Phytase from Bacillus subtilis BGS3: Purification, Characterization, and Evaluation of its Potential to Enhance Nutritional Availability in Food and Feed
Department of Microbiology, Mehsana Urban Institute of Science, Ganpat University, Mehsana, Gujarat India.
Corresponding Author E-mail: hss02@ganpatuniversity.ac.in
DOI : http://dx.doi.org/10.13005/bbra/3475
ABSTRACT:Since Bacillus subtilis species are generally regarded as safe (GRAS), they are widely exploited for diverse applications in the food and feed industries. The current study focuses on the purification and biochemical characterisation of phytase from B. subtilis BGS3 and further evaluates its potential to enhance the nutritional quality of food and feed products. The enzyme was purified to homogeneity, yielding an apparent molecular weight of 51 kDa. The purified enzyme exhibited maximum catalytic activity at 50 °C and pH 6.0. Among the tested metal ions, Ca²⁺, Mg²⁺, and Mn²⁺ caused a slight enhancement in phytase activity. Enzyme kinetic studies revealed a Vₘₐₓ of 651.83 µM min⁻¹ and a Kₘ of 0.086 mM. The in vitro efficacy of phytase on pearl millet flour hydrolysis resulted in the release of inorganic phosphate (Pi) and free amino acids up to 18 hours, while soluble protein content increased and phytic acid levels decreased up to 24 hours. The release of mineral ions occurred in the order Fe > Mg > Ca > Zn up to 24 hours. The dephytinization of phytic acid from food and feed products by the purified phytase revealed that maximum dephosphorylation occurred in the experimental diet, followed by brown bread, the standard diet, and was least in white bread.
KEYWORDS:Amelioration; Characterization; Dephytinization; Phytase; Phytic acid; Purification
Introduction
Plant-based staple crops such as beans and cereals are excellent sources of minerals, proteins, and essential amino acids for vegans. Phytic acid, which comprises about 5% of the dry weight of grains, serves as the primary storage form of inositol and phosphorus 1. However, these foods also contain antinutritional factors such as phytic acid (PA) 2. PA binds to proteins, metal cations, and microelements, forming complexes that hinder their absorption.
Monogastric animals (pigs, chickens, swine, and humans) lack the enzymes required to hydrolyse phytic acid 3; as a result, they are unable to utilise the bound nutrients efficiently, rendering phytic acid an antinutrient 4. Phytase enzymes, or the microorganisms that produce them, offer a practical way to reduce the antinutritional properties of PA. The benefits of phytase include an increase in protein and dialysable mineral ion content, along with improvements in the yield and overall quality of the final product, while maintaining the product’s inherent characteristics through the hydrolysis of PA 5. Iron and zinc deficiencies are widespread in underdeveloped regions. While insufficient dietary intake is a major contributing factor, antinutrients found in plant-based foods also significantly limit mineral bioavailability.6,7 These antinutritional factors are a major contributing cause of malnutrition and anaemia, particularly among growing children and pregnant or lactating women in developing countries.8
In the realm of baking, phytase has proven effective in enhancing micronutrient availability in whole rye and quinoa flours as well as in rye–wheat sourdough.9-11 A fungal phytase-based approach to processing soybean protein isolates (a common ingredient in meat alternatives, infant formulas, beverages, etc.) has been proven to release higher concentrations of readily available (dialysable) zinc and calcium, coupled with a decrease in phytate concentration.12 Studies have shown that the addition of phytase to sorghum-based foods, either before or after processing, leads to a reduction in phytate content.13,14 Traditional food processing methods such as germination, fermentation, soaking, and combined approaches can reduce phytic acid and phytate levels to some extent, although their effectiveness remains limited.
