Sebetralstat: First Oral Plasma Kallikrein Inhibitor for Acute Hereditary Angioedema
1Department of Pharmaceutical Chemistry, Bharati Vidyapeeth's College of Pharmacy, C.B.D. Belapur, Navi-Mumbai, Mumbai University, Maharashtra, India
2Department of Pharmaceutical Sciences, Pharmaceutical Research and Development Consultant, Mumbai, Maharashtra, India
Corresponding Author E-mail: vikrant.dandekar@bvcop.in
DOI : http://dx.doi.org/10.13005/bbra/3444
ABSTRACT:Hereditary angioedema (HAE) is a rare, potentially life-threatening disorder caused by excessive bradykinin generation. Current injectable on-demand therapies may delay timely treatment during attacks. This review summarizes the discovery, mechanism, and clinical development of sebetralstat (KVD900), the first oral plasma kallikrein inhibitor for acute HAE. Literature from 2010–2025 was evaluated across major databases and regulatory sources, with emphasis on phase 3 KONFIDENT trial outcomes. Sebetralstat consistently reduced time to symptom relief (median 1.6–1.8 h vs 6.7 h with placebo) and demonstrated favorable tolerability with predominantly mild, transient adverse events. FDA and MHRA approvals in 2025 position sebetralstat as a patient-centered, self-administered option offering rapid, effective treatment at attack onset. Remaining needs include pediatric evaluation, real-world cost-effectiveness, and assessment of its potential role beyond acute therapy.
KEYWORDS:Hereditary angioedema; Kallikrein inhibitor; On-demand therapy; Plasma kallikrein; Regulatory approval; Sebetralstat
Introduction
This review aligns with the journal’s focus on pharmacological innovation by critically evaluating the first oral plasma kallikrein inhibitor that transforms the treatment paradigm for hereditary angioedema. Beyond its mechanistic novelty, sebetralstat exemplifies how small-molecule design can substitute for injectable biologics, improving patient autonomy and adherence. The discussion further addresses regional and translational relevance, particularly for healthcare systems in low- and middle-income countries where self-administered oral therapies can substantially reduce dependence on infusion infrastructure, enhance treatment equity, and support cost-effective rare-disease management across Asia and similar resource-limited regions.
Hereditary angioedema (HAE) is a rare genetic disorder characterized by recurrent, non-pitting edema of the skin, gastrointestinal tract, and upper airways. Attacks are unpredictable and can become life-threatening when the airway is involved. Unlike histamine-mediated angioedema, HAE results from excessive bradykinin formation and therefore does not respond to corticosteroids, antihistamines, or epinephrine.1-8
Epidemiological studies suggest that HAE affects between 1 in 50,000 and 1 in 150,000 individuals worldwide, though underdiagnosis and misclassification likely contribute to variability in prevalence estimates. Most patients have mutations in the SERPING1 gene, which cause a deficiency or dysfunction of C1 esterase inhibitor (C1-INH). Other variants involving F12, PLG, ANGPT1, and KNG1 also contribute to HAE types with normal C1-INH levels. The disorder places a significant physical, emotional, and economic burden on patients due to the unpredictability and severity of attacks.9-12
Understanding the kallikrein–kinin cascade has led to targeted therapies that block bradykinin generation. Sebetralstat, the first oral plasma kallikrein inhibitor for acute HAE attacks, represents a meaningful shift away from injectable therapies. Its ease of self-administration may be especially valuable in settings with limited access to infusion facilities or trained medical staff.13-20
Pathophysiology of HAE
The pathophysiology of HAE is rooted in dysregulation of the kallikrein–kinin cascade. Under normal conditions, C1-INH plays a crucial role in controlling plasma kallikrein activity and regulating bradykinin formation. In patients with C1-INH deficiency or dysfunction, unchecked activation of plasma kallikrein results in excessive cleavage of high-molecular-weight kininogen into bradykinin. Bradykinin, acting via endothelial B2 receptors, increases vascular permeability and vasodilation, leading to tissue edema. This biochemical pathway explains why HAE is resistant to antihistamines and corticosteroids. The clinical phenotype corresponds directly to bradykinin-mediated vascular leakage: Cutaneous attacks: Swelling of extremities, face, or genitals, often impairing mobility or appearance. Abdominal attacks: Severe pain, vomiting, and diarrhea that mimic acute surgical conditions. Laryngeal attacks: Potentially fatal airway obstruction, historically accounting for significant mortality before effective therapies became available. Understanding this mechanistic cascade has been pivotal in identifying plasma kallikrein inhibitors such as sebetralstat, which interrupt bradykinin generation and provide a rational basis for targeted therapeutic intervention.21-27
Medicinal Chemistry and Mechanism of Action
Sebetralstat is an oral, selective, competitive, and reversible plasma kallikrein inhibitor. In Hereditary Angioedema (HAE), C1-INH deficiency or dysfunction leads to the uncontrolled activation of plasma kallikrein. Plasma kallikrein cleaves high-molecular-weight kininogen to generate excessive bradykinin, which causes the vascular leakage and edema characteristic of HAE attacks. Sebetralstat (KVD900) inhibits plasma kallikrein activity, thereby interrupting the generation of bradykinin and halting edema progression, leading to a reduction of angioedema attacks.28-31 The mechanism of Action of Sebetralstat in Hereditary Angioedema is illustrated in Fig. 1.
