Characterization of a Mannose-Binding Seed Lectin from Trigonella foenum-graecum with Broad Erythrocyte Agglutination
1Molecular Medicine and Microbial Genetics Laboratory, Department of Applied Genetics, Karnatak University, Pavatenagar, Karnataka, India.
2Regional Occupational Health Centre, ICMR Institute Devanahalli Bengaluru, Karnataka, India.
Corresponding Author E-mail:mmmgenetics@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3479
ABSTRACT:Lectins are carbohydrate-binding proteins widely distributed in leguminous plants and are known for their hemagglutination and diverse biological activities; in this study, a mannose-specific lectin was isolated and partially characterized from Trigonella foenum-graecum seeds by extraction with phosphate-buffered saline, partial purification through ammonium sulphate precipitation and dialysis, and characterization using hemagglutination, sugar inhibition assays, pH and temperature stability tests, and SDS-PAGE analysis. The lectin exhibited strong hemagglutination activity against human A, B, and O erythrocytes without strict blood-group specificity, and its activity was selectively inhibited by mannose, confirming its mannose specificity. The lectin remained stable within a pH range of 6–8 and at temperatures up to 70°C, while SDS-PAGE revealed a protein band of approximately 28–30 kDa. Overall, the findings indicate that Trigonella foenum-graecum seeds are a rich source of a stable mannose-specific lectin with significant potential applications in glycobiology and biomedical research.
KEYWORDS:Hemagglutination; pH; Sugar specificity; Temperature stability; Trigonella foenum-graecum
Introduction
Lectins, also known as carbohydrate-binding proteins, are a class of non-immune glycoproteins with the capacity to recognize and reversibly attach to carbohydrate moieties. Because of this special characteristic, lectins are involved in many biological processes, such as immunological responses, host-pathogen interactions, signal transduction, and cell recognition mechanisms.1 Lectin-glycan interactions have a crucial role in cellular communication and the development of illness, especially in infection biology and cancer research, according to recent studies.2, 3
In plants, lectins are extensively dispersed and are considered to play key roles in defensive mechanisms against insects, fungus, and bacteria, as well as in physiological processes like as seed germination, cell elongation, and symbiotic relationships.4 Plant lectins function as molecular recognition components, allowing plants to detect and react to biotic and environmental stress. Recent proteomic and functional studies have further established that plant lectins contribute to stress tolerance and adaptive responses under adverse environmental conditions.5
Based on their structural and functional traits, plant lectins are divided into a number of categories, with legume lectins being the largest and most thoroughly researched family.6 These lectins have a high selectivity for either simple or complex sugars and are usually oligomeric proteins with one or more carbohydrate-binding sites per subunit. Legume lectins have been widely used as molecular tools in glycobiology for the identification, separation, and characterization of glycoproteins and cell-surface glycans due to their characteristics.7 Legume lectins’ stability, oligomerization, and ligand-binding processes have all been better understood thanks to recent structural research, which has increased their biotechnological significance.8
In addition to their physiological functions in plants, lectins have garnered a lot of interest because of their potential in medicine. Many plant lectins have been shown to have immunomodulatory, anticancer, antiviral, and antifungal properties as well as the capacity to control lymphocyte proliferation and death.9 Recent studies have highlighted lectins’ potential as therapeutic agents and diagnostic biomarkers, especially in immune regulation and cancer targeting. As a result, a significant field of current research continues to be the purification and thorough characterisation of lectins from new and unexplored plant sources.10
Trigonella foenum-graecum (fenugreek), belongs to the family Fabaceae, is well known for its nutritional, medicinal, and therapeutic benefits. Rich in proteins, dietary fiber, alkaloids, saponins, and polyphenols, fenugreek seeds have long been used to treat digestive issues, diabetes, hypercholesterolemia, and inflammation.11 The pharmacological characteristics of fenugreek have been thoroughly studied, however lectins from its seeds have gotten relatively less attention. Previous studies have found mannose-specific lectins in fenugreek seeds, indicating their role in Rhizobium legume symbiosis and plant defense. Fenugreek seed proteins have attracted increased attention due to omics-based research that highlights their medicinal potential and functional variety.12
The significance of lectin ligand interactions in medicinal applications has also been highlighted by recent developments in lectin research. Porphyrins are frequently utilized as photosensitizers in photodynamic therapy (PDT) for the treatment of cancer, although their low selectivity toward tumor cells limits their therapeutic effectiveness.13 Lectin-mediated targeting techniques have been suggested to improve the specificity of porphyrin-based PDT because certain lectins can identify tumor-associated glycoconjugates. Thus, the creation of lectin-assisted medicinal systems requires an understanding of lectins’ structural stability, carbohydrate selectivity, and ligand-binding behavior.