Several probiotic strains belonging to the genera Lactobacillus, Bifidobacterium, and Enterococcus exhibit notable phytase activity.15 Specifically, Pediococcus acidilactici BNS5B has demonstrated a strong ability to dephosphorylate phytic acid, suggesting its potential application in cereal-based food and feed products.16 Phytase has become a standard additive in animal feed, demonstrating its positive impact on the overall quality of poultry and pigs, such as increased body weight and improved mineral assimilation through the hydrolysis of phytic acid and the release of bound micronutrients.17 In egg-laying hens, phytase supplementation in wheat–soybean–maize diets have been associated with increased egg production, improved shell quality, and enhanced egg content, likely due to the enzyme’s ability to liberate phytic acid, calcium, and other micronutrients.18 In a similar vein, studies have shown that phytase supplementation enhances laying hen performance and egg quality compared with a control group that did not receive phytase.19,20
One of the most important aspects of the commercial use and understanding of the structure–function relationship is the purification of the enzyme. Since Bacillus species are generally regarded as safe (GRAS), they are frequently exploited for the production of phytase.21 Concentrating the crude enzyme is the initial step in the purification of phytase, and this can be achieved through lyophilisation, ultrafiltration, or precipitation induced by organic solvents or salts, as studies have shown these methods to yield a highly purified enzyme. To further purify the concentrated protein to homogeneity, more than one chromatographic technique is typically employed.22 Despite the purification of phytase from various microbial sources, none of them possesses all the necessary characteristics required for commercial exploitation in the food and feed industries.
Phytases have been identified in a diverse range of microorganisms and are recognised for their numerous promising properties suitable for industrial applications in food and feed.15,23 Studies have shown that lactic acid bacteria producing functional phytase have the potential to improve phosphorus release from phytate in livestock, poultry, and fish diets, suggesting an approach to reduce reliance on inorganic phosphorus supplementation.24
Phytase-producing Pediococcus pentosaceus grown on fermented rice bran has been used to improve micronutrient bioavailability in Nile tilapia and has proven to be a cost-effective, environmentally sustainable approach for aquaculture feed by reducing waste and inorganic inputs.25 Another study reported that microbial phytase improved the digestibility of phytate phosphorus (phytate-P), overall phosphorus retention, growth, and feed conversion ratio (FCR) compared with the control group without phytase in a high plant meal inclusion diet for Atlantic salmon.26 In addition, studies have shown that supplementation with microbial phytase improves amino acid digestibility, mineral retention, and bone mineralisation, and also increases plasma inositol levels (a breakdown product) in monogastric mammals.27
However, an ideal phytase that can be utilised as both a feed and dietary supplement is still lacking. Therefore, continued efforts in enzyme purification and biochemical characterisation are essential to identify and develop novel phytases with desirable properties. Consequently, there is a growing need for novel industrial phytases that function more efficiently and reliably in the gastrointestinal tract of monogastric animals.17,28
Hence, the present study aims to utilise the bacterial strain Bacillus subtilis BGS3 for phytase purification and biochemical characterisation. The purified phytase was further employed to demonstrate its in vitro potential in the dephytinisation of pearl millet flour and various food and feed products, which may consequently enhance the bioavailability of nutrients.
Material and Methods
Microbial strain and preparation of crude extract
In the present study, the bacterial isolate Bacillus subtilis BGS3, previously identified and deposited with accession number PQ223664, was utilised for the purification of phytase.29 The fermentation medium used for phytase production consisted of rice bran (1.1 g %), CaCl₂ (0.05 %), casein (0.05 %), KH₂PO₄ (0.05 g %), K₂HPO₄ (0.04 g %), and MgSO₄·7H₂O (0.02 g %). The initial pH of the medium was adjusted to 7.30 The fermentation medium was inoculated with 5% of an overnight-grown culture of Bacillus subtilis BGS3 and incubated at 37 °C with shaking at 150 rpm. After 72 hours of incubation, the culture was centrifuged at 10,000 rpm for 20 minutes to separate the cells, and the resulting supernatant was collected and used as the crude enzyme extract.
