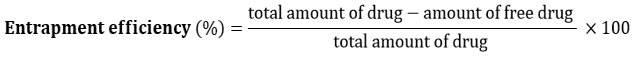Phytosomes, A versatile Phyto-Phospholipid Nanocarriers: A Review
1MET’s Institute Of Pharmacy, Bhujbal Knowledge City, Nashik, India.
2Department of Pharmaceutical Chemistry, MET’s Institute of Pharmacy, Bhujbal Knowledge City, Nashik, India.
Corresponding Author E-mail:Patilsonalisp21@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3445
ABSTRACT:Phytosomes are lipid based vesicular nanocarries that encapsulate phytoconstituents within phospholipid layers, designed to overcome the limitations of herbal extracts such as inadequate lipid solubility, restricted permeability, low bioavailability, and chemical instability, while improving the absorption, stability, and therapeutic efficacy of phytoconstituents or herbal extracts in pharmaceutical, nutraceutical, and cosmetic formulations. The review aims to provide an overview of phytosomes, including their complexation mechanism structural features, formulation methodologies, and characterization techniques for evaluation of this phyto-phospholipid complex. The review briefly summarizes phytosomes technology, including structural, molecular interaction between active agent and lipids, formulation techniques, storage conditions, stability considerations, and therapeutic applications, commercially available phytosome-based formulations, Challenges faced by technology emphasizing the role and importance of phytosomes as a promising platform for turning herbal active agents into effective and standardized dosage forms.
KEYWORDS:Absorption; Bioavailability; Encapsulation; Nanocarriers; Phytosomes; Phospholipid
Introduction
Medicinal plants have been serving as an integral part of the traditional medication system in different cultures as a source of therapeutic agents for treatment, mitigation of numerous diseases, disorders, or dermatological conditions.1 Herbal extracts offers a reservoir of bioactive phytoconstituents such as minerals, vitamins, flavonoids, terpenoids, tannins, glycosides, alkaloids, phenols, polyphenols, essential oils, etc. offering a diverse pharmacological effects, including antioxidant, anti-inflammatory, and anticancer properties. Most of the phytoconstituents are usually well-tolerated with potentially lesser side effects than synthetic compounds.2-4 However, the clinical utility of these compounds often hampered due to higher molecular weight and complex chemical structures giving rise to physicochemical barriers such as low lipid solubility, poor permeability across biological barriers, rapid metabolism, and restricted absorption, resulting in constrained systemic bioavailability upon oral and topical application.5-6
To combat these drawbacks associated, several nanotechnology based drug delivery system has been explored till date.4-5 One such technology named, phytosomes alternatively referred as herbosomes or phyto-phospholipid complex. Phytosomes are self- assembled phyto-phospholipid complex, recently emerging as a promising lipid-based vesicular drug delivery method to eliminate the shortcomings associated with conventional drug delivery approaches used for herbal actives agents. This food-grade biocompatible nanovesicles was pioneered and patented by Indena S.P.A, Italian Pharmaceutical and neutraceutical organisation to improve the solubility, permeability and bioavailability and pharmacokinetic characteristics of poorly absorbed herbal phytoconstituents. 5-7
Structural component and molecular arrangement of phytosomes vesicles
Phytosomes developed by complexation of phospholipids and phytoconstituents in suitable solvent system, typically in stoichiometric ratio of 1:1 to 1:2. Upon hydration, these complexes self-assemble into nano-vesicular or micellar structures size ranging from 50 to 300 nm.(8-9). The prerequisites for the development of phytosome molecular assemblies are phytoconstituents preferably hydrophilic agent (containing hydrogen-bonding groups –OH, –COOH, –NH₂, etc.),6-9 a phospholipid molecule (phosphatidylcholine, phosphatidylethanolamine, phosphatidylserine, etc), and suitable solvent system. 10-12 Sometimes cholesterol is incorporated additionally as a supportive film forming agent to improve membrane rigidity and to stabilize the phytosomal system.13 Additionally, buffered system such as saline phosphate buffer (pH 6.5-7.4), ethanol tris buffer (pH 6.5), distilled water, etc. used as a hydrating medium to form stable colloidal dispersions of phytosomes.15
The fundamental unit of phytosomes vesicles is phospholipid molecule, predominantly Phosphatidylcholine (PC). PC being amphiphilic in nature consists of hydrophilic polar head (phosphate moiety) and hydrophobic fatty acid non-polar tails (choline).10, 13-15 The polar head of PC binds non-covalently with water-soluble phytoconstituents via hydrogen bonding. While the lipophilic tails, forms protective envelope around the phytoconstituents, resulting in amphiphilic molecule (phytosomes vesicle) which can drastically enhances both, lipid solubility and membrane permeability of phytoconstituents as compared to its free form. This also helps to prevent degradation of phytoconstituents by the gastric environment. 9-10 The schematic representation phytosomes vesicle encapsulating phytoconstituents within phospholipid, demonstrated in figure 1.