Key pharmacokinetic attributes include:
Rapid absorption – near-complete PKa inhibition within ~15 minutes post-administration.
Favorable oral bioavailability – facilitating immediate, self-administered dosing at symptom onset.32-35
This represents a major leap from parenteral therapies, aligning treatment with patient convenience.
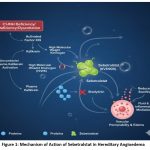 |
Figure 1: Mechanism of Action of Sebetralstat in Hereditary Angioedema |
Methodology
Search Strategy and Databases
A systematic literature search was conducted in accordance with the PRISMA 2020 guidelines to ensure comprehensive and reproducible identification of relevant studies. The following databases were searched:
PubMed/MEDLINE
Web of Science
Scopus
Google Scholar
The search strategy used combinations of keywords and Boolean operators: (“Sebetralstat” OR “KVD900” OR “EKTERLY”) AND (“Hereditary Angioedema” OR “HAE”) AND (“kallikrein inhibitor” OR “oral therapy” OR “on-demand treatment” OR “clinical trial” OR “FDA approval”). The reference lists of retrieved articles and review papers were also manually screened to identify any additional eligible sources.
Time Frame: The search covered literature published between January 2010 and October 2025, to include both preclinical development data and recent clinical/regulatory updates regarding sebetralstat and other plasma kallikrein inhibitors.
Inclusion Criteria
Studies were included if they met the following criteria:
Focused on hereditary angioedema and its on-demand or acute treatments.
Reported clinical, pharmacological, or mechanistic data on sebetralstat (KVD900) or related oral kallikrein inhibitors.
Published in peer-reviewed journals, clinical trial registries, or regulatory communications (FDA, MHRA).
Articles written in English and accessible in full text.
Exclusion Criteria
The following records were excluded:
Non-English language articles.
Conference abstracts without full data.
Animal studies lacking translational significance.
Editorials, commentaries, or press releases without verifiable primary data.
Data Extraction and Synthesis: Two independent reviewers screened titles and abstracts, retrieved full texts for eligible studies, and extracted data on trial design, population characteristics, intervention details, efficacy endpoints, and safety outcomes. Discrepancies were resolved by discussion and consensus. Quantitative synthesis was not attempted due to heterogeneity among studies; results were summarized narratively.
Bias Assessment
A formal risk-of-bias assessment was not performed in this review. Because this article is a narrative synthesis rather than a systematic review or meta-analysis, structured tools such as Cochrane RoB 2.0 or ROBINS-I were not applied. Instead, the review draws upon data from peer-reviewed clinical trials, regulatory assessments, and mechanistic studies, and the risk-of-bias evaluations provided in the original publications were considered qualitatively when interpreting the evidence.
PRISMA Flowchart: The Prism 2020 flowchart is depicted in Table 136-38
The flowchart summarizes the identification, screening, eligibility, and inclusion stages of the literature selection process used in this review.
Table 1: PRISMA 2020 flowchart
| Stage | Number of Records | Description |
| Records identified through database search | 246 | PubMed (102), Web of Science (54), Scopus (47), Google Scholar (43) |
| Additional records identified through a manual search | 18 | Cross-references and regulatory documents |
| Records after duplicates removed | 198 | —— |
| Records screened (title/abstract) | 198 | ——- |
| Full-text articles assessed for eligibility | 72 | ——– |
| Studies included in the qualitative synthesis | 35 | Final dataset for review |
Clinical Evidence
Konfident Trial (NCT05259917): Efficacy Summary
Design. Randomized, double-blind, three-way crossover trial in HAE-C1-INH (≥12 y); patients treated attacks with sebetralstat 300 mg, 600 mg, or placebo (≤2 doses/attack). Primary endpoint: time to beginning of symptom relief (PGI-C “a little better” at ≥2 consecutive time points within 12 h). Key secondary endpoints: time to reduction in severity (PGI-S) within 12 hrs. and time to complete resolution (PGI-S “none”) within 24 hrs.
Primary endpoint: time to the beginning of symptom relief
Median (IQR): 300 mg 1.61 h (0.78–7.04), 600 mg 1.79 h (1.02–3.79), placebo 6.72 h (1.34–>12). Adjusted p vs placebo: <0.0001 (300 mg) and 0.0013 (600 mg).
Median (95% CI) (EMA analyses): 300 mg 1.69 h (1.28–2.28) vs placebo 5.57 h (2.07–NE); 600 mg 2.01 h (1.38–2.46) vs placebo 7.32 h (…). (Both significant on Gehan/stratified testing; adjusted p<0.0001 and p=0.0013, respectively.)