Purification of enzyme
Crude enzyme was concentrated from the supernatant by gradually adding finely powdered ammonium sulfate to achieve 80% saturation at 20°C, followed by overnight incubation at 4°C. The pellets were harvested by centrifugation at 10,000 rpm for 15 minutes at 4 °C and resuspended in sodium acetate buffer (0.2 M, pH 5.5). The suspension was dialysed against the same buffer, with the buffer replaced after eight hours. The dialysed protein sample was then used for protein estimation and phytase assay.30,31 The Q-Sepharose anion-exchange column was equilibrated with 20 mM Tris-HCl buffer (pH 8.0) to obtain an anion exchanger at the desired pH. The dialysed sample was then passed through the column at a flow rate of 0.5 mL/min. The bound proteins were eluted using a segmented NaCl gradient ranging from 0.1 to 1.0 M in 20 mM Tris-HCl (pH 8.0). The eluted fractions were collected and analysed for protein content (by measuring absorbance at 280 nm) and phytase activity. The active fractions were pooled and further subjected to gel permeation chromatography using a Sephadex G-100 matrix, eluted with the same buffer at a flow rate of 20 mL/hr. Fractions of 2 mL each were collected and analysed for protein (O.D. at 280 nm) and enzyme activity (at 700 nm). The active fractions were then subjected to electrophoresis to assess the purity of the protein.
Analysis of molecular weight and purity of phytase
The purity of the protein was assessed using SDS–PAGE with a 12% resolving gel and a 5% stacking gel. The molecular weight of the protein was estimated by comparing its Rf value with those of molecular weight markers on a standard graph.32 Additionally, MALDI–TOF MS was employed to determine the intact mass of the protein by analysing the m/z values of ions generated from a protein–matrix mixture.33
Effect of pH and temperature on phytase activity
To study the effect of pH on purified phytase, 1 mL of 100 mM buffer solutions of varying pH—glycine-HCl (pH 2.0 and 3.0), sodium acetate (pH 4.0 and 5.0), sodium citrate (pH 6.0 and 7.0), Tris-HCl (pH 8.0), and glycine-NaOH (pH 9.0)—was mixed separately with 0.1 mL of purified phytase. The pH value showing maximum enzyme activity was subsequently used to assess the relative stability of the enzyme for up to 100 minutes, with measurements taken at 20-minute intervals.
To determine the optimal temperature, reaction mixtures containing the enzyme and substrate were incubated at different temperatures (ranging from 30°C to 90°C, at 10°C intervals) for 20 minutes, followed by enzyme assay. Thermal stability was further evaluated for up to 120 minutes, at 20-minute intervals, using the optimal
temperature obtained from the above experiment.
Effect of additives and metal ions
To study the effect of additives, 0.1 mL of 1% solutions of β-mercaptoethanol, EDTA, Tween 20, Tween 80, and SDS were each mixed with 0.1 mL of purified phytase. Similarly, 0.1 mL of different metal ions—Na⁺, K⁺, Zn²⁺, Mn²⁺, Ca²⁺, Fe³⁺, Co²⁺, and Mg²⁺—at a final concentration of 5 mM were added to separate enzyme samples. The mixtures were pre-incubated for 30 minutes at room temperature, after which the phytase assay was performed. The relative enzyme activity was then calculated with respect to the control.
Determination of the Michaelis–Menten constant (Kₘ) and Vₘₐₓ values
Phytase activity was measured by incubating 20 µM of purified phytase with varying concentrations of sodium phytate (0.05–0.5 mM) at 50 °C for 20 minutes. The enzyme’s kinetic parameters, Km and Vmax, were determined using a Lineweaver-Burk plot.34
Amelioration of pearl-millet flour nutrition using purified phytase from B. subtilis BGS3
A 10% (w/v) pearl millet flour suspension was prepared in sodium acetate buffer (0.1 M, pH 5.5) and treated with purified phytase at a constant ratio of 10 U/g of flour, then incubated at 50 °C under shaking conditions. A control was set up without the enzyme. Samples were withdrawn at fixed intervals of 6 hours up to 24 hours to analyse inorganic phosphate, amino acids, and protein content during dephytinisation.31,35,36 To analyze the phytic acid content release during dephytinization wade reagent was used for the colorimetric assay.37 All experiments represented with bars were conducted in triplicate, and the average values along with standard deviations (S.D.) are shown in the graphs.
