Exploring Neuroprotective Phytochemicals from Mucuna pruriens and Ginkgo biloba for Alzheimer’s Disease: A Molecular Docking and ADME Study
Department of Pharmaceutical Chemistry, Pravara Rural College of Pharmacy Pravaranagar, Tal-Rahata, Ahmednagar, Maharashtra, India.
Corresponding Author E-mail: rohit.bhor69@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3500
ABSTRACT:Alzheimer’s disease (AD) is a progressive neurodegenerative disorder characterized by cognitive decline, memory impairment, and neuronal loss, for which current therapies offer only symptomatic relief. In this context, the present study investigates the neuroprotective potential of selected phytochemical constituents from Mucuna pruriens (Kapikachu) and Ginkgo biloba using an integrated in silico approach involving molecular docking, physicochemical profiling, and ADME analysis. Molecular docking studies were performed against two key AD-related targets, acetylcholinesterase (AChE; PDB ID: 1EVE) and β-secretase (BACE-1; PDB ID: 1TQF), to evaluate binding affinity and interaction patterns. The results demonstrated that phytochemicals from Ginkgo biloba, particularly biflavonoids such as ginkgetin, bilobetin, and sciadopitysin, exhibited significantly stronger binding affinities toward both targets compared to constituents of Mucuna pruriens. In contrast, Kapikachu-derived compounds showed moderate interactions, suggesting a supportive role in cholinergic modulation and neuroprotection rather than direct enzyme inhibition. Overall, the integration of docking performance with pharmacokinetic evaluation highlights Ginkgo biloba as a promising source of multitarget lead compounds for Alzheimer’s disease therapy, while reinforcing the complementary neuroprotective value of Mucuna pruriens. The findings provide a strong theoretical basis for further experimental validation and lead optimization.
KEYWORDS:Alzheimer’s disease; ADME analysis; Ginkgo biloba; Molecular docking; Mucuna pruriens
Introduction
Ginkgo biloba, commonly known as maidenhair tree, is one of the oldest living tree species and has been extensively used in traditional Chinese medicine for the management of cognitive and neurological disorders. Standardized extracts of Ginkgo biloba, particularly EGb 761®, are rich in flavonoids (quercetin, kaempferol, isorhamnetin) and terpene lactones (ginkgolides and bilobalide), which contribute to its neuroprotective properties. In the context of Alzheimer’s disease, Ginkgo biloba has been widely investigated for its antioxidant, anti-inflammatory, anti-amyloidogenic, and cholinergic-modulating activities. The flavonoid fraction scavenges reactive oxygen species and reduces lipid peroxidation, thereby protecting neuronal membranes from oxidative stress–induced damage,1 a key pathological feature of neurodegeneration. Additionally, terpene lactones such as bilobalide have been reported to enhance mitochondrial function, improve cerebral blood flow, and protect against β-amyloid–induced neurotoxicity. Kapikachu (Mucuna pruriens (L.) DC.), a member of the family Fabaceae, was recognized as an important medicinal plant widely used in traditional systems of medicine, particularly Ayurveda. The plant was characterized as a tropical climbing legume,2 and its seeds were identified as the primary pharmacologically active part. Kapikachu was traditionally employed in the management of neurological disorders, reproductive dysfunction, and general debility. Phytochemical investigations revealed that Kapikachu seeds were rich in L-3,4-dihydroxyphenylalanine (L-DOPA), which constituted the major bioactive compound responsible for its neuropharmacological effects. In addition to L-DOPA,3 the plant was reported to contain alkaloids (mucunine, mucunadine), flavonoids, tannins, saponins, sterols, and phenolic compounds, contributing to its broad spectrum of biological activities. Pharmacological studies demonstrated that Kapikachu exhibited significant anti-Parkinsonian, neuroprotective, antioxidant, anti-inflammatory, aphrodisiac,4 and antidepressant activities3. From a pharmacokinetic perspective,5 Kapikachu-derived L-DOPA was reported to show favorable gastrointestinal absorption and central nervous system availability. However, proper processing of seeds was emphasized to eliminate irritant compounds and ensure safety. Overall, Kapikachu was established as a scientifically validated medicinal plant with strong relevance to neurodegenerative disease research. Its rich phytochemical composition and traditional therapeutic use supported its selection as a promising natural candidate for molecular docking, ADME evaluation, and further drug discovery studies targeting neurological disorders. Alzheimer’s disease (AD) is a progressive neurodegenerative disorder,6 the most common cause of dementia, characterized by gradual memory loss, cognitive impairment, and behavioral disturbances. Pathologically, AD is associated with extracellular accumulation of amyloid-β (Aβ) plaques, intracellular neurofibrillary tangles composed of hyperphosphorylated tau protein, synaptic dysfunction, and neuronal loss. Despite extensive research, current therapeutic options provide only symptomatic relief and fail to halt or reverse disease progression, highlighting the urgent need for novel and effective treatment strategies. At the molecular level, several enzymes and receptors play critical roles in the pathogenesis of Alzheimer’s disease, including acetylcholinesterase (AChE),7 β-site amyloid precursor protein cleaving enzyme 1 (BACE1), glycogen synthase kinase-3β (GSK-3β),8 and N-methyl-D-aspartate (NMDA) receptors.9 Modulation or inhibition of these targets is considered a promising approach for reducing amyloid deposition, preventing tau hyperphosphorylation, and restoring cholinergic neurotransmission. Therefore, identifying molecules capable of interacting effectively with these targets is a central focus of AD drug discovery. Molecular docking has emerged as a powerful in silico technique in the early stages of drug discovery,10 allowing prediction of the binding orientation, interaction pattern, and binding affinity of small molecules within the active site of target proteins. This computational approach provides valuable insights into ligand–protein interactions at the atomic level,11 thereby reducing time, cost, and reliance on extensive experimental screening. Docking studies are particularly advantageous in Alzheimer’s research,12 where multiple molecular targets and complex disease mechanisms necessitate rational and multi-target-based drug design strategies.