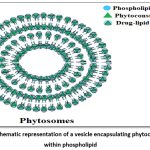 |
Figure 1: Schematic representation of a vesicle encapsulating phytoconstituents within phospholipid |
Methods for development of phytosomes vesicles
Phytosomes formulated by promoting a controlled molecular interaction between active phytoconstituents and PC under specific solvent system and processing conditions. Several methods used for development of phytosomes, including solvent evaporation, solvent injection, co-solvent lyophilisation, and thin-film hydration, anti-solvent precipitation, salting out techniques etc. The selection of appropriate method is generally based on the nature of phytoconstituent, desired vesicle size, polydispersity, stability, scalability requirements, dosage form and route of administration. A few of them as discussed below.15-17
Solvent Evaporation Method
Among all methods, this is most prevalent methods to develop lipid-based nano-carriers. The active ingredient and phospholipid solubilized into a suitable organic solvent and refluxed for 2–3 hours. Further rotary evaporator used to evaporate the solvent under pressure and film subsequently hydrated with phosphate buffer saline solution (pH 6.5 to 7.4) at temperature of 40 to 60°C. This method offers an excellent complexation efficiency.5-6, 19
Thin-Film Hydration Method
It is one of the most consistent and reliable methods. Here, PC and herbal concentrate blended in the preferred solvent system (based on the nature of active constituent and PC). This suspension subjected to continuous rotation in a rotary evaporator for a couple of hours. Subsequently, the solvent is evaporated under pressure, generating a delicate film of phytoconstituent-phospholipid complex inside the edges of a round-bottom flask. The film is further hydrated with a buffered system to produce multi-lamellar vesicles, whose sizes are subsequently reduced by sonication or membrane extrusion techniques,16-18
Solvent Injection Technique
Excellent method for complexation of heat-labile active constituents. The phyto-phospholipid complex is dispersed in alcohols such as ethanol or diethyl ether and is immediately introduced into an agitated aqueous phase (pH 6.5-7.0) by means of injection. This leads to the diffusion of solvent into the phyto-phospholipid complex, resulting in spontaneous phytosomes vesicle formation. While the residual solvent is extracted via vacuum evaporation. This strategy generates comparatively small unilamellar vesicles (80-150 nm) with minimal polydispersity, which is beneficial for thermolabile chemicals.19-20
Co-solvent Lyophilisation Method
The most efficient approach to retaining stability of phytosome vesicles over an extended period. A water-miscible co-solvent system such as ethanol-water or t-butanol-water, used to generate the phyto-phospholipid complex, which is subsequently mixed with 5-10% cryoprotectant agents like mannitol or trehalose and frozen at -80°C and lyophilize the resulting mixture to generate a free-flowing powder required to reconstitute before use. Due to micronized powder form, the phytosomes developed by this method exhibits exceptional stability (>24 months) along with significantly high drug loading capacity.19-21
Anti-solvent precipitation method
This is a fast and sustainable method for manufacturing phytosomes, especially for heat-sensitive phytoconstituents. In this manner, the phytoconstituent and phospholipid dispersed in the smallest possible amount of water-miscible organic solvent (ethanol or acetone). The resultant transparent mixture injected gradually onto a surplus volume of an anti-solvent solution (distilled water or a pH 6.5-7.4 phosphate buffer saline solution) in moderate stirring at 500-1000 rpm. A rapid fall in solvent strength produces immediate super saturation, resulting in the precipitation of nano-scale phytosomal particles via molecular complexation. The precipitate is gathered via centrifugation, rinsed to remove residual solvent, and dehydrated by means of air-drying or lyophilization to produce a fine powder that can quickly disperse into stable vesicles when hydrated.19, 21
Among all methods, thin film hydration and solvent evaporation are frequently utilised at laboratory as well as industrial level due to simplicity, high yield, and proven scalability in commercial phytosome products.19, 21 General method for preparation of phytosomes demonstrated in figure number 2.
