Anticancer Potential of Dioscorea villosa Derived Diosgenin in PA-1 Ovarian Cancer Cells: Mechanistic Insights into Apoptosis and PI3K/Akt Signaling
Department of Pharmacology, Navsahyadri Institute of Pharmacy, Pune, India
Corresponding Author E-mail:: ajaykalephd@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3480
ABSTRACT:Ovarian cancer is among the deadliest gynecological cancer, primarily largely attributed to delayed clinical detection, significant metastatic abilities, coupled with reduced responsiveness to chemotherapy. These challenges have sparked interest in bioactive compounds derived from plants as potentially safer and more effective treatments. Diosgenin and dioscin, both steroidal saponins sourced mainly from Dioscorea species, have shown promise as anticancer agents with diverse mechanisms of action. Existing experimental studies assessing the anticancer potential of diosgenin and dioscin in ovarian cancer are outlined, with focus on their biological effects and underlying mechanisms. Studies conducted in vitro and in vivo indicate that diosgenin can inhibit the growth, migratory activity, and invasive capacity of ovarian cancer cells while promoting programmed cell death and cell cycle arrest by modulating essential molecular pathways, including PI3K/Akt/mTOR, PTEN, NF-κB, and Bcl-2 family proteins. Likewise, dioscin demonstrates notable anticancer effects mediated through the induction of apoptosis and autophagy, reducing metastasis, reversing multidrug resistance, and improving chemosensitivity. Advances in nanocarrier delivery systems have also enhanced the bioavailability and effectiveness of these compounds. Despite the promising experimental results, clinical validation is still lacking. Overall, diosgenin and dioscin are promising natural options for ovarian cancer treatment, highlighting the need for additional studies on their mechanisms and clinical trials to support their development as viable anticancer therapies.
KEYWORDS:Apoptosis; Diosgenin; Ovarian cancer; PI3K/Akt signaling pathway; Steroidal saponins
Introduction
Ovarian cancer, a significant threat to women’s health globally, poses considerable challenges. According to recent global cancer statistics, there were approximately 324,603 new cases of ovarian cancer diagnosed worldwide in 2022, with an age-standardized incidence rate of about 6.7 per 100,000 women, making it one of the most commonly diagnosed malignancies in women globally. Additionally, ovarian cancer was responsible for an estimated 206,956 deaths in the same year, reflecting its high fatality burden among gynecological cancers. Ovarian cancer accounts for a substantial number of new cases and deaths annually and remains one of the leading causes of gynecological cancer related mortality worldwide. The high incidence and mortality rates are largely attributed to late stage diagnosis and the absence of reliable early detection methods. Its high mortality rate stems from the insidious nature of the disease, often lacking early symptoms, compounded by the lack of dependable early diagnostic tools, leading to diagnoses at advanced stages. Even with improvements in surgical and chemotherapeutic approaches, the five year survival rate of ovarian cancer patients remains disappointingly low. Standard treatment typically involves surgical removal of tumour, succeeded by chemotherapy employing platinum-based drugs, which can initially shrink tumour. However, recurrence is common, and chemoresistance frequently develops, resulting in treatment failures and poor patient outcomes. These issues underscore the pressing need for novel treatments that are both effective and less toxic.1-2
Recently, there’s been growing interest in natural products and plant-based compounds as potential cancer therapeutics. Phytochemicals, with their diverse structures, have the capacity to target numerous cellular processes, making them appealing for cancer therapy. Unlike traditional chemotherapy, many plant-based compounds exhibit reduced toxicity and can influence various signaling pathways involved in cancer growth, metastasis, and drug resistance. Notably, steroidal saponins have garnered attention for their promising anticancer properties.
Diosgenin and dioscin, both steroidal saponins primarily found in the Dioscorea plant genus, are of particular interest. Diosgenin, a steroidal sapogenin, serves as a crucial precursor for synthesizing various steroidal drugs and has been investigated for its favourable effects such as anti-inflammatory, antioxidant, and tumour suppressive activities. Similarly, dioscin, the glycosylated form of diosgenin, has demonstrated robust biological effects and significant anticancer activity in numerous studies.
