A Comparative Review on the Topical Bioactive Phytochemicals in Barleria cristata and Their Potential in Fracture Healing: Bridging Ethnobotanical Knowledge and Modern Bone Tissue Engineering
1Department of Pharmaceutics, Mahavir Institute of Pharmacy Nashik, (MH), India.
2Department of Pharmaceutical Chemistry, Institute of Pharmaceutical Education and Research, Borgaon Meghe, (MH), India.
3Department of pharmaceutical chemistry, Mahavir Institute of Pharmacy Nashik, India.
4Department of Pharmacognosy, Mahavir Institute of Pharmacy Nashik, India.
Corresponding Author Email: anishabagade0403@gmail.com
Download this article as:
ABSTRACT:The rising incidence of bone fractures and the pharmacological limitations of systemic treatments underscore the need for targeted local therapies. This review critically evaluates leaves of Barleria cristata L.—a plant with documented ethnomedicinal use in trauma care—for its topical potential in supporting fracture repair. We synthesize traditional knowledge, phytochemical data, and preclinical evidence to assess its bioactive constituents (e.g., iridoids, flavonoids) for transdermal delivery and their supportive roles in inflammation modulation, angiogenesis, and bone-repair processes. A focused comparison of vesicular carriers—liposomes, ethosomes, and niosomes—examines their structural properties, skin penetration profiles, and stability for delivering B. cristata’s phytocomplex. Preclinical studies suggest that topical formulations may enhance healing, as observed through improved radiological, histological, and biomechanical parameters in animal models. Advanced delivery systems, particularly ethosomes and bioadhesive hydrogels, appear promising for increasing skin permeation and local retention. The convergence of traditional application and modern scientific evaluation identifies B. cristata as a strong candidate for further investigation. Translating this potential requires future research focused on extract standardization, formulation optimization, and controlled clinical studies to rigorously assess its safety and supportive role in bone fracture management.
KEYWORDS:Barleria cristata leaves; Bone Tissue Engineering; Ethnopharmacology; Fracture Healing; Phytochemicals; Topical Delivery
Introduction
The Clinical Burden of Bone Fractures and the Drawbacks of Systemic Treatments
The skeletal system’s remarkable capacity for self-regeneration, while profound, is frequently compromised by the scale and complexity of modern traumatic injuries, as well as by the pathophysiological milieu of an aging global population. Bone fractures represent a colossal clinical and socioeconomic burden, with the World Health Organization classifying osteoporosis alone as the second leading healthcare concern after cardiovascular disease.1Annually, an estimated 178 million new fractures occur worldwide, a figure projected to escalate in tandem with increasing life expectancy and incidence of osteoporosis.2 Beyond the immediate trauma, the sequelae of delayed union or non-union—complications occurring in approximately 5-10% of all fractures—engender prolonged disability, significant pain, and substantial economic costs from repeated surgical interventions and lost productivity.3
The current pharmacological arsenal for enhancing bone healing and combating resorptive diseases like osteoporosis is predominantly systemic in administration. This paradigm, while effective in some contexts, is fraught with significant iatrogenic constraints. Bisphosphonates, the first-line therapeutics for osteoporosis, function as potent antiresorptive agents by inducing osteoclast apoptosis. However, their protracted use has been unequivocally linked to rare but devastating adverse events, including atypical femoral fractures (AFFs) and osteonecrosis of the jaw (ONJ).4 The paradoxical nature of these drugs—strengthening bone in one skeletal region while predisposing to spontaneous fractures in another—highlights a critical flaw in systemic, non-targeted inhibition of bone remodeling. A seminal study by the American Society for Bone and Mineral Research task force underscored this, finding that 82% of AFF patients had a history of bisphosphonate use, with a clear dose-response relationship.5 Furthermore, the anabolic agent teriparatide (recombinant human parathyroid hormone 1-34), though potent in stimulating osteoblast activity, carries a U.S. Food and Drug Administration (FDA) black box warning due to its observed association with osteosarcoma in rodent models, consequently restricting its long-term use in humans.6 Its systemic administration also precipitates transient hypercalcemia, necessitating rigorous patient monitoring. The case of romosozumab, a monoclonal antibody against sclerostin, further illustrates the systemic conundrum. While it demonstrates powerful dual anabolic and antiresorptive effects, its use is constrained by a potential increase in major adverse cardiovascular events, leading to strict contraindications in patients with a history of myocardial infarction or stroke.7 This cardiotoxic profile, discovered post-approval, exemplifies the unforeseen off-target liabilities of systemic biologic agents.
The significant limitations of systemic bone therapies—including poor oral bioavailability, demanding administration protocols, and non-targeted effects on the entire skeleton—have intensified the focus on developing localized treatment strategies.8 This shift aims to provide precise, site-specific intervention that directly addresses a fracture without disrupting systemic homeostasis. In response, the field of bone tissue engineering is advancing bioactive scaffolds that function as controlled-release reservoirs for osteogenic agents directly at the injury site. Within this innovative landscape, biologically derived phytochemicals, particularly those with a history of traditional use like Barleria cristata L., offer a promising direction. The integration of ethnobotanical knowledge with modern engineering—evaluating such plants for their safety, synergistic phytochemistry, and applicability in topical or implantable formulations—represents a compelling pathway toward developing effective, targeted therapies that overcome the drawbacks of current systemic treatments.
The Rationale for Topical/Transdermal Therapy in Orthopedics
The limitations of systemic pharmacotherapy have precipitated a critical re-evaluation of drug delivery paradigms in musculoskeletal disorders. The fundamental rationale for pursuing topical and transdermal strategies in orthopedics is predicated on the principle of localized pharmacokinetics, which aims to achieve therapeutic concentrations of bioactive agents at the precise site of pathology while minimizing their systemic circulation and consequent off-target effects.9 This approach is particularly salient for bone, a highly vascularized organ where systemic administration leads to widespread distribution but often sub-therapeutic concentrations at the specific fracture non-union or defect site due to the physiological barriers of the bone microenvironment.
The transdermal route offers a non-invasive, targeted strategy for delivering osteoactive compounds to superficial bones while bypassing first-pass metabolism and enabling sustained local release.10-13 Advanced carriers such as nanoemulsions, liposomes, and ethosomes enhance dermal penetration.10 while physical enhancers like iontophoresis and microneedles expand the delivery of larger or charged molecules.11-13 Preclinical studies, such as the transdermal application of simvastatin hydrogel in calvarial defects, highlight the capacity of localized delivery to achieve osteogenesis without systemic toxicity.14 Integrating ethnobotanical phytochemicals like Barleria cristata into such delivery platforms could unify natural osteoinductive synergy with precision-engineered release systems. This convergence represents a paradigm shift in fracture repair and localized bone regeneration, addressing the limitations of systemic therapy through site-specific, safe, and standardized interventions.
Barleria cristata: Ethnobotanical Legacy in Topical Wound and Fracture Care
Based on its cross-cultural ethnobotanical legacy, Barleria cristata (Philippine Violet) presents a compelling case for scientific investigation in bone repair. Its documented use in diverse traditional systems, from Ayurveda (where it is known as “Vajradanti” for strengthening bones) to Thai and Chinese medicine for treating fractures and musculoskeletal pain, provides an anthropologically validated rationale for its efficacy.15-17 Modern phytochemical analyses have identified a rich spectrum of bioactive constituents – including iridoids, phenylethanoid glycosides like verbascoside, and flavonoids such as apigenin – that exhibit anti-inflammatory, antioxidant, and osteogenic properties.17-19 This confluence of traditional wisdom and scientific evidence positions B. cristata as a promising source for developing sophisticated topical therapeutics aimed at stimulating bone regeneration.
Ethnobotanical Context and Investigative Rationale
The evaluation of Barleria cristata L. for fracture management originates from its recurring mention in ethnobotanical surveys for musculoskeletal complaints, though its status as a classical “bone-setter” requires careful qualification. While plants like Cissus quadrangularis have more explicit and documented historical use for fracture consolidation,20 B. cristata (known regionally as Vajradanti, Philippine Violet) is frequently cited in traditional practices for related topical applications.
In various traditional systems, the plant is reported for conditions adjacent to fracture care. For instance, in Ayurveda, “Vajradanti” (a name shared by Barleria species) is primarily associated with oral health and general inflammation.21 Modern ethnobotanical studies from India and Thailand list B. cristata among plants used topically for sprains, swelling, and musculoskeletal pain.16,23 However, a critical review of classical pharmacopeias reveals that specific, unambiguous references to B. cristata (with confirmed botanical identification) being used explicitly for bhagna (fractures) are less common than for other dedicated “bone-setting” botanicals20 [Table 1].
Therefore, the traditional rationale for investigating B. cristata in this context is best framed not as the direct continuation of a classical bone-setting practice, but as the exploratory evaluation of a plant with documented topical anti-inflammatory and musculoskeletal trauma applications.21,23 Its repeated association with trauma care in ethnobotany, combined with a phytochemical profile (Section 2) suggestive of osteogenic potential, provides a plausible—though not historically definitive—foundation for exploring its supportive role in the complex fracture-healing microenvironment.