 |
Figure 2: General method for preparation of phytosomes |
Characterization of Phytosomes
The uniformity and stability profile of phytosomes influenced by formulation and processing variables, including but not limited to: the type of solvent, stoichiometric ratio between active agent and PC, processing method, temperature, drying conditions, rotations per minute (rpm), etc. All mentioned parameters could significantly affect the stability of final product under storage conditions. Thus, these parameters can be studied, optimized and validated through suitable analytical methods to confirm molecular complexation, morphology, and performance metrics of the vesicles.23-24
Morphological analysis
Morphological examination of phytosomes vesicles employs electron microscopy techniques, such as digital microscopy, transmission electron microscopy (TEM), and scanning electron microscopy (SEM), to visualize phytosomes vesicle size, shape, and surface topology. In SEM, rays of three different electrons are projected: primary backscattered electrons, secondary electrons, and Auger electrons, along with X-rays, which strike directly onto the sample. Secondary electrons of SEM disclose the surface topography, backscattered electrons provide atomic number, and X-rays provide the elemental composition, while confirming the spherical or vesicular structures with sizes of 100-300 nm. On the other hand, TEM uses transmitted electrons to analyze the sample and provides high-resolution surface topography, comprehensive interior structure, and crystallography, which confirms lipid bilayer integrity and entrapment with active agents.25-26
Particle Size, Polydispersity Index (PDI) and Zeta Potential
The particle size and zeta potential of phytosomes can be analysed by dynamic light scattering technique using a method via photon correlation spectroscopy (PCS), which measures the hydrodynamic diameter in nanometres. The stability of phytosomes increases with the increase in electrostatic repulsion between the particles. Physical stability of the dispersion is predicted by a zeta potential, evaluated by a light scattering method, which helps to quantify surface charges. A value greater than 20 mV ensures electrostatic repulsion and high colloidal stability.22, 24-25
Entrapment Efficiency (%EE)
The amount of drug entrapped into phyto-phospholipid complex is evaluated by ultra-centrifugation technique. Phyto-phospholipid complex (or phytosomes) – 10 mg of drug are dispersed into a phosphate-buffered solution and subjected to centrifugation for a specific time at the desired speed (generally, the highest speed for the minimum time). Following separation of the supernatant layer via filtration, it is analysed by either HPLC or UV-Vis spectroscopy. Calculate drug entrapment by following formula:23-24
Thermal Analysis and Transition Temperature
thermal analysis techniques like differential scanning calorimetry (DSC) helps to determination of transition temperature of the phyto-phospholipid complex utilised to analyse the phase transition and thermal stability of phytosomes complex by changes appearance of new peaks and vanishing of old peaks, modifications to peak shape, peak temperature, and melting point, peak onset of action, relative peak area, enthalpy, or deletion of endothermic peaks usually associated with melting point of active agents and transition temperature of PC (30–50°C), indicates fluidity of vesicular membrane.22, 27-28
Crystallinity and Polymorphism
The X-ray diffraction (XRD) technique is employed to analyse the crystalline nature and polymorphic behaviour of phytosomes. A sharp crystalline peak of free phytoconstituents (2θ, at 10–30°), generally disappears or reduces due to phytosomes complexation, confirming molecular dispersion of active constituent within the phospholipid matrix.23-24, 28
Solubility and partition coefficient (log P) value
Phyto-phospholipid complexes are developed to alter the solubility of phytoconstituents. The solubility behaviour of the phyto-phospholipid complex typically determined by shake-flask or n-octanol/water systems.27-28
Infrared Spectroscopy (IR)/Fourier transform infrared spectroscopy (FTIR)
IR spectroscopy can help to analyse structural and chemical stability of phytosome complex, phospholipids, and plant drugs. The complex formation of phytosomes can be verified by comparing the IR spectrum of phytosome complex with the spectrum of phospholipid, phytoconstituents/drug, and its physical mixture separately. Complex formation indicated by the shifting or broadening of original characteristic peaks, which indicates typical interactions between drug and phospholipid.29
In-Vitro and In-Vivo Evaluations
The in-vivo and in-vitro models for phytosome evaluation can be selected based on the expected therapeutic action of the active phyto-constituents or medication included in phytosome formulation.
Storage conditions and Stability profile of phytosomes
Storage conditions
Phytosomes, being delicate structural aggregates, need to be stored carefully to prevent peroxidation of lipids, hydrolysis, and vesicle aggregation. Thus, to maintain stability and avoid deterioration, dried phytosomes should be stored at temperatures ranging from -20 to 30°C. While phytosomes in solution form should be stored at 4 ± 2°C in amber, airtight containers with silica gel desiccant (<60% relative humidity) protected from light. Light can accelerate the breakdown of phytosomes.24-25
Stability studies for phytosomes
Based on study parameters, the nature of phytoconstituent, phospholipid, and solvent used, a stability studies protocol should be developed based on ICH-Q2 (R1) guidelines, which generally includes long-term testing at 25 ± 2 °C/60 ± 5% relative humidity (RH) and accelerated testing at 40 ± 2 °C/75 ± 5% RH for 6–12 months. Stability studies help to investigate the phytochemical changes of phytosomes throughout storage and circulation. Over several months, stability could be determined by measuring mean vesicle size, zeta potential, size distribution, and trap efficiency.23-25
Merits of phytosomes technology
Enhanced solubility and absorption of phytoconstituents
Phytosomes vesicles improves the solubility and bioavailability phyto-active agents by drug-lipid complexation, tends to increase the aqueous solubility of phytoconstituents.31-32
Maryana et al. (2016)31 developed Silymarin-PC phytosomal complex with stoichiometric ratios of 1:5, 1:10, and 1:15, by adopting a thin-layer hydration technique, resulting in amphiphilic molecular complexes that enhance the restricted aqueous solubility of free silymarin.
Synergistic effect
A predominantly used phospholipid for phytosomes complexation is PC, an essential part of the cell membrane, PC nourishes skin. Thus when used in skin care preparation provides a synergistic effect (34-35). PC also has a potential hepatoprotective activity. When complexed with hepatoprotective agents such as silymarin, it shows a synergistic.33, 35-36
Chi et al. (2020)36 developed, Nanosuspensions of silybin–phospholipid phytosome (SPC-NP) and demonstrated that, SPCs-NP exhibits synergistic hepatoprotective efficacy beyond bioavailability enhancement of silybin, when compared to free silybin. The study showed marked increase in Cmax and AUC providing a stronger hepatoprotective activity in CCl₄-induced liver injury models (greater reduction in ALT/AST, MDA, and histological damage) resulting in superior overall liver protection that exceeds the sum of individual components.
