Degradable Magnesium-Based Alloy (Mg-Zn- Ca-Y) for Biomedical Applications
1Department of Mechanical Engineering, Pravara Rural Engineering College Loni, S.P. Pune University, Pune, India.
2Department of Mechanical Engineering, Dr. Vitthalrao Vikhe Patil College of Engineering, Ahmednagar, S.P. Pune University, Pune, India.
Corresponding Author E-mail:gholapss@pravaraengg.org.in
DOI : http://dx.doi.org/10.13005/bbra/3489
ABSTRACT:Magnesium (Mg) alloys have gained considerable attention as potential materials for biodegradable orthopaedic implants because of their mechanical compatibility with natural bone and excellent biocompatibility. Despite these advantages, their rapid corrosion in physiological environments restricts their long-term structural integrity. In the present study, Mg–4Zn–0.2Ca–xY (x = 3, 6, 9, and 12 wt.%) alloys were developed to evaluate the influence of Y (yttrium) content on microstructural characteristics, mechanical performance, and degradation behaviour. The alloys were synthesized using the casting technique and systematically examined through microstructural analysis, mechanical testing, and corrosion evaluation in simulated body fluid. The addition of Y promoted noticeable grain refinement and enhanced phase distribution, leading to improved mechanical properties and corrosion resistance. Among the investigated compositions, the Mg–4Zn–0.2Ca–6Y alloy demonstrated a more homogeneous microstructure along with superior strength and hardness compared to other variants. Furthermore, this composition exhibited a relatively slower and more uniform degradation rate, indicating improved stability under simulated physiological conditions. The outcomes of this study suggest that controlled incorporation of Y in Mg–Zn–Ca alloys can effectively tailor their structural and degradation properties. Therefore, the Mg–4Zn–0.2Ca–6Y alloy shows strong potential as a biodegradable material for orthopaedic implant applications requiring balanced mechanical reliability and controlled corrosion behaviour.
KEYWORDS:Biomedical; Degradation; Implant; Magnesium alloy; Yttrium, Zink
Introduction
Mg and its alloys have attracted significant attention as potential biodegradable materials for orthopaedic implants due to their favourable mechanical properties and biocompatibility. Compared with polymer-based biodegradable materials, Mg alloys possess higher mechanical strength, making them suitable for load-bearing implant applications. Additionally, their elastic modulus is closer to that of natural bone, which helps reduce stress shielding and supports bone regeneration during fracture healing.1 Mg is widely used in biomedical devices such as bone plates, screws, and vascular stents. However, the rapid degradation of Mg in physiological environments may cause premature implant failure, while hydrogen evolution during corrosion remains a major challenge for orthopaedic applications.2
Mg alloys exhibit low density, biodegradability, and mechanical properties comparable to natural bone, making them highly attractive for biomedical use. The Young’s modulus of cortical bone ranges from 10–27 GPa, whereas titanium alloys and 316L stainless steel possess significantly higher values of approximately 110 GPa and 193 GPa, respectively. Similarly, Mg alloys exhibit densities (1.74–2.02 g/cm³) close to that of natural bone (1.8–2 g/cm³), reducing stress concentration and improving implant compatibility.3 These alloys also offer a high strength-to-weight ratio, excellent biocompatibility, and favourable mechanical and thermal characteristics.
Recent studies have focused on alloying and surface modification strategies to control degradation behaviour and improve mechanical performance. Munir et al. 4 reported that scandium additions enhanced corrosion resistance and microstructural stability in Mg-based alloys while maintaining good cytocompatibility. Parfenov et al.5 demonstrated that ultrafine grain structures significantly improved corrosion resistance in Mg–Ca alloys. Surface modification techniques such as coatings and chemical treatments have also been widely explored to enhance implant performance. Chandra et al.6 reviewed various preparation and surface modification approaches for Mg alloys, while Li et al.7 demonstrated improved corrosion resistance and biocompatibility using polydopamine and hyaluronic acid coatings. Similarly, Nassif et al.8 highlighted the effectiveness of hydroxyapatite coatings in improving implant bioactivity and corrosion resistance.