Mechanism of Action of Kapikachu and Ginkgo biloba in Alzheimer’s Disease
The neuroprotective mechanism of Mucuna pruriens in Alzheimer’s disease was primarily attributed to its rich content of L-3,4-dihydroxyphenylalanine (L-DOPA) and associated antioxidant phytoconstituents. Alzheimer’s disease is characterized by cholinergic dysfunction,13 oxidative stress, β-amyloid plaque deposition, neuroinflammation, and progressive neuronal degeneration. Mucuna pruriens was reported to modulate several of these pathological pathways. The presence of L-DOPA was shown to enhance dopaminergic neurotransmission,14 which indirectly supported cognitive and behavioral functions impaired during neurodegeneration. In addition, antioxidant compounds present in the plant scavenged reactive oxygen species and reduced lipid peroxidation, thereby protecting neuronal membranes from oxidative damage,15 a key contributor to Alzheimer’s pathology. Experimental studies indicated that Mucuna pruriens reduced neuroinflammation by downregulating pro-inflammatory mediators, which contributed to the preservation of neuronal integrity. The plant extract was also suggested to inhibit acetylcholinesterase activity,16 leading to increased acetylcholine availability in synaptic clefts and improvement of cholinergic signaling, a central therapeutic target in Alzheimer’s disease. Overall, Mucuna pruriens was found to exert a multitarget neuroprotective effect in Alzheimer’s disease by modulating oxidative stress, neurotransmitter imbalance, neuroinflammation,17 and cholinergic dysfunction, supporting its potential role as a natural therapeutic candidate for Alzheimer’s disease management and molecular docking–based drug discovery studies.
Materials and Methods
Preparation of protein and in-silico molecular docking
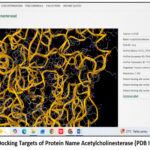 |
Figure 1(a): Docking Targets of Protein Name Acetylcholinesterase (PDB ID: 1eve).
|
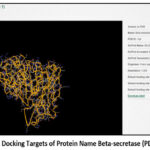 |
Figure 1(b): Docking Targets of Protein Name Beta-secretase (PDB ID: 1tqf)
|
The above image illustrates the three-dimensional crystal structure of acetylcholinesterase (AChE) with the Protein Data Bank (PDB) ID: 1EVE, given in Figure 1(a) and Figure 1(b), which was used as a molecular docking target in Alzheimer’s disease research. The protein structure was displayed in a ribbon representation,18 highlighting the complex folding pattern composed of α-helices and loops that formed the active and peripheral binding sites of the enzyme. The protein was annotated in UniProt as ACHE_TORCA,19 with the accession number P04058 and taxonomic ID 7787. The crystal structure was resolved at a resolution of 2.50 Å,20 providing detailed insight into the enzyme’s three-dimensional conformation. The default binding site was identified within the active-site gorge of the enzyme and was located at the coordinates X = 2.861, Y = 6.432, and Z = 7.892.21-22 This structural information was used as a docking target for evaluating potential acetylcholinesterase inhibitors in Alzheimer’s disease research.
Preparation of molecules
Levodopa consisted of a catechol-substituted aromatic ring linked to an amino acid side chain, and it was recorded in DrugBank under the ID DB00173. Lecithin (phosphatidylcholine) was identified as a glycerophospholipid composed of a glycerol backbone esterified with long-chain fatty acids and a choline phosphate head group,23 and it was listed in DrugBank with the ID DB00163. Glutathione was characterized as a tripeptide composed of glutamic acid, cysteine,20 and glycine containing a reactive thiol group, and its DrugBank identification number was DB00143. Oleic acid was observed as a monounsaturated long-chain fatty acid with a cis double bond and a terminal carboxylic acid group, and it was registered in DrugBank as DB00125. Tyrosine was described as an aromatic amino acid bearing a phenolic hydroxyl group attached to a benzene ring, and its DrugBank ID was DB00159. Valine was found to be a branched-chain aliphatic amino acid with an isopropyl side chain,24 and it was indexed in DrugBank under the ID DB00165. Serotonin possessed an indole ring system with a hydroxyl group and an ethylamine side chain, and it was documented in DrugBank with the ID DB00260. Linoleic acid was identified as a polyunsaturated fatty acid containing two cis double bonds within an 18-carbon chain, and it was assigned the DrugBank ID DB00146. Phytochemical Constituents of Kapikachu (Mucuna pruriens (L.) DC.) and Ginkgo biloba were given in Figure 2(a) and Figure 2(b).
Drug likeness characters and ADME/toxicity analysis
Drug likeness and ADME/toxicity analysis were performed for the selected molecules. The structure of the selected molecules was screened to predict whether the molecules obey Lipinski’s rule of five. The physicochemical properties of the selected molecules were predicted and the analysis was performed using ADMET lab 2.0 programme https://www.swissadme.ch/. The pharmacodynamics properties such as, Caco-2 permeability, mutagenicity, carcinogenicity, Cytochrome (CYP) inhibitory capability, human intestinal absorption, and blood brain barrier permeability were analysed.