The Silymarin phytosomes developed by Maryana et. al. (2016) 31, aiming to enhance the bioavailability of silymarin also demonstrated the synergistic role of phospholipid.
Topical/dermal application
Phytosomes complex tends to improve the lipophilicity highly water soluble active agents by developing stable phyto-phospholipid complexes. This leads significant enhancement in penetration ability of such agents via lipid rich skin barrier, indicating that the technique is appropriate for topical and transdermal medication administration, making it a viable approach for cosmetic preparations.34, 37-38
High drug entrapment efficiency
The rate of drug-lipid complexation in the phyto-phospholipid complex is quite remarkable as compared to liposomes.5, 38
Reduced dose and side effects
As targeted drug delivery can be achieved in phyto-phospholipid complexes, the dose required and dosing frequency can be reduced.40-41
Maiti et al. (2007)9 conducted a study on curcumin phytosomes and revealed that, in comparison to free curcumin, the curcumin-phospholipid complex was beneficial in producing therapeutic outcomes with lesser dosage and fewer adverse effects.
Enhanced stability
Encapsulation of phytosonstituents into phospholipid molecule developed a protective layer/envelop around active constituents, protects from gastric environment, environmental factors etc.
Effionora and Nadira (2018)42 formulated phytosomes loaded with maltodextrin gun Arabic microsphere for enhancing absorption and stability of Camellia sinensis extract (green tea extract).
Targeted drug delivery
Phytoconstituents complexed in phospholipids can help in achieving targeted drug delivery.
Nandayasa et al. (2023) 43 demonstrated that phytosomes technology could be used as targeted drug delivery for diseases like cancer.
Zhao and Liu (2016) 44, developed on Ginko biloba extract-loaded phytosomes showed targeted drug delivery to the brain, strengthening the therapeutic efficacy in neurodegenerative disorders.
Typical advantages of phytosomes are highlighting figure number 3.
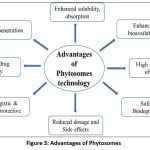 |
Figure 3: Advantages of Phytosomes |
Therapeutic applications of phytosomes technology
Wound healing
Tafish et al., (2023)30 developed CAR (carvacrol) loaded phytosomal formulation thin-film hydration, salting out and co-solvency method by using phospholipon 90 H and Lipoid S 100. Further study results demonstrated that its effective wound-healing ability has been increased after complexation with lipids compared to its free form of CAR.
Cancer treatment
the applicability of various herbal agents has been increased by applying phytosomes technology to enhance multiple factors such as solubility, absorption, targeted delivery, etc. Various phytosomes formulation developed as an anti-cancer targets are extensively discussed by Gaikwad SS, et al., (2023).20
Breast cancer
Alhakamy et al., (2022)46 paired quercetin (QRT) and scorpion venom peptides (SV) into phytosomes as an anticancer medicine. This phytosomes to aid in cellular intake and antiproliferative action against lung cancer cell line-derived human breast cancer cell (MCF-7). The results of the study indicated that a QRT formulation might be a possible curative approach to treating breast cancer.
Cosmeceutical applications
Phytosomes have gained attention in the cosmeceutical and neutraceutical industries due to a marked enhancement in solubility, bioavailability, and stability of phytoactive agents (34, 37-38). Indena, S.P.A., Italy48-49 has developed phytosomes with cosmetics application Meriva®, A potent anti-oxidant agent.
Obesity management:
Menshawe et al., (2018) created a nano lipo-vesicle phytosomal thermo gel from soybean and examined it for anti-obesity properties. Author, discovered that the soy phytosomal thermo gel had a local anti-obesity effect on the abdomen of experimental male albino rats and a small systemic influence on lipid profiles (47). Greenselect® Phytosome, Indena, S.P.A., for weight loss management.48-49
Hepatoprotective effect
The Silybin-Phosphatidylcholine and Vitamin E complex was developed by Loguercio et al. (2019)35 to treat non-alcoholic fatty liver disease and investigated its hepatoprotective activity along with antioxidant activity. Maryana et. al., (2016) 31, also demonstrated the synergistic role of phospholipid.