Long-term in vivo investigations have further confirmed the potential of Mg alloys for biomedical applications. Oshibe et al.9 reported that anodized WE43 Mg alloys exhibited good corrosion resistance and biocompatibility during long-term implantation. Bakhsheshi-Rad et al.10 developed multifunctional coatings incorporating Ta₂O₅ and PCL/MgO–Ag to enhance antibacterial and corrosion-resistant properties. Rahman et al.11 and Hanas et al.12 demonstrated that surface treatments and polymer coatings can effectively regulate degradation rates and improve implant stability.
Apart from composition and surface modification, the fabrication technique significantly influences the performance of Mg-based implants. Conventional processing methods such as casting, powder metallurgy, and additive manufacturing affect microstructure, mechanical behaviour, and corrosion characteristics. In the present study, stir-cast Mg–Zn–Ca–Y alloys were developed to evaluate the effect of Y addition on microstructure, mechanical properties, and degradation behaviour. The study aims to optimise Y content to achieve a balanced combination of mechanical strength and controlled biodegradation suitable for orthopaedic implant applications.
Materials and Methods
Bottom pouring Stir casting
Several casting methods, such as gravity casting, vacuum casting, rotary centrifugal casting, squeeze casting, and extrusion casting, are available in an automatic bottom-pouring stir casting furnace intended for melting Mg, zinc, calcium, and Y metal alloys. Any metal with melting point less than 1000° C unlike conventional casting process, the melt pours from the bottom of this machine into the mould. Casting machine that allows mixing the alloying material in the base metal and pouring it into the mould with ease and without loss in the melt temperature. The Y concentration up to 12 wt.% was selected to evaluate its strengthening and corrosion resistance benefits in Mg-based alloys while remaining within ranges reported in biomedical studies. Previous research indicates that controlled Y addition improves microstructural stability and degradation behaviour; however, excessive release of Y³⁺ ions may cause cytotoxic effects and influence osteoblast activity. Therefore, this concentration range was chosen to identify an optimal balance between mechanical performance and biological safety (Witte et al., 2008; Myrissa et al., 2016; Rahman et al., 2020). Four distinct alloy samples were prepared with varying concentrations of Zn, Ca, and Y maximum acceptable limit of Y in body is 12% that’s why it can vary from 3-12% as detailed in Table 1.
Table 1: Materials Percentage
| Sample Number | Mg (wt. %) | Zn (wt. %) | Ca (wt. %) | Y (wt. %) |
| 1 | 92.8 | 4.0 | 0.2 | 3 |
| 2 | 89.8 | 4.0 | 0.2 | 6 |
| 3 | 86.8 | 4.0 | 0.2 | 9 |
| 4 | 83.8 | 4.0 | 0.2 | 12 |
Adding Y in controlled amounts (3, 6, 9, 12 wt.%) to Mg-4Zn-0.2Ca-Y alloys can improve mechanical properties and corrosion behavior, addressing limitations observed in previously studied Mg alloys. The selected Y concentrations were chosen based on prior studies indicating that this range balances enhanced strength, refined microstructure, and potential biocompatibility, which are critical for biomedical applications. Choosing 3, 6, 9, and 12 wt.% Y is to systematically study the effect of increasing Y content on microstructure, mechanical properties, and corrosion behavior, and to identify an optimal concentration that enhances performance for biomedical applications without compromising biocompatibility.
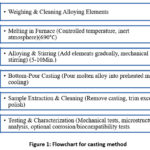 |
Figure 1: Flowchart for casting method.
|
As shown in the figure 1 steps are used to prepare samples the bottom-pouring stir casting furnace has a primary furnace that, while the stirring process is ongoing, automatically pours molten metal composites into casting equipment.13 The system, designed for Mg alloys, includes a cylindrical primary furnace capable of heating the melt to 1000°C, utilizing, Kanthal APM heating elements. It is equipped with a leak-proof lid, lock, and a bottom-positioned valve for pouring molten composites directly into casting equipment during active stirring.