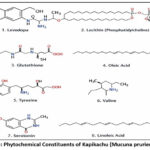 |
Figure 2(a): Phytochemical Constituents of Kapikachu (Mucuna pruriens (L.) DC.).
|
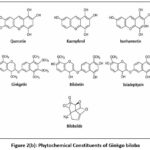 |
Figure 2(b): Phytochemical Constituents of Ginkgo biloba.
|
Results
Physicochemical Properties of Selected Phytochemical Constituents
The physicochemical properties of the selected phytochemical constituents were compared based on hydrogen-bonding capacity, molecular refractivity (MR), topological polar surface area (TPSA), and lipophilicity descriptors. Levodopa exhibited a relatively high number of hydrogen bond acceptors (5) and donors (4) along with a moderate TPSA (103.78 Ų), indicating good polar interaction potential but low lipophilicity, as reflected by its negative tom-additive Calculated Logarithm of the Octanol/Water Partition Coefficient (−2.74). Glutathione showed the highest hydrogen-bonding capability with 7 acceptors and 5 donors and possessed the largest TPSA (197.62 Ų), suggesting strong hydrophilicity and limited membrane permeability. In contrast, Lecithin demonstrated a markedly higher molecular refractivity (218.47) and strong lipophilicity, with elevated intrinsic Logarithm of the Partition Coefficient (octanol/water) is (4.58) and tom-additive Calculated Logarithm of the Octanol/Water Partition Coefficient (13) values, indicating its bulky and highly hydrophobic nature. Oleic acid and linoleic acid displayed low TPSA values (37.3 Ų) and minimal hydrogen-bonding capacity, while their high lipophilicity suggested favorable membrane permeability but reduced aqueous solubility. Among the amino acids, tyrosine and valine showed moderate hydrogen-bonding properties and lower MR values, with negative XLOGP3 values indicating hydrophilic behavior. Serotonin exhibited balanced properties with moderate MR, TPSA, and lipophilicity, suggesting a favorable compromise between polarity and membrane permeability mentions in Table 1 and graph of TPSA in Figure 3.
In the present study, the physicochemical parameters (#H-bond acceptors, #H-bond donors, MR, TPSA, iLOGP, and XLOGP3) were generated using in silico prediction tools and were compared descriptively between phytochemicals of Mucuna pruriens and Ginkgo biloba. No inferential statistical test (e.g., t-test or ANOVA) was initially applied because these values represent deterministic computational outputs rather than experimentally replicated measurements. To assess the pharmacokinetic relevance of the selected phytochemical constituents, a rule-based comparison was performed using established drug-likeness and oral bioavailability criteria rather than statistical testing. According to Lipinski’s rule of five, acceptable compounds should possess ≤5 hydrogen bond donors, ≤10 hydrogen bond acceptors, and a LogP value ≤5. Additionally, the Veber rule suggests that a topological polar surface area (TPSA) ≤140 Ų is associated with favorable oral bioavailability, while compounds with TPSA <90 Ų are generally considered more suitable for blood–brain barrier (BBB) penetration, which is particularly relevant in Alzheimer’s disease therapy. Within this framework, glutathione (TPSA 197.62 Ų) markedly exceeded the Veber threshold, suggesting limited membrane permeability and poor oral absorption potential despite its strong hydrogen-bonding capacity. Similarly, the biflavonoids ginkgetin and bilobetin exhibited TPSA values greater than 150 Ų, indicating restricted passive BBB penetration, although their strong docking affinities may compensate through high target binding efficiency. In contrast, oleic acid (TPSA 37.3 Ų) satisfied Veber and BBB criteria, implying favorable membrane permeability; however, its elevated lipophilicity (high LogP) may present challenges related to solubility and metabolic stability. Levodopa (TPSA 103.78 Ų) demonstrated borderline compliance with CNS permeability guidelines, which is consistent with its known ability to cross the BBB via active transport mechanisms rather than passive diffusion.
Table 1: Phytochemical constituent’s comparison table for Kapikachu and Ginkgo biloba including #H-bond acceptors, #H-bond donors, MR, TPSA, iLOGP and XLOGP3
| Phytochemical Constituents | #H-bond acceptors | #H-bond donors | MR | TPSA | iLOGP | XLOGP3 |
| Kapikachu (Mucuna pruriens (L.) DC.), | ||||||
| Levodopa | 5 | 4 | 49.55 | 103.78 | 0.72 | -2.74 |
| Lecithin | 8 | 0 | 218.47 | 121 | 4.58 | 13 |
| Glutathione | 7 | 5 | 70.37 | 197.62 | 0.32 | -4.5 |
| Oleic Acid – | 2 | 1 | 89.94 | 37.3 | 4.01 | 7.64 |
| Tyrosine | 4 | 3 | 47.52 | 83.55 | 0.84 | -2.26 |
| Valine | 3 | 2 | 30.63 | 63.32 | 1.03 | -2.26 |
| Serotonin | 2 | 3 | 52.8 | 62.04 | 1.18 | 1.49 |
| Linoleic Acid | 2 | 1 | 89.46 | 37.3 | 1.18 | 1.49 |
| Ginkgo biloba | ||||||
| Quercetin | 7 | 5 | 78.03 | 131.36 | 1.63 | 1.54 |
| Kaempfol | 6 | 4 | 76.01 | 111.13 | 1.7 | 1.9 |
| Isorhamnetin | 7 | 4 | 82.5 | 120.36 | 2.35 | 1.87 |
| Ginkgetin | 10 | 4 | 155.91 | 159.8 | 3.94 | 5.69 |
| Bilobetin | 10 | 5 | 151.44 | 170.8 | 3.39 | 5.36 |
| Sciadopitysin | 10 | 3 | 160.38 | 148.8 | 4.65 | 6.02 |
| Bilobalide | 8 | 2 | 71.2 | 119.36 | 0.81 | -0.27 |
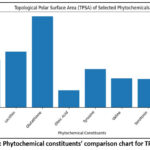 |
Figure 3: Phytochemical constituents’ comparison chart for TPSA data
|
ADME and CYP Inhibition Profile
The ADME-related properties of the selected phytochemical constituents were comparatively evaluated in terms of gastrointestinal (GI) absorption, blood–brain barrier (BBB) permeability, P-glycoprotein (P-gp) substrate behavior, and cytochrome P450 (CYP) enzyme inhibition. Levodopa exhibited high GI absorption and BBB permeability while showing no P-gp substrate liability and no inhibitory effect on the major CYP isoforms, suggesting a favorable pharmacokinetic profile. In contrast, lecithin demonstrated low GI absorption but was BBB permeant and acted as a P-gp substrate, with inhibitory activity against CYP2C9 and CYP3A4, indicating a higher potential for drug–drug interactions. Glutathione showed low GI absorption despite being BBB permeant and did not inhibit any of the evaluated CYP enzymes, reflecting a limited interaction profile. Oleic acid exhibited high GI absorption and BBB permeability and was identified as a CYP1A2 and CYP2C9 inhibitor, suggesting possible metabolic interaction risks. Tyrosine and valine both displayed high GI absorption but lacked BBB permeability and CYP inhibition, indicating good absorption with minimal metabolic interference. Serotonin and linoleic acid showed high GI absorption and BBB permeability without P-gp substrate behavior or CYP inhibition, suggesting balanced pharmacokinetic characteristics. Overall, the comparative analysis highlighted distinct differences among the constituents, particularly in GI absorption, BBB permeation, and CYP inhibition potential given in Table 2, which may influence their biological activity and suitability as therapeutic candidates.