In view of these considerations, the present study focuses on the isolation and partial characterization of a lectin from the seeds of T. foenum-graecum. The lectin is characterized with particular emphasis on hemagglutination activity, carbohydrate specificity, and stability under varying pH and temperature conditions. This comprehensive analysis contributes to a deeper understanding of fenugreek seed lectins and supports their potential applications in glycobiology, biotechnology, and therapeutic research.
Materials And Methods
Collection of Plant Material and Chemicals
Fresh 25g seeds of Trigonella foenum‑graecum were collected during March 2023 from Dharwad, India (15°26′28.5″N, 74°59′2.1″E). The seeds were washed thoroughly, air-dried, and immediately processed for lectin extraction. For hemagglutination studies, Human blood samples were collected through the civil Hospital, Dharwad. Analytical grade chemicals used were of. Ammonium sulfate, NaCl, dialysis membrane (MWCO 50 kDa), pH buffers, SDS- chemicals were procured from Himedia (India) & Sigma-Aldrich (USA). Additional reagents other electrophoresis-grade chemicals were purchased from Sisco Research Laboratories (SRL) and Himedia. Sugars used for inhibition assays. All glassware and plasticware used were of sterilizable laboratory quality.
Extraction of Lectin from Trigonella foenum‑graecum Seeds
Lectin extraction was carried out following a modified protocol described by.14 Approximately 20 g of Dried seed coat free Trigonella foenum‑graecum seed were fine powdered and separated through 50 µm sieve. Then, powder was mixed with 20 mL of phosphate-buffered saline (PBS; 100 mM, pH 7.2) supplemented with 200 mM EDTA and 2 mM PMSF to inhibit proteolysis. The homogenate was kept under gentle stirring at 4 °C overnight. The mixture was filtered through muslin cloth and centrifuged to remove insoluble debris at 5000 rpm for 20 min at 4 °C. For subsequent purification supernatant was stored at 4 °C for.
Purification of Crude Extract & Estimation Protein concentration
Low-molecular-weight contaminants were removed by dialysis. After adding the crude extract to a 50 kDa dialysis tube, it was dialyzed against regular saline for 24 hours at 4 °C with sporadic buffer changes. The dialyzed extract was gradually saturated to 0–100% saturation at 4°C to precipitate ammonium sulphate. Centrifugation was used for 30 minutes at 6000 rpm to collect the precipitated protein fraction. To get rid of any remaining ammonium sulphate, the pellet was completely dialyzed and resuspended in regular saline. The partially purified lectin sample used for additional characterization was this ammonium sulphate fraction (ASF).15 The protein was estimated using BSA as the standard readings at 280 nm as per .16
Trypsinized Erythrocytes Preparation
Samples A, B, and O of human blood were drawn into tubes filled with 4% sodium citrate. After pelleting the erythrocytes for five minutes at 1500 rpm, they were rinsed three times with isotonic saline and then with PBS. At 660 nm, the densely packed erythrocytes were adjusted to OD 2.5. Erythrocytes were trypsinized by incubating them with 0.025% trypsin for one hour at 37 °C. The final erythrocyte suspension was adjusted to OD 3.5 at 660 nm after excess trypsin was eliminated by repeated saline washing. Hemagglutination and inhibition tests were performed using this suspension.17
Haemagglutination Assay
Using trypsinized human erythrocytes and a two-fold serial dilution experiment, the hemagglutination activity of Trigonella foenum-graecum lectin was evaluated in a 96-well U-bottom microplate. As a positive control, 100 μL of standard lectin and 100 μL of RBC suspension were added to the first well in each row. As a negative control, each row’s last well was filled with 100 μL of RBC suspension and 100 μL of saline. RBCs, saline, and lectin solution (100 μL each) were added to each intermediate well. 10 μL of the normalized crude freeze-dried lectin was added to the first well to start the test, and it was serially diluted in the following wells. After an hour of room temperature incubation, the plate was visually examined for agglutination. The highest and lowest dilutions exhibiting agglutination were noted. One hemagglutinating unit (1 HAU) was the lowest lectin concentration that could cause agglutination, and the hemagglutination titer was the maximum dilution that still resulted in visible hemagglutination.