A secondary pre-heating furnace with a dual-stage opening is located above the primary furnace, enabling pre-heating of alloy and metal granules up to 900°C.14 A variable-speed stirrer, capable of reaching up to 3000 RPM and fitted with interchangeable blades, is positioned at the top of the system. This ensures robust stirring of the molten metal, preventing dross formation. The furnace design allows for the addition of metal matrix components during the stirring process.15 A control panel adjacent to the stirrer houses electrical and electronic components to manage the furnace and its attachments. The entire system, including the furnaces, stirrer, and control panel, is enclosed in a rigid movable frame fitted with wheels and brakes for mobility and stability. Gas cylinders with appropriate regulators and connections supply argon and SF6 gases for system operation. The furnace is also equipped with moulds/die castings at the bottom of the primary cylindrical furnace, enabling direct conveyance of molten metal alloys into the moulds16. These moulds are compatible with a number of casting techniques, such as extrusion, rotary centrifugal, squeeze, vacuum, and gravity casting. However, the thermo-casting setup is used independently and not in combination with other casting moulds.
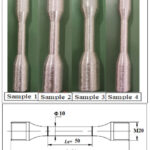 |
Figure 2: Samples prepared by casting method
|
Samples as shown in Figure.2 manufactured by bottom stir casting method, with specimen size as diameter 10mm and length as 100mm.
Table 2: Chemical composition of samples
| Specimen 1 | ||||||
| Element | Zn% | Ca% | Y% | Cr% | Si% | Mg% |
| Composition | 4.06 | 0.22 | 3.02 | 0.004 | 0.003 | 92.693 |
| Specimen 2 | ||||||
| Element | Zn% | Ca% | Y% | Cr% | Si% | Mg% |
| Composition | 4.05 | 0.22 | 6.04 | 0.004 | 0.003 | 89.683 |
| Specimen 3 | ||||||
| Element | Zn% | Ca% | Y% | Cr% | Si% | Mg% |
| Composition | 4.05 | 0.24 | 9.07 | 0.002 | 0.002 | 86.636 |
| Specimen 4 | ||||||
| Element | Zn% | Ca% | Y% | Cr% | Si% | Mg% |
| Composition | 4.06 | 0.22 | 12.01 | 0.004 | 0.002 | 83.704 |
The alloying composition of the synthesized Mg–Zn–Ca–Y alloys was evaluated through a systematic multi-stage procedure to ensure accuracy and homogeneity of the final chemical constituents. Initially, the weighed elemental inputs were calculated according to the intended wt.% ratios, and high-purity Mg, Zn, Ca, and Y were melted under a controlled protective atmosphere to minimize oxidation. After casting and solidification, the alloy compositions were quantitatively verified using spectroscopic and microscopic techniques.
Chemical composition of sample is as mention in the above table no.2 which has been tested in Hyderabad metallurgical lab, Material Testing Services, Metallurgical & NDT Materials test has been conducted with the help of ASTM (American Society for Testing and Materials) standard.
Mechanical Testing
Compression Test
The compression test is a vital mechanical evaluation method used to examine how materials react under compressive loads. During this test, a sample is subjected to a compressive force using specialized fixtures on a universal testing machine.17 In this study, four distinct samples with different material compositions were fabricated through casting to identify the composition that exhibits the highest load-bearing capacity.18
Tensile Test
An important method for determining the tensile strength, yield strength, and ductility of metallic materials is tensile testing. This test determines the force needed to fracture a sample, as well as its elongation or stretching behaviour until failure. For composite materials, tensile tests are often carried out using simple tension or flat-sandwich tension setups, adhering to standards such as ASTM. The results are typically presented as stress-strain curves, which serve as a basis for calculating the tensile modulus of test sample.