ICP-MS was used to detect the release of divalent metal ions resulting from dephytinisation. For metal ion measurement, test samples were collected from the dephytinisation apparatus over a 24-hour period.38
Dephytinization of food and feed products using purified phytase from B. subtilis BGS3
Purified phytase from B. subtilis BGS3 was utilised for the amelioration of food and feed products. The effectiveness of the purified phytase obtained from B. subtilis BGS3 was evaluated by estimating the amount of orthophosphate present in the samples both before and after phytase treatment.39 White bread (refined flour), brown bread (whole wheat flour), and a standard diet (Vetmido cattle booster) were procured from the local market in Ahmedabad, India. A modified, phytic-acid-rich dough was prepared in the laboratory. To analyse the dephytinisation efficacy of the purified phytase on various food and feed samples, 1 g of each sample was processed. A 1000 µL neutralised sample was treated with 1 U of phytase for 20 minutes at 50 °C. A spectrophotometric method was used to quantify the amount of free phosphorus and phytic acid released.16
All experiments were conducted in triplicate, and the average values along with their standard deviations are shown in the graphs. A one-way ANOVA was used to determine the significance of phytase production. Using Tukey’s test (post hoc), significance was assessed at the 95% confidence level with a P-value of 0.05. Identical letters in the graphs indicate that the corresponding values are statistically similar and show no significant variation.
Results
Purification of phytase from B. subtilis BGS3
Phytase from Bacillus subtilis BGS3 was purified using the three-step procedure outlined in Table 1. The crude extract exhibited 1.5 U of phytase activity and contained 1.143 mg of protein. Following ammonium sulphate precipitation and dialysis, the dialysed fraction retained phytase activity, yielding 0.101 mg of total protein with a specific activity of 11.18 U/mg. The precipitated and eluted fractions were then applied to an anion-exchange column and eluted at a concentration of 0.5 M NaCl, resulting in a specific activity of 41.31 U/mg. Fractions displaying the highest phytase activity were subsequently subjected to gel filtration chromatography using a Sephadex G-100 column to obtain a highly purified enzyme preparation.
Overall, the three-step purification protocol achieved a 46.04-fold purification with a final specific activity of 64.12 U/mg protein and a yield of 32.22%. Comparable purification efficiencies have been reported previously; for example, phytase from Bacillus licheniformis PFBL-03 was purified in three steps, achieving 39-fold purification with a 10% yield.40 Similarly, phytases from Bacillus cereus (isolated from Achatina fulica), Bacillus nealsonii ZJ0702, and Citrobacter freundii have been purified to 12.8-fold (1.6% yield), 44-fold (5.7% yield), and 41.3-fold (9.3% yield), respectively.41-43
Table 1: Summary of steps involved in the purification of phytase from B.subtilis BGS3
| Steps | Protein Conc.(mg/mL) | Enzyme Activity(U/mL) | Specific Activity(U/mg) | Yield (%) | Purification fold |
| Crude | 1.143 | 1.59 | 1.40 | 100 | 1 |
| Partially Purified | 0.101 | 1.13 | 11.18 | 70.98 | 8.03 |
| Ion-Exchange Ch. | 0.023 | 0.95 | 41.31 | 59.68 | 29.66 |
| Gel permeation Ch. | 0.008 | 0.51 | 64.12 | 32.22 | 46.04 |
Determination of molecular weight of phytase
The purified samples from each step were analysed using SDS–PAGE to assess purity and estimate molecular mass, and by MALDI-TOF MS to determine the precise molecular mass of the phytase. SDS–PAGE indicated that the purified enzyme is a monomeric protein with an approximate molecular mass of 47.25 kDa (Figure 1), whereas MALDI-TOF MS analysis revealed a molecular mass of 51 kDa (Figure 2). The MALDI-TOF MS result suggests a true difference in the actual mass of the protein, as this technique typically provides a more accurate determination than the approximate values inferred from SDS–PAGE. This discrepancy may be attributed to protein folding or anomalous migration during electrophoresis, which can affect apparent mobility in the gel. MALDI-TOF MS, in contrast, measures the exact mass-to-charge ratio of ionised molecules and therefore provides a more precise molecular weight than SDS–PAGE, which offers only an estimated mass based on gel migration.