Commercially available phytosomes
The fields of cosmetics, neutraceuticals, and pharmaceuticals all make extensive use of phytosome technology. Few of the phytosomes developed and marketed by Indena S.P.A., Italy discussed in table 1.48-49
Table 1: Commercially available phytosomes48-4
| Product Name | Active Ingredient | Category | Therapeutic Uses |
| Siliphos® | Silybin, present in Silybum marianum or milk thistle | Antioxidant,Hepatoprotective | support and improves liver health,detoxification,
hepatoprotection |
| Greenselect® Phytosome | Catechin, from green tea | Antioxidant,Anti-inflammatory,
Weight loss |
For management of weight, support metabolic demands, antioxidant and anti-inflammatory |
| Meriva® | Curcuminoids, obtained from Curcuma longa | Antioxidant,Anti-inflammatory | Used in management of joint health,inflammatory response, antioxidant support |
| Casperome® | A mixture of triterpenoid from gum resin and Boswellia serrate | Anti-inflammatory modulation | Manages joint and gut health, healthy inflammatory response |
| Quercefit® | Quercetin from Sophora japonica L. | Supports nutrition, endothelial function and histamine modulation,Anti-inflammatory,
antioxidant, |
improves seasonal immune health and provides anti-inflammatory effect,anti-oxidant |
| Vazguard® | Bergamot extract, polyphenolic extract obtained from Citrus bergamia | Enhances lipid-lowering effects | Supports health in metabolic syndrome, lipid profile management and cardiovascular health, |
| Ubiqsome® | Nonbotanical compounds madeup ofCo-enzyme Q10 | Antioxidant, energy production, supports electron transport chain | Antioxidant, engaged in energy generation, supports cardiovascular and cellular health |
| Hawthorn Phytosome | Flavonoids from Crataegus species | Anti-hypertensive and cardioprotective and antioxidant | Support CVS (cardioprotective),antihypertensive |
| Oleaselect™ Phytosome | Polyphenols from Olea Europaea or olive leaf | Antihyperlipidemic, anti-inflammatory,cardioprotective | Antihyperlipidemic,anti-inflammatory,
cardioprotective |
Future scope
Phytosome technology is constantly improving in terms of stability and formulation diversity. Novel approaches for phyto-phospholipid complexation, drug-lipid ratio optimization, and methods for complex creation to boost stability are among the several developments. Phytosome approaches have also been investigated in terms of different formulations by combining them with nanotechnology and other innovative drug delivery systems. Munot et al. (2022) 51 studied quercetin nanocochleated liposomes and discovered increased bioavailability, antioxidant activity, and anticancer potential.
As phytosome-based innovative formulations continue to be created, regulatory agencies should focus on establishing guidelines for developing and commercializing phytosomes and their products.
Limitations of phytosomes technology
Despite the numerous benefits, phytosomes technology-based products might encounter limitations such as lipid peroxidation, phytoconstituent leaching from the lipid membrane, inconsistency in formulation quality and large-scale formulation standardization. As the drawbacks associated with phytosomes technology is not yet completely documented, clinical evidence for this investigation is minimal.8, 16
Conclusion
Phytosomes technology plays a significant role enhancing the applicability of phytoconstituents with poor bioavailability by forming stable phytoconstituents-lipid complexes. This complexation enhances the solubility due to lipid encapsulation, improves absorption and prolongs circulation of phytoconstituents within the systemic circulation. Despite several benefits, challenges remain with large-scale formulation standardization and the limited availability of clinical data compared to conventional drug delivery systems. In order to validate therapeutic claims, future research should focus on developing regulatory guidelines, refining formulation techniques, and conducting well-designed clinical trials. Overall, phytosomes offer an ideal platform for converting phytoconstituents into safe and efficiency dosage forms with a strong potential to bridge a gap between traditional herbal medication and a modern drug delivery technologies.
Acknowledgement
The authors extend their gratitude to Dr. Sanjay S. Kshirsagar, Principal, MET’s Institute of Pharmacy, Bhujbal Knowledge City, Nashik, for his continuous encouragement, valuable advice, and consistent backing during the drafting of this review article. We are also thankful to the management of MET’s Institute of Pharmacy for providing the necessary facilities, access to scholarly publications, and a suitable research atmosphere to enable this work. Finally, the corresponding author, Ms. Sonali R. Patil extends her heartfelt gratitude to her research mentor, Dr. Santosh Subhash Chhajed, for his critical scientific contributions, rigorous editing, and constant motivation.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Sonali Ravindra Patil: Conceptualization, Methodology, Data Collection, literature survey, investigation, formal analysis, Writing – Original Draft, Review & Editing
Santosh Subhash Chhajed: Conceptualization, Visualization, Supervision, critical revision of the manuscript for important intellectual content.