Proportion reaching endpoint ≤12 h: 300 mg 75.9%, 600 mg 78.9%, placebo 48–51%
Key secondary endpoints:
Time to reduction in severity ≤12 h:
– Median (IQR): 300 mg 9.27 h (1.53–>12), 600 mg 7.75 h (2.19–>12), placebo >12 h
(6.23–>12).
Adjusted p vs placebo: 0.004 (300 mg), 0.003 (600 mg):
– EMA table shows Gehan LS mean differences vs placebo: 300 mg −51.22 (95% CI −83.13 to −19.31), 600 mg −50.65 (−81.82 to −19.48); both significant.
Time to complete resolution ≤24 h:
– Median (IQR): 300 mg >24 h (16.6–NE), 600 mg 24.0 h (10.6–NE), placebo >24 h (NE–NE). Adjusted p vs placebo: 0.002 (300 mg), <0.001 (600 mg).
– Complete resolution ≤24 h (proportion): 300 mg 42.5%, 600 mg 49.5%, placebo 27.4%
Subgroup analyses (primary endpoint; medians by subgroup):
Baseline severity (PGI-S)—None/Mild: 300 mg 1.70 h, 600 mg 1.82 h; Moderate: 1.56 h and 2.11 h; Severe/very severe: 1.41 h and 1.51 h.
Attack location (pooled)—Mucosal: 300 mg 1.29 h, 600 mg 1.32 h; Subcutaneous: 1.65 h and 2.12 h.
Treatment paradigm—On-demand only: 300 mg 1.35 h, 600 mg 1.77 h; On-demand + LTP: 1.85 h and 2.03 h.
Geography—Europe: 300 mg 1.78 h, 600 mg 1.77 h; US: 1.28 h and 1.77 h; Asia/Pacific: 1.70 h and 2.30 h.
Dose-to-dose (exploratory) comparisons
In congress supplement analyses (not powered for 600 mg vs 300 mg), hazard ratios (HR, 95% CI) for 600 mg vs 300 mg were:
Beginning of symptom relief ≤12 h HR 1.09 (0.77–1.53);
Reduction in severity ≤12 h HR 0.94 (0.62–1.43);
Complete resolution ≤24 h HR 0.80 (0.52–1.25).
Subgroup HRs by baseline severity and treatment paradigm were likewise centered near 1 with wide Cis.39-43
Long-Term Outcomes
Extension studies evaluating over 1,700 treated attacks confirmed the reproducibility of clinical benefit. Patients consistently experienced symptom relief within 1–1.5 hours, including those with abdominal and laryngeal attacks—sites of heightened morbidity and mortality. Importantly, real-world use revealed that patients often self-administered sebetralstat within minutes of symptom onset, supporting feasibility and practicality in routine care.44- 45
Safety Profile
Across all clinical trials—including the pivotal KONFIDENT Phase 3 and its long-term extension studies—sebetralstat demonstrated a consistent and favorable safety profile. No treatment-related serious adverse events (SAEs), thromboembolic events, or cardiovascular abnormalities were observed.
Adverse events (AEs) were predominantly mild to moderate, transient, and self-resolving. The overall AE incidence was comparable to placebo. No clinically relevant effects on laboratory parameters, ECG, or vital signs were detected. No QTc prolongation, hepatotoxicity, or hypersensitivity reactions were observed. Adverse events reported in Phase 1 to Phase 3 clinical trials were depicted in Table 2.46-47
Table 2: Summary of Adverse Events (All Phase 1–3 Studies)
| Category | Adverse Event | Sebetralstat (300–600 mg, pooled) |
Placebo | Severity / Notes |
| General | Headache | 3.1 % | 2.4 % | Mild; transient; no discontinuations |
| Fatigue | 2.3 % | 1.7 % | Mild; self-limited | |
| Gastrointestinal | Nausea | 2.1 % | 1.5 % | No vomiting or discontinuation reported |
| Dyspepsia | 1.8 % | 1.2% | Mild; resolved spontaneously | |
| Neurological | Dizziness | 1.6% | 1.4% | Mild; transient |
| Dermatologic / Vasomotor | Flushing | 1.3% | 1.0% | Transient erythema; non-serious |
| Musculoskeletal | Back pain / myalgia | 1.1% | 0.9% | No functional limitation |
| Serious Adverse Events (SAE) | None deemed treatment-related | —– | —— | No deaths or thrombotic events reported |
| Laboratory / ECG Findings | No clinically meaningful trends | ——- | —— | Stable hematology, hepatic, and renal profiles |
Sebetralstat has demonstrated a favorable safety profile. Headache was the most commonly reported adverse event (~3%). Other mild, transient effects included dyspepsia, nausea, fatigue, back pain, and flushing. Importantly, no significant cardiovascular or thrombotic complications were identified during trials.48-50
The safety data reinforce sebetralstat’s tolerability for on-demand use in HAE. The absence of severe or cumulative toxicity, along with oral dosing convenience, underscores its suitability for early self-administration at attack onset. These findings position sebetralstat as a patient-centric alternative to injectable kallikrein inhibitors and complement long-term prophylactic regimens.