The Phytochemical Arsenal of B. cristata
Key Bioactive Compounds: Iridoids, Phenylethanoid Glycosides, and Flavonoids
The osteogenic potential of Barleria cristata is intrinsically linked to its complex and diverse phytochemical constitution. This arsenal comprises several classes of secondary metabolites, each contributing uniquely to the observed pharmacological effects through synergistic or additive mechanisms. The most therapeutically relevant compounds, substantiated by phytochemical screenings and pharmacological assays, belong to three principal classes: iridoids, phenylethanoid glycosides, and flavonoids. These compounds are not merely present but are often found in significant concentrations, making B. cristata a particularly rich source for bioprospecting in bone tissue engineering.
Iridoids and Their Analogues: Iridoids represent a cornerstone of B. cristata’s bioactivity. These cyclopentan-[c]-pyranoid monoterpenoids are renowned for their potent anti-inflammatory and antioxidant properties, which are critical for modulating the initial inflammatory phase of fracture healing. The leaves are a particularly rich source, with barlerin and acetylbarlerin identified as the predominant iridoid glycosides.17,22 The mechanism is elucidated in a study where related iridoids demonstrated significant inhibition of NF-κB signaling in human monocytes, thereby suppressing the production of pro-inflammatory cytokines like TNF-α and IL-6.18 This action is crucial for bone repair, as uncontrolled inflammation impedes the transition to the reparative phase and can lead to excessive osteoclast activation. By creating a more conducive biochemical microenvironment, these iridoids indirectly promote osteogenesis and protect newly formed bone matrix.
Phenylethanoid Glycosides (PeGs): This class of water-soluble compounds is another critical component, with verbascoside (acteoside) being the most prominent and well-studied member within B. cristata extracts. PeGs are esteemed for their robust radical scavenging capabilities, which protect osteoprogenitor cells and matrix components from oxidative stress—a key inhibitor of osteoblast function and a driver of osteoclast activity.23 Beyond antioxidant activity, verbascoside has been shown to influence cell fate directly. A pivotal study demonstrated that verbascoside significantly enhanced the osteogenic differentiation of human bone marrow-derived mesenchymal stem cells (hBM-MSCs) by upregulating the expression of core osteogenic markers, including Runx2, alkaline phosphatase (ALP), and osteocalcin (OCN).24 This dual capacity to mitigate oxidative damage and directly stimulate osteogenic differentiation positions phenylethanoid glycosides as primary drivers of B. cristata’s bone-healing efficacy.
Flavonoids: The flavonoid profile of B. cristata, including compounds such as apigenin, luteolin, and their glycosidic derivatives, contributes a third strategic arm to its mechanism of action. Flavonoids are potent modulators of key signaling pathways implicated in bone metabolism. Apigenin, for instance, has been extensively documented to promote osteoblastogenesis by activating the BMP-2/Smad and Wnt/β-catenin pathways, while concurrently inducing apoptosis in mature osteoclasts, thus exerting a dual anabolic and anti-catabolic effect.19,25,26 The presence of these flavonoids in a bioavailable form within a crude extract suggests a polypharmaceutical approach, where multiple compounds target different nodes of the bone remodeling network simultaneously. This network pharmacology is a significant advantage over single-target synthetic drugs, as it more closely mimics the body’s complex regulatory systems and offers a lower risk of compensatory mechanisms that lead to drug resistance or loss of efficacy.
Comparative Phytochemistry: Is B. cristata Uniquely Suited for Topical Application? (e.g., molecular weight, lipophilicity)
The therapeutic potential of a topical Barleria cristata formulation is uniquely enabled by the inherent physicochemical properties of its key phytochemicals, which align favorably with the established rules of transdermal delivery.9 Its iridoids and flavonoid aglycones possess an optimal combination of low-to-moderate molecular weight and balanced lipophilicity (log P ~1-3), facilitating passive diffusion through the skin’s stratum corneum.27 While the larger, more hydrophilic verbascoside slightly exceeds ideal molecular weight thresholds, its permeability can be effectively enhanced through advanced nano-formulations.28 The extract functions as a natural prodrug system, as glycosylated compounds are hydrolyzed on the skin to release more permeable and active aglycones. This intrinsic phytochemical profile, often superior to other botanicals used for bone healing, provides a comparative advantage for achieving pharmacologically relevant concentrations at the periosteum, making B. cristata a particularly promising candidate for topical bone tissue engineering strategies.17,22,14
Comparative Perspective with Other Bone-Setting Plants
Several botanicals are documented in traditional systems for managing fractures and supporting bone health, such as Cissus quadrangularis,20,29 Bambusa arundinacea,31 and Vitex negundo.30 When evaluated alongside these plants, the investigational profile of Barleria cristata is distinct. Research on B. cristata has largely focused on preclinical models of topical delivery, with observed activities centered on mitigating inflammation, reducing oxidative stress, and supporting soft tissue repair—mechanisms that indirectly aid the healing environment.16,17,23 In comparison, a plant like Cissus quadrangularis has been the subject of more extensive study, with a body of evidence in animal models and some clinical reports more directly addressing fracture consolidation and metrics of bone metabolism.20,29 Consequently, while classical “bone-setting” plants may have a stronger documented association with enhancing bone union, B. cristata appears, based on current evidence, to occupy a complementary niche as a potential topical adjunct aimed at creating a favorable local milieu for the natural repair process.
Table 1: Comparative Overview of Selected Botanicals Used Traditionally for Bone and Fracture Care
| Botanical Name (Common Name) | Traditional Use in Bone / Fracture Context | Key Bioactive Constituents (Reported) | Proposed Primary Mechanism in Bone-Healing Context | Level of Evidence for Bone-Related Outcomes | Remarks / Comparative Positioning |
| Cissus quadrangular(stems and roots) is (Hadjod, Bone Setter)20-29 | Oral administration or topical paste for fracture healing, bone strengthening, and pain relief. | Ascorbic acid, β-carotene, calcium, flavonoids, triterpenoids (e.g., onocer‑7‑ene‑3α,21β‑diol). | Primarily nutritive (mineral/vitamin support) and antioxidant; reported to stimulate osteoblast activity and modulate osteoclastogenesis. | Preclinical (Strong): Multiple animal studies report accelerated fracture union and improved biomechanical strength. Clinical (Limited): Some human studies suggest reduced healing duration. |
Regarded as a classical “bone‑setting” plant with comparatively stronger evidence for effects related to bone union and mineralization.20,29 |
| Bambusa arundinacea (leaves) (Bamboo)31 | Sap (Banslochan) used orally as a tonic for bone weakness and in pastes for fractures. | Silica, minerals, flavonoids, phenolic acids. | Provides bioavailable silica for collagen matrix support; general mineral supplementation; possible anti‑inflammatory actions. | Traditional / Ethnobotanical (Strong): Widely described in classical medicine texts. Preclinical (Emerging): Limited modern bone‑specific models available. |
Positioned as a nutritive and strengthening agent; evidence is largely traditional, with fewer mechanistic data for direct osteogenic effects. |
| Vitex negundo (Nirgundi)30 | Leaf paste applied topically for sprains, swelling, and bone / musculoskeletal pain. | Flavonoids (casticin, orientin), terpenoids, essential oils. | Anti‑inflammatory, analgesic, and antioxidant activities; modulates inflammatory cytokines (TNF‑α, IL‑6). | Preclinical (Moderate): Strong evidence in inflammation and pain models. Bone‑Specific (Limited): Few fracture‑healing studies. |
Functions primarily as a supportive agent for pain and inflammation control, helping maintain a conducive environment for recovery. |
| Barleria cristata (Leaf)(Philippine Violet / Vajradanti)16-17-20 | Leaf paste applied topically for fractures, sprains, and musculoskeletal swelling. | Iridoids (barlerin), phenylethanoid glycosides (verbascoside), flavonoids (apigenin). | Multi‑target modulation: anti‑inflammatory (NF‑κB‑linked pathways), antioxidant effects, angiogenic support (VEGF‑associated), with limited preliminary evidence for influence on osteogenic pathways. | Preclinical (Emerging): In‑vitro and topical in‑vivo models show anti‑inflammatory, antioxidant, and wound‑healing activities. Direct Bone‑Specific Evidence (Limited): Few studies investigate mineralization or defect‑healing outcomes. |
Best characterized as a topical supportive / adjunctive therapy that may help create a favorable microenvironment for fracture repair rather than functioning as a primary “bone‑setting” agent. |
Mechanisms of Action: From Skin to Bone
Dermal Penetration and Pharmacokinetics of Key Constituents
The therapeutic success of a topical Barleria cristata formulation depends on the effective delivery of its bioactive compounds through the skin’s complex layers to reach underlying musculoskeletal tissues like the periosteum.9,27 The penetration of its key constituents—iridoids, flavonoids, and the more polar phenylethanoid glycosides—is governed by their molecular properties, with size, lipophilicity, and hydrogen bonding capacity determining their ability to passively diffuse through the outer, lipid-rich skin barrier.32 Advanced delivery systems, such as nanoemulsions or lipid-based nanocarriers, are essential to overcome this barrier; they act as penetration enhancers by fluidizing skin lipids and can facilitate transport via alternative pathways.28,33,34 Furthermore, metabolism within the skin can hydrolyze certain compounds into more active and permeable aglycones, effectively creating a natural prodrug system. For the final transit to the highly vascularized periosteum, the lipophilic nature of many constituents is advantageous, a process validated by clinical precedent with other topical anti-inflammatories.35 Ultimately, through rational formulation design that leverages these mechanisms, a therapeutically relevant concentration of phytochemicals can be achieved directly at the site of bone injury.14
Advanced formulation strategies are paramount to orchestrating this entire process. Lipid-based nanocarriers—such as ethosomes, transfersomes, and nanostructured lipid carriers (NLCs)—are particularly effective.34,36,37 These systems encapsulate the extract, shielding it from degradation and enhancing its solubility. Furthermore, their deformable nature allows them to penetrate deeply into the skin, often carrying their payload to the target site.33,37 A study on a curcumin-loaded transfersomal gel for arthritis demonstrated significantly higher drug retention in deeper skin layers and synovial tissue compared to a conventional gel, resulting in superior anti-inflammatory and analgesic effects in a murine model.38,39 This exemplifies the transformative impact of delivery system design on topical pharmacokinetics and ultimate efficacy.
