AI-Enabled Ocular Drug Delivery Systems for the Management of Eye Infections
Research and Development-Biotechnology, KG College of Arts and Science, Coimbatore, Tamil Nadu, India.
Corresponding Author E-mail- g.neethirajan@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3487
ABSTRACT:Eye infection is a major health issue in the world, and it is considered a major cause of preventable visual impairment. Various eye infection therapies are available, but the delivery of these drugs is a challenging task due to anatomical and physiological limitations, leading to poor drug bioavailability and therapeutic response. The traditional eye infection drug delivery system has limitations, such as precorneal loss of the drug, frequent instillation, and poor patient compliance. Recent developments in ophthalmology have introduced various novel eye infection drug delivery systems, such as nanoparticles, niosomes, in situ gelling systems, contact lens-based drug delivery systems, micro-needle technology, and eye infection inserts and implants, including stimuli-sensitive systems. Along with these developments, artificial intelligence (AI) has been integrated into ophthalmology, enabling data science approaches for eye infection therapy, such as formulation optimization, eye infection pharmacokinetics, and selection of anti-microbial agents. Significant potential lies in AI-assisted modelling and machine learning techniques in the development, efficiency, and application of advanced drug delivery systems for the eyes. This review aims to critically discuss the latest developments in advanced drug delivery systems for the eyes in the management of eye infections and the role of AI in improving the accuracy of diagnosis, therapeutic targeting, and therapeutic outcomes. Overall, the application of advanced drug delivery systems and AI has immense potential in improving therapeutic outcomes in eye infections.
KEYWORDS:Artificial Intelligence; Antimicrobial Therapy; Eye Infections; Nanoparticles-in-situ Gels; Niosomes Ocular Drug Delivery; Sustained Release Systems
Introduction
Ocular infections are a noteworthy public healthiness issue and a primary cause of preventable visual impairment. Infections such as conjunctivitis, keratitis, blepharitis, and endophthalmitis can be caused by bacterial, viral, fungal, or parasitic organisms and can rapidly progress if not properly managed, leading to corneal scarring, vision loss, and blindness.1 The growing problem of antimicrobial resistance and recurrent infections further makes the proper management of ocular infectious diseases challenging.2
Pharmacotherapy of ocular infections is made difficult by the functional and biological barricades of the eye, which are naturally designed to defend the eye from external injury. These obstacles, such as tear turnover, blinking, nasolacrimal drainage, corneal epithelial tight junctions, conjunctiva blood movement, and the blood-retinal blockage, greatly reduce the absorption and retention of ocular drugs.3,4 Consequently, the conventional ocular dosage paperwork inclusive of eye drops, suspensions, and ointments commonly result in much less than 5% of the drug achieving the goal tissues. This low bioavailability of the drug requires common dosing, that could lead to terrible patient compliance and unfavourable reactions.5
Topical administration has been the most frequently used route for the management of anterior segment ocular infections, as it is non-invasive and easy to perform. However, the rapid elimination of drugs from the precorneal surface and poor permeability across the cornea have been found to limit the therapeutic efficacy to a great extent.6 Systemic administration is less effective for localized ocular infections and may also cause systemic toxicity. While invasive routes of administration, such as intravitreal injections, are more effective for posterior segment infections but pose risks of complications and are less acceptable to patients.7
As a result of these challenges, there has been intense research aimed at the development of advanced ocular drug delivery systems. This can provide enhanced drug residence time, improved corneal and conjunctival penetration, and sustained and site-specific antimicrobial delivery. It has become one of the main aims in ocular therapy. Recent developments in ocular drug delivery have thus been directed towards nanotechnology-based carrier systems. These include as nanoparticles, liposomes, niosomes, in-situ gelling systems, drug-delivering contact lenses, microneedle-based ocular drug delivery systems, ocular inserts, biodegradable implants and smart delivery systems.8-12
Among these systems, niosomal vesicles have received increasing attention in recent years due to their improved chemical stability, cost-effectiveness, ease of formulation, and proven potential to improve ocular bioavailability and therapeutic outcomes in infectious conditions.13
Parallel to the development of drug delivery systems, the application of artificial intelligence (AI) has also received importance in the field of pharmaceuticals and ophthalmology. Machine learning and modelling have been increasingly used to optimize formulation development, ocular pharmacokinetics, antimicrobial selection, and personalized therapeutic approaches.14, 15 The combination of AI and ocular drug delivery systems provides new avenues to efficiently develop treatments minimize trial-and-error approaches for ocular infections is shown in Figure 1.