Regulatory Milestones
United States (FDA).51-53
Product/Name: EKTERLY® (sebetralstat) tablets, 300 mg.
Application: NDA 219301 (new drug application).
Approval date: July 3, 2025
Indication: “Treatment of acute attacks of hereditary angioedema (HAE) in adult and pediatric patients aged ≥12 years.”
Recommended dose: 600 mg (two 300-mg tablets) at earliest recognition of an HAE attack; a second 600-mg dose ≥3 h later if response is inadequate/worsening; max 1,200 mg/24 h.
Additional confirmation: Listed on FDA’s Novel Drug Approvals 2025 page as “Ekterly (sebetralstat) — 07/03/2025 — To treat acute attacks of hereditary angioedema.”
United Kingdom (MHRA).54-55
Authority action: Marketing authorization granted by MHRA.
Approval date: July 15, 2025.
Indication & population: “Treatment of HAE attacks in adults and adolescents aged 12 years and older.”
Notability: First and only MHRA-approved oral, on-demand HAE treatment at time of decision.
Background (pre-licence): Earlier EAMS scientific opinion and Public Assessment Report (PAR) published March 24, 2025, supporting early access before full approval.
Table 3 depicts the regulatory status of sebetralstat in USA & UK.
Table 3: Regulatory status of sebetralstat in USA & UK
| Regulator |
Document (primary source) | Date |
| FDA (CDER) | NDA Approval Letter – NDA 219301 (EKTERLY, sebetralstat) | Jul 3, 2025 |
| FDA (label/USPI) | Full Prescribing Information (USPI) | Rev. 07/2025 |
| FDA (listing) | Novel Drug Approvals 2025 | Updated Oct 7, 2025 |
| MHRA | MHRA Press Release (“MHRA approves sebetralstat | Jul 15, 2025 |
| MHRA (pre-approval) |
EAMS Public Assessment Report (PAR) | Mar 24, 2025 |
Clinical Impact and Future Perspectives
Clinical Impact
The approval of sebetralstat represents a major advancement in the management of HAE:
Accessibility: Oral administration eliminates dependence on injections and infusion centers, broadening access.
Timeliness: Rapid onset of action enables treatment at the earliest signs of an attack, reducing morbidity.
Quality of life: Patients gain autonomy, reducing the anxiety associated with unpredictable attacks.
Healthcare burden: Decreased emergency visits and hospitalizations are anticipated to lower overall healthcare costs.
Future Perspectives
Several avenues warrant further exploration:
Pediatric Development – Trials are needed to evaluate dosing, safety, and efficacy in children under 12 years.
Prophylactic Potential – While sebetralstat is approved for acute use, investigations into its suitability for short-term prophylaxis (e.g., perioperative settings) may broaden indications.
Combination Approaches – Integration with long-acting biologics or gene therapies could offer comprehensive, individualized management strategies.
Global Access and Equity – Ensuring affordability and availability, particularly in low-resource settings, will be critical to maximizing public health benefits.
Precision Medicine – Genotypic and biomarker-driven stratification may optimize patient selection and outcomes.
Overall, sebetralstat exemplifies the successful translation of molecular pathophysiology into a transformative clinical innovation, setting the stage for a new era in HAE therapy. Sebetralstat represents not only a therapeutic innovation but also a paradigm shift in rare-disease pharmacotherapy. Its oral administration aligns modern treatment with patient needs for autonomy, speed, and convenience. As further data emerge, particularly in pediatrics and prophylaxis, sebetralstat is likely to reshape standard care algorithms for HAE.56-60
In line with the journal’s mission to advance innovative pharmacotherapy and regional applicability, this work underscores sebetralstat’s role as a first-in-class oral kallikrein inhibitor offering a tangible solution to accessibility challenges in rare-disease care. The drug’s molecular precision, safety, and ease of administration highlight how pharmacological innovation can bridge the gap between global discovery and regional clinical implementation, especially within emerging healthcare economies.
Cost-effectiveness and accessibility in low-resource settings
Sebetralstat may also offer advantages in terms of cost-effectiveness and accessibility, particularly in low-resource healthcare settings where infusion facilities, trained personnel, and injectable storage requirements may be limited. The oral formulation eliminates the need for specialized administration infrastructure and may reduce the frequency of emergency visits associated with delayed treatment of attacks. Although formal pharmacoeconomic analyses are still needed, the simplified delivery model has the potential to improve treatment equity, reduce indirect healthcare costs, and broaden access to timely on-demand therapy in underserved regions.