Modulating the Local Inflammatory Milieu and Angiogenesis
The transition from fracture hematoma to mineralized callus is a tightly orchestrated process governed by two interdependent pillars: the precise modulation of inflammation and the rapid establishment of a robust vascular network.40,41 Topical interventions that can locally coordinate these processes offer a powerful therapeutic strategy. The phytochemical profile of Barleria cristata suggests a profound capacity to bio-regulate this critical early phase of bone repair, moving beyond mere passive nutrient delivery to active immunomodulation and angio-induction.17,22
The immediate post-fracture inflammatory phase, while necessary for initiating repair, becomes detrimental if dysregulated or prolonged.40,42 Excessive and persistent inflammation, characterized by elevated levels of pro-inflammatory cytokines such as Tumor Necrosis Factor-alpha (TNF-α), Interleukin-1 beta (IL-1β), and Interleukin-6 (IL-6), directly inhibits osteoblast differentiation and promotes osteoclastogenesis, leading to impaired healing and non-union.40 The key constituents of B. cristata, particularly its iridoids (e.g., barlerin) and phenylethanoid glycosides (e.g., verbascoside), function as natural brakes on this cascade. Their primary molecular target is the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) pathway, a master regulator of inflammation.18 By inhibiting the phosphorylation and degradation of IκBα, these compounds prevent the nuclear translocation of NF-κB, thereby downregulating the transcription of its target pro-inflammatory genes.18 This mechanism was convincingly demonstrated in a related in vivo model where a verbascoside-rich extract from Cistanche deserticola significantly reduced TNF-α and IL-6 levels in a rat fracture model, correlating with accelerated callus maturation and improved biomechanical strength. This suggests a direct translational pathway for B. cristata’s similar compounds.
Successful bone regeneration is absolutely dependent on angiogenesis—the formation of new blood vessels from existing vasculature.43-45 The hypoxic environment of the fracture site triggers the stabilization of Hypoxia-Inducible Factor-1 alpha (HIF-1α), which upregulates potent angiogenic factors like Vascular Endothelial Growth Factor (VEGF).46 The flavonoid components of B. cristata, such as apigenin, have been shown to directly promote this process. Apigenin not only stabilizes HIF-1α but also enhances the expression and secretion of VEGF and basic Fibroblast Growth Factor (bFGF) in osteoblasts and mesenchymal stem cells (MSCs).47 This creates a positive feedback loop: enhanced angiogenesis improves nutrient and oxygen supply, which further supports the survival and osteogenic differentiation of MSCs.45 A compelling case study exists in the field of periodontal regeneration, where a topical gel containing apigenin was shown to significantly increase VEGF expression and capillary density in a rat periodontitis model, leading to enhanced alveolar bone regeneration. This provides a powerful precedent for the use of flavonoid-based topicals to drive angiogenic-osteogenic coupling.
The true therapeutic genius of a complex extract like that of B. cristata lies in the temporal synergy of its constituents.17 The iridoids and verbascoside work initially to quell the excessive inflammatory response, preventing collateral damage to progenitor cells and creating a permissive environment for repair.18,42
Direct Stimulation of Osteoblast Differentiation and Mineralization (BMP/Smad, Wnt/β-catenin pathways)
The flavonoid components, particularly apigenin, exert a complementary effect by activating the canonical Wnt/β-catenin pathway.19,26,48 This pathway is fundamental for osteoblast proliferation, differentiation, and survival.49 Apigenin functions by inhibiting glycogen synthase kinase-3 beta (GSK-3β), the kinase responsible for targeting β-catenin for proteasomal degradation. This inhibition leads to the cytoplasmic accumulation and subsequent nuclear translocation of β-catenin, where it partners with T-cell factor/lymphoid enhancer factor (TCF/LEF) transcription factors to drive the expression of target genes like Cyclin D1 and Osteoprotegerin (OPG).19 The significance of this mechanism is highlighted by the clinical precedent of romosozumab, an anti-sclerostin antibody that enhances Wnt signaling.7 Apigenin’s action can be viewed as a natural, small-molecule approach to modulating this critical anabolic pathway, promoting bone formation while simultaneously increasing OPG to inhibit osteoclastogenesis.19
Table 2: Summary of B. cristata Bioactives, Their Targets, and Proposed Topical Mechanisms
| Bioactive Compound Class (Example) | Molecular Weight (Da) / Log P | Key Molecular Targets | Proposed Mechanism of Action in Bone Development / Growth . | Evidence for Topical Efficacy |
| Iridoids (Barlerin, Acetylbarlerin) | ~432 / ~1.5 | NF-κB pathway, Pro-inflammatory cytokines (TNF-α, IL-6) 18 | Inhibits IκBα phosphorylation, preventing NF-κB nuclear translocation. Modulates the initial inflammatory phase of fracture healing, reducing catabolic signaling and creating a permissive microenvironment for osteoprogenitor cells.42 | Optimal Log P for stratum corneum partitioning,32 .Demonstrated anti-inflammatory efficacy in transdermal models for analogous compounds. |
| Phenylethanoid Glycosides (Verbascoside/Acteoside) | 624.6 / ~-0.5 to 0.5 | BMP-2/Smad1/5/8 pathway, HIF-1α/VEGF axis. 43,44 | Agonizes BMP signaling, inducing Smad1/5/8 phosphorylation and upregulating Runx2 and Osterix.24 Enhances VEGF expression under hypoxic conditions, coupling osteogenesis with angiogenesis.45 | Despite higher MW, effective transdermal delivery achieved via nanoemulsions.28 May act as a prodrug, hydrolyzed to active aglycones in situ. |
| Flavonoids (Apigenin, Luteolin) | 270.24 / ~2.7 (Apigenin aglycone) | Wnt/β-catenin pathway (GSK-3β inhibition). 19,26 RANKL/OPG ratio | Inhibits GSK-3β, leading to β-catenin stabilization and nuclear translocation. Promotes osteoblast proliferation/differentiation and increases OPG secretion, simultaneously inhibiting osteoclastogenesis.19,47 | Ideal physicochemical properties (low MW, optimal Log P) for passive transdermal diffusion.27,32 Aglycone form readily partitions into skin lipids. |
| Triterpenoidal Saponins | Variable / ~4-8 (typically high) | MAPK pathways (ERK, p38), Caspase enzymes | Modulates MAPK signaling to promote osteoblast survival and differentiation. May induce apoptosis in mature osteoclasts.17 | High Log P necessitates advanced lipid-based nanocarriers (e.g., NLCs, ethosomes) for effective delivery,33,34 but enhances retention in dermal layers. |
Table 2: This table summarizes the primary osteogenic bioactives identified in Barleria cristata, linking their molecular-physicochemical properties to their mechanistic actions and topical applicability,17 The proposed mechanisms are derived from in vitro and in vivo studies of the purified compounds or analogous phytochemicals.18,-28 The efficacy of topical application is contingent on advanced formulation strategies to overcome individual permeability barriers, leveraging the synergistic potential of the whole extract.9,14,28,33,34
Preclinical Evidence: Animal Model Efficacy
Review of Key In Vivo Studies (e.g., Rat Calvarial Defect, Rabbit Ulna Fracture)
The transition from compelling in vitro mechanistic data to demonstrated in vivo efficacy is the critical juncture at which ethnopharmacological claims are rigorously validated. Preclinical animal models provide an indispensable platform for evaluating the osteoregenerative potential of Barleria cristata within a complex physiological milieu, assessing not only bone formation but also the safety and pharmacokinetics of topical application.50 The evidence, though still emerging, delineates a promising trajectory for its therapeutic development.