Research indicates that both diosgenin and dioscin combat cancer through multiple mechanisms, positioning them as promising candidates for cancer treatment. Preclinical studies suggest that diosgenin effectively suppresses the proliferation of various cancer cell lines including ovarian cancer, by inducing cell death, inducing cell cycle arrest while inhibiting cell migration and invasiveness. These effects are associated with the regulation of critical signaling cascades, including PI3K/Akt/mTOR, NF-κB, and Bcl-2 family proteins, as well as the activation of tumour suppressor mechanisms like PTEN.3
Likewise, dioscin aids in tumour suppression by enhancing cell death and autophagy, preventing metastasis, reversing multidrug resistance, and boosting sensitivity to chemotherapy. The ability of these compounds to simultaneously target multiple pathways is particularly advantageous in complex diseases like ovarian cancer. However, the clinical use of diosgenin and dioscin is presently constrained by issues including low solubility, limited bioavailability, and a lack of sufficient clinical studies. Emerging advancements in drug delivery methods, particularly nanocarrier systems, hold promise in addressing these challenges by enhancing the efficacy of these compounds in vivo.
Consequently, a comprehensive understanding of the anticancer mechanisms, experimental evidence, and future potential of diosgenin and dioscin is essential. This review endeavours to provide a detailed overview of current preclinical findings concerning the anticancer properties of diosgenin and dioscin, specifically in the context of ovarian cancer, focusing on their mechanisms of action, therapeutic significance, and their potential as viable natural leads for developing new ovarian cancer treatments.4-6
Methodology
A comprehensive literature search was performed to collect relevant studies on the anticancer potential of diosgenin and dioscin in ovarian cancer, with a special focus on PA-1 ovarian cancer cells. Electronic databases including PubMed, ScienceDirect, and Google Scholar were searched systematically. The search was conducted using combinations of keywords such as diosgenin, dioscin, dioscorea, PA-1 cells, ovarian cancer, apoptosis, cell cycle arrest, PI3K/Akt/mTOR, metastasis, autophagy, and chemoresistance. Articles published up to 2025 were considered for inclusion.
The selected literature mainly included original research articles, in vitro studies, in vivo studies, and mechanistic studies reporting the biological effects and molecular pathways of diosgenin and dioscin in ovarian cancer models. Studies were included if they were published in English and contained experimental evidence related to ovarian cancer, PA-1 cell line, apoptosis, signaling pathways, metastasis, or drug resistance. Articles were excluded if they were unrelated to ovarian cancer, lacked mechanistic relevance, were not available as full text, or were non-scientific reports. The final set of included studies was analysed and summarized to highlight key mechanisms and therapeutic potential.
Lack of Focused Research in PA-1 Ovarian Cancer Cells
While diosgenin and dioscin from Dioscorea villosa have been extensively studied for their anticancer properties in various cancer types, there is a notable lack of focused research on their effects in PA-1 ovarian cancer cells, a model that closely represents human ovarian teratocarcinoma. Most existing studies are limited to general ovarian cancer cell lines or other cancer types, leaving a gap in understanding the specific mechanistic actions, apoptotic pathways, and PI3K/Akt signaling modulation in PA-1 cells. This review addresses this gap by critically evaluating the preclinical evidence of diosgenin’s activity in PA-1 cells and highlighting potential molecular targets for future translational studies.7-8
Origin of PA-1 Cells
The PA-1 cell line is a well-known human ovarian cancer cell line derived from a teratocarcinoma in a female patient. These cells display an epithelial-like appearance and have genetic and phenotypic traits associated with ovarian cancers. PA-1 cells are noted for their high growth rate, consistent culture stability, and reliable reactions to anticancer treatments, which makes them ideal for research purposes. Given their human origin and tumorigenic characteristics, PA-1 cells serve as a biologically relevant model for exploring the cellular and molecular aspects of ovarian cancer pathophysiology.9-10