Table 4: Comparison of Key Therapies for Hereditary Angioedema (HAE)
| Drug (Type) | Route of Administration | Indication | Onset of Relief | Key Advantages | Limitations |
| Sebetralstat (Oral plasma kallikrein inhibitor) | Oral tablet | On-demand (acute attacks) | 1–2 hours | First oral on-demand option; rapid absorption; patient-friendly; no injection | Prophylaxis role not yet confirmed; limited pediatric data |
| Icatibant (B2 receptor antagonist) | Subcutaneous injection | On-demand (acute attacks) | ~2 hours | Effective; established use | Injection-site pain; training required; cost |
| Berotralstat (Oral plasma kallikrein inhibitor) | Oral capsule (daily) | Long-term prophylaxis | Preventive | Convenient prophylaxis; non-invasive | Not for on-demand use; breakthrough attacks possible |
| Lanadelumab (Monoclonal antibody) | Subcutaneous injection (2–4 weeks) | Long-term prophylaxis | Preventive | Potent; durable prophylaxis | Injection; high cost; breakthrough attacks may occur |
| C1-INH replacement plasma/recombinant | IV or SC injection | On-demand & prophylaxis | 30–90 minutes (IV) | Addresses underlying deficiency | IV/SC access required; cost; less convenient |
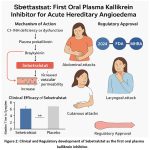 |
Figure 2: Clinical and Regulatory development of Sebetralstat as the first oral plasma kallikrein inhibitor. |
Comparison with Existing on-Demand Therapies
A comparison with established on-demand treatments highlights how sebetralstat fits within the current therapeutic landscape. Icatibant, a bradykinin B2 receptor antagonist administered subcutaneously, provides effective symptom relief but is frequently associated with injection-site pain and requires patient training for proper use. Ecallantide, a plasma kallikrein inhibitor, must be administered in a clinical setting due to the risk of hypersensitivity reactions, which can delay treatment during an acute attack. In contrast, sebetralstat offers a non-invasive oral option with a rapid onset of relief and a favorable safety profile, facilitating timely self-administration at symptom onset. However, direct head-to-head studies are lacking, and further comparative research is needed to establish its performance relative to injectable agents across diverse clinical scenarios.
Conclusion
Sebetralstat has demonstrated clinically meaningful and statistically significant improvements in time to symptom relief for acute HAE attacks. In the KONFIDENT Phase 3 trial, symptom relief occurred in approximately 1.6–1.8 hours with sebetralstat compared with 6.7 hours with placebo. Most adverse events were mild and similar in frequency to placebo, supporting its suitability for on-demand use. The oral formulation provides a convenient alternative to injectable on-demand therapies. However, data remain limited to patients aged 12 years and older, and long-term or prophylactic use has not yet been established. Additional studies are needed to compare sebetralstat with existing therapies, evaluate its use in younger children, and assess its effectiveness in real-world practice.
Regulatory approvals by the FDA and MHRA in 2025 confirm the robustness of current evidence. As further data emerge, sebetralstat may play an important role in expanding access to rapid, patient-controlled treatment for HAE attacks, particularly in resource-limited settings.
Overall, current evidence supports sebetralstat as an effective and generally well-tolerated oral option for managing acute HAE attacks. Its broader clinical and pharmacoeconomic impact should be defined through longitudinal and comparative studies aligned with real-world patient outcomes.
Acknowledgement
The authors would like to thank Bharati Vidyapeeth’s College of Pharmacy, Navi-Mumbai, for invaluable support and resources.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Vikrant Dandekar: Conceptualization, Methodology, Writing Original Draft.
Milind Sathe: Data Collection, Analysis, Writing Review & Editing.
References
- Longhurst H, Cicardi M. Hereditary angio-oedema. Lancet 2012;379(9814):474–81. doi:10.1016/S0140-6736(11)60935-5.
CrossRef - Bork K, Hardt J, Schicketanz KH, Ressel N. Clinical studies of sudden upper airway obstruction in patients with hereditary angioedema due to C1 esterase inhibitor deficiency. Arch Intern Med. 2003;163(10):1229–35. doi:10.1001/archinte.163.10.1229.
CrossRef - de Maat S, Hofman ZLM, Maas C. Hereditary angioedema: the plasma contact system out of control. J Thromb Haemost 2018;16(9):1674–85. doi: 10.1111/jth.14209.
CrossRef - Bork K, Meng G, Staubach P, Hardt J. Hereditary angioedema: new findings concerning symptoms, affected organs, and course. Am J Med. 2006;119(3):267–74. doi:10.1016/j.amjmed.2005.09.064
CrossRef - Busse PJ, Christiansen SC. Hereditary angioedema. N Engl J Med 2020;382(12):1136–48. doi: 10.1056/NEJMra1808012
CrossRef - Zuraw BL. Clinical Practice: Hereditary angioedema. N Engl J Med. 2008; 359(10):1027–36. doi: 10.1056/NEJMcp0803977
CrossRef - Hofman ZLM, de Maat S, Suffritti C, et al. Cleaved kininogen as a biomarker for bradykinin release in hereditary angioedema. J Allergy Clin Immunol. 2017;140(6):1700–3. doi: 10.1016/j.jaci.2017.07.012.
CrossRef - Kaplan AP. Enzymatic pathways in the pathogenesis of hereditary angioedema: the role of C1 inhibitor therapy. J Allergy Clin Immunol 2010;126(5):918–25. doi: 10.1016/j.jaci.2010.08.012.
CrossRef - Zuraw BL, Christiansen SC. HAE pathophysiology and underlying mechanisms. Clin Rev Allergy Immunol 2016; 51(2):216–29. doi:10.1007/s12016-016-8561-8.