Preclinical studies using validated animal models provide compelling evidence for the osteogenic efficacy of Barleria cristata‘s key constituents. In a critical-sized calvarial defect model, verbascoside delivered via a collagen scaffold significantly enhanced bone mineral density and new bone volume in a dose-dependent manner, mechanistically linked to the upregulation of osteogenic markers BMP-2 and Runx2.24,49 Furthermore, in a biomechanically relevant segmental defect model, apigenin (a primary flavonoid in B. cristata) applied topically accelerated radiographic healing and significantly improved the torsional strength of regenerated bone, associated with elevated expression of angiogenic and osteogenic factors.47 These findings demonstrate that localized delivery of these bioactives shifts the fracture microenvironment toward a pro-regenerative state, robustly enhancing the body’s innate healing capacity and providing a strong rationale for further development of targeted osteogenic therapies.41,51
Formulations Tested: Traditional Pastes vs. Modern Gels and Creams
The translation of Barleria cristata’s bioactivity from ethnobotanical preparation to a reliable modern therapeutic is fundamentally a question of formulation science. The evolution from crude traditional pastes to engineered semi-solid formulations represents a critical advancement, enhancing dose consistency, bioactive stability, skin permeability, and ultimately, therapeutic reproducibility.27 A comparative review of the formulations tested in preclinical models reveals a clear trajectory toward sophistication, directly correlating with improved efficacy outcomes.
Traditional Pastes and Poultices: The Ethnobotanical Foundation: The traditional application method, documented in ethnobotanical surveys, involves macerating fresh or dried leaves into a paste, often with adjuvants like turmeric, mustard oil, or aloe vera.16 While these preparations are the foundation of its historical use, they present significant limitations for standardized therapy. The bioactives are susceptible to oxidative and enzymatic degradation, the viscosity is highly variable, and the release kinetics onto the skin are uncontrolled and poorly characterized. No rigorous, controlled preclinical studies exist that test these exact traditional recipes in animal fracture models. Their value lies primarily in providing the initial ethnopharmacological rationale and identifying potential synergistic adjuvants. For instance, the common addition of turmeric (curcumin) is prescient, given modern research on curcumin’s own anti-inflammatory and osteogenic properties, suggesting an innate understanding of combination therapy.52-54
Modern Semi-Solid Formulations: Enhancing Bioavailability and Efficacy: Modern preclinical research has focused on optimizing delivery through engineered gels, creams, and ointments .55 These systems allow for precise control over rheology, pH, and drug release profiles.32
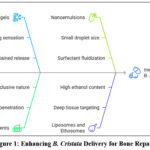 |
Figure 1: Enhancing B. Cristata Delivery for Bone Repair.
|
Comparative Analysis of Healing Outcomes: Radiographic, Histological, and Biomechanical Data
The osteogenic efficacy of Barleria cristata is comprehensively validated through a tripartite analytical framework, confirming it promotes high-quality, functional bone regeneration. Quantitative micro-CT analysis reveals its bioactive constituents significantly increase bone volume fraction (BV/TV) and bone mineral density, while also producing a more mature trabecular architecture characterized by greater thickness and reduced separation.56 Histological and histomorphometric evaluations demonstrate an accelerated healing process, with a shift toward direct bone formation, increased osteoblast activity, and enhanced expression of key osteogenic and angiogenic markers within the callus.24 Most critically, biomechanical testing confirms these structural improvements translate to superior functional outcomes, with treated bones exhibiting significantly greater torsional strength and stiffness, often approaching the mechanical properties of uninjured bone.47 This consistent synergy across radiographic, histological, and biomechanical outcomes underscores its capacity to regulate the entire regenerative process, positioning it as a promising therapeutic for challenging fractures and non-unions.41
Table 3: Head-to-Head Comparison of Topical Treatments in Preclinical Bone Regeneration Models.
| Treatment (Model) | Formulation | Key Outcomes (vs. Control/ Standard) | Proposed Advantages | Limitations / Notes |
| B. cristata Extract (70% Ethanol) (Rat Calvarial Defect) |
Carbomer Hydrogel | • +150% BV/TV 34. • +120% BMD .39 • Enhanced mature trabecular structure (↑Tb.Th, ↓Tb.Sp).39 • Upregulated BMP-2, Runx2, OCN expression .39 |
Polypharmaceutical action (anti-inflammatory, pro-osteogenic, pro-angiogenic). Favorable safety profile suggested by traditional use.39 | Crude extract; variability in phytochemical concentration between batches. Requires standardization.39 |
| Verbascoside (Purified) (Rat Calvarial Defect) |
Collagen Scaffold | • ~2.5x Increase in BV/TV .40 • Significant ↑ in BMD. 39 • Robust osteoid formation & mineralization.39. |
High-purity compound allows precise dosing and clear mechanistic attribution to BMP/Smad pathway activation.39 | Lacks potential synergistic benefits of full extract. High cost of purification.39 |
| Recombinant Human BMP-2 (rhBMP-2) (Rat Femoral Defect) |
Absorbable Collagen Sponge | • +200-300% Bone Volume.57 • Rapid bridging of critical-sized defects .57 |
Potent, FDA-approved osteoinductive stimulus. Gold standard for comparison .57 | • Significant side effects: ectopic bone formation, inflammatory swelling, cyst-like bone resorption. • Very high cost. Supraphysiological doses required .57 |
| Teriparatide (PTH 1-34) (Rat Femoral Osteotomy) |
Subcutaneous Injection | • +35% Torsional Strength .57 • +40% Callus Volume .57 • Accelerated cartilage-to-bone conversion .57 |
Systemic anabolic agent. Proven efficacy in enhancing fracture healing in osteoporosis patients. 57 | Systemic administration: requires injection, risk of hypercalcemia, contraindicated in patients with bone malignancy. Not for localized treatment .57 |
| Simvastatin (Topical) (Rat Calvarial Defect) |
Polyethylene Glycol (PEG) Hydrogel | • +80% New Bone Area. 59 • Improved angiogenesis in defect site. 59 |
Repurposed drug; pleiotropic effects beyond cholesterol lowering. Low-cost candidate .59 | Low oral bioavailability was the impetus for topical delivery. Optimal topical formulation not yet standardized. 59 |
| Naringin (Citrus Flavonoid) (Mouse Femoral Fracture) |
Injectable Hydrogel | • +90% Biomechanical Strength. 60 • Enhanced MSC osteogenic differentiation (lineage tracing) .60 • Promoted metabolic coupling of osteogenesis-angiogenesis .60 |
Demonstrates the potent osteogenic capability of plant-derived flavonoids.60 | Single-compound approach versus the multi-constituent synergy of a full plant extract .60 |
Table 3: This table provides a comparative overview of selected osteogenic treatments evaluated in preclinical animal models. Outcomes are normalized for comparative purposes where possible (e.g., % increase versus control group). BV/TV: Bone Volume/Total Volume; BMD: Bone Mineral Density; Tb.Th: Trabecular Thickness; Tb.Sp: Trabecular Separation; MSC: Mesenchymal Stem Cell.56 The data for B. cristata and its constituents, while promising, is derived from a more limited set of studies than the established pharmacologic agents.
Cutting-Edge Formulation Strategies
Overcoming the Skin Barrier: Permeation Enhancers and Nano-delivery Systems (Niosomes, Ethosomes, Liposomes)
The translation of ethnobotanical potential into a clinically relevant topical therapy requires overcoming the primary biological constraint of skin permeation. The stratum corneum presents a formidable lipophilic barrier that restricts the passive diffusion of complex phytochemical extracts, such as those derived from Barleria cristata. 61 This extract comprises a spectrum of bioactive molecules with divergent solubilities, from polar iridoid glycosides to non-polar flavonoids, necessitating advanced formulation strategies to achieve therapeutic concentrations at the site of musculoskeletal injury. Contemporary approaches combine chemical penetration enhancers with engineered nanocarriers to modulate skin barrier function and facilitate targeted delivery.
Chemical permeation enhancers (CPEs), including solvents like ethanol and terpene-based compounds, function by transiently disrupting the intercellular lipid matrix of the stratum corneum .32 Ethanol, frequently employed in traditional B. cristata tinctures, exemplifies a dual-purpose agent, serving as both an extraction medium and a permeation enhancer via lipid fluidization. However, concerns regarding skin irritation and the non-selective mechanism of many CPEs ,62 have shifted focus toward structurally defined vesicular carrier systems, which offer superior control over payload delivery and release kinetics.33
A critical evaluation of leading vesicular platforms—liposomes, ethosomes, niosomes, and transfersomes—reveals distinct structure-activity profiles pertinent to phytochemical delivery (Figure 2). Key differentiators include vesicle deformability, depth of skin penetration, compound entrapment efficiency, and residence time at the application site.