Scanning Electron Microscopy (SEM) and Energy-Dispersive X-ray Spectroscopy (EDS)
SEM was utilized to examine the surface morphology and fracture characteristics of the alloy samples. The samples were cleaned, dried, and coated with a thin conductive layer prior to analysis to enhance imaging quality. EDS, attached to the SEM, was used to determine the elemental composition and distribution within the samples. Microstructural characterization and elemental verification of the developed alloys were performed using the combined SEM–EDS analysis.
Testing of Degradation Rate
In vivo biodegradation test
In vivo corresponds to experiments, processes carried out overall living organism, such as humans, plants, or animals in a laboratory setting.19 This approach allows researchers to observe biological processes in their natural context within the living system.
In vitro biodegradation test
In vitro means in glass. This phrase describes tests or experiments that are possible to carry out in a controlled laboratory setting, usually away from living things. Since in vitro studies do not involve live organisms, they are widely used in medical research and experimentation. Hanks’ solution is often used for conducting in vitro degradation tests on materials. The following blends can be used to make a solution with a salt balance for Hanks’ solution: NaCl – 8.0 g/L; MgSO4 -0.1 g/L;CaCl2 – 0.14 g/L; D-Glucose -1.0 g/L, NaHPO4 – 0.05 g/L; KH2PO4 – 0.06 g/L; and KCl-0.4 g/L. The test for immersion, performed with Hanks’ solution, is used to observe how materials react biochemically, particularly in relation to Mg samples.20 These tests are designed to simulate physiological conditions, with the solution temperature typically maintained at 37°C .21
Insertion of sample in the solution
Four different samples are put in different four beakers. Hank’s solution is filled in all the four beakers. Ensure that sample is completely dipped in the solution. The samples of test and degradation are shown in Figure 3 and 4.
 |
Figure 3: Four Samples prepared for degradation
|
 |
Figure 4: Samples kept for degradation test
|
Degradation of samples
Table 3 mention bellow represents the weight, diameter and length of all four samples.
Table 3: Size of Samples
| Samples | Weight in (gms) | Diamter in (mm) | Length in (mm) |
| 1 | 7 | 18 | 15 |
| 2 | 7 | 18 | 15 |
| 3 | 7 | 18 | 15 |
| 4 | 7 | 18 | 15 |
After insertion these all samples in hanks solution weights of all samples has been monitored daily.
Results
Compression Test
Four Mg–4Zn–0.2Ca–xY alloy samples were fabricated using the casting technique to evaluate load-bearing capability. The compressive strength values obtained for each composition are summarized in Table 4.
Table 4: Compressive Strength
| Sample Number | Chemical Composition | Compressive Strength |
| 1 | Mg-4Zn-0.2Ca-3Y | 892.542 MPa |
| 2 | Mg-4Zn-0.2Ca-6Y | 962.443 MPa |
| 3 | Mg-4Zn-0.2Ca-9Y | 964.989 MPa |
| 4 | Mg-4Zn-0.2Ca-12Y | 1080.000 MPa |
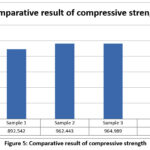 |
Figure 5: Comparative result of compressive strength
|
Figure 5 illustrates the compressive strength variation with Y addition. The results indicate a progressive increase in compressive strength with increasing Y content, with Sample 4 exhibiting the highest strength.
Tensile Test
Tensile testing was conducted to evaluate the tensile behaviour of the developed alloys. The obtained tensile strength values are listed in Table 5.