CrossRef - Longhurst H, et al. Prevention of hereditary angioedema attacks with a subcutaneous C1 inhibitor. N Engl J Med. 2017; 376(12):1131–40. doi: 10.1056/NEJMoa1613627.
CrossRef - Aberer W, Maurer M, Bouillet L, et al. Breakthrough attacks in patients with hereditary angioedema receiving long-term prophylaxis are responsive to icatibant: findings from the Icatibant Outcome Survey. Allergy Asthma Clin Immunol 2017; 13(1):31. doi:10.1186/s13223-017-0203-z.
CrossRef - Maetzel A, Smith MD, Duckworth EJ, Hampton SL, Donatis GMD, Murugesan N, et al. KVD900, an oral on-demand treatment for hereditary angioedema: phase 1 study results. Allergy Clin Immunol 2022; 149(6):2034–42. doi: 10.1016/j.jaci.2021.10.038.
CrossRef - Duckworth EJ, Murugesan N, Li L, et al. Pharmacological suppression of the kallikrein-kinin system with KVD900: an orally available plasma kallikrein inhibitor for the on-demand treatment of hereditary angioedema. Clin Exp Allergy 2022;52(9):1059–69. doi: 10.1111/cea.14122.
CrossRef - Craig TJ, Busse PJ, Gower RG, et al. Long-term outcomes in hereditary angioedema: disease control, attack frequency, and patient-reported quality of life. Ann Allergy Asthma Immunol. 2020;125(3):304–311. doi: 10.1016/j.anai.2020.05.028.
CrossRef - Caballero T, Farkas H, Bouillet L, et al. Classification, pathophysiology, and diagnosis of angioedema: consensus report from the HAE International Working Group. Allergy. 2019;74(10):1969–1986. doi:10.1111/all.13810.
CrossRef - Lumry WR, Johnston DT, Banerji A, et al. Healthcare utilization and disease burden in hereditary angioedema: insights from a global patient registry. Allergy Asthma Proc. 2023;44(2):150–160. doi:10.2500/aap.2023.44.220090
CrossRef - Veronez CL, Moreno AS, Constantino-Silva RN, et al. Genetic and clinical profile of Brazilian patients with hereditary angioedema due to C1 inhibitor deficiency. Clin Immunol. 2018; 197:110–117. doi: 10.1016/j.clim.2018.09.006
CrossRef - Maurer M, Magerl M, Ansotegui I, Aygören-Pürsün E, Betschel S, Bork K, et al. The international WAO/EAACI guideline for the management of hereditary angioedema – 2021 revision and update. Allergy. 2022;77(7):1961–1990. doi:10.1111/all.15140
CrossRef - Bork K, Aygören-Pürsün E, Bas M, et al. Hereditary angioedema: new diagnostic and therapeutic developments. Allergy. 2021;76(12):3634–3649. doi:10.1111/all.14968.
CrossRef - Banerji A, Riedl MA, Bernstein JA. Advances in understanding and managing hereditary angioedema: 2023 update. J. Allergy Clin Immunol Pract. 2023;11(4):1235–1249. doi: 10.1016/j.jaip.2022.12.031.
CrossRef - Riedl MA, Farkas H, Aygören-Pürsün E, et al. Oral sebetralstat for on-demand treatment of hereditary angioedema attacks. N Engl J Med. 2024;391(1):32–42. doi:10.1056/NEJMoa2314192.
CrossRef - Cohn DM, Aygören-Pürsün E, Bernstein JA, Farkas H, Lumry WR, Maurer M, et al. Evaluation of patient-reported outcome measures for on-demand treatment of hereditary angioedema attacks and design of KONFIDENT, a phase 3 trial of sebetralstat. Clin Transl Allergy 2023; 13(1): e12288. doi:10.1002/clt2.12288.
CrossRef - Kaplan AP, Joseph K. The bradykinin-forming cascade in hereditary angioedema: a current understanding of pathophysiologic mechanisms. Allergy Asthma Proc. 2023;44(1):5–13. doi:10.2500/aap.2023.44.220054
- Longhurst HJ, Craig TJ. Plasma kallikrein–kinin system and the bradykinin B2 receptor: central mediators of angioedema pathogenesis. J Allergy Clin Immunol. 2022;150(5):1176–1187. doi: 10.1016/j.jaci.2022.07.006
CrossRef
- de Maat S, Maas C. Factor XII–driven contact activation: a link between inflammation and hereditary angioedema. Front Immunol. 2021;12: 731846. doi:10.3389/fimmu.2021.731846.