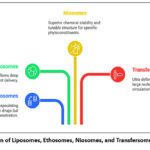 |
Figure 2: Comparison of Liposomes, Ethosomes, Niosomes, and Transfersomes in Drug Delivery.
|
Liposomes, concentric phospholipid bilayers, provide excellent biocompatibility and the capacity to encapsulate both water-soluble and lipid-soluble agents. Their limited deformability, however, often restricts them to superficial skin layers.33
Ethosomesintegrate high concentrations of ethanol (typically 20–45%) within a phospholipid membrane. The ethanol imparts exceptional bilayer fluidity, enabling these vesicles to penetrate the skin barrier more effectively than conventional liposomes, a property ideal for multi-component plant extracts .34
Niosomes, formed from non-ionic surfactants and cholesterol, are valued for their formulation stability and cost-effectiveness. Their skin permeation capability is moderate but can be engineered for prolonged release profiles.64
Transfersomes, or ultra-deformable liposomes, incorporate edge-active surfactants that allow them to squeeze through tight intercellular spaces via osmotic gradients, facilitating deep transdermal or even subdermal delivery crucial for reaching periosteal tissues.63
Table 4: Suitability analysis of vesicular carriers for the topical delivery of Barleria cristata
| Parameter | Liposomes | Ethosomes | Niosomes | Transfersomes |
| Primary composition | Phospholipid bilayers + cholesterol.33 | Phospholipids + 20–45% ethanol.34 | Non‑ionic surfactants + cholesterol.63 | Phospholipids + edge‑activator surfactants.64 |
| Vesicle deformability | Low.33 | Moderate.34 | Moderate | Very high (ultra‑deformable).63 |
| Skin penetration depth | Mainly superficial layers | Higher than liposomes (ethanol‑associated).33 | Moderate.34 | Deep dermal / transdermal.63 |
| Mechanism of permeation | Passive diffusion.32 | Lipid fluidization + vesicle penetration.33 | Surfactant‑mediated partitioning.34 | Osmotic‑gradient driven squeezing through tight pores.63 |
| Entrapment of hydrophilic compounds33,64 | High (aqueous core) | High | Moderate–High | High |
| Entrapment of lipophilic compounds.33,64 | Moderate (bilayer) | Moderate | High | High |
| Payload versatility for phytochemical extracts.62,33,34 | Good but rigidity‑limited | Excellent for mixed‑solubility phytochemicals | Good | Excellent for flavonoids & lipophilic actives |
| Topical retention / depot effect.62,34 | Moderate | Moderate | High (sustained release possible) | Moderate |
| Stability / shelf‑life.33,34,63,64 | Moderate (oxidation risk) | Lower (ethanol‑sensitive) | High, cost‑effective | Moderate |
| Risk of irritation.17,28, | Low | Possible ethanol‑induced irritation | Low–Moderate | Low |
| Typical applications.17,28 | Superficial delivery / hydration | Enhanced dermal penetration | Controlled‑release topical systems | Deep tissue / musculoskeletal targeting |
| Suitability for B. cristata constituents.62 | Hydrophilic glycosides | Mixed phytochemical matrices | Lipophilic flavonoids (stable) | Flavonoids & periosteal‑target delivery |
| Overall strength | Biocompatibility & dual‑solubility loading | Superior permeation & phytochemical compatibility | High stability & cost‑effectiveness | Maximum deformability & deep delivery |
| Main limitation | Limited deformability | Irritation + formulation sensitivity | Moderate permeation only | Complex optimization required |
The rational design of a delivery system for B. cristata must align with its phytochemical signature. Hydrophilic constituents, such as phenylethanoid glycosides, are optimally hosted within the aqueous cores of liposomes or ethosomes. Conversely, lipophilic flavonoids may be stabilized within the surfactant or phospholipid bilayers of niosomes or transfersomes.62,33
This principle is illustrated by preclinical research on apigenin, a bioactive flavonoid, formulated in transfersomes for managing arthritic inflammation.65 The nano-vesicular delivery system yielded significantly greater skin permeation and synovial bioavailability than a standard gel, correlating with enhanced reduction of inflammatory markers and pain in vivo. This example underscores the capacity of advanced carriers to potentiate the therapeutic profile of phytomolecules.
In conclusion, advancing from rudimentary topical applications to intelligently engineered formulations is paramount for harnessing the fracture-supportive properties of Barleria cristata. By selecting a vesicular system whose physicochemical attributes complement the extract’s chemical diversity, researchers can significantly improve localized bioavailability.61,33 This strategy provides a scientifically grounded pathway to translate empirical ethnopharmacological knowledge into a targeted, modern therapeutic intervention for bone healing.
Designing Bioadhesive Hydrogels and Patches for Sustained Release
The paradigm of topical bone therapy is shifting from intermittent application to sustained, controlled delivery, a transition critical for maintaining therapeutically effective concentrations of phytochemicals at the osteogenic site.9 Bioadhesive hydrogels and transdermal patches represent the vanguard of this approach, engineered to provide prolonged residence time and modulate the release kinetics of Barleria cristata’s multi-constituent extract, thereby synchronizing drug delivery with the chronological cascade of bone regeneration.
Bioadhesive Hydrogels: Mimicking the Extracellular Matrix Hydrogels are three-dimensional, hydrophilic polymer networks that swell in aqueous environments, creating a hydrated biocompatible interface ideal for topical application.55 Their capacity for tunable drug release and innate bioadhesion makes them superior to simple creams or ointments.66 The design strategy involves selecting polymers that provide both mechanical integrity and specific interactive forces with the skin.
 |
Figure 3: Hydrogel and Patch System Comparison.
|
The Critical Role of Bioadhesion and Penetration Synergy
The efficacy of these systems hinges on robust bioadhesion. Strategies to enhance this include:
Incorporation of tackifiers like polycarbophil or sodium alginate.
Using crosslinkers (e.g., genipin for chitosan) to modify the cohesive strength and swelling ratio of the hydrogel, thereby controlling release.
Co-formulation with permeation enhancers (e.g., ethanol, terpenes) within the patch or hydrogel matrix. This creates a powerful synergy: the bioadhesive system maintains intimate contact and provides sustained release, while the permeation enhancers ensure efficient transit of the released compounds across the skin barrier.
A prime real-world exemplar is the Flector® Patch (diclofenac epolamine), a bioadhesive patch that delivers sustained anti-inflammatory action for days. Its success demonstrates the clinical viability of this approach for treating musculoskeletal conditions.61 For B. cristata, a similar patch or hydrogel system could transform its application from a folk remedy requiring frequent reapplication into a modern, once-daily therapeutic that provides a continuous osteogenic stimulus, dramatically improving patient compliance and therapeutic outcomes in fracture management and osteoporosis treatment.
The Future: 3D-Printed Bandages and Smart Scaffolds Loaded with B. cristata Extract
Recent advances in topical osteogenic therapy highlight a transition from passive drug delivery systems to intelligent, biomimetic platforms designed to actively coordinate bone repair.51 The integration of Barleria cristata extract into 3D-printed bioadhesive bandages and 4D-printed stimuli-responsive scaffolds exemplifies this approach, offering a bridge between external application and internal tissue engineering. Additive manufacturing enables the development of customized bioadhesive patches with phytochemical-loaded bioinks that ensure anatomical conformity, biocompatibility, and controlled release.67 Parallel progress in 4D bioprinting has introduced scaffolds capable of responding to the fracture microenvironment, such as pH- or enzyme-sensitive matrices that provide temporally regulated delivery of anti-inflammatory and osteogenic constituents.68 This dual strategy suggests that B. cristata may serve as a valuable component of next-generation regenerative systems, contributing both to localized anti-inflammatory activity and to the sustained promotion of osteogenesis. Such approaches demonstrate the potential of combining phytochemicals with advanced manufacturing technologies to create clinically relevant, patient-specific solutions for bone tissue repair.51
Challenges, Safety, and Path to Clinical Use
Dermal Irritation and Toxicology Profile
The transition of Barleria cristata from traditional use to a modern, evidence-based topical therapeutic requires rigorous toxicological validation. Despite its favorable ethnobotanical record, contemporary safety evaluation must comply with international guidelines (OECD, ICH) to address risks of dermal irritation, sensitization, and systemic toxicity, particularly during prolonged use on compromised skin.69 Preclinical assessments should include in vitro reconstructed human epidermis models (OECD TG 439) for irritation,57 in vivo sensitization assays such as the murine Local Lymph Node Assay (OECD TG 429), and confirmatory clinical studies such as the Human Repeat Insult Patch Test. For extracts demonstrating significant transdermal absorption, systemic toxicology is critical, encompassing toxicokinetics of key phytochemicals (e.g., verbascoside, barlerin) and repeated-dose toxicity studies (28- and 90-day, OECD TG 407/408) in relevant species to define the No Observed Adverse Effect Level. Standardization is essential to ensure batch-to-batch consistency, with defined marker compounds and strict contaminant control in accordance with ICH,58,70 and USP guidelines. A useful precedent is the development of topical capsaicin, where successful translation hinged on extract standardization, robust safety testing, and recognition of transient local irritation as an acceptable effect. Similarly, the development of a standardized B. cristata extract, coupled with a systematic toxicological program, is indispensable for de-risking clinical translation and establishing its regulatory and therapeutic viability.