Justification for Usage in Ovarian Malignancy Research
PA-1 cells are frequently utilized in ovarian cancer studies due to their representation of several key characteristics of malignant cells, including resistance to apoptotic cell death, dysregulated cell cycle progression, and modified intracellular signaling pathways. Notably, PA-1 cells express significant molecular targets commonly associated with ovarian cancer, like BCL-2 family proteins, elements of the PI3K/Akt signaling cascade and caspase enzymes. This renders them particularly useful for assessing compounds that function by inducing apoptosis and inhibiting survival pathways. Furthermore, PA-1 cells are responsive to both traditional chemotherapy agents and experimental phytochemicals, allowing for comparative analyses between established drugs and new therapeutic candidates. Their sensitivity to steroidal compounds also enhances their appropriateness for studying plant-derived substances, such as diosgenin and other steroidal saponins found in Dioscorea villosa root extract.11-12
Importance of In-Vitro Anticancer Screening
In-vitro models are essential for the preliminary evaluation of anticancer compounds, with PA-1 cells serving as a dependable and consistent screening system. These cells are frequently used in cytotoxicity tests (such as MTT and SRB), assays for detecting apoptosis (including Annexin V/PI and caspase activity), cell cycle analysis, and measurements of oxidative stress. The capability of PA-1 cells to exhibit quantifiable apoptotic and cell cycle reactions enables accurate evaluation of the effectiveness and mechanisms of various compounds. For research involving Dioscorea villosa root extract, PA-1 cells provide an excellent model to investigate mitochondrial-mediated apoptosis, the regulation of BCL-2 family proteins, inhibition of the PI3K/Akt pathway, and cytotoxicity induced by oxidative stress.13-15 Thus, the PA-1 cell line is a scientifically significant and effective in-vitro model for assessing plant-derived anticancer agents and exploring their potential therapeutic effects against ovarian cancer.
Dioscorea villosa phytochemistry overview
Dioscorea villosa L. or wild yam, is a medicinal plant celebrated for its diverse phytochemical composition, especially its steroidal saponin content. These bioactive compounds are believed to underpin many of the plant’s beneficial biological activities, including anti-inflammatory, antioxidant, and tumour suppressive effects. Diosgenin and dioscin, two steroidal saponins found in Dioscorea villosa, have garnered considerable scientific interest due to their structural resemblance to endogenous steroids and their varied biological activities.
Steroidal Saponins
Steroidal saponins are naturally derived glycosides consisting of a steroidal aglycone linked to one or multiple sugar units. Within Dioscorea villosa, these compounds are vital secondary metabolites that enhance the plant’s therapeutic capabilities. They possess surface-active traits and can interact with cellular membranes, affecting membrane permeability and intracellular signaling. In the field of cancer research, steroidal saponins have been shown to influence essential pathways linked to cell growth, programmed cell death, and oxidative stress, making them compelling candidates for anticancer drug development.16-17
Diosgenin
Diosgenin is a steroidal sapogenin obtained through the hydrolysis of steroidal saponins and is regarded as the primary bioactive compound in Dioscorea villosa. Its structure closely resembles that of natural steroid hormones, allowing it to interact with various molecular targets. Pharmacological research has indicated that diosgenin manifests anticancer properties through several mechanisms, including promoting apoptosis, regulating BCL-2 family proteins, suppressing the PI3K/Akt signaling cascade and inducing cell-cycle arrest. In ovarian cancer models, diosgenin has demonstrated the ability to decrease cellular viability and trigger mitochondrial mediated apoptosis, highlighting its relevance in cancer treatment research.18-20