References
- Liu JK. Natural products in cosmetics. Nat Prod Bioprospect. 2022;12(1):40. Doi: 10.1007/s13659-022-00363-y
CrossRef - Chandorkar N, Tambe S, Amin P, et al. Alpha Arbutin as a Skin-Lightening Agent: A Review. International Journal of Pharmaceutical Research. 2021;13:3502-3510. Doi: 10.31838/ijpr/2021.13.2.446
CrossRef - Rana L, Harwansh RK, Deshmukh R. Recent updates on phytopharmaceuticals-based novel phytosomal systems and their clinical trial status: a translational perspective. Crit Rev Ther Drug Carrier Syst. 2025;42(1):1-45. Doi: 10.1615/CritRevTherDrugCarrierSyst.v42.i1.10
CrossRef - Goyal N, Jerold F. Biocosmetics: technological advances and future outlook. Environ Sci Pollut Res Int. 2023;30(10):25148-25169. Doi: 10.1007/s11356-021-17567-3
CrossRef - Lu M, Qiu Q, Luo X, et al. Phyto-phospholipid complexes (phytosomes): A novel strategy to improve the bioavailability of active constituents. Asian Journal of Pharmaceutical Sciences. 2019;14(3):265-274. Doi: 10.1016/j.ajps.2018.05.011
CrossRef - Barani M, Sangiovanni E, Angarano M, et al. Phytosomes as innovative delivery systems for phytochemicals: a comprehensive review of literature. Int J Nanomedicine. 2021;16:6983-7022. Doi: 10.2147/IJN.S318416
CrossRef - Pandita A, Sharma P. Pharmacosomes: an emerging novel vesicular drug delivery system for poorly soluble synthetic and herbal drugs. ISRN Pharmaceutics. 2013:348186. Doi: 10.1155/2013/348186
CrossRef - Semalty A, Semalty M, Singh D, et al. Preparation and characterization of phospholipid complexes of naringenin for effective drug delivery. J Incl Phenom Macrocycl Chem. 2010;67:253-260. Doi: 10.1007/s10847-009-9705-8
CrossRef - Maiti K, Mukherjee K, Gantait A. Curcumin–phospholipid complex: preparation, therapeutic evaluation and pharmacokinetic study in rats. Int J Pharm. 2007;330(1-2):155-163. Doi: 10.1016/j.ijpharm.2006.09.025
CrossRef - Jing L, Xuling W, Ting Z, et al. A review on phospholipids and their main applications in drug delivery systems. Asian Journal of Pharmaceutical Sciences. 2015;10(2):81-98. Doi: 10.1016/j.ajps.2014.09.004
CrossRef - Drescher S, Hoogevest VP. The phospholipid research center: current research in phospholipids and their use in drug delivery. Pharmaceutics. 2020;12(12):1235. Doi: 10.3390/pharmaceutics12121235
CrossRef - Babazadeh A, Jafari SM, Shi B. Encapsulation of food ingredients by nanophytosomes. Lipid-based nanostructures for food encapsulation purposes. 2019:405-443. Doi: 10.1016/B978-0-12-815673-5.00010-6
CrossRef - Briuglia ML, Rotella C, McFarlane A, et al. Influence of cholesterol on liposome stability and on in vitro drug release. Drug Delivery and Translational Research. 2015;5(3):231-242. Doi: 10.1007/s13346-015-0220-8
CrossRef - Singh R, Kumar R, Sharma PK. Phytosome: recent advance in herbal delivery system. Asian J Pharm Clin Res. 2022;15(6):23-30. https://ajprd.com/index.php/journal/article/view/185
- Saroha K, Waliyan P, Pahwa R, et al. Phytosomes: A Promising Strategy for Enhanced Therapeutic Benefits of Phytochemicals. International Journal of Research in Pharmaceutical Sciences. 2021;11:1-7. Doi: 10.26452/ijrps.v11iSPL4.4656
CrossRef - Sakure K, Patel A, Pradhan M, et al. Recent trends and future prospects of phytosomes: a concise review. Indian J Pharm Sci. 2024;86(3):1234-1245. Doi: 10.36468/pharmaceutical-sciences.1813
CrossRef - Maryana W, Rachmawati H, Mudhakir D. Formation of Phytosome Containing Silymarin Using Thin Layer-Hydration Technique Aimed for Oral Delivery. Materials Today: Proceedings. 2016;3(3):855-866. Doi: 10.1016/j.matpr.2016.02.019
CrossRef - Aniket, Kumari A, Kumari P, et al. Formulation and Evaluation of Topical Soy-Phytosome Cream. Indian J Pharm Pharmacol. 2015;2(2):105-112.
- Shabanpour S. Phytosome: A Novel Drug Delivery Approach in Herbal Medicine. Pharmaceutical Science. 2025. Doi: 10.5772/intechopen.1005766
CrossRef - Gaikwad SS, Morade YY, Kothule AM, et al. Overview of phytosomes in treating cancer: Advancement, challenges, and future outlook. Heliyon. 2023;9(6):e16561. Doi: 10.1016/j.heliyon.2023.e16561
CrossRef - Mukesh VK, Jameel AM. Unlocking the potential of phytosomes: a review of formulation techniques, evaluation methods, and emerging applications. Acta Materia Medica. 2024;3(4):509-520. Doi: 10.15212/AMM-2024-0055
CrossRef - Freag MS, Elnaggar YS, Abdallah OY. Lyophilized phytosomal nanocarriers as platforms for enhanced diosmin delivery: optimization and ex vivo permeation. International Journal of Nanomedicine. 2013;8:2385-2397. Doi: 10.2147/IJN.S45231
CrossRef - Rani A, Kumar S, Khar RK. Murraya koenigii extract loaded phytosomes prepared using antisolvent precipitation technique for improved antidiabetic and hypolidemic activity. Indian J Pharm Educ Res. 2022;56:s326-s338. Doi: 10.5530/ijper.56.2s.103
CrossRef - Sundaresan N, Ilango K. Development and characterization of a nano-drug delivery system containing vasaka phospholipid complex to improve bioavailability using quality by design approach. Research in Pharmaceutical Sciences. 2021;16(1):103-117. Doi: 10.4103/1735-5362.305193
CrossRef - Nandayasa WW, Febriyenti, Lucida H. Optimization and Characterization of Quercetin Vitamin C Nano-Phytosome Formulation. Int J Appl Pharm. 2023;15:51-55. Doi: 10.22159/ijap.2023.v15s1.47507
CrossRef - Das MK, Kalita B. Design and evaluation of phyto-phospholipid complexes (phytosomes) of Rutin for transdermal application. J Appl Pharm Sci. 2014;4(10):51-57. Doi: 10.7324/JAPS.2014.40110
CrossRef - Habbu, Madagundi S, Shastry R, et al. Preparation and Evaluation of Antidiabetic Activity of Allium cepa-Phospholipid Complex (Phytosome) in Streptozotocin Induced Diabetic Rats. RGUHS J Pharm Sci. 2016;5(4):132-141. Doi: 10.5530/rjps.2015.4.3
CrossRef - Elfiyani, Rahmah, Radjab NS, et al. Garlic Extract Phytosome: Preparation And Physical Stability. Int J App Pharm. 2024;16(1):118-125.