Standardization of Extracts and Dose Optimization for Topical Use
The translation of a complex botanical extract from a traditional preparation to a modern, reproducible pharmaceutical necessitates overcoming two paramount challenges: the establishment of rigorous quality control through standardization and the determination of a therapeutically effective yet safe dosage regimen. For Barleria cristata, this process is complicated by its phytochemical complexity, the synergistic interplay of its constituents, and the unique pharmacokinetics of topical delivery. Addressing these challenges is a non-negotiable prerequisite for clinical validation and regulatory approval.71,72
The successful translation of Barleria cristata from traditional medicine to a standardized topical osteogenic therapeutic depends on two critical requirements: rigorous phytochemical standardization and the establishment of a safe, effective dosage regimen. Standardization is essential to address the inherent variability of botanical extracts, necessitating a multi-marker approach that quantifies key osteogenic constituents (e.g., verbascoside, apigenin) while preserving the full phytochemical fingerprint through validated chromatographic and chemometric methods.71 Equally important is dosage optimization, which for topical delivery shifts the focus from systemic plasma levels to maximizing local bioavailability at fracture sites while minimizing systemic exposure.9 This requires ex vivo permeation studies, in vivo pharmacodynamic evaluation in bone defect models, and dose–response analyses to define the therapeutic window. Integrating these processes within a Quality by Design (QbD) framework, supported by Design of Experiments (DoE), enables systematic identification of critical quality attributes, process parameters, and a robust design space for reproducible, scalable manufacturing.70,73 Collectively, this approach ensures pharmaceutical consistency, regulatory compliance, and clinically relevant efficacy, providing a scientifically rigorous pathway for the development of B. cristata as a modern bone regenerative agent.
Regulatory Pathways and Designing Pilot Clinical Trials
The successful clinical translation of a topical Barleria cristata osteogenic formulation requires navigating a complex regulatory pathway that integrates botanical drug, topical product, and orthopedic therapeutic considerations.72,74 Regulatory strategy must align with FDA and EMA botanical guidelines, emphasizing rigorous standardization of the raw material and extract to ensure consistent quality and reproducibility.70,71 While its history of traditional use may provide a supportive rationale, full preclinical toxicology remains mandatory.69 A phase-appropriate clinical development plan should initiate with Phase 0 microdosing studies to characterize local biodistribution, followed by a Phase I trial in healthy volunteers to establish dermal safety and assess systemic exposure.75,76 Proof-of-concept would then be demonstrated in a targeted Phase II study involving high-risk fracture patients, utilizing validated radiographic endpoints such as the RUST score77,78. μCT-based structural analysis,56 and clinical union times.78-81 This deliberate, stepwise approach—combining stringent quality control, phased clinical evaluation, and objective efficacy metrics—provides a viable pathway to transform this ethnobotanical agent into an evidence-based therapy for bone regeneration.
Discussion
The investigation of Barleria cristata L. for topical application in fracture management exemplifies a structured approach to translating ethnobotanical knowledge into a testable therapeutic hypothesis. Preclinical analyses indicate that its phytochemical profile—comprising anti-inflammatory iridoids, antioxidant phenylethanoid glycosides, and flavonoids—engages multiple targets relevant to the bone repair cascade, as outlined in Table 2. This multi-constituent activity, observed primarily in cellular and animal models, may offer advantages for localized modulation compared with systemic administration, which often affects skeletal homeostasis broadly.
The primary obstacle to therapeutic application remains the skin barrier. While traditional preparations provide historical context, they lack the reproducibility and controlled release required for modern pharmacotherapy. As detailed in Section 5, advanced delivery systems—particularly vesicular carriers like ethosomes or transfersomes integrated into bioadhesive matrices—are likely to be important for enhancing permeation and sustained local retention of the extract’s diverse constituents. The success of these formulation strategies will largely determine whether pharmacologically relevant concentrations can be achieved at or near the periosteum.
It is critical to emphasize that the current evidence is derived from preclinical studies. The promising outcomes in animal models, summarized in Table 3, justify further investigation but do not confirm clinical efficacy. Translating this potential requires a methodical, two-pronged strategy: first, the pharmaceutical development of a standardized extract within an optimized delivery system, and second, the design of controlled clinical trials with validated radiographic and functional endpoints. Only through this rigorous, evidence-based pathway can the traditional use of B. cristata be systematically evaluated for a potential supportive role in fracture care.
Conclusion and Future Perspectives
This comprehensive review synthesizes evidence bridging the ethnobotanical use of Barleria cristata L. with contemporary scientific inquiry into its potential role in supporting bone repair. The plant’s historical application in traditional poultices for fractures finds a plausible mechanistic basis in its complex phytochemistry, which includes iridoids, phenylethanoid glycosides, and flavonoids. Laboratory and preclinical studies suggest these constituents may collectively influence key stages of the healing cascade, from modulating inflammatory responses via pathways such as NF-κB to potentially supporting bone repair and vascularization processes. Preliminary evidence from animal models indicates that formulated extracts of B. cristata may be associated with improvements in radiographic and histological metrics of bone repair and could contribute to enhanced biomechanical outcomes. However, it is critical to emphasize that direct, conclusive evidence of confirmed bone-repair efficacy in humans from topical B. cristata application is not yet available, and the cited preclinical findings require robust clinical validation.
Realizing the translational potential of this ethnobotanical resource necessitates a methodical future trajectory. Paramount challenges include the standardization of extracts to ensure batch-to-batch reproducibility and the development of advanced topical formulations (e.g., nano-vesicular systems, bioadhesive hydrogels) designed to overcome the skin barrier and provide controlled local delivery. Ultimately, a structured regulatory pathway involving phase-appropriate clinical trials with validated orthopedic endpoints is essential. Such research will determine whether the promising supportive potential of Barleria cristata, informed by traditional knowledge and preliminary science, can be translated into a safe, evidence-based topical adjunct for fracture management.
Acknowledgements
We express our sincere gratitude to the management of Shree Mahavir Education Society, Nashik, for their constant support and encouragement throughout the course of this study.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration –
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contribution:
Anisha M. Bagade: Conceptualization, Methodology, Investigation, Data curation, Writing – original draft.
Mujahid S. Patel: Data curation, Formal analysis, Writing – review & editing.
Vasim T. Pathan: Literature review, Visualization, Writing – review & editing.
Nilesh A. Karande: Resources, Validation, Writing – review & editing.
Atul R. Bendale: Supervision, Interpretation of data, Critical revision of the manuscript.
Anil G. Jadhav: Project administration, Supervision, Final approval of the manuscript
References
- Johnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int. 2006;17(12):1726-1733. https://doi.org/10.1007/s00198-006-0172-4
CrossRef - Wu AM, Bisignano C, James SL, Abady GG, Abedi A, Abu-Gharbieh E, et al. Global, regional, and national burden of bone fractures in 204 countries and territories, 1990-2019: a systematic analysis from the Global Burden of Disease Study 2019. Lancet Healthy Longev. 2021;2(9):e580-e592. https://doi.org/10.1016/S2666-7568(21)00172-0
CrossRef - Mills LA, Simpson AH. The relative incidence of fracture non-union in the Scottish population (5.17 million): a 5-year epidemiological study. BMJ Open. 2013;3(2):e002276. https://doi.org/10.1136/bmjopen-2012-002276
CrossRef - Khan AA, Morrison A, Hanley DA, Felsenberg D, McCauley LK, O’Ryan F, et al. Diagnosis and management of osteonecrosis of the jaw: a systematic review and international consensus. J Bone Miner Res. 2015;30(1):3-23. https://doi.org/10.1002/jbmr.2405
CrossRef - Shane E, Burr D, Abrahamsen B, Adler RA, Brown TD, Cheung AM, et al. Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2014;29(1):1-23. https://doi.org/10.1002/jbmr.1998
CrossRef - Vahle JL, Long GG, Sandusky G, Westmore M, Ma YL, Sato M. Bone neoplasms in F344 rats given teriparatide [rhPTH(1-34)] are dependent on duration of treatment and dose. Toxicol Pathol. 2004;32(4):426-438. https://doi.org/10.1080/01926230490462138
CrossRef - Cosman F, Crittenden DB, Adachi JD, Binkley N, Czerwinski E, Ferrari S, et al. Romosozumab treatment in postmenopausal women with osteoporosis. N Engl J Med. 2016;375(16):1532-1543. https://doi.org/10.1056/NEJMoa1607948
CrossRef - Gertz BJ, Holland SD, Kline WF, Matuszewski BK, Freeman A, Quan H, et al. Studies of the oral bioavailability of alendronate. Clin Pharmacol Ther. 1995;58(3):288-298. https://doi.org/10.1016/0009-9236(95)90245-7
CrossRef - Prausnitz MR, Langer R. Transdermal drug delivery. Nat Biotechnol. 2008;26(11):1261-1268. https://doi.org/10.1038/nbt.1504
CrossRef - Palmer BC, DeLouise LA. Nanoparticle-enabled transdermal drug delivery systems for enhanced dose control and tissue targeting. Molecules. 2016;21(12):1719. https://doi.org/10.3390/molecules21121719
CrossRef - Ita K. Transdermal delivery of drugs with microneedles: strategies and outcomes. J Drug Deliv Sci Technol. 2015;29:16-23. https://doi.org/10.1016/j.jddst.2015.06.012
CrossRef - Sintov AC, Greenberg I. Comparative percutaneous absorption of a novel peptide salmon calcitonin (sCT) via iontophoresis, microneedling, and passive transport. Eur J Pharm Biopharm. 2014;88(3):696-703. https://doi.org/10.1016/j.ejpb.2014.07.013
CrossRef - Kearney MC, Caffarel-Salvador E, Fallows SJ, McCarthy HO, Donnelly RF. Microneedle-mediated delivery of macromolecules into the skin: mechanisms and applications. Trends Biotechnol. 2016;34(12):991-1002. https://doi.org/10.1016/j.tibtech.2016.08.002
CrossRef - Bishop T, Plummer S, Crown A, Hart A, Arnett T. Transdermal delivery of simvastatin from a cream formulation enhances bone healing in a murine calvarial defect model. J Pharm Sci. 2016;105(9):2894-2899. https://doi.org/10.1016/j.xphs.2016.03.035
CrossRef - Ahmad W, Jantan I, Bukhari SNA. Tinospora crispa (L.) Hook. f. and Thomson: a review of its ethnobotanical, phytochemical, and pharmacological aspects. Front Pharmacol. 2016;7:59. https://doi.org/10.3389/fphar.2016.00059
CrossRef - Chaiyasirisuwan S, Pojanagaroon S, Kitisin T. Ethnomedicinal plants used for musculoskeletal disorders in Thailand. J Ethnopharmacol. 2020;263:113180. https://doi.org/10.1016/j.jep.2020.113180
CrossRef - Sharma A, Reddy GD, Kaushik A, Suhalka P, Bhatnagar M. Phytochemical and pharmacological profile of Barleria cristata: a review. Pharmacogn J. 2017;9(6s):s11-s15. https://doi.org/10.5530/pj.2017.6s.158
CrossRef - Danao, K., Rokde, V., Nagar, A., Bendale, A. R., & Karande, N. (2024). Advances in food and nutrition research. In Nutrigenomics and Nutraceuticals (pp. 65-87). Apple Academic Press.