Table 2: Phytochemical constituents’ comparison table for Kapikachu (Mucuna pruriens) and Ginkgo biloba, including GI absorption, BBB permeant, Pgp substrate, CYP1A2 inhibitor, CYP2C19 inhibitor, CYP2C9 inhibitor, CYP2D6 inhibitor, and CYP3A4 inhibitor
| Phytochemical
Constituents |
GI absorption | BBB permeant | Pgp substrate | CYP1A2 inhibitor | CYP2C19 inhibitor | CYP2C9 inhibitor | CYP2D6 inhibitor | CYP3A4 inhibitor |
| Kapikachu (Mucuna pruriens (L.) DC.), | ||||||||
| Levodopa | High | Yes | No | No | No | No | No | No |
| Lecithin | Low | Yes | Yes | No | No | Yes | No | Yes |
| Glutathione | Low | Yes | No | No | No | No | No | No |
| Oleic Acid – | High | Yes | No | Yes | No | Yes | No | No |
| Tyrosine | High | No | No | No | No | No | No | No |
| Valine | High | No | No | No | No | No | No | No |
| Serotonin | High | Yes | No | No | No | No | No | No |
| Linoleic Acid | High | Yes | No | No | No | No | No | No |
| Ginkgo biloba | ||||||||
| Quercetin | High | No | No | Yes | No | No | Yes | Yes |
| Kaempfol | High | No | No | Yes | No | No | Yes | Yes |
| Isorhamnetin | High | No | No | Yes | No | No | Yes | Yes |
| Ginkgetin | Low | No | No | No | No | Yes | No | No |
| Bilobetin | Low | No | No | No | No | Yes | No | No |
| Sciadopitysin | Low | No | No | No | No | No | No | No |
| Bilobalide | High | No | Yes | No | No | No | No | No |
Drug-Likeness and Oral Bioavailability Assessment:
The drug-likeness properties of the selected phytochemical constituents were comparatively analysed using Lipinski, Ghose, Veber, Egan, and Muegge filters along with predicted bioavailability scores. Levodopa complied with Lipinski, Ghose, Veber, and Egan criteria without violations, although it showed two Muegge violations, while maintaining a moderate bioavailability score of 0.55. Lecithin exhibited multiple violations across Ghose (four) and Muegge (three) filters, along with single violations in Lipinski, Veber, and Egan rules, indicating poor drug-likeness despite a moderate bioavailability score. Glutathione demonstrated acceptable compliance with Lipinski rules but showed violations in Ghose, Veber, Egan, and Muegge filters, which was reflected in its notably low bioavailability score of 0.11. Oleic acid showed one violation each in Lipinski, Ghose, Veber, Egan, and Muegge criteria and displayed the highest bioavailability score (0.85), suggesting favorable absorption characteristics despite partial rule non-compliance. Tyrosine and serotonin fully satisfied Lipinski, Ghose, Veber, and Egan rules, with limited Muegge violations, and both maintained moderate bioavailability scores of 0.55. Valine complied with Lipinski, Veber, and Egan criteria but showed multiple Ghose and Muegge violations, although its predicted bioavailability remained moderate. Linoleic acid exhibited complete compliance with Lipinski, Ghose, Veber, and Egan rules and only a single Muegge violation, accompanied by a bioavailability score of 0.55. Overall, the comparative evaluation highlighted variability in Table 3 including drug-likeness and bioavailability among the phytochemicals, with oleic acid showing the most favorable bioavailability and glutathione the least favorable profile.