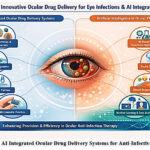 |
Figure 1: AI Integrated Ocular Drug Delivery Systems for Anti-Infective Therapy
|
Schematic picture of superior drug shipping structures such as nanoparticles, niosomes, in situ gels, and microneedles that are included with artificial intelligence to optimize drug formulations, tailor treatments to individual sufferers, and maximize drug efficacy.
The reason of this narrative assessment is to significantly verify the latest developments in novel ocular drug transport structures for the remedy of eye infections. This focuses on nanotechnology-based delivery systems, niosome carriers, and smart delivery systems. In addition, this review will also discuss the future prospects of artificial intelligence in improving ocular drug distribution and antimicrobial therapy.
Barriers in Drug Delivery
Ocular infections are difficult to control because of the special anatomical and physiological boundaries of the attention, which restriction the penetration and retention of the drug on the infection website online.
Pre-corneal Barriers – Rapid turnover of tears, blinking, and nasolacrimal drainage cause rapid clearance of topically implemented tablets, as a result reducing the house time of the drug at the ocular surface and its bioavailability.16
Cornea and Conjunctiva Barriers-The multi-layered structure of the cornea inhibits the permeation of many antimicrobials, In addition, the clearance of drugs is further increased by the blood and lymphatic circulation of the conjunctiva.17
Posterior Segment Barriers-The blood-retinal barrier restricts the penetration of systemically administered tablets into the posterior section, making the remedy of extreme infections like endophthalmitis extra tough18 and it has been shown in Figure 2.
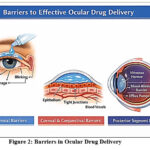 |
Figure 2: Barriers in Ocular Drug Delivery
|
It depicts diverse limitations in ocular drug transport, which include precorneal, corneal/conjunctival, and posterior segment limitations that restrict drug absorption, retention, and delivery to the focused sites in the attention.
Nanotechnology-Based Drug Distribution Systems
Nanotechnology has been recognized as a revolutionary tool in the delivery of ocular drugs. This technology provides several benefits, including controlled release of drugs, increased permeability, and improved stability of the formulation.
Polymeric/Lipid Nanoparticles-Polymeric nanoparticles and lipid nanocarriers improve corneal retention and antimicrobial activity. Chitosan-based nanoparticles have mucoadhesive and antimicrobial properties, leading to improved therapeutic effects.19, 20
Liposomes-Liposomes are biocompatible vesicular carriers which might be capable of encapsulating water-soluble in addition to fats-soluble pills. These vendors have proven extended house time in the attention and decreased toxicity.21
Solid Lipid Nanoparticles and Nanostructured Lipid Carriers-Solid lipid nanoparticles (SLNs) and nanostructured lipid providers (NLCs) have shown advanced drug loading capability, stability, and sustained launch traits, which lead them to appropriate for the remedy of chronic ocular infections.22
In Situ Gelling Systems
In-situ gelling systems change from sol to gel in response to biological stimuli, creating a gel structure that increases the ocular residence time.23 Thermo-sensitive and pH-sensitive gels containing antimicrobials have demonstrated high efficacy in conjunctivitis and keratitis infections.24
Contact Lens-Mediated Drug Delivery
Drug-releasing contact lenses facilitate sustained ocular delivery of antimicrobials, with newer developments like molecular imprinting and vitamin E diffusion barriers for controlled release.25, 26
Microneedle-Based and Minimally Invasive Approaches
Microneedle-based drug distribution systems facilitate targeted drug transport to the deeper tissues of the eye with minimal invasiveness.27, 28
Ocular Inserts and Implants
Ocular inserts and biodegradable implants facilitate sustained antimicrobial delivery, enhancing patient compliance and drug efficacy.29, 30
Intelligent and Stimuli-Responsive Drug Delivery
Stimuli-responsive drug transport structures are engineered to launch antimicrobial capsules in reaction to precise environmental stimuli, together with pH changes or enzymatic reactions that allows you to goal the delivery of the medicine to the web page of infection.31, 32
Niosome-Based Drug Delivery
Niosomes are non-ionic surfactant vesicular drug delivery systems that are formed by amphiphilic molecules, which self-assemble to form bilayers that can entrap both hydrophilic and lipophilic drugs. Niosomes provide better chemical stability, ease of preparation, and flexibility in formulation compared to liposomes, making them very useful for ocular drug delivery systems.33
Regarding eye infections, niosome-based delivery systems have shown great potential in enhancing corneal penetration, precorneal dwelling time, and sustained release of antimicrobial drugs. The vesicular nature of niosomes permits to engage higher with the corneal epithelium, therefore improving drug absorption and reducing nasolacrimal drainage and systemic absorption. Various studies have proven that niosomal formulations have higher antimicrobial pastime in comparison to conventional eye drops in cases of bacterial and fungal eye infections.34
In precise, the work of Neethirajan35 has suggested a complete evaluate of niosome-primarily based methods in ocular drug shipping, highlighting the benefits of niosomes in improving ocular bioavailability, efficacy, and affected character compliance.35 This particular observe highlighted the flexibility of niosomes in handing over an extensive variety of ophthalmic drugs and the significance of niosomes in the treatment of ocular infections.
