural Biofunctional Health-Promoting Dairy Foods. Microbiol. 2017; 8. https://doi.org/10.3389/fmicb.2017.00846
CrossRef - Maftei N M, Raileanu C R, Balta A A, et al. The Potential Impact of Probiotics on Human Health: An Update on Their Health-Promoting Properties. Microorganisms. 2024; 12(2): 234. https://doi.org/10.3390/microorganisms12020234
CrossRef - López-Yerena A, De Santisteban Villaplana V, Badimon L, Vilahur G, Padro T. Probiotics: a potential strategy for preventing and managing cardiovascular disease. Nutrients. 2024; 17(1): https://doi.org/10.3390/nu17010052
CrossRef - Friques A G F, Arpini C M, Kalil I C, et al. Chronic administration of the probiotic kefir improves the endothelial function in spontaneously hypertensive rats. Transl. Med. 2015; 13(1). https://doi.org/10.1186/s12967-015-0759-7
CrossRef - Abdi M, Ghaleh H E G, Ranjbar R. Lactobacilli and Bifidobacterium as anti-atherosclerotic agents. DOAJ. 2022; 25(8): 934–946. https://doi.org/10.22038/ijbms.2022.63860.14073
- Dong Y, Xu R, Chen X, et al. Characterization of gut microbiota in adults with coronary atherosclerosis. PeerJ. 2023; 11: e15245.
CrossRef - Pavlidou E, Fasoulas A, Mantzorou M, Giaginis C. Clinical Evidence on the Potential Beneficial Effects of Probiotics and Prebiotics in Cardiovascular Disease. J. Mol. Sci. 2022; 23(24): 15898. https://doi.org/10.3390/ijms232415898
CrossRef - Gadelha C J M U, Bezerra A N. Effects of probiotics on the lipid profile: systematic review. J Vasc Bras. 2019; 18: e20180124. https://doi.org/10.1590/1677-5449.180124
CrossRef - Tsai C, Lin P, Hsieh Y, Zhang Z, Wu H, Huang C. Cholesterol-lowering potentials of lactic acid bacteria based on bile-salt hydrolase activity and effect of potent strains on cholesterol metabolism in vitro and in vivo. Sci World J. 2014; 2014: 690752. https://doi.org/10.1155/2014/690752
CrossRef - Chen L, Wang X, Wang S, Liu W, Song Z, Liao H. The impact of gut microbiota on the occurrence, treatment, and prognosis of ischemic stroke. Dis. 2025: 106836. https://doi.org/10.1016/j.nbd.2025.106836
CrossRef - Renukadevi J, Velmurugan R, Nandhinidevi G, Karthikha VS, Vaishnavi VS. Exploring the neuroprotective role of probiotics in the therapeutic interventions of cognitive decline. Appl. Pharm. Res. 2024; 12(5): 1-5.https://doi.org/10.69857/joapr.v12i5.648
CrossRef - Jia Q, Li H, Zhou H, et al. Role and effective therapeutic target of gut microbiota in heart failure. Ther. 2019; 2019: 1–10. https://doi.org/10.1155/2019/5164298
CrossRef - Nurgaziyev M, Kozhakhmetov S, Jarmukhanov Z, et al. Impact of probiotics and polyphenols on adults with heart failure: a systematic review and meta-analysis. Eur J Med Res. 2025; 30(1): e2538.https://doi.org/10.1186/s40001-025-02538-y
CrossRef - Xu H, Wang J, Cai J, et al. Protective effect of Lactobacillus rhamnosus GG and its supernatant against myocardial dysfunction in obese mice exposed to intermittent hypoxia is associated with activation of the Nrf2 pathway. Int J Biol Sci. 2019; 15(11): 2471–2483. https://doi.org/10.7150/ijbs.36465
CrossRef - Ayesha I E, Monson N R, Klair N, et al. Probiotics and their Role in the Management of Type 2 Diabetes mellitus (Short-Term versus Long-Term Effect): A Systematic Review and Meta-Analysis. Cureus. 2023; 15(10): e46741. https://doi.org/10.7759/cureus.46741
CrossRef - Li S, Liu Z, Zhang Q, et al. The Antidiabetic Potential of probiotics: A review. Nutrients. 2024; 16(15): 2494. https://doi.org/10.3390/nu16152494