CrossRef - Suffritti C, Zanichelli A, Maggioni L, Bonanni E, Cugno M. Complement and contact system interplay in hereditary angioedema: new targets for treatment. Front Immunol. 2020; 11:545448. doi:10.3389/fimmu.2020.545448
- Farkas H, Veszeli N, Kajdácsi E, et al. Novel insights into the regulation of bradykinin generation: endothelial activation and genetic modifiers in hereditary angioedema. Int Immunopharmacol. 2019; 78:106080. doi: 10.1016/j.intimp.2019.106080
CrossRef - Davie RL, Edwards HJ, Evans DM, Hodgson ST, Stocks MJ, Smith AJ, et al. Sebetralstat (KVD900): a potent and selective small molecule plasma kallikrein inhibitor featuring a novel P1 group as a potential oral on-demand treatment for hereditary angioedema. J Med Chem. 2022; 65(19):13629–45. doi: 10.1021/acs.jmedchem.2c00921.
CrossRef - Levy RJ, Lumry WR, McNeil DL, Li HH, Campion M, Horn PT, et al. EDEMA4: a phase 3, double-blind study of subcutaneous ecallantide treatment for acute attacks of hereditary angioedema. Ann Allergy Asthma Immunol 2010; 104(6):523–9. doi: 10.1016/j.anai.2010.04.012.
CrossRef - Murugesan N, Donatis GMD, Lee DK, et al. Discovery of orally bioavailable kallikrein inhibitors: optimization of amidine surrogates for improved permeability and metabolic stability. Bioorg Med Chem Lett. 2021; 50:128336. doi: 10.1016/j.bmcl.2021.128336
CrossRef - Patel S, Murugesan N, Smith AJ, et al. Molecular modeling and kinetic analysis of sebetralstat binding to plasma kallikrein: insights into reversible, competitive inhibition and selectivity against related serine proteases. Eur J Med Chem. 2024; 258:115708. doi: 10.1016/j.ejmech.2024.115708.
- Zuraw B, Lumry WR, Johnston DT, et al. Oral once-daily berotralstat for the prevention of hereditary angioedema attacks: a randomized, double-blind, placebo-controlled phase 3 trial. J Allergy Clin Immunol 2021; 148(1):164–72. doi: 10.1016/j.jaci.2020.10.015.
CrossRef - Aygören-Pürsün E, Zanichelli A, Cohn D, et al. Investigational oral plasma kallikrein inhibitor sebetralstat for on-demand treatment of hereditary angioedema. Lancet 2023; 401(10375):458–66. doi:10.1016/S0140-6736(22)02406-0.
CrossRef - Staller K, Lembo A, Banerji A, Bernstein JA, Shah ED, Riedl MA. Consider hereditary angioedema in the differential diagnosis for unexplained recurring abdominal pain. J Clin Gastroenterol 2022; 56(8):740–7. doi: 10.1097/MCG.0000000000001744.
CrossRef - Donatis GMD, Rushbrooke LJ, Lee DK, Duckworth EJ, Stocks MJ. Preclinical ADME and plasma protein binding characterization of KVD900: implications for oral bioavailability and kallikrein inhibition kinetics. Xenobiotica 2021;51(11):1250–1261. doi:10.1080/00498254.2021.1918412
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi:10.1136/bmj.n71.
CrossRef - Page MJ, Moher D, Bossuyt PM, et al. PRISMA 2020 explanation and elaboration: updated guidance and examples for reporting systematic reviews. BMJ. 2021;372:n160. doi:10.1136/bmj.n160
CrossRef - Haddaway NR, Page MJ, Pritchard CC, McGuinness LA. PRISMA2020: an R package and Shiny app for producing PRISMA 2020-compliant flow diagrams. Syst Rev. 2022;11(1):4. doi:10.1186/s13643-021-01826-4.
- Rubinstein E, Stolz LE, Sheffer AL, Stevens C, Bousvaros A. Abdominal attacks and treatment in hereditary angioedema with C1-inhibitor deficiency. BMC Gastroenterol 2014; 14(1):71. doi: 10.1186/1471-230X-14-71.
CrossRef - KalVista Pharmaceuticals. KalVista Pharmaceuticals announces FDA approval of EKTERLY® (sebetralstat), first and only oral on-demand treatment for hereditary angioedema. Press Release. July 7, 2025. Accessed July 7, 2025. Available from: https://ir.kalvista.com/news-releases/news-release-details/kalvista-pharmaceuticals-announces-fda-approval-ekterlyr.
- Girolami JP, Bouby N, Richer-Giudicelli C, Alhenc-Gelas F. Kinins and kinin receptors in cardiovascular and renal diseases. Pharmaceuticals 2021; 14(3):240. doi: 10.3390/ph14030240.
CrossRef - Ponczek MB, Shamanaev A, LaPlace A, et al. The evolution of factor XI and the kallikrein-kinin system. Blood Adv 2020; 4(24):6135–47. doi: 10.1182/bloodadvances.2020002456.
CrossRef - Petersen RS, Bordone L, Riedl MA, Tachdjian R, Craig TJ, Lumry WR, et al. A phase 2 open-label extension study of prekallikrein inhibition with donidalorsen for hereditary angioedema. Allergy 2023; 79(3):724–34. doi: 10.1111/all.15948.