Niosomes are a versatile machine learning technology that may be utilized together with new technologies. Surface modification with mucoadhesive polymers, the design of in-situ gelling structures, and the application of artificial intelligence-assisted formulation development are some of the likely strategies to similarly boost the niosomal structures. AI modelling can be employed to optimize surfactant formulation, cholesterol concentration, vesicle size, and drug release characteristics, making it possible to develop infection-targeting niosomal ocular formulations and their swift translation from the laboratory to the clinic shown in Figure 3.
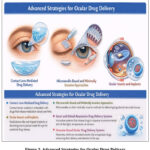 |
Figure 3: Advanced Strategies for Ocular Drug Delivery
|
(A) Contact lens-based totally drug shipping systems, (B) microneedle-based drug transport structures, (C) ocular inserts/implants, (D) stimuli-responsive drug delivery systems, and (E) niosome-primarily based drug transport systems.
Translational and Regulatory Issues – Despite the life of promising preclinical facts, the interpretation of advanced ocular drug transport systems into the scientific placing remains a challenge. The regulatory issues associated with sterility, biocompatibility, scalability, and lengthy-time period toxicity must be addressed to facilitate approval and commercialization
Future Prospects
The future approaches in ocular drug delivery may include multifunctional systems that can simultaneously target antimicrobial, anti-inflammatory, and tissue regeneration requirements. The use of artificial intelligence to provide personalized dosing and infection-responsive drug delivery may also improve future prospects.
Artificial Intelligence in Ocular Drug Delivery for Eye Infections
Artificial intelligence and machine learning are gaining popularity in pharmaceutical research and the field of ophthalmology, providing novel solutions for improving ocular drug delivery and eye infections. AI and ML allow for data-driven formulation design, predictive modelling, and personalized treatment, which can overcome several limitations associated with traditional trial-and-error approaches.
AI-Assisted Design and Optimization of Drug Delivery Systems-
AI algorithms, such as neural networks and deep learning, have been used to optimize the formulation parameters of ocular drug delivery systems, such as particle size, drug loading, release rates, and stability. In nanoparticle and hydrogel-based ocular formulations, AI algorithms can predict physicochemical properties and in vitro performance as a function of polymer composition and processing conditions, which can greatly accelerate development time and costs.36, 37
Machine learning algorithms have also been used to simulate corneal permeability and ocular pharmacokinetics, which can be used to make rational choices of drug carriers and excipients for improved ocular bioavailability.38
AI in Antimicrobial Selection and Resistance Prediction
AI software is increasingly being used to analyse large clinical and microbiological databases to predict susceptibility patterns and antimicrobial resistance. In the context of ocular infections, AI-assisted diagnostic tools can be used to rapidly identify pathogens and make informed choices of antimicrobial agents and regimens, which can help to minimize the use of empirical therapy and the development of resistance.39
Integration of AI-assisted diagnostic results with sophisticated drug delivery systems provides the potential for personalized ocular treatment depending on the type and severity of the infection.
AI-Assisted Smart and Responsive Ocular Drug Delivery Systems
AI can improve the capabilities of smart and responsive ocular drug delivery systems by providing real-time monitoring and responsive drug release. Smart contact lenses and implantable devices with biosensors and AI algorithms have the potential to provide responsive drug release based on changes in ocular biomarkers such as pH, inflammatory mediators, or microbial concentrations.40, 41
These closed-loop systems have immense potential for the accurate treatment of chronic and periodic infections of the eye is shown in Figure 4.