CrossRef - Portincasa P, Khalil M, Graziani A, et al. Gut microbes in metabolic disturbances: promising role for therapeutic manipulations? Eur J Intern Med. 2023; 119: 13–30.https://doi.org/10.1016/j.ejim.2023.10.002
CrossRef - Yu Y, Ding Y, Wang S, Jiang L. Gut microbiota dysbiosis and its impact on type 2 diabetes: from pathogenesis to therapeutic strategies. 2025; 15(6): 397.https://doi.org/10.3390/metabo15060397
CrossRef - Farida E, Nuraida L, Giriwono P E, Jenie, B S L. Lactobacillus rhamnosus Reduces Blood Glucose Level through Downregulation of Gluconeogenesis Gene Expression in Streptozotocin-Induced Diabetic Rats. J. Food Sci. 2020; 2020: 1–12. https://doi.org/10.1155/2020/6108575
CrossRef - Zeng Z, Guo X, Zhang J, Yuan Q, Chen S. Lactobacillus paracasei modulates the gut microbiota and improves inflammation in type 2 diabetic rats. Food Funct. 2021; 12(15): 6809–6820.
CrossRef - Le T K C, Hosaka T, Le T T T, et al. Oral administration of Bifidobacterium improves insulin resistance, induces adiponectin, and prevents inflammatory adipokine expressions. Biomed. Res. 2014; 35(5): 303–310. https://doi.org/10.2220/biomedres.35.303
CrossRef - Zhao D, Zhu H, Gao F, et al. Antidiabetic effects of selenium-enriched Bifidobacterium longum DD98 in type 2 diabetes model of mice. Food Funct. 2020; 11(7): 6528–6541. https://doi.org/10.1039/d0fo00180e
CrossRef - Lee Y S, Lee D, Park G S, et al. Lactobacillus plantarum HAC01 ameliorates type 2 diabetes in high-fat diet and streptozotocin-induced diabetic mice in association with modulating the gut microbiota. Food Funct. 2021; 12(14): 6363-6373.
CrossRef - Pegah A, Abbasi-Oshaghi E, Khodadadi I, Mirzaei F, Tayebinia H. Probiotic and resveratrol normalize GLP-1 levels and oxidative stress in the intestine of diabetic rats. Metab Open. 2021; 10: 100093. https://doi.org/10.1016/j.metop.2021.100093.
CrossRef - Du X, Liu M, Li J, et al. Bifidobacterium animalisSupplementation Improves Intestinal Barrier Function and Alleviates Antibiotic-Associated Diarrhea in Mice. Foods. 2025; 14(10): 1704. https://doi.org/10.3390/foods14101704
CrossRef - Simon M, Strassburger K, Nowotny B, et al. Intake of Lactobacillus reuteri improves incretin and insulin secretion in glucose-tolerant humans: a proof of concept. Diabetes Care. 2015; 38(10): 1827–1834. https://doi.org/10.2337/dc14-2690
CrossRef - Toejing P, Khampithum N, Sirilun S, Chaiyasut C, Lailerd N. Influence of Lactobacillus paracasei HII01 supplementation on glycemia and inflammatory biomarkers in type 2 diabetes: a randomized clinical trial. 2021; 10(7): 1455. https://doi.org/10.3390/foods10071455
CrossRef - Ayesha I E, Monson N R, Klair N, et al. Probiotics and Their Role in the Management of Type 2 Diabetes Mellitus (Short-Term Versus Long-Term Effect): A Systematic Review and Meta-Analysis. Cureus. 2023; 15(10): e46741. https://doi.org/10.7759/cureus.46741
CrossRef - Nikolaidis C G, Gyriki D, Stavropoulou E, et al. Modulating the Gut Microbiome in Type 2 Diabetes: Nutritional and Therapeutic Strategies. Nutrients. 2026; 18(1): 89. https://doi.org/10.3390/nu18010089
Croseef - Viana M D M, Santos S S, Cruz A B O, et al. Probiotics as antioxidant strategy for managing diabetes mellitus and its complications. 2025; 14(7): 767. https://doi.org/10.3390/antiox14070767
CrossRef - Geng J, Ni Q, Sun W, Li L, Feng X. The links between gut microbiota and obesity and obesity related diseases. Pharmacother. 2022; 147: 112678.