 |
Figure 1: Chemical structure of diosgenin
|
Dioscin
Dioscin is a glycosylated derivative of diosgenin and is another significant steroidal saponin found in Dioscorea villosa. It demonstrates diverse biological activities, encompassing anticancer, anti-inflammatory, and antioxidant effects. Dioscin has been shown to exert cytotoxic effects by promoting apoptosis, modulating levels of reactive oxygen species, and disrupting survival signaling pathways. Its capacity to increase oxidative stress and trigger apoptotic processes positions dioscin as a valuable compound in the investigation of plant-derived anticancer therapies.21
Phytochemical Constituents of Dioscorea villosa
Table 1: Major phytochemical constituents of Dioscorea villosa and their bioactivities
| Compound | Chemical Class | Reported Bioactivity | Reference |
| Diosgenin | Steroidal sapogenin | Induces apoptosis, inhibits PI3K/Akt, causes cell cycle arrest | Sethi et al., 2019 |
| Dioscin | Steroidal saponin (glycoside of diosgenin) | Promotes apoptosis, oxidative stress mediated cytotoxicity | Guo & Ding, 2018 |
| Yamogenin | Steroidal sapogenin | Antioxidant, cytotoxic in cancer cells | Carbohydrate Research, 2013 |
| Other saponins | Steroidal sapogenin | Anti-inflammatory, antioxidant | Carbohydrate Research, 2013 |
Mechanism of Action
The anticancer properties of Dioscorea villosa root extract are mainly attributed to its high content of steroidal saponins, particularly diosgenin and dioscin. These active phytochemicals exert potent cytotoxic effects through various linked molecular pathways that collectively suppress the proliferation and viability of ovarian cancer cells. Studies from both laboratory and computational research indicate that compounds from Dioscorea villosa target critical features of cancer, including resistance to programmed cell death, abnormal intracellular signaling, uncontrolled cell cycle progression, and oxidative stress imbalances. These findings underscore the considerable therapeutic potential of Dioscorea villosa derived compounds in treating ovarian cancer.
Apoptosis induction
Apoptosis is a form of programmed cell death that is tightly regulated process, is frequently disrupted in ovarian cancer, facilitating tumor growth, progression, and resistance to chemotherapy. Diosgenin has been observed to induce apoptosis in ovarian cancer cells primarily by activating the intrinsic mitochondrial pathway. This treatment alters mitochondrial membrane potential disruption, leading to the release of cytochrome c into the cytoplasm, activating caspases that result in DNA fragmentation and cell death. Studies conducted on the PA-1 ovarian malignancy cell line consistently demonstrated that diosgenin induced apoptosis was associated with a notable reduction in cell viability and distinct morphological alterations in apoptotic cells, such as cell shrinkage, chromatin condensation, and membrane blebbing .22-23
Regulation of bcl-2 family protein
BCL-2 family proteins are crucial regulators of mitochondrial membrane permeability and influencing cell fate. Ovarian cancer cells often exhibit elevated levels of apoptosis inhibiting proteins such as BCL-2, which promote cell viability and contribute to treatment resistance. Diosgenin has been shown to adjust the expression of BCL-2 family proteins through the suppression of apoptosis inhibiting proteins and the enhancement of pro-apoptotic proteins, including Bax. This alteration in the Bax/BCL-2 ratio promotes apoptosis and increases the susceptibility of ovarian cancer cells, including PA-1 cells, to programmed cellular demise.
Suppression of the PI3k/AKT signaling pathway
The PI3K/Akt pathway plays a central role in regulating cell growth, survival, and metabolism and is frequently hyperactivated in ovarian malignancies. Continuous activation of this pathway enhances tumor aggressiveness and fosters resistance to apoptosis. Diosgenin has been reported to inhibit PI3K/Akt signaling by reducing Akt phosphorylation, thereby blocking pro-survival pathways. This inhibition leads to decreased cell proliferation and heightened apoptotic responses. Molecular docking studies suggest that diosgenin may directly bind to PI3K and related proteins, elucidating its molecular inhibitory capabilities.