Sugar Specificity Assay
Assays for sugar inhibition were carried out as previously mentioned.18 Each chosen sugar was serially diluted two times in regular saline. Each sugar dilution received a fixed amount of lectin (4 HAU) and was incubated for one hour at 37 °C. Following incubation, 50 µL of 2% trypsin-treated erythrocytes were added, and the solutions were allowed to sit at room temperature for half an hour. The inhibitory concentration was defined as the lowest sugar concentration that totally prevented hemagglutination.
Temperature & pH Effect Stability of Lectin
The purpose of the pH stability test was to determine the ideal pH for the highest activity of lectins found in the partly purified plant lectin seeds. Using buffers with pH values between 1 and 10. After 30 minutes of room temperature incubation with 100 µl of various buffer solutions, 100 µl of lectin was tested for agglutination using 2% erythrocyte. To assess thermal stability, 100 µL aliquots of lectin were incubated for 30 minutes at temperatures between 30 and 90 °C. Samples were cooled to room temperature after heat treatment, and the standard test was used to measure hemagglutination activity.19
SDS–Polyacrylamide Gel Electrophoresis (SDS-PAGE)
Lectin purity and molecular weight were analyzed by SDS-PAGE following Laemmli’s discontinuous buffer system.20 A 4% stacking gel and a 10% resolving gel were made. Each well contained about 20 µg of protein. Electrophoresis was carried out at 100 V for resolving and 70 V for stacking. 0.1% Coomassie Brilliant Blue R-250 (50% methanol, 10% acetic acid) was used to stain the gels, and they were destained until a distinct band resolution was achieved. For calibration, pre-stained molecular weight markers (20–140 kDa) were employed. The Bio-Rad Protocol Bulletin 6210 was used to calculate approximate molecular weights.
Analysis of statistics
Excel Office-2019 was used to perform the statistical analysis. A statistically significant p-value was defined as < 0.05. Every experiment was conducted in triplicate, and the mean ± standard deviation (SD) is used to express the results. The SD is shown by error bars in the graphs.
Results
Sample colection
Figure 1 shows collection of seeds sample Trigonella foenum‑graecum, from Dharwad was selected for screening due to its traditional medicinal relevance and limited prior research on its seed’s proteins. Preliminary assays showed strong hemagglutination activity against all blood groups, making the seed extract a suitable candidate for further lectin characterization.
Agglutination Assay
Trigonella foenum‑graecum exhibited agglutination with all tested erythrocytes and showed no strict discrimination among blood groups. As shown in Table 1, the hemagglutination was observed with all erythrocytes. In Fig. 2, A, B, O group erythrocytes formed characteristic carpet patterns up to dilutions of 5 each beyond these points, button formations appeared, indicating the loss of agglutination. Although Trigonella foenum‑graecum did not exhibit absolute blood group specificity, it demonstrated affinity, with the titre observed against Blood group all erythrocytes (1:32). Based on these findings, all group erythrocytes were used for all subsequent analyses, as they provided maximum hemagglutination activity. Protein estimation of the dialyzed extract using the lowry method revealed a total protein concentration of 15 mg/mL Table 2.