Table 3: Phytochemical constituents’ comparison table for Kapikachu (Mucuna pruriens) and Ginkgo biloba, including Lipinski #violations; Ghose #violations; Veber #violations; Egan #violations; Muegge #violations, and Bioavailability Score
| Phytochemical
Constituents |
Lipinski #violations | Ghose #violations | Veber #violations | Egan #violations | Muegge #violations | Bioavailability Score |
| Kapikachu (Mucuna pruriens (L.) DC.), | ||||||
| Levodopa | 0 | 0 | 0 | 0 | 2 | 0.55 |
| Lecithin | 1 | 4 | 1 | 1 | 3 | 0.55 |
| Glutathione | 0 | 1 | 2 | 1 | 2 | 0.11 |
| Oleic Acid – | 1 | 1 | 1 | 1 | 1 | 0.85 |
| Tyrosine | 0 | 0 | 0 | 0 | 2 | 0.55 |
| Valine | 0 | 3 | 0 | 0 | 2 | 0.55 |
| Serotonin | 0 | 0 | 0 | 0 | 1 | 0.55 |
| Linoleic Acid | 0 | 0 | 0 | 0 | 1 | 0.55 |
| Ginkgo biloba | ||||||
| Quercetin | 0 | 0 | 0 | 0 | 0 | 0.55 |
| Kaempfol | 0 | 0 | 0 | 0 | 0 | 0.55 |
| Isorhamnetin | 0 | 0 | 0 | 0 | 0 | 0.55 |
| Ginkgetin | 1 | 3 | 1 | 1 | 2 | 0.55 |
| Bilobetin | 1 | 2 | 1 | 1 | 2 | 0.55 |
| Sciadopitysin | 1 | 3 | 1 | 2 | 1 | 0.55 |
| Bilobalide | 0 | 1 | 0 | 0 | 0 | 0.55 |
Molecular docking results:
The molecular docking results revealed distinct differences in binding affinities between the phytochemical constituents of Kapikachu and Ginkgo biloba against the Alzheimer’s disease–related targets acetylcholinesterase (AChE; PDB ID: 1EVE) and β-secretase (BACE-1; PDB ID: 1TQF). Among the Kapikachu-derived compounds, levodopa, valine, serotonin, and linoleic acid demonstrated moderate binding affinities toward AChE, with docking scores ranging from −7.0 to −7.1 kcal/mol. In contrast, their interaction with β-secretase was comparatively weaker, showing scores between −3.6 and −6.9 kcal/mol. Lecithin exhibited the lowest affinity for both targets, particularly against β-secretase (−3.6 kcal/mol). Overall, Kapikachu phytoconstituents displayed moderate and relatively uniform interactions, suggesting limited target specificity, especially toward β-secretase. In comparison, Ginkgo biloba constituents showed significantly stronger binding affinities toward both enzymes. Ginkgetin emerged as the most potent inhibitor, with docking scores of −14.3 kcal/mol (AChE) and −9.8 kcal/mol (β-secretase), followed closely by bilobetin and sciadopitysin. Flavonoids such as kaempferol and isorhamnetin also exhibited strong AChE inhibition, with scores of −10.1 and −9.5 kcal/mol, respectively. Bilobalide, although showing strong AChE interaction (−11.8 kcal/mol), displayed comparatively weaker binding toward β-secretase (−6.2 kcal/mol), indicating possible selectivity toward AChE. Comparative analysis clearly indicates that Ginkgo biloba phytochemicals possess superior binding affinities toward both AChE and β-secretase when compared to Kapikachu constituents. The markedly lower docking scores of biflavonoids such as ginkgetin, bilobetin, and sciadopitysin suggest their potential as dual-target inhibitors, which is a desirable feature in Alzheimer’s disease therapy. In contrast, Kapikachu compounds may contribute supportive or adjunctive effects rather than strong enzymatic inhibition. The docking results highlight Ginkgo biloba, particularly its biflavonoid constituents, as a more promising source of lead compounds for Alzheimer’s disease management through simultaneous inhibition of AChE and β-secretase. These findings support further ADME, molecular dynamics, and in vitro validation studies to confirm their therapeutic potential. Phytochemical constituent’s comparison table for Kapikachu and Ginkgo biloba including Lipinski #violations; Ghose #violations; Veber #violations; Egan #violations; Muegge #violations, and Bioavailability Score were given in Table 4.
Table 4: Phytochemical constituents’ comparison table by 1eve (Acetylcholinesterase) and 1tqf (Beta Secretase) for Kapikachu and Ginkgo biloba
| Sr.No. | Phytochemical Constituents | 1eve (Acetylcholinesterase) | 1tqf
(Beta Secretase) |
| Mucuna pruriens | |||
| 1 | Levodopa- | -7.1 | -6.9 |
| 2 | Lecithin | -5.7 | -3.6 |
| 3 | Glutathione | -6.7 | -6.7 |
| 4 | Oleic Acid | -6.7 | -6.1 |
| 5 | Tyrosine | -7.0 | -6.6 |
| 6 | Valine | -7.1 | -4.7 |
| 7 | Serotonin | -7.1 | -6.5 |
| 8 | Linoleic Acid | -7.1 | -5.5 |
| Ginkgo biloba | |||
| 1 | Quercetin | -7.0 | -7.4 |
| 2 | Kaempfol | -10.1 | -7.3 |
| 3 | Isorhamnetin | -9.5 | -7.4 |
| 4 | Ginkgetin | -14.3 | -9.8 |
| 5 | Bilobetin | -12.0 | -10.1 |
| 6 | Sciadopitysin | -11.8 | -9.8 |
| 7 | Bilobalide | -11.8 | -6.2 |
Discussion
The present study provides a comparative in silico evaluation of phytochemical constituents from Mucuna pruriens and Ginkgo biloba against two major therapeutic targets implicated in Alzheimer’s disease—acetylcholinesterase (AChE) and β-secretase (BACE-1). When interpreted in the context of previously published literature, the findings demonstrate strong agreement with established pharmacological and computational evidence supporting the multitarget potential of Ginkgo-derived flavonoids, while reinforcing the supportive neuroprotective role of Mucuna phytochemicals. The docking results revealed that biflavonoids from Ginkgo biloba, particularly ginkgetin, bilobetin, and sciadopitysin, exhibited markedly stronger binding affinities toward both AChE and BACE-1 compared to Kapikachu-derived compounds. The observed docking score of −14.3 kcal/mol for ginkgetin against AChE is notably lower than the typical docking range reported for flavonoid–AChE interactions (−8 to −11 kcal/mol) in earlier computational studies. Previous reports have demonstrated that Ginkgo flavonoids exert significant anti-cholinesterase and antioxidant effects, contributing to cognitive enhancement in dementia models. Clinical and experimental evaluations of standardized Ginkgo extracts (EGb 761®) have shown improvements in memory and cognitive performance, primarily through modulation of cholinergic transmission and reduction of oxidative stress. The present docking outcomes