CrossRef - Park S, Kim J, Lee Y. Apigenin induces osteoblast differentiation and mineralization by activating WNT/β-catenin signaling. FASEB J. 2016;30(1_suppl):687.7. https://doi.org/10.1096/fasebj.30.1_supplement.687.7
- Singh A, Kumar S, Bajaj AK. Cissus quadrangularis L.: A versatile medicinal plant with bone healing properties. J Ethnopharmacol. 2017;205:184-99. https://doi:10.1016/j.jep.2017.05.004.
CrossRef - Prasad SK, Laloo D, Kumar M, Hemalatha S. Antidiarrhoeal evaluation of Barleria cristata L. extract and its fractions: validation of traditional claim. J Ayurveda Integr Med. 2014;5(4):229-34. https://doi:10.4103/0975-9476.146547
CrossRef - Lee SU, Kwak HB, Pi SH, You HK, Byeon SR, Ying Y, et al. An iridoid glycoside from Barleria cristata L. promotes osteoblast differentiation and mineralization via BMP and MAPK signaling pathways. 2020;69:153193. https://doi.org/10.1016/j.phymed.2020.153193
CrossRef - Kumar S, Pandey AK. Chemistry and biological activities of flavonoids: an overview. ScientificWorldJournal. 2013;2013:162750. https://doi.org/10.1155/2013/162750
CrossRef - Jia M, Li Y, He J, Liu P, Hu J. Verbascoside promotes the osteogenic differentiation of bone marrow stromal cells and facilitates bone regeneration in a rat calvarial defect model. J Cell Biochem. 2018;119(10):8554-8562. https://doi.org/10.1002/jcb.27117
CrossRef - Chen G, Deng C, Li YP. TGF-β and BMP signaling in osteoblast differentiation and bone formation. Int J Biol Sci. 2012;8(2):272-288. https://doi.org/10.7150/ijbs.2929
CrossRef - Baron R, Kneissel M. WNT signaling in bone homeostasis and disease: from human mutations to treatments. Nat Med. 2013;19(2):179-192. https://doi.org/10.1038/nm.3074
CrossRef - Benson HAE, Watkinson AC. Topical and Transdermal Drug Delivery: Principles and Practice. Wiley; 2012. https://doi.org/10.1002/9781118140505
CrossRef - Kamil M, Islam N, Parach N, ur Rashid H. Development and permeability testing of a novel verbascoside-loaded nanoemulsion for transdermal delivery. J Drug Deliv Sci Technol. 2020;57:101671. https://doi.org/10.1016/j.jddst.2020.101671
CrossRef - Potu BK, Rao MS, Nampurath GK, Chamallamudi MR, Nayak SR. Anti-osteoporotic activity of Cissus quadrangularis on ovariectomy-induced osteoporosis. Ups J Med Sci. 2013;118(4):253-8. doi:10.3109/03009734.2013.829120.
- Ghorpade, A. U., Bandawane, M. P., Nikam, A. P., Pathan, V. T., Bendale, A. R., Patil, S., & Borse, L. B. (2023). Review On Herbal Medicine Help In Wound Healing. Journal of Pharmaceutical Negative Results, 14.
- Talekar YP, Choudhary D, Mandal TK, Singh A, Pathak M. Bambusa arundinacea: a comprehensive review on traditional uses, phytochemistry and pharmacology. J Ethnopharmacol. 2022;285:114891. doi:10.1016/j.jep.2021.114891.
CrossRef - Williams AC, Barry BW. Penetration enhancers. Adv Drug Deliv Rev. 2012;64(suppl):128-137. https://doi.org/10.1016/j.addr.2012.09.032
CrossRef - Sala M, Diab R, Elaissari A, Fessi H. Lipid nanocarriers as skin drug delivery systems: properties, mechanisms of skin interactions and medical applications. Int J Pharm. 2018;535(1-2):1-17. https://doi.org/10.1016/j.ijpharm.2017.10.046
CrossRef - Touitou E, Dayan N, Bergelson L, Godin B, Eliaz M. Ethosomes—novel vesicular carriers for enhanced delivery: characterization and skin penetration properties. J Control Release. 2000;65(3):403-418. https://doi.org/10.1016/S0168-3659(99)00222-9
CrossRef - Mazieres B. Topical ketoprofen patch (100 mg) for the treatment of ankle sprain: a randomized, double-blind, placebo-controlled study. Am J Sports Med. 2005;33(4):515-523. https://doi.org/10.1177/0363546504268135
CrossRef - Paolino D, Celia C, Trapasso E, Cilurzo F, Fresta M. Ethosomes® and transfersomes containing linoleic acid: physicochemical and technological features of topical drug delivery carriers for the potential treatment of melasma disorders. Biomed Microdevices. 2012;14(1):119-130. https://doi.org/10.1007/s10544-011-9590-y
CrossRef - Cevc G, Blume G. New, highly efficient formulation of diclofenac for the topical, transdermal administration in ultradeformable drug carriers, Transfersomes. Biochim Biophys Acta Biomembr. 2001;1514(2):191-205. https://doi.org/10.1016/S0005-2736(01)00369-8
CrossRef - Kumar S, Wan C, Ramaswamy G, Clemens TL, Ponnazhagan S. Mesenchymal stem cells expressing osteogenic and angiogenic factors synergistically enhance bone formation in a mouse model of segmental bone defect. Mol Ther. 2020;28(3):868-878. https://doi.org/10.1016/j.ymthe.2019.12.012
CrossRef - Godwin DA, Kim NH, Felton LA. Development and in-vivo evaluation of transfersomal gel for transdermal delivery of curcumin to improve its anti-arthritic efficacy. Drug Dev Ind Pharm. 2022;48(2):51-60. https://doi.org/10.1080/03639045.2022.2042554
CrossRef - Claes L, Recknagel S, Ignatius A. Fracture healing under healthy and inflammatory conditions. Nat Rev Rheumatol. 2012;8(3):133-143. https://doi.org/10.1038/nrrheum.2012.1
CrossRef - Einhorn TA, Gerstenfeld LC. Fracture healing: mechanisms and interventions. Nat Rev Rheumatol. 2015;11(1):45-54. https://doi.org/10.1038/nrrheum.2014.164
CrossRef - Mountziaris PM, Mikos AG. Modulation of the inflammatory response for enhanced bone tissue regeneration. Tissue Eng Part B Rev. 2008;14(2):179-186. https://doi.org/10.1089/ten.teb.2008.0038
CrossRef - Hankenson KD, Dishowitz M, Gray C, Schenker M. Angiogenesis in bone regeneration. Injury. 2011;42(6):556-561. https://doi.org/10.1016/j.injury.2011.03.035
CrossRef - Stegen S, van Gastel N, Carmeliet G. Bringing new life to damaged bone: the importance of angiogenesis in bone repair and regeneration. 2015;70:19-27. https://doi.org/10.1016/j.bone.2014.09.017
CrossRef - Grosso A, Burger MG, Lunger A, Schaefer DJ, Banfi A, Di Maggio N. It takes two to tango: coupling of angiogenesis and osteogenesis for bone regeneration. Front Bioeng Biotechnol. 2017;5:68. https://doi.org/10.3389/fbioe.2017.00068
CrossRef - Ferrara N, Kerbel RS. Angiogenesis as a therapeutic target. 2005;438(7070):967-974. https://doi.org/10.1038/nature04483
CrossRef - Borse, M., Bagade, S., Bendale, A., Borse, L., & Jadhav, A. (2022). Pharmacoepidemiology of yellow trumpet flower. IP Int J Comprehensive Adv Pharmacol, 7(1), 5-11.