CrossRef - Anwar E, Farhana N. Formulation and evaluation of phytosome-loaded maltodextrin-gum Arabic microsphere system for delivery of camellia sinensis extract. J Young Pharm. 2018;10(2):s56-s62. Doi: 10.5530/jyp.2018.2s.11
CrossRef - Tafish AM, El-Sherbiny M, Al-Karmalawy AA, et al. Carvacrol-loaded phytosomes for enhanced wound healing: molecular docking, formulation, DoE-aided optimization, and in vitro/in vivo evaluation. International Journal of Nanomedicine. ;18:5749-5780. Doi: 10.2147/IJN.S421617
CrossRef - Maryana W, Rachmawati H, Mudhakir D. Formation of phytosome containing silymarin using thin layer-hydration technique aimed for oral delivery. Materials Today: Proceedings. 2016;3(3):855-866. Doi: 10.1016/j.matpr.2016.02.019
CrossRef - Barzaghi N, Crema F, Gatti G, et al. Pharmacokinetic studies on IdB 1016, a silybin-phosphatidylcholine complex, in healthy human subjects. Eur J Drug Metab Pharmacokinet. 1990;15:333-338. Doi: 10.1007/BF03190223
CrossRef - David JC, Steven HZ. Lecithin and Choline in Human Health and Disease. Nutrition Reviews. 1994;52(10):327-339. Doi: 10.1111/j.1753-4887.1994.tb01357.x
CrossRef - Rahman SH. Formulation and evaluation of Cassia auriculata flower extract-loaded phytosomal cream to enhance the topical bioavailability. International Journal of Green Pharmacy. 2021;15(4). Doi: 10.22377/ijgp.v15i4.3194
- Loguercio C, Andreone P, Brisc C, et al. Silybin combined with phosphatidylcholine and vitamin E in patients with nonalcoholic fatty liver disease: a randomized controlled trial. Free Radical Biology and Medicine. 2012;52(9):1658-1665. Doi: 10.1016/j.freeradbiomed.2012.02.008
CrossRef - Chi C et al. Phytosome-nanosuspensions for silybin-phospholipid complex with increased bioavailability and hepatoprotection efficacy. European Journal of Pharmaceutical Sciences. 2020;144:105212. Doi: 10.1016/j.ejps.2020.105212
CrossRef - Yasmiwar S, Chaerunisa A, Yohana, et al. Phytosome drug delivery system for natural cosmeceutical compounds: Whitening agent and skin antioxidant agent. Journal of Advanced Pharmaceutical Technology & Research. 2021;12(4):327-334. Doi: 10.4103/japtr.JAPTR_100_20
CrossRef - Puglia C, Offerta A, Tirendi GG, et al. Design of solid lipid nanoparticles for caffeine topical administration. Drug Delivery. 2016;23(1):36-40. Doi: 10.3109/10717544.2014.903011
CrossRef - Belcaro G, Ledda A, Hu S, et al. Greenselect phytosome for borderline metabolic syndrome. Evidence-Based Complementary and Alternative Medicine. 2013;2013:869061. Doi: 10.1155/2013/869061
CrossRef - Pawar HA, Bhangale BD. Phytosome as a novel biomedicine: a microencapsulated drug delivery system. Journal of Bioanalysis & Biomedicine. 2015;7. Doi: 10.4172/1948-593x.1000116
CrossRef - Singh B, Awasthi R, Ahmad A, et al. Phytosome: most significant tool for herbal drug delivery to enhance the therapeutic benefits of phytoconstituents. Journal of Drug Delivery and Therapeutics. 2018;8(1):98-102. Doi: 10.22270/jddt.v8i1.1559
CrossRef - Anwar E, Farhana N. Formulation and evaluation of phytosome-loaded maltodextrin-gum Arabic microsphere system for delivery of Camellia sinensis extract. Journal of Young Pharmacists. 2018;10(2s):S56. Doi: 10.5530/jyp.2018.2s.11
CrossRef - Nandayasa W, Febriyenti H. Optimization and characterization of quercetin vitamin C Nano-Phytosome Formulation. International Conference on Contemporary Science and Clinical Pharmacy. ;:51. ISSN- 0975-7058
CrossRef - Huang Z, Brennan CS, Zhao H, et al. Fabrication and assessment of milk phospholipid-complexed antioxidant phytosomes with vitamin C and E: A comparison with liposomes. Food Chemistry. 2020;324:126837. Doi: 10.1016/j.foodchem.2020.126837
CrossRef - Karimi N, Ghanbarzadeh B, Hamishehkar H, et al. Phytosome as novel delivery system for nutraceutical materials. International Journal of Current Microbiology and Applied Sciences. 2015;4(6):152-159. ISSN: 2319-7706
- Alhakamy NA, Fahmy UA, Eldin SMB, et al. Scorpion venom-functionalized quercetin phytosomes for breast cancer management: in vitro response surface optimization and anticancer activity against MCF-7 cells. Polymers. 2021;14(1):93. https://www.mdpi.com/2073-4360/14/1/93#