CrossRef - Chen X, You L, Jia Y. The role of probiotics in adolescents’ obesity. Cell. Infect. Microbiol. 2025; 15: 1546627. https://doi.org/10.3389/fcimb.2025.1546627
CrossRef - Liu B, Liu X, Liang Z, Wang J. Gut microbiota in obesity. World J. Gastroenterol. 2021; 27(25): 3837–3850. https://doi.org/10.3748/wjg.v27.i25.3837
CrossRef - Augustynowicz G, Lasocka M, Szyller H P, et al. The role of gut microbiota in the development and treatment of obesity and Overweight: a literature review. Clin. Med. 2025; 14(14): 4933. https://doi.org/10.3390/jcm14144933
CrossRef - Shirouchi B, Nagao K, Umegatani M, et al. Probiotic Lactobacillus gasseri SBT2055 improves glucose tolerance and reduces body weight gain in rats by stimulating energy expenditure. Br J Nutr. 2016; 116(3): 451–458.https://doi.org/10.1017/s0007114516002245
CrossRef - Schellekens H, Torres-Fuentes C, Van De Wouw M, et al. Bifidobacterium longum counters the effects of obesity: partial successful translation from rodent to human. 2020; 63: 103176. https://doi.org/10.1016/j.ebiom.2020.103176
CrossRef - Jang A, Jung D, Lee T, et al. Lactobacillus plantarum NCHBL-004 modulates high-fat diet–induced weight gain and enhances GLP-1 production for blood glucose regulation. Nutrition. 2024; 128: 112565. https://doi.org/10.1016/j.nut.2024.112565
CrossRef - Park S S, Lee Y J, Song S, et al. Lactobacillus acidophilus NS1 attenuates diet-induced obesity and fatty liver. J Endocrinol. 2018; 237(2): 87–100.
CrosRef - Sun M, Wu T, Zhang G, et al. Lactobacillus rhamnosus LRa05 improves lipid accumulation in mice fed with a high fat diet via regulating the intestinal microbiota, reducing glucose content and promoting liver carbohydrate metabolism. Food Funct. 2020; 11(11): 9514–9525.https://doi.org/10.1039/d0fo01720e
CrossRef - Shang H, Sun J, Chen Y Q. Clostridium butyricum1 modulates lipid profile, insulin resistance and colon homeostasis in obese mice. PLoS One. 2016; 11(4): e0154373. https://doi.org/10.1371/journal.pone.0154373
CrossRef - Mercado-Monroy J, Falfán-Cortés R N, Muñóz-Pérez V M, Gómez-Aldapa C A, Castro-Rosas J. Probiotics as modulators of intestinal barrier integrity and immune homeostasis: a comprehensive review. J Sci Food Agric.