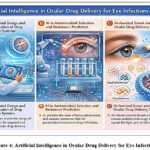 |
Figure 4: Artificial Intelligence in Ocular Drug Delivery for Eye Infections
|
The software of synthetic intelligence in (A) drug delivery device optimization, (B) selection of anti-infective retailers and prediction of resistance, and (C) development of smart drug delivery systems which are conscious of specific stimuli.
Clinical Decision Support and Personalized Ocular Therapy
Clinical choice assist structures and advanced modelling strategies are an increasing number of being utilized in ophthalmology for the diagnosis and remedy of ocular sicknesses. When blended with advanced ocular drug delivery structures, those fashions may be used to expand personalised remedy regimens primarily based on affected person-particular parameters such as tear film dynamics, ocular surface disease, remedy history, and compliance.
Clinical Decision Support and Personalized Ocular Therapy
Clinical decision assist structures primarily based on AI are increasingly more being used within the subject of ophthalmology for the diagnosis and management of various ocular illnesses. When incorporated with state-of-the-art ocular drug delivery structures, AI can assist in the development of personalised treatment plans for sufferers, deliberating diverse affected person-unique parameters inclusive of tear movie dynamics, ocular floor ailment, preceding remedy reaction, and compliance.42
While most of the state-of-the-art ocular drug transport systems are nevertheless within the experimental segment, the continuing improvements in sensor era, facts acquisition, and regulatory guidelines are expected to expedite their transition into clinical exercise.
Conclusion
Innovative ocular drug transport structures are a modern modality for the remedy of eye infections, which successfully pass the anatomical and physiological constraints hindering the efficacy of traditional ophthalmic arrangements. Recent trends in nanotechnology-primarily based transport systems, in-situ gelling structures, contact lens-based totally drug delivery, micro-needle-primarily based systems, ocular inserts, and implantable machine learning technologies have shown great potential in improving the bioavailability and sustained web site-particular antimicrobial action of drugs. These progressive systems no longer only enhance the efficacy of remedy however additionally decrease the frequency of dosing, decrease systemic absorption, and enhance affected person compliance, which can be critical for the successful remedy of both acute and chronic ocular infections.
The aggregate of sophisticated computational resources and predictive modelling abilities further improves ocular drug delivery making an allowance for the rational layout of formulations, the optimization of drug launch profiles and personalised remedy strategies. These abilities have the capacity to offer predictions concerning drug-tissue interactions, dosing, and healing consequences, therefore minimizing the need for trial-and-mistakes strategies and growing the performance of therapeutic improvement. When mixed with clever and stimuli-responsive transport systems, those abilities permit for the development of adaptive, closed-loop delivery systems that may be precipitated by using contamination-related or patient-specific physiological indicators.
Despite those encouraging tendencies, translation into the medical institution is presently restricted. Approaches to deal with demanding situations which includes regulatory approval, protection validation, standardization, and integration into the prevailing medical infrastructure ought to be cautiously considered. Preclinical and scientific validation, in addition to collaboration among ophthalmologists, pharmaceutical scientists, engineers, and computational professionals, might be vital to facilitate the interpretation of laboratory research into the clinic.
The integration of superior drug shipping platforms, clever stimuli-responsive structures, and computationally guided optimization strategies is a paradigm shift inside the control of ocular infection. Further studies and validation of these incorporated technologies might be necessary to allow extra specific, green and affected person-friendly ocular healing procedures
Acknowledgement
The authors acknowledge the support the management of KG College of Arts and Science (KGCAS) for institutional support.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Authors Contributions
Neethirajan. Guruswamy: Conceptualization, Methodology, Writing – Original Draft.
Shalini Devaraj: Data Collection, Analysis, Writing – Review & Editing.
References
- World Health Organization. Global data on visual impairments. WHO; 2023.