CrossRef - Menshawe SF, Ali AA, Rabeh MA, et al. Nanosized soy phytosome-based thermogel as topical anti-obesity formulation: an approach for acceptable level of evidence of an effective novel herbal weight loss product. International Journal of Nanomedicine. 2018;:307-318. Doi: 10.2147/IJN.S153429
CrossRef - Indena, Technical paper issue 0.1: https://share.google/4lm6y0S4ofb2ZlGFO
- Indena, Products list: https://www.indena.com/indena_files/2020/02/Indena-product_list_2020.pdf
- Riva A, Allegrini P, Franceschi F, et al. A novel boswellic acids delivery form (Casperome®) in the management of musculoskeletal disorders: a review. European Review for Medical & Pharmacological Sciences. 2017;21(22):5258-5263. Doi:
- Munot N, Kandekar U, Giram PS, et al. A comparative study of quercetin-loaded nanocochleates and liposomes: formulation, characterization, assessment of degradation and in vitro anticancer potential. Pharmaceutics. 2022;14(8):1601. Doi: 10.3390/pharmaceutics14081601
CrossRef - Bhia M, Motallebi M, Abadi B, et al. Naringenin Nano-Delivery Systems and Their Therapeutic Applications. Pharmaceutics. 2021;13(2):291. Doi: 10.3390/pharmaceutics13020291
CrossRef - Marjan T, Kimia S, Mohammad SD, et al. Phytosomes: A promising nanocarrier system for enhanced bioavailability and therapeutic efficacy of herbal products. Phytomedicine Plus. 2025;5(2):100779. Doi: 10.1016/j.phyplu.2025.100779
CrossRef - Metkari V, Shah R, Salunkhe N, et al. QBD approach for the design, optimization, development, and characterization of naringenin-loaded phytosomes to enhance solubility and oral bioavailability. Journal of Pharmaceutical Innovation. 2023;18:2083-2097. Doi: 10.1007/s12247-023-09775-w
CrossRef - Khan S, Kotkar P. Phytosomes in Nanovesicle Herbal Formulation: A Contemporary Advancement in Natural Medicine. Journal of Pharmaceutical Advanced Research. 2023;6(10):1940-1947. e – ISSN: 2581-6160
- Telange DR, Patil AT, Pethe AM, et al. Formulation and characterization of an apigenin-phospholipid phytosome (APLC) for improved solubility, in vivo bioavailability, and antioxidant potential. European Journal of Pharmaceutical Sciences. 2017;108:36-49. Doi: 10.1016/j.ejps.2016.12.009
CrossRef - Chaudhary K, Rajora A. Phytosomes: a critical tool for delivery of herbal drugs for cancer: Phytosomes: Advancing Herbal Medicine Delivery. Phytochemistry Reviews. 2025;24(1):165-195. Doi: 10.1007/s11101-024-09947-7
CrossRef - Deleanu M, Toma L, Sanda GM, et al. Formulation of phytosomes with extracts of ginger rhizomes and rosehips with improved bioavailability, antioxidant and anti-inflammatory effects in vivo. Pharmaceutics. 2023;15(4):1066. Doi: 10.3390/pharmaceutics15041066
CrossRef - Allam AN, Komeil IA, Abdallah OY. Curcumin phytosomal softgel formulation: Development, optimization and physicochemical characterization. Acta Pharmaceutica. 2015;65(3):285-297. Doi: 10.1515/acph-2015-0029
CrossRef - Chettupalli AK, Bukke SP, Vardhan J, et al. Polyherbal formulations and phytosome-based delivery in diabetic wound healing: an integrative review. Journal of Biomaterials Science, Polymer Edition. 2025;:1-28. Doi: 10.1080/09205063.2025.2568684
CrossRef - Mane K, Baokar S, Bhujbal A, et al. Phyto-phospholipid complexes (phytosomes): a novel approach to improve the bioavailability of active constituents. Journal of Advanced Scientific Research. 2020;11(03):68-78. https://sciensage.info/index.php/JASR/article/view/508
- Koppula S, Shaik B, Maddi S. Phytosomes as a new frontier and emerging nanotechnology platform for phytopharmaceuticals: Therapeutic and clinical applications. Phytotherapy Research. 2025;39(5):2217-2249. Doi: 10.1002/ptr.8465
CrossRef - Govindaram LK, Bratty MA, Alhazmi HA, et al. Formulation, biopharmaceutical evaluation and in-vitro screening of polyherbal phytosomes for breast cancer therapy. Drug Development and Industrial Pharmacy. 2022;48(10):552-565. Doi: 10.1080/03639045.2022.2138911
CrossRef
Accepted on: 01-12-2025
Second Review by: Dr. Sura Maan Salim
Final Approval by: Dr. Ali Elshafei