align with these findings by demonstrating strong theoretical stabilization of ligand–enzyme complexes within the catalytic gorge of AChE. The superior binding performance of biflavonoids can be explained through structure–activity relationship principles. Their extended aromatic systems enable π–π stacking interactions with aromatic residues in the active site, while multiple hydroxyl and methoxy groups facilitate hydrogen bonding. Such interaction networks increase binding stability and reduce calculated binding energy. Similar trends have been reported in previous molecular docking studies targeting BACE-1, where polyphenolic compounds with higher molecular rigidity and aromatic surface area exhibited enhanced inhibitory potential. The ability of ginkgetin and bilobetin to demonstrate strong docking scores against both AChE and BACE-1 suggests possible dual-target inhibition, which is increasingly considered a promising strategy given the multifactorial pathogenesis of Alzheimer’s disease. In contrast, phytochemicals from Mucuna pruriens, including levodopa, serotonin, and tyrosine, demonstrated moderate docking affinities clustered around −7.0 kcal/mol for AChE and weaker interactions for BACE-1. These results are consistent with earlier pharmacological studies indicating that Mucuna primarily exerts neuroprotective and dopaminergic effects rather than strong anti-amyloid or anti-secretase activity. The therapeutic relevance of Mucuna has been well documented in Parkinson’s disease due to its natural L-DOPA content; however, its role in Alzheimer’s disease appears to be more supportive, focusing on antioxidant activity, neurotransmitter modulation, and reduction of oxidative stress. Therefore, the moderate docking results observed in the present study align with its established pharmacodynamic profile. The ADME and drug-likeness analysis further differentiates the two plant sources. While Ginkgo biflavonoids exhibited strong binding affinities, some compounds showed predicted cytochrome P450 inhibition, which is consistent with earlier reports of potential herb–drug interactions associated with Ginkgo extracts. Conversely, levodopa demonstrated favorable gastrointestinal absorption and blood–brain barrier permeability without significant CYP inhibition, corroborating its known clinical pharmacokinetic profile. This contrast highlights an important translational consideration: strong docking affinity does not always equate to optimal pharmacokinetic suitability. Interestingly, although certain compounds such as oleic acid displayed high predicted bioavailability, their docking scores were comparatively moderate, emphasizing the importance of integrating binding affinity with pharmacokinetic predictions. This integrative evaluation supports the modern drug discovery paradigm, where multitarget interaction and ADME balance are both essential for therapeutic success. When compared with existing experimental and computational literature, the present findings substantiate that Ginkgo biloba phytochemicals possess stronger theoretical multitarget inhibitory potential against Alzheimer’s-related enzymes, whereas Mucuna pruriens contributes complementary neuroprotective mechanisms. These results reinforce the concept that plant-based therapies may offer synergistic or adjunctive benefits in neurodegenerative disease management. Future validation through molecular dynamics simulations, in vitro enzyme inhibition assays, and in vivo cognitive models will be necessary to confirm the translational relevance of these computational predictions.
 |
Table 5: Phytochemical constituents’ comparison of 3D images table by 1eve (Acetylcholinesterase) and 1tqf (Beta Secretase) for Kapikachu and Ginkgo biloba generated by 1 Click Docking Software
|
The present study provides a comprehensive in silico comparison of selected phytochemical constituents from Mucuna pruriens (Kapikachu) and Ginkgo biloba against two key Alzheimer’s disease–associated targets, acetylcholinesterase (AChE) and β-secretase (BACE-1). By integrating molecular docking results with physicochemical profiling, ADME prediction, and drug-likeness evaluation, the study offers theoretical insights into the multitarget therapeutic potential and pharmacokinetic feasibility of these natural compounds. Molecular docking analysis revealed clear differences in binding behavior between Kapikachu and Ginkgo biloba phytoconstituents. Compounds derived from Mucuna pruriens, including levodopa, serotonin, valine, tyrosine, and linoleic acid, demonstrated moderate binding affinities toward AChE, with docking scores clustered around −7.0 kcal/mol. These interactions suggest a supportive inhibitory potential against cholinergic dysfunction, which is a hallmark of Alzheimer’s disease. However, their comparatively weaker binding toward BACE-1 indicates limited efficacy in directly modulating amyloidogenic pathways. This observation aligns with the known pharmacological role of Kapikachu, where neuroprotection and neurotransmitter modulation are more prominent than direct enzyme inhibition. In contrast, Ginkgo biloba phytochemicals exhibited markedly stronger binding affinities toward both AChE and BACE-1. Biflavonoids such as ginkgetin, bilobetin, and sciadopitysin emerged as the most potent ligands, demonstrating exceptionally low docking scores for both targets. The enhanced binding efficiency of these compounds can be attributed to their structural complexity, extended aromatic systems, and abundance of hydrogen bond donors and acceptors, which collectively enable multiple stabilizing interactions within the enzyme active sites. The ability of these molecules to interact simultaneously with both cholinergic and amyloidogenic targets supports their potential role as dual-target inhibitors, a highly desirable strategy in Alzheimer’s disease management. The physicochemical analysis further supports these findings by