- Gaudana R, Ananthula HK, Parenky A, Mitra AK. Ocular drug delivery. AAPS J. 2010;12(3):348–360. DOI: 10.1208/s12248-010-9183-3
CrossRef - Urtti A. Challenges and obstacles of ocular pharmacokinetics. Adv Drug Deliv Rev. 2006;58(11):1131–1135. DOI: 10.1016/j.addr.2006.07.008
CrossRef - Del Amo EM, Rimpelä AK, Heikkinen E, et al. Pharmacokinetic aspects of retinal drug delivery. Prog Retin Eye Res. 2017;57:134–185. DOI: 10.1016/j.preteyeres.2016.12.001
CrossRef - Kaur IP, Kanwar M. Ocular preparations: the formulation approach. Drug Dev Ind Pharm. 2002;28(5):473–493. DOI: 10.1081/DDC-120003445
CrossRef - Urtti A. Challenges and obstacles of ocular pharmacokinetics and drug delivery. Adv Drug Deliv Rev. 2021;171:125–148. doi:10.1016/j.addr.2020.11.006
CrossRef - Cunha-Vaz J. The blood–retinal barriers system: basic concepts and clinical evaluation. Exp Eye Res. 2004;78(3):715–721. DOI: 10.1016/j.exer.2003.09.003
CrossRef - Diebold Y, Calonge M. Applications of nanoparticles in ophthalmology. Prog Retin Eye Res. 2010;29(6):596–609. DOI: 10.1016/j.preteyeres.2010.08.002
CrossRef - de Campos AM, Sánchez A, Alonso MJ. Chitosan nanoparticles: a new vehicle for the improvement of the delivery of drugs to the ocular surface. Int J Pharm. 2001;224(1–2):159–168. DOI: 10.1016/S0378-5173(01)00762-8
CrossRef - Bochot A, Fattal E. Liposomes for ocular drug delivery: a state of the art. Drug Deliv. 2012;19(1):1–15. DOI: 10.3109/10717544.2011.640216
- Seyfoddin A, Al-Kassas R. Solid lipid nanoparticles and nanostructured lipid carriers as ocular drug delivery systems. Int J Pharm. 2013;448(2):369–383. DOI: 10.1016/j.ijpharm.2013.02.045
CrossRef - Almeida H, Amaral MH, Lobão P, Lobo JM. In situ gelling systems: a strategy to improve the bioavailability of ophthalmic pharmaceutical formulations. J Pharm Pharm Sci. 2014;17(3):396–421. DOI: 10.18433/J3MW2F
- Gratieri T, Gelfuso GM, Rocha EM, Sarmento VH, de Freitas O, Lopez RF. A poloxamer/chitosan in situ forming gel with prolonged retention time for ocular delivery. Eur J Pharm Sci. 2010;41(3–4):441–448. DOI: 10.1016/j.ejps.2010.07.007
CrossRef - Ciolino JB, Hoare TR, Iwata NG, et al. A drug-eluting contact lens. Invest Ophthalmol Vis Sci. 2009;50(7):3346–3352. DOI: 10.1167/iovs.08-3155
CrossRef - Phan CM, Subbaraman LN, Jones L. Contact lenses for antimicrobial drug delivery. Expert Opin Drug Deliv. 2014;11(4):537–546. DOI: 10.1517/17425247.2014.884555
CrossRef - Jiang J, Gill HS, Ghate D, McCarey BE, Patel SR, Edelhauser HF. Coated microneedles for drug delivery to the eye. Invest Ophthalmol Vis Sci. 2007;48(9):4038–4043. DOI: 10.1167/iovs.07-0219
CrossRef - Kim YC, Park JH, Prausnitz MR. Microneedles for ocular drug delivery. Adv Drug Deliv Rev. 2012;64(14):1547–1568. DOI: 10.1016/j.addr.2012.05.005
CrossRef - Yasukawa T, Ogura Y, Kimura H, et al. Drug delivery systems for vitreoretinal diseases. Prog Retin Eye Res. 2004;23(3):253–276. DOI: 10.1016/j.preteyeres.2004.02.001
CrossRef - Patel S, Cholkar K, Agrahari V, Mitra AK. Advances in ocular drug delivery: Role of implants and inserts. Pharmaceutics. 2021;13(2):171. DOI: 10.3390/pharmaceutics13020171
CrossRef - Alvarez-Lorenzo C, Concheiro A. Smart drug delivery systems: from fundamentals to clinical applications. Chem Commun. 2014;50(58):7743–7765. DOI: 10.1039/C4CC01408D
CrossRef - Mitra AK, Cholkar K, Mandal A. Emerging nanotechnologies for diagnostics, drug delivery, and medical devices. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2012;4(3):247–259. DOI: 10.1002/wnan.1165
CrossRef - U.S. Food and Drug Administration. Ophthalmic drug products: guidance for industry. FDA; 2022.