Table 1: Agglutination study of lectins of Trigonella foenum‑graecum seeds with human erythrocytes.
| Sl. No | Erythrocytes | Agglutination |
| 1 | A | ++ |
| 2 | B | ++ |
| 3 | O | ++ |
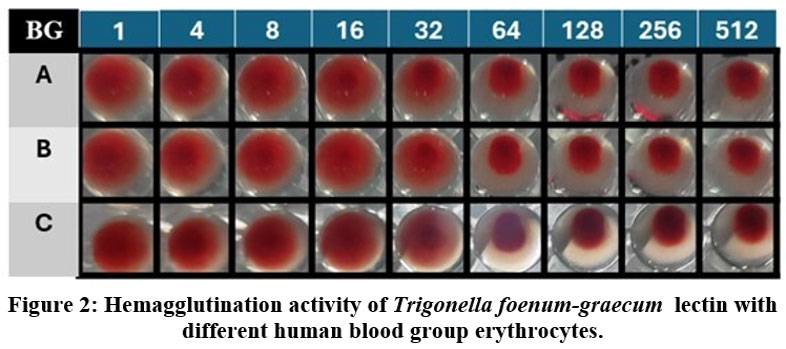 |
Figure 2: Hemagglutination activity of Trigonella foenum‑graecum lectin with different human blood group erythrocytes.
|
Table 2: Trigonella foenum‑graecum Protein concentration
| Protein (mg/ml-1) | HAU/ml
(A) |
HAU/ml
(B) |
HAU/ml
(O) |
SA
(A) |
SA
(B) |
SA
(O) |
|
| Dialysed extract | 15 | 1:32 | 1:32 | 1:32 | 2.13 | 2.13 | 2.13 |
Note: HAU-Hemagglutination Unit, SA-Specific activity
Sugar specificity assay
To evaluate the carbohydrate-binding preference of the lectin, a sugar inhibition assay was performed using a panel of carbohydrates (Table 3). Among all the tested carbohydrates, only mannose was able to inhibit hemagglutination, with a minimum inhibitory concentration (MIC) of 400 mM. None of the other monosaccharides or disaccharides produced any detectable inhibition. These findings indicate that the lectin exhibits a strong affinity for simple sugars, particularly mannose, while showing no recognition of more complex carbohydrates. This monosaccharide specificity may also contribute to its selective interaction with certain human blood groups.
Table 3: Sugar Inhibition activity against T. foenum‑graecum lectin
| Sl. No | Sugar | The lowest concentration needed to prevent hemagglutination (mM) |
| 1 | Fructose | No |
| 2 | Sucrose | No |
| 3 | Maltose | No |
| 4 | Lactose | No |
| 5 | Mannose | Inhibition (400mM) |
Temperature and pH Effect
Lectin was extracted in a variety of buffers with pH values ranging from 1 to 14 to determine the ideal pH for lectin activity. Up to a pH of 6 to 8, the hemagglutination activity remained stable. However, when the pH dropped below 6, activity gradually decreased. As shown in Fig. 3 (a), agglutination activity was lost below pH 6 and above pH 8. The hemagglutination activity was measured after lectin was extracted and incubated at various temperatures ranging from 10 to 100oC for an hour to assess the stability of lectin activity at various temperatures. Lectin from T. foenum-graecum seeds exhibits 100% agglutination between temperatures ranging from 50 to 70ºC when tested after being heated for one hour at temperatures above 70ºC, as shown in Fig. 3 (b). The activity was constant at 50ºC, but as the temperature rose, it declined and eventually stopped above 80ºC. The results showed that lectin was stable for a considerable amount of time, but following heating to 90ºC, lectin activity was eliminated. At room temperature, unheated lectin, however, continued to be active for a few days. The inhibition of the extract’s proteases could be the cause of this. Additionally, lectin activity remained constant at room temperature for at least seven days.