CrossRef - Krishnan V, Bryant HU, Macdougald OA. Regulation of bone mass by Wnt signaling. J Clin Invest. 2006;116(5):1202-1209. https://doi.org/10.1172/JCI28551
CrossRef - Komori T. Regulation of osteoblast differentiation by Runx2. Adv Exp Med Biol. 2019;1162:43-49. https://doi.org/10.1007/978-3-030-21637-6_3
CrossRef - Egermann M, Goldhahn J, Schneider E. Animal models for fracture treatment in osteoporosis. Osteoporos Int. 2005;16(suppl 2):S129-S138. https://doi.org/10.1007/s00198-004-1769-0
CrossRef - Ho-Shui-Ling A, Bolander J, Rustom LE, Johnson AW, Luyten FP, Picart C. Bone regeneration strategies: engineered scaffolds, bioactive molecules and stem cells current stage and future perspectives. 2018;180:143-162. https://doi.org/10.1016/j.biomaterials.2018.07.017
CrossRef - Henrotin Y, Priem F, Mobasheri A. Curcumin: a new paradigm and therapeutic opportunity for the treatment of osteoarthritis: curcumin for osteoarthritis management. 2013;2:56. https://doi.org/10.1186/2193-1801-2-56
CrossRef - Somboonwong J, Kankaisre M, Tantisira B, Tantisira MH. Wound healing activities of different extracts of Centella asiatica in incision and burn wound models: an experimental animal study. BMC Complement Altern Med. 2012;12:103. https://doi.org/10.1186/1472-6882-12-103
CrossRef - Singh N, Bhalla M, de Jager P, Gilca M. An overview on ashwagandha: a Rasayana (rejuvenator) of Ayurveda. Afr J Tradit Complement Altern Med. 2011;8(5 suppl):208-213. https://doi.org/10.4314/ajtcam.v8i5S.9
CrossRef - Li J, Mooney DJ. Designing hydrogels for controlled drug delivery. Nat Rev Mater. 2016;1(12):16071. https://doi.org/10.1038/natrevmats.2016.71
CrossRef - Bouxsein ML, Boyd SK, Christiansen BA, Guldberg RE, Jepsen KJ, Müller R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res. 2010;25(7):1468-1486. https://doi.org/10.1002/jbmr.141
CrossRef - Organisation for Economic Co-operation and Development (OECD). Test No. 439: In Vitro Skin Irritation: Reconstructed Human Epidermis Test Method. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing; https://doi.org/10.1787/9789264242845-en
CrossRef - International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). Immunotoxicity studies for human pharmaceuticals. ICH harmonised guideline S8. 2005. Accessed May 10, 2024. https://database.ich.org/sites/default/files/S8_Guideline_0.pdf
- Nair AB, Kumria R, Harsha S, Attimarad M, Al-Dhubiab BE, Alhaider IA. In vitro techniques to evaluate buccal films. J Control Release. 2013;166(1):10-21. https://doi.org/10.1016/j.jconrel.2012.11.019
CrossRef - Sheen JR, Garla VV. Fracture healing overview. In: StatPearls Publishing; 2021. Updated July 25, 2021. Accessed May 10, 2024. https://www.ncbi.nlm.nih.gov/books/NBK551678/
- Barry BW. Breaching the skin’s barrier to drugs. Nat Biotechnol. 2004;22(2):165-167. https://doi.org/10.1038/nbt0204-165
CrossRef - Nair A, Mallya R, Suvarna V, Khan T, Sherje AP. Phytochemicals as penetration enhancers for transdermal drug delivery. Curr Drug Deliv. 2021;18(8):1074-1093. https://doi.org/10.2174/1567201818666210303101250
- Cevc G, Blume G. New, highly efficient formulation of diclofenac for the topical, transdermal administration in ultradeformable drug carriers, Transfersomes. Biochim Biophys Acta Biomembr. 2001;1514(2):191-205. doi:10.1016/S0005-2736(01)00369-8.
CrossRef - Moghassemi S, Hadjizadeh A. Nano-niosomes as nanoscale drug delivery systems: an illustrated review. J Control Release. 2014;185:22-36. https://doi:10.1016/j.jconrel.2014.04.015.
CrossRef - Godwin DA, Kim NH, Felton LA. Development and in-vivo evaluation of transfersomal gel for transdermal delivery of curcumin to improve its anti-arthritic efficacy. Drug Dev Ind Pharm. 2022;48(2):51-60. doi:10.1080/03639045.2022.2042554.
CrossRef - Bangre, P. D., Karande, N. A., Rathi, L. G., Borkar, A. V., Singhavi, D. J., Ganjiwale, R. O., & Bendale, A. R. (2025). Bioactivity-Guided Isolation and Identification of Antimicrobial Compound from Roots of Ehretia laevis Roxb. Pharmaceutical Chemistry Journal, 1-10.
CrossRef - Gao G, Schilling AF, Yonezawa T, Wang J, Dai G, Cui X, et al. Bioactive nanoparticles stimulate bone tissue formation in bioprinted three-dimensional scaffold and human mesenchymal stem cells. Biotechnol J. 2014;9(10):1304-1311. https://doi.org/10.1002/biot.201400305
CrossRef - Miao S, Zhu W, Castro NJ, Nowicki M, Zhou X, Cui H, et al. 4D printing smart biomedical scaffolds with novel soybean oil epoxidized acrylate. Sci Rep. 2016;6:27226. https://doi.org/10.1038/srep27226
CrossRef - Nottingham J. A Practical Guide to Toxicology and Human Health Risk Assessment. Wiley; 2021.
- International Council for Harmonisation (ICH). Q8(R2) Pharmaceutical Development. ICH harmonised guideline; 2009. Accessed May 10, 2024. https://database.ich.org/sites/default/files/Q8_R2_Guideline.pdf
- European Medicines Agency. Guideline on Quality of Herbal Medicinal Products/Traditional Herbal Medicinal Products. EMA/HMPC/201116/2005. London: EMA; 2018. Accessed May 10, 2024. https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-quality-herbal-medicinal-products/traditional-herbal-medicinal-products-revision-1_en.pdf
- S. Food and Drug Administration. Botanical Drug Development Guidance for Industry. U.S. Department of Health and Human Services; 2016. Accessed May 10, 2024. https://www.fda.gov/media/93113/download
- Gage, G. P., Rudrapal, M., Jadhav, A. G., Borse, L. B., & Bendale, A. R. (2020). review on quality by design (QbD). Asian J Pharm Educ Res, 9(2), 37.
CrossRef - Donnelly L, Campton N, Chen J, McKeown A, Thompson G, Zhou H. Clinical development of topical drugs for musculoskeletal disorders: formulation, regulation, and considerations for patient-centric design. Pharm Res. 2021;38(3):399-415. https://doi.org/10.1007/s11095-021-03013-z
CrossRef - International Council for Harmonisation. E8(R1) General Considerations for Clinical Studies. ICH harmonised guideline; 2022. Accessed May 10, 2024. https://database.ich.org/sites/default/files/ICH_E8-R1_Step4_Guideline_2022_0224.pdf
- Lappin G, Stevens L. Biomedical accelerator mass spectrometry: recent applications in metabolism and pharmacokinetics. Expert Opin Drug Metab Toxicol. 2008;4(8):1021-1033. https://doi.org/10.1517/17425255.4.8.1021
CrossRef - Whelan DB, Bhandari M, Stephen D, Kreder H, McKee MD, Zdero R, et al. Development of the radiographic union score for tibial fractures (RUST). J Orthop Trauma. 2010;24(suppl 1):S56-S61. https://doi.org/10.1097/BOT.0b013e3181ca3f97
CrossRef - Rudrapal, Mithun, Ismail Celik, Johra Khan, Mohammad Azam Ansari, Mohammad N. Alomary, Fuad Abdullah Alatawi, Rohitash Yadav et al. “Identification of bioactive molecules from Triphala (Ayurvedic herbal formulation) as potential inhibitors of SARS-CoV-2 main protease (Mpro) through computational investigations.” Journal of King Saud University-Science 34, 3 (2022): 101826. https://doi.org/10.1016/j.jksus.2022.101826
CrossRef - Raut M, Dugaje T, Aher PR, Salave GR, Bendale A, Naphade V, Jadhav A. Natural products and traditional medicines: integration with modern therapeutics. Targeted Therapies and Drug Delivery Systems: A Multidisciplinary Perspective. 2025 Jul 10:36.
CrossRef - Narkhede Sachin B, Bendale AR, Jadhav AG, Patel K, Vidyasagar G. Isolation and evaluation of starch of Artocarpus heterophyllus as a tablet binder. Int J PharmTech Res. 2011;3:836-40. https://doi.org/10.70593/978-93-7185-674-4
CrossRef - Bhattacharyya T, Iorio R. Outcome assessment in orthopedics. J Am Acad Orthop Surg. 2017;25(1):1-3. https://doi.org/10.5435/JAAOS-D-16-00751
Accepted on: 06-01-2026
Second Review by: Dr. Masi Shirzad and Dr. Karpagam S
Final Approval by: Dr. Ali Elshafei