Cell cycle arrest
Additionally, unchecked cellular division cycle progression is a typical characteristic of cancer cells. Diosgenin has been reported to halt the progression of the cellular division cycle at specific phases, usually G0/G1 or G2/M, depending on the cellular context. This action involves the regulation of cyclins, cyclin-dependent kinases, and checkpoint proteins, limiting cancer cellular proliferation while enhancing apoptotic potential. In PA-1 ovarian malignancy cells, diosgenin’s ability to disrupt cell cycle progression significantly enhances its overall anti-proliferative effects.24-26
Modulation of oxidative stress
Oxidative stress has a multifaceted impact on cancer development, affecting both tumor growth and response to treatment. Diosgenin has been shown to affect the intracellular concentrations of reactive oxygen species (ROS), resulting in ROS mediated cytotoxicity in cancer cells. High ROS levels can cause mitochondrial dysfunction and activate apoptotic signaling pathways. By selectively generating oxidative stress in cancer cells while minimizing harm to normal cells, Dioscorea villosa derived compounds further underscore their therapeutic potential in combating ovarian cancer.27-28
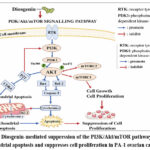 |
Figure 2: Diosgenin-mediated suppression of the PI3K/Akt/mTOR pathway triggers mitochondrial apoptosis and suppresses cell proliferation in PA-1 ovarian cancer cells
|
Limitations
Current evidence is largely based on in vitro experiments primarily conducted on PA-1 ovarian malignancy cells.
Lack of in-vivo validation limits understanding of efficacy, toxicity, and pharmacokinetics.
Low bioavailability of diosgenin due to poor solubility and rapid metabolism restricts clinical translation.
Future Perspectives
Conduct in-vivo animal studies to confirm anticancer efficacy and safety.
Develop nano-formulations (nanoparticles, liposomes) to improve stability, solubility, and targeted delivery.
Explore combination therapy with conventional chemotherapeutic agents to improve therapeutic effectiveness and counteract resistance.
Investigate molecular synergy and dose optimization for improved therapeutic outcomes.
Discussion
Ovarian malignancy continues to be among the deadliest gynecological cancers, largely owing to delayed detection and resistance to chemotherapy. Despite advances in surgical management and platinum-based chemotherapeutic regimens, long-term survival rates remain unsatisfactory, primarily due to tumor recurrence and the development of chemoresistance. This highlights the urgent need for alternative or adjunct therapeutic strategies with improved safety and efficacy profiles. Plant derived steroidal saponins from Dioscorea villosa, particularly diosgenin and dioscin, exhibit notable anticancer activity. Diosgenin triggers programmed cell death in ovarian malignancy cells by regulating apoptotic proteins and suppressing the PI3K/Akt pathway, a key driver of tumor progression. Mechanistically, diosgenin has been reported to modulate the balance between pro-apoptotic and anti-apoptotic proteins, thereby activating intrinsic apoptotic pathways. Suppression of PI3K/Akt signaling is particularly significant, as aberrant activation of this pathway contributes to enhanced cell survival, proliferation, and resistance to chemotherapy in ovarian cancer. Inhibiting Akt phosphorylation may therefore sensitize malignant cells to apoptosis and reduce their proliferative capacity. Dioscin further enhances anticancer activity through the induction of cell-cycle arrest and oxidative stress mediated cytotoxicity, with relative selectivity toward cancer cells. Increased reactive oxygen species (ROS) production can disrupt mitochondrial integrity and activate apoptotic cascades. Its relative selectivity toward malignant cells may offer an advantage over conventional chemotherapeutic agents that often produce systemic toxicity. Although current evidence is largely preclinical, these results underscore the potential of Dioscorea villosa bioactive compounds as effective therapeutic or adjunct agents. Nevertheless, additional in vivo and clinical investigations are needed to validate their efficacy, safety, and optimal dosing for ovarian cancer treatment.