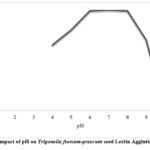 |
Figure 3: (a). Impact of pH on Trigonella foenum-graecum seed Lectin Agglutination Activity
|
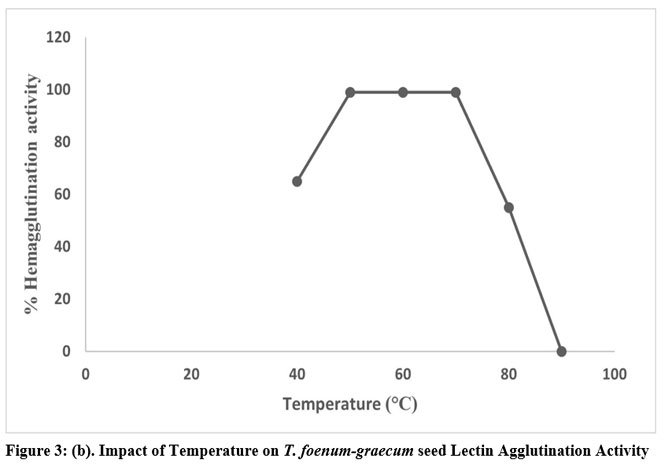 |
Figure 3: (b). Impact of Temperature on T. foenum-graecum seed Lectin Agglutination Activity
|
Gel Electrophoresis
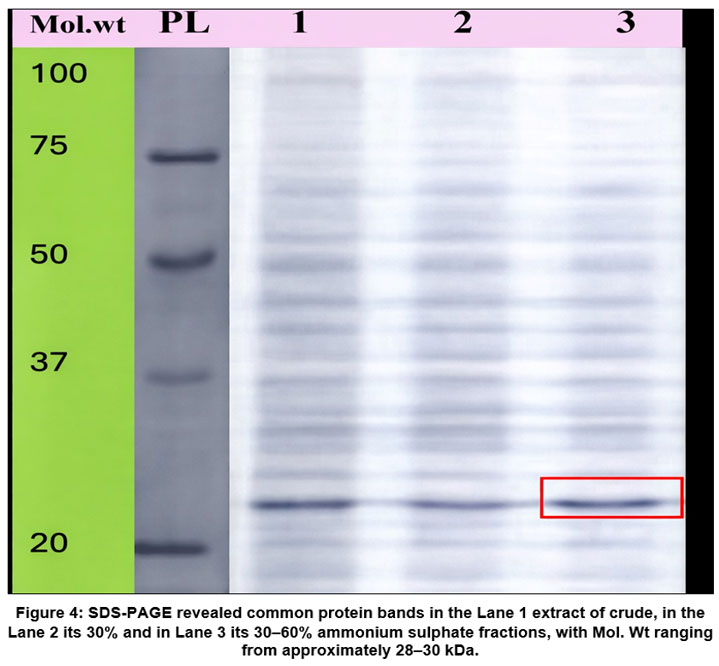
SDS–polyacrylamide gel electrophoresis was employed to analyze the protein composition of both the crude extract and the ammonium sulphate precipitated fractions to assess the degree of purification. As shown in Figure 4, the protein profile of the 30–60% ammonium sulphate fraction (Lane 3) exhibited a substantial reduction in the number of detectable bands when compared with the crude sample (Lane 1), indicating effective partial purification. Notably, a consistent protein band with an approximate molecular mass of 28 kDa was present across all fractions analyzed. This recurring band is likely associated with the lectin protein.
Discussion
The current work reports the isolation and partial characterization of a lectin from Trigonella foenum-graecum seeds, with particular emphasis on hemagglutination activity, carbohydrate specificity, and physicochemical stability. The observed biochemical properties are consistent with those reported for several legume lectins, reinforcing the classification of the isolated protein as a typical leguminous lectin.
The lectin showed high hemagglutination activity against human erythrocytes from blood groups A, B, and O, with no stringent blood-group specificity. Similar broad erythrocyte agglutination has been observed for lectins isolated from various leguminous plants, including Vigna mungo, Tamarindus indica, and Oryza sativa. These lectins detect conserved carbohydrate determinants on erythrocyte membranes rather than ABO-specific antigens.21,22 The lectin may interact with common glycan structures, such as mannose-rich oligosaccharides found on erythrocyte surface glycoproteins, according to this non-selective agglutination.
The lectin’s ability to bind mannose was confirmed by sugar inhibition experiments, which showed that mannose selectively inhibited hemagglutination. Many legume lectins have mannose specificity, which has been linked to their functions in symbiosis, plant defence, and the identification of microbial glycoconjugates.23. The validity of the current findings is supported by reports of similar mannose-specific inhibitory patterns for lectins from fenugreek seeds and other Fabaceae members..24 Another possibility is that the lectin identifies low-affinity structurally similar sugars that were not found in the experiment conditions. Nonetheless, a major mannose specificity is strongly supported by the lack of inhibition by other studied carbs, even at greater doses.