Table 5: Tensile Strength
| Sample Number | Chemical Composition | Tensile strength |
| 1 | Mg-4Zn-0.2Ca-3Y | 315.382 MPa |
| 2 | Mg-4Zn-0.2Ca-6Y | 353.452 MPa |
| 3 | Mg-4Zn-0.2Ca-9Y | 372.678 MPa |
| 4 | Mg-4Zn-0.2Ca-12Y | 477.847 MPa |
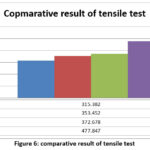 |
Figure 6: comparative result of tensile test
|
Figure 6 shows that tensile strength increases with increasing Y concentration, with Sample 4 demonstrating the highest tensile strength.
Degradation test
All the samples are kept for degradation to check their degradation rate.
In vitro degradation tests were performed to evaluate corrosion behaviour. The degradation results are presented in Figure 7. Sample 2 exhibited the slowest degradation rate and remained structurally stable for approximately 6.5 months. Electrochemical analysis indicated that increasing Y content shifts corrosion potential (Ecorr) toward more positive values and reduces corrosion current density (icorr), suggesting improved corrosion resistance. Weight variation trends revealed cyclic formation and breakdown of protective Mg(OH)₂ and oxide layers during immersion.
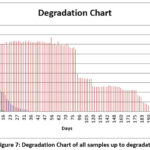 |
Figure 7: Degradation Chart of all samples up to degradation
|
The degradation data for all samples are presented in figure 7. The degradation tests were performed in vitro. The requirements for degradable implant materials vary depending on the implant location. Key mechanical properties expected from implants include ductility, strength, and longevity. Strength is critical to prevent sudden failure, while ductility plays a key role if the implant experiences plastic deformation during surgery. For implants to be successful, fatigue strength and strength retention are vital to ensure safe and effective performance. Implants must maintain their strength, aid in bone healing, and avoid failure before the healing process is completed.
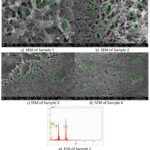 |
Figure 8: SEM images of all samples after 15 days and EDS of sample 2
|
SEM images (Figure 8) show fracture morphology differences among samples. Sample 1 displayed a porous and brittle fracture surface, whereas Samples 2–4 exhibited ligament-like structures and micro-dimples, indicating mixed ductile–brittle fracture behaviour.
Discussions
The degradation characteristics of magnesium-based implants play a vital role in their biomedical performance; as such implants must retain adequate mechanical strength during the bone healing period while gradually degrading afterwards. Among the investigated alloy compositions, the Mg–4Zn–0.2Ca–6Y alloy exhibited the most favourable degradation performance, indicating that the addition of Y significantly alters corrosion behaviour through microstructural refinement.
Y enhances grain boundary stability by reducing electrochemical inhomogeneity, thereby limiting localized corrosion processes. The segregation of Y at grain boundaries decreases anodic dissolution by reducing micro-galvanic interactions between different phases. Furthermore, Y promotes the formation of thermodynamically stable intermetallic compounds, including Mg₁₂YZn and Mg₂₄Y₅, which contribute to improved structural integrity and corrosion resistance. The presence of Y also strengthens grain boundary cohesion by enhancing atomic bonding between adjacent grains, resulting in improved resistance to crack initiation and propagation. In addition, Y interacts with detrimental impurity elements such as iron, nickel, and copper, thereby mitigating impurity-induced corrosion acceleration. The refinement of microstructure associated with Y incorporation further ensures a more uniform solute distribution, minimizing localized compositional gradients that typically promote corrosion activity. Moreover, Y influences passive film formation by facilitating the development of Y-enriched oxide layers, which demonstrate greater stability and protective capability compared to conventional Mg(OH)₂ films.
Scanning electron microscopy observations revealed that increasing Y concentration altered the fracture morphology from predominantly brittle behaviour to a mixed ductile–brittle fracture mode, indicating enhanced mechanical reliability and energy absorption capacity.
However, an alternative interpretation should also be considered. At higher Y concentrations, excessive formation of intermetallic phases may occur, which could potentially act as cathodic sites and promote localized galvanic corrosion. Such phase accumulation may also reduce ductility by introducing brittle constituents within the microstructure. Therefore, while moderate Y addition improves overall alloy performance, excessive Y content may negatively influence corrosion behaviour and mechanical properties, suggesting the existence of an optimal Y concentration range.