Conclusion
Ovarian cancer poses considerable therapeutic challenges arising from delayed diagnosis, aggressive nature, and common resistance to chemotherapy. While traditional chemotherapy is effective, it is often hampered by systemic side effects and diminishing long-term effectiveness. Bioactive compounds derived from plants, especially those from Dioscorea villosa, present promising alternatives owing to their multimodal mechanisms and relative safety. The steroidal saponins diosgenin and dioscin have demonstrated strong anticancer properties in ovarian malignancy through in vitro and in silico research, including studies on PA-1 cell line models. These compounds promote apoptosis via mitochondrial pathways, modulate BCL-2 family proteins, PI3K/Akt signaling, trigger halting of the cellular division cycle, and manage oxidative stress, all contributing to the reduction of cancer cell growth. Importantly, herbal bioactive compounds may also enhance the efficacy of modern chemotherapeutic agents. Several studies suggest that phytoconstituents such as diosgenin can sensitize cancer cells to platinum-based drugs like Cisplatin and taxane based agents such as Paclitaxel by modulating drug resistance pathways, including downregulation of P-glycoprotein and inhibition of survival signaling cascades. This synergistic interaction may reduce the required dose of chemotherapeutic drugs, thereby minimizing toxicity and adverse effects. Furthermore, antioxidant and anti-inflammatory properties of herbal compounds may help protect normal cells from chemotherapy-induced oxidative damage, improving overall treatment tolerance. Globally, ovarian cancer continues to show a substantial incidence and mortality rate, particularly due to late stage diagnosis and limited early detection strategies, emphasizing the urgent need for improved therapeutic options. Understanding these mechanisms underscores the potential of Dioscorea villosa derived compounds as natural anticancer treatments. Unlike many conventional chemotherapeutic drugs that typically act on a single molecular target and are associated with significant systemic toxicity, herbal-derived compounds often exhibit multi-targeted mechanisms of action with comparatively lower adverse effects, which may contribute to better tolerability and reduced development of resistance. Further comprehensive preclinical and clinical studies are necessary to confirm these effects in vivo and assess their clinical relevance, ultimately aiding the development of new plant based approaches for managing ovarian cancer.
Acknowledgement
The authors would like to acknowledge the Department of Pharmacology, Navsahyadri Institute of Pharmacy, for providing the necessary facilities to conduct this review.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest-
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contribution
Harshada Pandit : Conceptualization, literature review, and manuscript preparation including Abstract, Introduction, Methodology, Lack of Focused PA-1 Cell Line, Origin of PA-1 Cell Line, Importance of In-Vitro Anticancer Screening, Dioscorea villosa Phytochemistry Overview, Steroidal Saponins, Diosgenin, Mechanism of Action, Discussion, and Conclusion.
Kishor Otari : Supervision, critical revision of the manuscript for important intellectual content, and overall guidance.
Ajay Kale : Literature collection, reference management, manuscript formatting, and proofreading.
All authors contributed to the conception, design, literature search, and writing of the manuscript. All authors have read and approved the final manuscript.
References
- Havasi A, Cainap SS, Havasi AT, Cainap C. Ovarian cancer insights into platinum resistance and overcoming it. Medicina (Kaunas). 2023;59(3):544. doi:10.3390/medicina59030544
CrossRef - Madar S, Amor RE, Furman-Assaf S, Friedman E. Innovative approaches to early detection of cancer-transforming screening for breast, lung, and hard-to-screen cancers. Cancers (Basel). 2025;17(11):1867. doi:10.3390/cancers17111867.