- Chauhan A, Radke C. Ophthalmic drug delivery systems. Int J Pharm. 2021;603:120710. DOI: 10.1016/j.ijpharm.2021.120710
CrossRef - Agatonovic-Kustrin S, Beresford R. Basic concepts of artificial neural network modeling in pharmaceutical analysis. J Pharm Biomed Anal. 2000;22(5):717–727. DOI: 10.1016/S0731-7085(99)00272-1
CrossRef - Wu Z, Ramsundar B, Feinberg EN, et al. MoleculeNet: a benchmark for molecular machine learning. Chem Sci. 2018;9(2):513–530. DOI: 10.1039/C7SC02664A
CrossRef - Wong JKW, Wong JG. Predictive modeling of ocular drug permeability using machine learning. Adv Drug Deliv Rev. 2021;171:128–145. DOI: 10.1016/j.addr.2020.11.002
CrossRef - Ting DSW, Pasquale LR, Peng L, et al. Artificial intelligence and ophthalmology: a review. Br J Ophthalmol. 2019;103(2):167–175. DOI: 10.1136/bjophthalmol-2018-313173
CrossRef - Ciolino JB, Hudson SP, Mobbs AN, et al. A prototype smart contact lens for controlled drug delivery. Adv Mater. 2014;26(23):4082–4087. DOI: 10.1002/adma.201304522
- Kim J, Campbell AS, de Ávila BEF, Wang J. Wearable biosensors for healthcare monitoring. Nat Biotechnol. 2019;37(4):389–406. DOI: 10.1038/s41587-019-0045-y
CrossRef - Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44–56. DOI: 10.1038/s41591-018-0300-7
CrossRef - Torchilin VP. Multifunctional, stimuli-sensitive nanoparticulate systems for drug delivery. Nat Rev Drug Discov. 2014;13(11):813–827. DOI: 10.1038/nrd4333
CrossRef - Calonge M, Diebold Y, Sáez V, et al. Applications of nanotechnologies in ocular drug delivery. Prog Retin Eye Res. 2018;64:1–38. DOI: 10.1016/j.preteyeres.2017.10.003
CrossRef - Uchegbu IF, Vyas SP. Non-ionic surfactant based vesicles (niosomes) in drug delivery. Int J Pharm. 1998;172(1–2):33–70. DOI: 10.1016/S0378-5173(98)00162-5
CrossRef - Pardakhty A, Moazeni E. Niosomes as a drug delivery system: a review. Int J Pharm. 2013;11(2):123–136. DOI: 10.62472/kjps.v16.i27.210-229
CrossRef - Neethirajan G. Niosome-based approaches in ocular drug delivery: advances, challenges, and future perspectives. J Pharm Res Integr Med Sci. 2025;2(9):135–149. doi:10.64063/3049-1681.vol.2.issue9.12
CrossRef - Topol EJ. Preparing the healthcare workforce to deliver the digital future. Lancet. 2019;394(10192):239–240. DOI: 10.1016/S0140-6736(19)31244-5
- Rajkomar A, Dean J, Kohane I. Machine learning in medicine. N Engl J Med. 2019;380(14):1347–1358. DOI: 10.1056/NEJMra1814259
CrossRef - Esteva A, Robicquet A, Ramsundar B, et al. A guide to deep learning in healthcare. Nat Med. 2019;25(1):24–29. DOI: 10.1038/s41591-018-0316-z
CrossRef - Gulshan V, Peng L, Coram M, et al. Development and validation of a deep learning algorithm for detection of eye disease. JAMA. 2016;316(22):2402–2410. DOI: 10.1001/jama.2016.17216
CrossRef - Liu Y, Chen PC, Krause J, et al. How to read articles that use machine learning. JAMA. 2019;322(18):1806–1816. DOI: 10.1001/jama.2019.16489
CrossRef - He J, Baxter SL, Xu J, Xu J, Zhou X, Zhang K. The practical implementation of artificial intelligence technologies in medicine. Nat Med. 2019;25(1):30–36. DOI: 10.1038/s41591-018-0307-0
CrossRef - Bohr A, Memarzadeh K. The rise of artificial intelligence in pharmaceutical sciences. Drug Discov Today. 2020;25(3):718–724. DOI: 10.1016/j.drudis.2019.10.008
CrossRef
Accepted on: 20-03-2026
Second Review by: Dr Sunil Chaudhry
Final Approval by: Dr. Eugene A. Silow