Conclusions
Mg–4Zn–0.2Ca–xY alloys with varying Y concentrations were successfully fabricated and evaluated for biodegradable implant applications. The following conclusions can be drawn:
Y addition significantly refines microstructure and enhances mechanical properties. Both compressive and tensile strengths increased with increasing Y content.
Y improves corrosion resistance by promoting stable intermetallic phase formation and enhancing protective oxide film stability.
The Mg–4Zn–0.2Ca–6Y alloy demonstrated the most balanced degradation behaviour, maintaining structural integrity while enabling controlled biodegradation.
Excessive Y content may promote intermetallic phase coarsening, which can reduce ductility and increase brittleness.
Although Y improves mechanical and corrosion performance, potential cytotoxicity associated with excessive Y³⁺ ion release should be carefully controlled for safe biomedical applications.
Overall, Mg–4Zn–0.2Ca–6Y alloy demonstrates strong potential as a biodegradable orthopaedic implant material due to its balanced mechanical strength and controlled degradation behaviour.
Acknowledgement
The author gratefully acknowledges Department of Mechanical Engineering, Dr. Vitthalrao Vikhe Patil College of Engineering, Ahmednagar and Department of mechanical engineering, PREC loni and SPPU Pune for providing the necessary facilities to carry out this study.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement-
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Snehal Sainath Gholap: Conceptualization, Methodology, Writing – Original Draft
Ravindra Rambhau Navthar: Data Collection, Analysis, Writing – Review & Editing
References
- Kraus T, Fischerauer SF, Hänzi AC, Uggowitzer PJ, Löffler JF, Weinberg AM. Magnesium alloys for temporary implants in osteosynthesis: In vivo studies of their degradation and interaction with bone. Acta Biomater. 2012;8(3):1230–1238.
CrossRef - Myrissa A, Agha NA, Lu Y, Martinelli E, Eichler J, Szakács G. In vitro and in vivo comparison of binary Mg alloys and pure Mg. Mater Sci Eng C. 2016;61:865–874.
CrossRef - Zha M, Wang SC, Jia HL, Yang Y, Ma PK, Wang HY. Effect of minor Ca addition on microstructure and mechanical properties of a low-alloyed Mg–Al–Zn–Sn alloy. Mater Sci Eng A. 2023;862:144457.
CrossRef - Munir K, Lin J, Wen C, Wright PFA, Li Y. Mechanical, corrosion, and biocompatibility properties of Mg–Zr–Sr–Sc alloys for biodegradable implant applications. Acta Biomater. 2019;94:1–15.
- Parfenov EV, Kulyasova OB, Mukaeva VR, Mingo B, Farrakhov RG, Cherneikina YV. Influence of ultra-fine grain structure on corrosion behaviour of biodegradable Mg–1Ca alloy. Corros Sci. 2019;163:108303.
CrossRef - Chandra G, Pandey A. Preparation strategies for Mg alloys for biodegradable orthopaedic implants and other biomedical applications: A review. IRBM. 2022;43(3):229–249.
CrossRef - Li JA, Chen L, Zhang XQ, Guan SK. Enhancing biocompatibility and corrosion resistance of biodegradable MgZn–Y–Nd alloy by PDA/HA coating for cardiovascular applications. Mater Sci Eng C. 2020;109:110607.
CrossRef - Nassif N, Ghayad I. Corrosion protection and surface treatment of magnesium alloys used for orthopedic applications. Adv Mater Sci Eng. 2013;2013:532896.
CrossRef - Oshibe N, Marukawa E, Yoda T, Harada H. Degradation and interaction with bone of magnesium alloy WE43 implants: A long-term in vivo rat tibia study. J Biomater Appl. 2019;33(9):1157–1167.