highlighting structural features that influence binding and bioavailability. Compounds with balanced molecular weight, moderate topological polar surface area, and optimal lipophilicity exhibited more favorable docking behavior. Excessively polar molecules, such as glutathione, despite their strong hydrogen-bonding capacity, displayed limited docking efficiency and poor predicted oral bioavailability. Conversely, highly lipophilic molecules such as lecithin showed suboptimal docking scores and multiple drug-likeness violations, suggesting limited translational potential despite membrane permeability advantages. ADME and cytochrome P450 inhibition profiling provided additional layers of differentiation among the compounds. Kapikachu constituents such as levodopa and serotonin exhibited favorable gastrointestinal absorption, blood–brain barrier permeability, and minimal CYP inhibition, indicating a lower risk of metabolic drug–drug interactions. These properties support their potential use as adjunctive neuroprotective agents rather than primary enzyme inhibitors. On the other hand, several Ginkgo biloba flavonoids demonstrated CYP inhibitory activity, particularly against CYP1A2, CYP2D6, and CYP3A4, which may raise concerns regarding metabolic interactions. Nevertheless, their strong binding affinities and acceptable bioavailability scores suggest that structural optimization or dosage modulation could mitigate these limitations. Drug-likeness and oral bioavailability assessment further emphasized the importance of integrating pharmacokinetic considerations with docking performance. While some compounds exhibited excellent binding affinities, violations of multiple drug-likeness filters may limit their direct applicability as oral therapeutics. Oleic acid, despite partial rule violations, displayed the highest predicted bioavailability, underscoring that strict compliance with all filters is not always necessary for pharmacological relevance. Overall, compounds that balanced strong docking performance with acceptable ADME profiles emerged as the most promising theoretical leads. Taken together, the findings of this study indicate that Ginkgo biloba, particularly its biflavonoid constituents, represents a more potent source of lead molecules for Alzheimer’s disease therapy through simultaneous inhibition of AChE and BACE-1. In contrast, Mucuna pruriens phytochemicals appear to contribute primarily through neuroprotective, antioxidant, and neurotransmitter-modulating mechanisms rather than strong enzymatic inhibition. Phytochemical constituent’s comparison of 3D images table by 1eve (Acetylcholinesterase) and 1tqf (Beta Secretase) for Kapikachu and Ginkgo biloba generated by 1 Click Docking Software were given in Table 5. The integration of molecular docking, physicochemical analysis, and ADME prediction provides a robust theoretical framework for prioritizing natural compounds for further investigation.
Conclusion
The present in silico study provides a systematic comparative evaluation of phytochemical constituents from Mucuna pruriens (Kapikachu) and Ginkgo biloba against key molecular targets implicated in Alzheimer’s disease, namely acetylcholinesterase (AChE) and β-secretase (BACE-1). Molecular docking results demonstrated that phytochemicals from Ginkgo biloba, particularly biflavonoids such as ginkgetin, bilobetin, and sciadopitysin, exhibited significantly stronger binding affinities toward both AChE and BACE-1 when compared to Kapikachu-derived compounds. These findings suggest a pronounced dual-target inhibitory potential, which is a desirable characteristic for addressing the multifactorial pathology of Alzheimer’s disease. In contrast, phytochemicals from Mucuna pruriens showed moderate binding affinities, indicating a supportive role in cholinergic modulation and neuroprotection rather than direct enzyme inhibition. Overall, the study highlights Ginkgo biloba as a more potent source of multitarget anti-Alzheimer’s agents, while reinforcing the complementary neuroprotective value of Mucuna pruriens. The integration of docking performance with pharmacokinetic prediction strengthens the reliability of lead identification and prioritization. While the findings are based on computational predictions, they provide a strong theoretical foundation for future molecular dynamics simulations, in vitro enzyme inhibition studies, and in vivo validation. Thus, the present work contributes valuable insights toward the rational development of plant-based, multitarget therapeutic strategies for Alzheimer’s disease.
Acknowledgement
The authors are thankful to Pravara Rural College of Pharmacy, Pravaranagar.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from other Sources
Not Applicable
Author Contributions
Anerao Abhishek Parmeshwar: In silico ADMET (Absorption, Distribution, Metabolism, Excretion)
Shweta Vijaykant Bairagi: Drawing of Tafenoquine drug derivatives
Sahil Babasaheb Jadhav: SWISS ADMET Software Data
Harshali Narayan Anap: Docking images of 1-Click Docking software
Rohit Jaysing Bhor: Research drafting
Arati Dattatray Jadhav: Collect information of research introduction
Aniket Kakasaheb Jadhav: 1-Click Docking software
Tejal kailas Anarthe:1-Click Docking software
References
- Aggarwal BB, Prasad S, Sung B, Krishnan S, Guha S. Prevention and treatment of Alzheimer’s disease by natural products. J Neuroimmune Pharmacol. 2013;8(1):34-45. doi:10.1007/s11481-012-9405-3
- Ballard C, Gauthier S, Corbett A, Brayne C, Aarsland D, Jones E. Alzheimer’s disease. Lancet. 2011;377(9770):1019-1031. doi:10.1016/S0140-6736(10)61349-9
CrossRef - Behl C, Moosmann B. Antioxidant neuroprotection in Alzheimer’s disease as a preventive and therapeutic approach. Free Radic Biol Med. 2002;33(2):182-191. doi:10.1016/S0891-5849(02)00883-3
CrossRef - Bharathi P, Anuradha CV. Protective role of Mucuna pruriens in neurodegenerative disorders. J Herb Med Toxicol. 2010;4(1):1-8.