CrossRef - Ortiz M, Wabel E, Horibata S. Mechanisms of chemotherapy resistance in ovarian cancer. Cancer Drug Resist. 2022;5:304-316. doi:10.20517/cdr.2021.147
CrossRef - Ren Y, Xu R, Wang Y, Su L, Su J. Global, regional, and national burden of ovarian cancer in women aged 45+ from 1990 to 2021 and projections for 2050: a systematic analysis based on the 2021 global burden of disease study. J Cancer Res Clin Oncol. 2025;151(8):225. doi:10.1007/s00432-025-06277-9
CrossRef - Fang F, Zhang X, Fang Y. Diosgenin inhibits proliferation and migration of ovarian cancer cells and induces apoptosis via upregulation of PTEN. Chem Biol Drug Des. 2024. doi:10.1111/cbdd.14459
CrossRef - Wang MY, Zhang YN, Ni SL, Sun M, Wu QL, Wu XL, Chen Q, Wang SJ. The anticancer activity of dioscin: an update and future perspective. Med Oncol. 2025;42:63. doi:10.1007/s12032-024-02572-6
CrossRef - Pandey P, Ramniwas S, Baldaniya L, Lakhanpal S, Mahmood D, et al. An updated review of molecular mechanisms implicated with the anticancer potential of diosgenin and its nanoformulations. Drug Des Devel Ther. 2025;19:2205-2227. doi:10.2147/DDDT.S502322
CrossRef - Liu L, Bao Y, Chen Y, Ding M, Liang H, Li B, et al. Anticancer effects and mechanisms of genus Dioscorea: a review. Phytomedicine. 2025;148:157328. doi:10.1016/j.phymed.2025.157328
CrossRef - Basavarajappa DS, Niazi SK, Bepari A, Assiri RA, Hussain SA, Muzaheed, Nayaka S, Hiremath H, Rudrappa M, Hugar A, et al. Efficacy of Penicillium limosum strain AK-7 derived bioactive metabolites on antimicrobial, antioxidant, and anticancer activity against human ovarian teratocarcinoma (PA-1) cell line. Microorganisms. 2023;11(10):2480. doi:10.3390/microorganisms11102480.
CrossRef - Sethi G, Shanmugam MK, Warrier S, Merarchi M, Arfuso F, Kumar AP, et al. Proapoptotic and anticancer properties of diosgenin: a comprehensive and critical review. Nutrients. 2019;10(5):645. doi:10.3390/nu10050645
CrossRef - Teekaraman D, Elayapillai SP, Viswanathan MP, Jagadeesan A. Quercetin inhibits human metastatic ovarian cancer cell growth and modulates components of the intrinsic apoptotic pathway in PA-1 cell line. Chem Biol Interact. 2019;300:91-100. doi:10.1016/j.cbi.2019.03.012
CrossRef - Reddy PRK, Thiruvanavukkarasu P, Rajesh S, Karunakaran S, Hari R. Effect of ethanolic extract of Carica papaya leaves on cytotoxicity and apoptotic potential in human ovarian cancer PA-1 cells. Pharmacogn Mag. 2020;16(5 Suppl):524-530. doi:10.4103/pm.pm_117_20
CrossRef - Arulmozhi A, Jayaprakash A, Behera A, Malakondaiah S, Dharmalingam Jothinathan MK. Evaluation of in vitro anticancer activity in ovarian cancer PA-1 cells and zebrafish embryo toxicity of Parmelia perlata ethanolic extract. Uttar Pradesh J Zool. 2024;45(13):28-40. doi:10.56557/upjoz/2024/v45i134133
CrossRef - Guo X, Ding X. Dioscin suppresses the viability of ovarian cancer cells by regulating the VEGFR2 and PI3K/AKT/MAPK signaling pathways. Oncol Lett. 2018;15(6):9537-9542. doi:10.3892/ol.2018.8454
CrossRef - Choi J, Jeong M, Lee K, Kim J, Lee WH, Park I, Kwon HC, Choi J. Sedum middendorffianum Maxim induces apoptosis and inhibits the invasion of human ovarian cancer cells via oxidative stress regulation. Antioxidants (Basel). 2023;12(7):1386. doi:10.3390/antiox12071386
CrossRef - Rinne N, Christie EL, Ardasheva A, Kwok CH, Demchenko N, Low C, Tralau-Stewart C, Fotopoulou C, Cunnea P. Targeting the PI3K/AKT/mTOR pathway in epithelial ovarian cancer: therapeutic treatment options for platinum-resistant ovarian cancer. Cancer Drug Resist. 2021. doi:10.20517/cdr.2021.05
CrossRef - Bandopadhyay S, Anand U, Gadekar VS, Jha NK, Gupta PK, Behl T, Kumar M, Radha, Shekhawat MS, Dey A. Dioscin: a review on pharmacological properties and therapeutic values. Biofactors. 2022. doi:10.1002/biof.1815
CrossRef - Xiong C, Yan B, Xia S, Yu F, Zhao J, Bai H. Tilianin inhibits human ovarian cancer (PA-1) cell proliferation by blocking the cell cycle, inducing apoptosis, and inhibiting the JAK2/STAT3 signaling pathway. Saudi J Biol Sci. 2021;28(9):4900-4907. doi:10.1016/j.sjbs.2021.06.033
CrossRef - Gupta AK, Sharma M, Gupta R, Kumar S. Insights into the phytochemistry and pharmacology of Dioscorea species: a review. J Ethnopharmacol. 2021;273:113957. doi:10.1016/j.jep.2021.113957
CrossRef - Bhandari MR, Kaul S, Dhar MK. A systematic review on ethnobotany, phytochemistry, and pharmacology of Dioscorea bulbifera S Afr J Bot. 2024;170:367-393. doi:10.1016/j.sajb.2024.05.014.