The lectin demonstrated excellent hemagglutination activity in the pH range of 6-8 and retained activity at 70 °C, showing significant physicochemical stability. Several plant lectins have been shown to be stable, which is commonly attributed to their compact tertiary structure and substantial hydrogen bonding within carbohydrate-binding domains.25,26 The decrease of activity at extreme pH values and higher temperatures could be due to conformational alterations or partial denaturation that affects the carbohydrate-binding site integrity. An alternate explanation for thermal stability could be oligomeric subunit association, which has been shown to improve resistance to heat-induced unfolding in legume lectin27
SDS-PAGE examination revealed a prominent protein band of around 28-30 kDa, which is consistent with the molecular mass range described for bean lectins.28 Mannose-specific lectins from Trigonella foenum-graecum and other leguminous species have been found to have comparable molecular weights.29 The existence of a continuous band across partially purified fractions indicates lectin protein enrichment; nevertheless, isolectins or closely related lectin variants with similar molecular weights may coexist in the preparation. To rule out this option, further purification and mass spectrometric analysis are required.
From a functional perspective, the biochemical properties reported in this study suggest prospective applications of the fenugreek seed lectin in glycobiology and medicinal research. In cancer treatment, such as lectin-assisted photodynamic therapy, mannose-specific lectins have been investigated as targeting molecules, antibacterial agents, and diagnostic tools..30 However, it is now unable to rule out other theories on biological activity, such as indirect hemagglutination mediated by non-lectin proteins or the synergistic effects of small pollutants. Confirming the lectin’s inherent biological functions will require sophisticated purification and functional testing.
Overall, the present findings are in strong agreement with previously reported studies on legume lectins and extend existing knowledge by providing additional data on lectins from T. foenum-graecum. The study lays a foundation for future investigations involving complete purification, structural elucidation, and evaluation of biological activities, which will further clarify the functional significance and application potential of this lectin.
Conclusion
This study effectively isolated and partially described a sugar-specific lectin from Trigonella foenum-graecum seeds. The lectin showed strong hemagglutination activity for human erythrocytes A, B, and O, indicating general erythrocyte identification rather than strict blood-group specificity. Sugar inhibition assays confirmed that the lectin is mannose-specific, demonstrating its capacity to bind carbohydrates selectively. According to physicochemical characterization, the lectin is stable over a wide pH range, with optimal activity at neutral pH. It also retains functional integrity at moderately high temperatures, losing activity only at higher thermal settings. A molecular mass of roughly 28–30 kDa was found by SDS-PAGE analysis, which is in line with the typical subunit size of legume lectins. Reduced protein heterogeneity indicates that the lectin fraction was enriched after partial purification using ammonium sulfate precipitation. All these results point to T. foenum-graecum seeds as a rich source of a stable, mannose-specific lectin with characteristics common to legume lectins. This lectin’s robust hemagglutination activity and biochemical stability point to its possible use in glycobiological studies and diagnostic procedures. To investigate its potential biomedical and biotechnological significance, more research comprising thorough purification, structural clarification, and biological activity assessment is necessary.
Acknowledgement
The authors like to acknowledge the assistance of the Department of Applied Genetics, Karnatak University Dharwad, & Regional Occupational Health Centre, ICMR Institute Devanahalli Bengaluru, Karnataka, India. for giving us access to their laboratory facilities during the research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions-
Suresh Basavaraj Arakera: Supervision, Writing review & editing.
Vinuta Hanamant Mane: Conceptualization, Methodology, Writing – Original Draft.
Shubhangi Pingle Conceptualization, Data Collection, Analysis Writing – review & editing.