CrossRef - Ülker İ, Yıldıran H, Aktürk M Y. Probiotics’ effect on nutritional status, appetite hormones, and inflammation in pre-obese women. J Adv Res Health Sci.https://doi.org/10.26650/jarhs2024-1347787
CrossRef - Wiciński M, Gębalski J, Gołębiewski J, Malinowski B. Probiotics for the Treatment of Overweight and Obesity in Humans—A Review of Clinical Trials. Microorganisms. 2020; 8(8): 1148. https://doi.org/10.3390/microorganisms8081148
CrossRef - Huang C, Hao W, Wang X, Zhou R, Lin Q. Probiotics for the treatment of ulcerative colitis: a review of experimental research from 2018 to 2022. Microbiol. 2023; 14: 1211271. https://doi.org/10.3389/fmicb.2023.1211271
CrossRef - Shen Z, Zhu C, Quan Y, et al. Relationship between intestinal microbiota and ulcerative colitis: mechanisms and clinical application of probiotics and fecal microbiota transplantation. World J Gastroenterol. 2018; 24(1): 5–14. https://doi.org/10.3748/wjg.v24.i1.5
CrossRef - Gou H Z, Zhang Y L, Ren L F, Li Z J, Zhang L. How do intestinal probiotics restore the intestinal barrier? Microbiol. 2022; 13: 929346.
CrossRef - Ma X Y, Son Y H, Yoo J W, Joo M K, Kim D H. Tight junction protein expression-inducing probiotics alleviate TNBS-induced cognitive impairment with colitis in mice. Nutrients. 2022; 14(14): 2975.
CrossRef - Wu Y, Jha R, Li A, et al. Probiotics (Lactobacillus plantarum HNU082) supplementation relieves ulcerative colitis by affecting intestinal barrier functions, immunity-related gene expression, gut microbiota, and metabolic pathways in mice. Microbiol Spectr. 2022; 10(6): e0165122.https://doi.org/10.1128/spectrum.01651-22
CrossRef - Markowiak-Kopeć P, Śliżewska K. The effect of probiotics on the production of short-chain fatty acids by human intestinal microbiome. 2020; 12(4): 1107.https://doi.org/10.3390/nu12041107
CrossRef - Hijová E. Probiotics in IBD: Evidence and Perspectives on Patient Health and Disease Management. J. Mol. Sci. 2025; 26(18): 9065. https://doi.org/10.3390/ijms26189065
CrossRef - Štofilová J, Kvaková M, Kamlárová A, Hijová E, Bertková I, Guľašová Z. Probiotic-Based Intervention in the Treatment of Ulcerative Colitis: Conventional and New Approaches. Biomedicines. 2022; 10(9): 2236.https://doi.org/10.3390/biomedicines10092236
CrossRef - Dziedzic A, Saluk J. Probiotics and Commensal Gut Microbiota as the Effective Alternative Therapy for Multiple Sclerosis Patients Treatment. J. Mol. Sci. 2022; 23(22): 14478. https://doi.org/10.3390/ijms232214478
CrossRef - Tukaram K S, Sopanrao S R, Kacharu L O. Unveiling the therapeutic potential of probiotics: a review. J Future Foods.https://doi.org/10.1016/j.jfutfo.2025.09.011.
CrossRef - Renukadevi J, Sneha P, Mridula DS, Nimithasree K, Sanjay V. Bioengineered Isoflavone–Probiotic Functional Foods for Sustainable Modulation of Metabolic and Reproductive Health in PCOS. Food Bioeng. 2026.https://doi.org/10.1002/fbe2.70044.
CrossRef - Monteagudo-Mera A, Rastall R A, Gibson G R, Charalampopoulos D, Chatzifragkou A. Adhesion mechanisms mediated by probiotics and prebiotics and their potential impact on human health. Appl Microbiol Biotechnol. 2019; 103(16): 6463–6472. https://doi.org/10.1007/s00253-019-09978-7
CrossRef - Yuan L, Yang C, Han Y, Yang F, Tu H. Beyond antibiotics: probiotics as a promising ally against Helicobacter pylori. Pharmacol. 2025; 16:1620870.https://doi.org/10.3389/fphar.2025.1620870
CrossRef - Midolo P D, Lambert J R, Hull R, Luo F, Grayson M L. In vitro inhibition of Helicobacter pylori NCTC 11637 by organic acids and lactic acid bacteria. J Appl Bacteriol. 1995; 79(4): 475–479.