CrossRef - Bouabdallah S, Al-Maktoum A, Amin A. Steroidal saponins: naturally occurring compounds as inhibitors of the hallmarks of cancer. Cancers (Basel). 2023;15(15):3900. doi:10.3390/cancers15153900
CrossRef - Sobolewska D, Galanty A, Grabowska K, Makowska-Wąs J, Wróbel-Biedrawa D, Podolak I. Saponins as cytotoxic agents: an update (2010–2018). Part I—steroidal saponins. Phytochem Rev. 2020;19:139-189.
CrossRef - Bhardwaj N, Tripathi N, Goel B, Jain SK. Anticancer activity of diosgenin and its semisynthetic derivatives: role in autophagy-mediated cell death and induction of apoptosis. Mini Rev Med Chem. 2021;21(13):1646-1665. doi:10.2174/1389557521666210105111224
CrossRef - Mazzio E, Almalki A, Darling-Reed SF, Soliman KF. Effects of wild yam root (Dioscorea villosa) extract on the gene expression profile of triple-negative breast cancer cells. Cancer Genomics Proteomics. 2021;18(6):735-755. doi:10.21873/cgp.20294
CrossRef - Elsori D, Alshabrmi FM, Alharbi AM, Saeed M, Singh A, Pandey P, Rab SO, Khan F. Anticancer potential of natural bioactive compound saponins in glioblastoma multiforme. Front Pharmacol. 2025. doi:10.3389/fphar.2025.1712599
CrossRef - Palanisamy CP, Cui B, Zhang H, Panagal M, Paramasivam S, Chinnaiyan U, Jeyaraman S, Murugesan K, Rostagno M, Sekar V, Natarajan SP. Anti-ovarian cancer potential of phytocompounds and extracts from South African medicinal plants and their role in the development of chemotherapeutic agents. Am J Cancer Res. 2021;11(5):1828-1844.
- Wang P, Wang Y, Liu S, Wang K, Yao Y, Liu W, Li D. Yamogenin-induced cell cycle arrest, oxidative stress, and apoptosis in human ovarian cancer cell line. Molecules. 2022;27(23):8181. doi:10.3390/molecules27238181
CrossRef - Wang Q, Zhang L, Xiao Y, Ghaemnezhad A. Diosgenin promotes cisplatin-induced apoptosis through oxidative DNA damage in A549 non-small cell lung cells. Cell Biochem Funct. 2022;40(7). doi:10.1002/cbin.11862.
CrossRef
Abbreviation
Akt: Protein Kinase B; BCL-2: B-cell lymphoma 2; mTOR: Mammalian target of rapamycin; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; PA-1: Human ovarian teratocarcinoma cell line; PI3K: Phosphoinositide 3-kinase; PTEN: Phosphatase and tensin homolog; ROS: Reactive oxygen species; SRB: Sulforhodamine B; DNA: Deoxyribonucleic acid; Bax: Bcl-2-associated X protein; CDK4: Cyclin-dependent kinase 4; MDR: Multidrug resistance; RTK: Receptor tyrosine kinase; PDK1: Phosphoinositide dependent kinase-1.
Accepted on: 08-03-2026
Second Review by: Dr Sunil Chaudhry
Final Approval by: Dr. Eugene A. Silow