CrossRef - KalVista Pharmaceuticals. Press Release: Phase 3 KONFIDENT Trial Meets All Endpoints for Sebetralstat as First Oral On-Demand Therapy for Hereditary Angioedema. 13 Feb 2024. Accessed 18 Feb 2024. Available from:https://ir.kalvista.com/news-releases/news-release-details/kalvista-pharmaceuticals-reports-phase-3-konfident-trial-meets
- U.S. Food and Drug Administration (FDA). Clinical Review and Summary Basis of Approval – NDA 219301 (EKTERLY®, sebetralstat). Silver Spring (MD): FDA Center for Drug Evaluation and Research; 2025. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/nda/ 2025/219301Orig 1s000ChemR.pdf.
- KalVista Pharmaceuticals. KalVista Pharmaceuticals reports phase 3 KONFIDENT trial meets all endpoints for sebetralstat as first oral on-demand therapy for hereditary angioedema. Press Release. February 13, 2024. Accessed February 18, 2024. Available from: https://ir.kalvista.com/news-releases/news-release-details/kalvista-pharmaceuticals-reports
- Morabito C, Cohen T, Gunsior M, et al. Support for STAR-0215 administered every three- or six-months for hereditary angioedema: phase 1a results. Ann Allergy Asthma Immunol 2023; 131(5 Suppl): S32–3. doi: 10.1016/j.anai.2023.08.107.
CrossRef - Ionis Pharmaceuticals. Ionis announces positive topline results from phase 3 OASIS-HAE study of investigational donidalorsen in patients with hereditary angioedema. Press Release. 2024. Accessed February 18, 2024. Available from: https://ir.ionispharma.com/news-releases/news-release-details/ionis-announces-positive-topline-results-phase-3-oasis-hae-study.
- Papanikolaou E, Bosio A. The promise and the hope of gene therapy. Front Genome Ed 2021; 3:618346. doi:10.3389/fgeed.2021.618346.
CrossRef - Anzalone AV, Randolph PB, Davis JR, et al. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 2019; 576:149–57. doi:10.1038/s41586-019-1711-4.
CrossRef - U.S. Food and Drug Administration (FDA). Approval Letter – NDA 219301: EKTERLY® (sebetralstat) tablets, 300 mg. Center for Drug Evaluation and Research; July 3, 2025. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2025/219301Orig1s000ltr
- U.S. Food and Drug Administration (FDA). Clinical Pharmacology and Biopharmaceutics Review – NDA 219301 (EKTERLY® [sebetralstat]). July 2025. Available from: https://www.accessdata.fda.gov/ drugsatfda_docs/nda/2025/219301Orig1s000ClinPharmR
- KalVista Pharmaceuticals. Press Release: FDA Approves EKTERLY® (sebetralstat), the First and Only Oral On-Demand Therapy for Hereditary Angioedema (HAE). July 7, 2025. Accessed July 2025. Available from: https://ir.kalvista.com/news-releases/news-release-details/kalvista-pharmaceuticals-announces-fda-approval-ekterlyr.
- Medicines and Healthcare products Regulatory Agency (MHRA). Public Assessment Report: EKTERLY® (sebetralstat) – UK Marketing Authorization. London: MHRA; July 15, 2025. Available from: https://www.gov.uk/mhra-par-ekterly.
- Medicines and Healthcare products Regulatory Agency (MHRA). EAMS Scientific Opinion – Sebetralstat (EKTERLY®) for the Treatment of Acute Attacks of Hereditary Angioedema. March 24, 2025. Available from: https://www.gov.uk/government/publications/sebetralstat
- Baker AH, Herzog RW. Did dendritic cell activation, induced by adenovirus-antibody complexes, play a role in the death of Jesse Gelsinger? Mol Ther 2020; 28:704–6. doi: 10.1016/j.ymthe.2020.02.010.
CrossRef - Gower RG, Wilber M. Considerations for transition from subcutaneous to oral prophylaxis in the treatment of hereditary angioedema. Allergy Asthma Clin Immunol 2021; 17:100.doi:10.1186/s13223-021-00603-9.
CrossRef - Beyaz S, Demir S, Oztop N, Colakoglu B, Buyukozturk S, Gelincik A. How satisfactory is on-demand icatibant from the patients’ perspective in real life? Allergy Asthma Proc 2022; 43:148–54. doi:10.2500/aap.2022.43.210104.
CrossRef - Federici C, Perego F, Borsoi L, Crosta V, Zanichelli A, Gidaro A, et al. Costs and effects of on-demand treatment of hereditary angioedema in Italy: a prospective cohort study of 167 patients. BMJ Open 2018; 8:e022291. doi:10.1136/bmjopen-2018-022291.
CrossRef - Betschel SD, Caballero T, Jones DH, Longhurst HJ, Manning M, van Kooten S, et al. The complexities of decision-making associated with on-demand treatment of hereditary angioedema (HAE) attacks. Allergy Asthma Clin Immunol 2024; 20:43. doi:10.1186/s13223-024-00903-w.
CrossRef
Accepted on: 01-12-2025
Second Review by: Dr. Sarraa Dhiaa Kasim
Final Approval by: Dr. Eugene A. Silow