References
- Sharon N. Lectins: Carbohydrate-specific Reagents and Biological Recognition Molecules. Journal of Biological Chemistry. 2007;282(5):2753-2764. doi:10.1074/JBC.X600004200
CrossRef - Ideo H, Tsuchida A, Takada Y. Lectin-Based Approaches to Analyze the Role of Glycans and Their Clinical Application in Disease. International Journal of Molecular Sciences. 2024;25(18). doi:10.3390/ijms251810231
CrossRef - Peters K, Peters M. The Role of Lectin Receptors and Their Ligands in Controlling Allergic Inflammation. Front Immunol. 2021;12. doi:10.3389/fimmu.2021.635411
CrossRef - Bellande K, Bono JJ, Savelli B, Jamet E, Canut H. Plant Lectins and Lectin Receptor-Like Kinases: How Do They Sense the Outside? International Journal of Molecular Sciences. 2017;18(6):1164. doi:10.3390/ijms18061164
CrossRef - Bezerra RP, Conniff AS, Uversky VN. Comparative study of structures and functional motifs in lectins from the commercially important photosynthetic microorganisms. Biochimie. 2022;201:63-74. doi:10.1016/j.biochi.2022.07.004
CrossRef - De Coninck T, Van Damme EJM. Review: The multiple roles of plant lectins. Plant Science. 2021;313:111096. doi:10.1016/j.plantsci.2021.111096
CrossRef - Bojar D, Meche L, Meng G, et al. A Useful Guide to Lectin Binding: Machine-Learning Directed Annotation of 57 Unique Lectin Specificities. ACS Chem Biol. 2022;17(11):2993-3012. doi:10.1021/acschembio.1c00689
CrossRef - Olvera-Lucio FH, Riveros-Rosas H, Quintero-Martínez A, Hernández-Santoyo A. Tandem-repeat lectins: structural and functional insights. Glycobiology. 2024;34(7):cwae041. doi:10.1093/glycob/cwae041
CrossRef - Tsaneva M, Van Damme EJM. 130 years of Plant Lectin Research. Glycoconj J. 2020;37(5):533-551. doi:10.1007/s10719-020-09942-y
CrossRef - Mazalovska M, Kouokam JC. Plant-Derived Lectins as Potential Cancer Therapeutics and Diagnostic Tools. BioMed Research International. 2020;2020(1):1631394. doi:10.1155/2020/1631394
CrossRef - Ruwali P, Pandey N, Jindal K, Singh RV. Fenugreek (Trigonella foenum-graecum): Nutraceutical values, phytochemical, ethnomedicinal and pharmacological overview. South African Journal of Botany. 2022;151:423-431. doi:10.1016/j.sajb.2022.04.014
CrossRef - Faisal Z, Irfan R, Akram N, et al. The multifaceted potential of fenugreek seeds: From health benefits to food and nanotechnology applications. Food Science & Nutrition. 2024;12(4):2294-2310. doi:10.1002/fsn3.3959
CrossRef - Itoo AM, Paul M, Padaga SG, Ghosh B, Biswas S. Nanotherapeutic Intervention in Photodynamic Therapy for Cancer. ACS Omega. 2022;7(50):45882-45909. doi:10.1021/acsomega.2c05852
CrossRef - Rajender O, Pallavi H, Sultana R. Purification and characterization of a lectin from Trigonella foenum-graecum (fenugreek) seeds and its porphyrin binding studies. J Plant Biochem Biotechnol. 2024;33(3):341-352. doi:10.1007/s13562-024-00894-0
CrossRef - Osman M, Awadallah A, Konozy E. Isolation, Purification and Partial Characterization of Three Lectins from Tamarindus indica Seeds with a Novel Sugar Specificity. International journal of plant research. 2016;6:13-19.