CrossRef - Bakhsheshi-Rad HR, Ismail AF, Aziz M, Hadisi Z, Omidi M, Chen X. Antibacterial activity and corrosion resistance of Ta₂O₅ thin film and electrospun PCL/MgO–Ag coatings on biodegradable Mg alloy implants. Ceram Int. 2019;45(9):11883–11892.
CrossRef - Rahman M, Dutta NK, Roy Choudhury N. Magnesium alloys with tunable interfaces as bone implant materials. Front Bioeng Biotechnol. 2020;8:564.
CrossRef - Hanas T, Sampath Kumar TS, Perumal G, Doble M. Tailoring degradation of AZ31 alloy by surface pretreatment and electrospun PCL fibrous coating. Mater Sci Eng C. 2016;65:43–50.
CrossRef - Pahlevanzadeh F, Bakhsheshi-Rad HR, Ismail AF, Aziz M, Chen XB. Development of PMMA-Mon-CNT bone cement with superior mechanical and biological properties for bone-defect treatment. Mater Lett. 2019;240:9–12.
CrossRef - Kiani F, Wen C, Li Y. Prospects and strategies for magnesium alloys as biodegradable implants from crystalline to bulk metallic glasses and composites: A review. Acta Biomater. 2020;103:1–23.
CrossRef - El-Mahallawy N, Palkowski H, Klingner A, Diaa A, Shoeib M. Effect of 1.0 wt.% Zn addition on microstructure, mechanical properties, and biocorrosion behaviour of Mg–Sn–Mn alloy. Mater Today Commun. 2020;24:100999.
CrossRef - Vinogradov A, Merson E, Myagkikh P, Linderov M, Brilevsky A, Merson D. Attaining high functional performance in biodegradable Mg–Zn–Ca alloys: Challenges and prospects. Materials. 2023;16(3):1324.
CrossRef - Kasaeian-Naeini M, Sedighi M, Hashemi R, Delavar H. Microstructure, mechanical properties, and fracture toughness of ECAPed magnesium–hydroxyapatite composites for bioabsorbable implants. Ceram Int. 2023;49(12):17074–17090.
CrossRef - Verma A, Ogata S. Magnesium-based alloys for reinforcing biopolymer composites and coatings: A critical overview. Adv Ind Eng Polym Res. 2023;6(4):341–355.
CrossRef - Zhang Q, Zhou T, Guo F, Liu D. In vitro corrosion behavior of Mg–Sn–Zn–Zr alloys in Hank’s solution. J Mater Eng Perform. 2023;32(12):8299–8313.
CrossRef - Savaedi Z, Motallebi R, Mirzadeh H, Aghdam RM, Mahmudi R. Superplasticity of fine-grained magnesium alloys for biomedical applications: A comprehensive review. Curr Opin Solid State Mater Sci. 2023;27(5):101058.
CrossRef - Liu Z, Wang T, Xu Y, Liang C, Li G, Guo Y. Double-layer calcium phosphate/siloxane composite coating for enhanced corrosion resistance and biocompatibility of magnesium alloys. Prog Org Coat. 2023;177:107417.a
CrossRef
Abbreviations List
Ca – Calcium
CaCl₂ – Calcium Chloride
EDX – X-ray Spectroscopy
FE-SEM – Field Emission Scanning Electron Microscopy
HAp – Hydroxyapatite
HBSS – Hank’s Balanced Salt Solution
KCl – Potassium Chloride
Mg – Magnesium
NaCl – Sodium Chloride
NaHCO₃ – Sodium Hydrogen Carbonate
SBF – Simulated Body Fluid
SEM – Scanning Electron Microscopy
UTM – Universal Testing Machine
XRD – X-ray Diffraction
Y – Yttrium
Zn – Zinc
Accepted on: 17-02-2026
Second Review by: Dr. Ramya Sri Sura
Final Approval by: Dr. Ali Elshafei