- Butterfield DA, Halliwell B. Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nat Rev Neurosci. 2019;20(3):148-160. doi:10.1038/s41583-019-0132-6
CrossRef - Cummings JL, Tong G, Ballard C. Treatment combinations for Alzheimer’s disease: Current and future pharmacotherapy options. J Alzheimers Dis. 2019;67(3):779-794. doi:10.3233/JAD-180766
CrossRef - Ellman GL, Courtney KD, Andres V Jr, Featherstone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961;7(2):88-95. doi:10.1016/0006-2952(61)90145-9
CrossRef - Ferreira LG, dos Santos RN, Oliva G, Andricopulo AD. Molecular docking and structure-based drug design strategies. Molecules. 2015;20(7):13384-13421. doi:10.3390/molecules200713384
CrossRef - Reynoso-García MF, Nicolás-Álvarez DE, Tenorio-Barajas AY, Reyes-Chaparro A. Structural bioinformatics applied to acetylcholinesterase enzyme inhibition. Int J Mol Sci. 2025;26(8):3781. doi:10.3390/ijms26083781.
CrossRef - Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science. 2002;297(5580):353-356. doi:10.1126/science.1072994
CrossRef - Howes MJR, Perry NSL. The role of phytochemicals in the treatment and prevention of dementia. Drugs Aging. 2011;28(6):439-468. doi:10.2165/11589260-000000000-00000
CrossRef - Lane RM, Potkin SG, Enz A. Targeting acetylcholinesterase and butyrylcholinesterase in dementia. Int J Neuropsychopharmacol. 2006;9(1):101-124. doi:10.1017/S1461145705005833
CrossRef - Lipinski CA, Lombardo F, Dominy BW, Feeney PJ. Experimental and computational approaches to estimate drug-likeness and permeability. Adv Drug Deliv Rev. 2001;46(1-3):3-26. doi:10.1016/S0169-409X(00)00129-0
CrossRef - Müller WE, Eckert A, Eckert GP, Leuner K. Ginkgo biloba extract EGb 761® in the treatment of dementia. Int J Clin Pharmacol Ther. 2010;48(8):479-491.
- Perry EK, Pickering AT, Wang WW, Houghton PJ, Perry NS. Medicinal plants and Alzheimer’s disease. J Altern Complement Med. 1999;5(4):367-382. doi:10.1089/acm.1999.5.367
CrossRef - Raskin J, Cummings J, Hardy J, Schuh K, Dean RA. Neurobiology of Alzheimer’s disease: Integrated molecular perspectives. J Alzheimers Dis. 2015;46(3):531-544. doi:10.3233/JAD-150018
CrossRef - Selkoe DJ, Hardy J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol Med. 2016;8(6):595-608. doi:10.15252/emmm.201606210
CrossRef - Singh A, Naidu PS, Kulkarni SK. Neuroprotective potential of Mucuna pruriens in Parkinson’s disease. Phytother Res. 2004;18(9):706-712. doi:10.1002/ptr.1535
CrossRef - Sliwoski G, Kothiwale S, Meiler J, Lowe EW Jr. Computational methods in drug discovery. Pharmacol Rev. 2014;66(1):334-395. doi:10.1124/pr.112.007336
CrossRef - Trott O, Olson AJ. AutoDock Vina: Improving docking speed and accuracy. J Comput Chem. 2010;31(2):455-461. doi:10.1002/jcc.21334
CrossRef - Veber DF, Johnson SR, Cheng HY, Smith BR, Ward KW, Kopple KD. Molecular properties influencing oral bioavailability. J Med Chem. 2002;45(12):2615-2623. doi:10.1021/jm020017n
CrossRef - Escamilla S, Sáez-Valero J, Cuchillo-Ibáñez I. NMDARs in Alzheimer’s disease: Between synaptic and extrasynaptic membranes. Int J Mol Sci. 2024;25(18):10220. doi:10.3390/ijms251810220.
CrossRef - Liu W, Li Y, Zhao T, et al. The role of N-methyl-D-aspartate glutamate receptors in Alzheimer’s disease: From pathophysiology to therapeutic approaches. Prog Neurobiol. 2023;231:102534. doi:10.1016/j.pneurobio.2023.102534.
CrossRef - Wang J, Ferruzzi MG, Ho L, et al. Brain-targeted proanthocyanidin metabolites for Alzheimer’s disease. J Neurosci. 2012;32(15):5144-5150. doi:10.1523/JNEUROSCI.6434-11.2012
CrossRef
Abbreviations List
AD: Alzheimer’s Disease; Aβ: Amyloid Beta; Ach: Acetylcholine; AChE: Acetylcholinesterase; ADME: Absorption, Distribution, Metabolism, and Excretion; BBB: Blood–Brain Barrier; BACE-1: β-Site Amyloid Precursor Protein Cleaving Enzyme: 1 (β-Secretase); CNS: Central Nervous System; CYP: Cytochrome P450; GI: Gastrointestinal; HBD: Hydrogen Bond Donor; HBA: Hydrogen Bond Acceptor; iLOGP: Intrinsic Logarithm of Partition Coefficient; MR: Molecular Refractivity; NMDA: N-Methyl-D-Aspartate; PDB: Protein Data Bank; P-gp: P-glycoprotein; PSA: Polar Surface Area; QSAR: Quantitative Structure–Activity Relationship; ROS: Reactive Oxygen Species; SAR: Structure Activity Relationship; TPSA: Topological Polar Surface Area; XLOGP3: Calculated Logarithm of Octanol/Water Partition Coefficient.
Accepted on: 02-03-2026
Second Review by: Dr. Randa Salahand Dr. Ankur Vashi
Final Approval by: Dr. Eugene A. Silow