- Lowry OliverH, Rosebrough NiraJ, Farr AL, Randall RoseJ. PROTEIN MEASUREMENT WITH THE FOLIN PHENOL REAGENT. Journal of Biological Chemistry. 1951;193(1):265-275. doi:10.1016/S0021-9258(19)52451-6
CrossRef - Zhang W, Peumans WJ, Barre A, et al. Isolation and characterization of a jacalin-related mannose-binding lectin from salt-stressed rice (Oryza sativa) plants. Planta. 2000;210(6):970-978. doi:10.1007/s004250050705
CrossRef - Kurokawa T, Tsuda M, Sugino Y. Purification and characterization of a lectin from Wistaria floribunda seeds. Journal of Biological Chemistry. 1976;251(18):5686-5693. doi:10.1016/S0021-9258(17)33112-5
CrossRef - Sanji AS, J. M, Kennedy JF, et al. Purification and characterization of lectin from Phyllanthus reticulatus (PRL) plant fruit inducing cytotoxic effect on T47D and SKBR3 breast cancer cell lines. International Journal of Biological Macromolecules. 2025;307:142006. doi:10.1016/j.ijbiomac.2025.142006
CrossRef - Laemmli UK. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature. 1970;227(5259):680-685. doi:10.1038/227680a0
CrossRef - Konozy EHE, Osman ME fadil M. Plant lectin: A promising future anti-tumor drug. Biochimie. 2022;202:136-145. doi:10.1016/j.biochi.2022.08.002
CrossRef - E.A. Pereira M, Kabat EA, Lotan R, Sharon N. Immunochemical studies on the specificity of the peanut (Arachis hypogaea) Agglutinin. Carbohydrate Research. 1976;51(1):107-118. doi:10.1016/S0008-6215(00)84040-9
CrossRef - Naeem A, Ahmad E, Ashraf MohdT, Khan RH. Purification and characterization of mannose/glucose-specific lectin from seeds of Trigonella foenumgraecum. Biochemistry (Moscow). 2007;72(1):44-48. doi:10.1134/S0006297907010051
CrossRef - Nasi A, Picariello G, Ferranti P. Proteomic approaches to study structure, functions and toxicity of legume seeds lectins. Perspectives for the assessment of food quality and safety. Journal of Proteomics. 2009;72(3):527-538. doi:10.1016/j.jprot.2009.02.001
CrossRef - Zhao J, He S, Tang M, et al. Low-pH induced structural changes, allergenicity and in vitro digestibility of lectin from black turtle bean (Phaseolus vulgaris L.). Food Chemistry. 2019;283:183-190. doi:10.1016/j.foodchem.2018.12.134
CrossRef - Naithani S, Komath SS, Nonomura A, Govindjee G. Plant lectins and their many roles: Carbohydrate-binding and beyond. Journal of Plant Physiology. 2021;266:153531. doi:10.1016/j.jplph.2021.153531
CrossRef - Tang C, Jiang X, Liu C, Washburn BK, Sathe SK, Rao Q. Effect of temperature on structural configuration and immunoreactivity of pH-stressed soybean (Glycine max) agglutinin. Food Chemistry. 2024;442:138376. doi:10.1016/j.foodchem.2024.138376
CrossRef - Peumans WJ, Van Damme EJ. Lectins as plant defense proteins. Plant Physiol. 1995;109(2):347-352. doi:10.1104/pp.109.2.347
CrossRef - Lis H, Sharon N. LECTINS AS MOLECULES AND AS TOOLS. Annual Review of Biochemistry. 1986;55(Volume 55, 1986):35-67. doi:10.1146/annurev.bi.55.070186.000343
CrossRef - Mitra N, Srinivas VR, Ramya TNC, Ahmad N, Reddy GB, Surolia A. Conformational Stability of Legume Lectins Reflect Their Different Modes of Quaternary Association: Solvent Denaturation Studies on Concanavalin A and Winged Bean Acidic Agglutinin. Biochemistry. 2002;41(29):9256-9263. doi:10.1021/bi020240m
CrossRef
Abbreviations List
ASF – Ammonium Sulphate Fraction
BSA – Bovine Serum Albumin
EDTA – Ethylenediaminetetraacetic Acid
HAU – Hemagglutination Unit
ICMR – Indian Council of Medical Research
MIC – Minimum Inhibitory Concentration
OD – Optical Density
PBS – Phosphate-Buffered Saline
PDT – Photodynamic Therapy
PMSF – Phenyl methyl sulfonyl Fluoride
RBC – Red Blood Cells
SD – Standard Deviation
SDS – Sodium Dodecyl Sulphate
SDS-PAGE – Sodium Dodecyl Sulphate–Polyacrylamide Gel Electrophoresis
Accepted on: 12-02-2026
Second Review by: Dr. Makhabbah Jamilatun
Final Approval by: Dr. Eugene A. Silow