CrossRef - Wu S, Xu Y, Chen Z, et al. Lactiplantibacillus plantarum ZJ316 reduces Helicobacter pylori adhesion and inflammation by inhibiting the expression of adhesin and urease genes. Mol Nutr Food Res. 2023; 67(18): 2300241.
CrossRef - Gebremariam H G, Qazi K R, Somiah T, et al. Lactobacillus gasseri Suppresses the Production of Proinflammatory Cytokines in Helicobacter pylori-Infected Macrophages by Inhibiting the Expression of ADAM17. Immunol. 2019; 10: 2326. https://doi.org/10.3389/fimmu.2019.02326
CrossRef - Dash D, Mishra V, Panda M K, Pathak S K. Effects of Lactobacillus on Helicobacter pylori: a promising frontier in the era of antibiotic resistance. Probiotics Antimicrob Proteins. 2024: 1-20.
CrossRef - Auclair-Ouellet N, Tremblay A, Kassem O, Caballero-Calero S E, Bronner S, Binda S. Probiotics as Adjuvants to Standard Helicobacter pylori Treatment: Evidence for the Use of Lacidofil®, an Established Blend of Thoroughly Characterized Strains. Microorganisms. 2025; 13(10): 2223.https://doi.org/10.3390/microorganisms13102223
CrossRef - Gawron A J, Shah S C, Altayar O, et al. AGA Technical Review on Gastric Intestinal Metaplasia—Natural History and Clinical Outcomes. Gastroenterology. 2020; 158(3): 705-731.https://doi.org/10.1053/j.gastro.2019.12.001
CrossRef - Umeano L, Iftikhar S, Alhaddad S F, et al. Effectiveness of probiotic use in alleviating symptoms of irritable bowel syndrome: a systematic review. 2024; 16(4): e58306. https://doi.org/10.7759/cureus.58306
CrossRef - Dudek-Wicher R, Junka A, Paleczny J, Bartoszewicz M. Clinical trials of probiotic strains in selected disease entities. J. Microbiol. 2020; 2020: 1–8. https://doi.org/10.1155/2020/8854119
CrossRef - Jiang W, Wu J, Zhu S, Xin L, Yu C, Shen Z. The role of short chain fatty acids in irritable bowel syndrome. J Neurogastroenterol Motil. 2022; 28(4): 540–548. https://doi.org/10.5056/jnm22093
CrossRef - Satish Kumar L, Pugalenthi L S, Ahmad M, Reddy S, Barkhane Z, Elmadi J. Probiotics in Irritable Bowel Syndrome: A Review of Their Therapeutic Role. Cureus. 2022; 14(4): e24240. https://doi.org/10.7759/cureus.24240
CrossRef - Dimidi E, Christodoulides S, Scott S M, Whelan, K. Mechanisms of action of probiotics and the gastrointestinal microbiota on gut motility and constipation. Nutr. 2017; 8(3): 484–494. https://doi.org/10.3945/an.116.014407
CrossRef - Horvat I B, Gobin I, Kresović A, Hauser G. How can probiotic improve irritable bowel syndrome symptoms? World J. Gastrointest. Surg. 2021; 13(9): 923–940. https://doi.org/10.4240/wjgs.v13.i9.923
CrossRef - Oh J H, Jang Y S, Kang D, Chang D K, Min Y W. Efficacy and Safety of New Lactobacilli Probiotics for Unconstipated Irritable Bowel Syndrome: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2019; 11(12): 2887. https://doi.org/10.3390/nu11122887
CrossRef - Ishaque S M, Khosruzzaman S M, Ahmed D S, Sah M P. A randomized placebo-controlled clinical trial of a multi-strain probiotic formulation (Bio-Kult(R)) in the management of diarrhea-predominant irritable bowel syndrome. BMC Gastroenterol.2018; 18(1): 71. https://doi.org/10.1186/s12876-018-0788-9
CrossRef - Zhang J, Zhang X, Cheng X, et al. Probiotics in inflammatory bowel diseases: emphasis on mechanisms and clinical application. Front Med. 2025; 12: 1620079. https://doi.org/10.3389/fmed.2025.1620079
CrossRef - Shan Y, Lee M, Chang E B. The gut microbiome and inflammatory bowel diseases. Annu Rev Med. 2021; 73: 455–468. https://doi.org/10.1146/annurev-med-042320-021020.
CrossRef - Wang X, Zhang P, Zhang X. Probiotics regulate gut microbiota: an effective method to improve immunity. 2021; 26(19): 6076. https://doi.org/10.3390/molecules26196076.
CrossRef - Roy S, Dhaneshwar S. Role of prebiotics, probiotics, and synbiotics in management of inflammatory bowel disease: current perspectives. World J Gastroenterol. 2023; 29(14): 2078–2100. https://doi.org/10.3748/wjg.v29.i14.2078
CrossRef - Gao H, Li Y, Sun J, et al. Saccharomyces boulardiiAmeliorates Dextran Sulfate Sodium-Induced Ulcerative Colitis in Mice by Regulating NF-κB and Nrf2 Signaling Pathways. Oxid Med Cell Longev. 2021; 2021: 1622375. https://doi.org/10.1155/2021/1622375
CrossRef - Sivananthan K, Petersen A M. Review of Saccharomyces boulardii as a treatment option in IBD. Immunopharmacol Immunotoxicol. 2018; 40(6): 465–475. https://doi.org/10.1080/08923973.2018.1469143
CrossRef - Han X, Lee A, Huang S, Gao J, Spence J R, Owyang C. Lactobacillus rhamnosus GG prevents epithelial barrier dysfunction induced by interferon-gamma and fecal supernatants from irritable bowel syndrome patients in human intestinal enteroids and colonoids. Gut Microbes. 2018; 10(1): 59–76. https://doi.org/10.1080/19490976.2018.1479625
CrossRef - Guo S, Chen S, Ma J, et al. Escherichia coli Nissle 1917 Protects Intestinal Barrier Function by Inhibiting NF-κB-Mediated Activation of the MLCK-P-MLC Signaling Pathway. Mediators Inflamm. 2019; 2019: 1–13. https://doi.org/10.1155/2019/5796491.
CrossRef - Kumar M, Hemalatha R, Nagpal R, et al. Probiotic approaches for targeting inflammatory bowel disease: an update on advances and opportunities in managing the disease. J. Probiotic Prebiotics. 2016; 11(3-4): 99.
- Ahire J J, Jakkamsetty C, Kashikar M S, Lakshmi S G, Madempudi R S. In Vitro Evaluation of Probiotic Properties of Lactobacillus plantarum UBLP40 Isolated from Traditional Indigenous Fermented Food. Probiotics Antimicrob Proteins. 2021; 13(5): 1413–1424. https://doi.org/10.1007/s12602-021-09775-7
CrossRef - Huang Z, Xie L, Huang L. Regulation of host immune responses by Lactobacillus through aryl hydrocarbon receptors. Microecol. 2023; 16: 100081.https://doi.org/10.1016/j.medmic.2023.100081.
CrossRef - Mitra A K, Asala A F, Malone S, Mridha M K. Effects of probiotics in adults with gastroenteritis: a systematic review and meta-analysis of clinical trials. Diseases. 2023; 11(4): 138. https://doi.org/10.3390/diseases11040138.
CrossRef - Basso P J, Câmara N O, Sales-Campos H. Microbial-based therapies in the treatment of inflammatory bowel disease–an overview of human studies. Pharmacol. 2019; 9: 1571. | https://doi.org/10.3389/fphar.2018.01571.
CrossRef
Accepted on: 05-03-2026
Second Review by: Dr. J.Renuka Devi
Final Approval by: Dr. Ali Elshafei