 |
Figure 1: SDS-PAGE of protein sample during various steps of purification |
M: Protein molecular weight marker; Lane 1: Crude extract of protein; Lane 2: Partially purified protein sample; Lane 3: Sample after ion-exchange chromatography; Lane 4: Sample after sephadex G-100 column chromatography.
 |
Figure 2: Intact mass analysis of purified phytase |
Phytases, particularly those from Bacillus species, typically exhibit molecular masses ranging from 37 to 48 kDa. Several studies support this observation, phytase purified from the probiotic strain B. subtilis P6 showed a molecular mass of 40 kDa44, while Dan et al. purified a monomeric phytase of 40–42 kDa from B. licheniformis ONF2.45 Other reported molecular masses include 43 kDa for phytase from B. nealsonii ZJ070242, 45 kDa from Bacillus sp.46, 44 kDa from Bacillus sp. DS1147 and Bacillus sp. KHU-1048, 47.5 kDa from Bacillus sp. MD49, and 46 kDa from B. subtilis CF9250. Phytases from non-Bacillus species also fall within this range, such as the 45 kDa phytase from Klebsiella pneumoniae 9-3B.51
However, some Bacillus phytases possess lower molecular masses, such as the 38 kDa enzymes from B. subtilis natto N-7752, B. subtilis MJA53, and B. licheniformis PFBL-03.40 Additionally, low-molecular-mass phytases have been reported by Parhamfar et al., who identified a 28 kDa phytase from Geobacillus stearothermophilus DM1254, and by Patki et al., who described phytases in the 16–22 kDa range from Bacillus species.55
Conversely, several high-molecular-mass phytases have also been documented. Abdolshahi et al. purified a 73 kDa phytase from Bacillus sp. strain LA1256, while Neira-Voelma et al. reported a phytase of 89 kDa from Aspergillus niger.57
Effect of pH
Phytase activity peaked at pH 6, and within the pH range of 5 to 7 the purified enzyme retained at least 80% of its maximal activity. However, activity decreased sharply below pH 5 and above pH 7 (Figure 3). These findings indicate that the purified phytase is most stable and active under mildly acidic conditions. Time-dependent stability analysis at pH 6 showed that the purified phytase maintained up to 50% relative stability for 100 minutes (Figure 4).
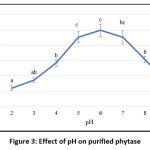 |
Figure 3: Effect of pH on purified phytase |
Phytases exhibiting optimal activity within the pH range of 3.5 to 6 are classified as acidic phytases.58 Phytases from microorganisms belonging to the genera Bacillus, Aspergillus, Lactobacillus, and Pseudomonas have been shown to possess a broad pH activity range, with optimal pH values between 2.5 and 6.5 for Bacillus spp., Enterobacter spp., and Lactobacillus plantarum.59 The present findings agree with earlier reports, as phytase from B. subtilis BGS3 also performed optimally at pH 6. Similar observations have been reported for B. subtilis P6, which exhibited an optimum pH of 6 and maximal activity between pH 5 and 7.35
Further studies have documented comparable results, including purified phytase from B. cereus EME48 with a pH optimum of 5.5.60, Bacillus sp. DM12 with an optimum of pH 4.554, Mitsuokella jalaludinii with an optimum range of pH 4.0–5.061, and Selenomonas ruminantium JY35 with an optimum of pH 4.0–5.5.62 Additional studies on Bacillus phytases have reported moderately acidic pH optima, such as pH 6.5 for B. subtilis (natto) N-77,52 and pH 7.0 for B. subtilis VTT E-68013 and Bacillus sp. DS11.47
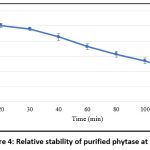 |
Figure 4: Relative stability of purified phytase at pH 6 |
Effect of temperature
As shown in Figure 5, the purified phytase exhibited maximal activity at 50 °C. However, the thermal stability analysis conducted over a period of 120 minutes at 50 °C (Figure 6) demonstrated that the enzyme remained relatively stable for up to 100 minutes at this temperature, retaining approximately 50% residual activity. These findings are consistent with earlier reports on purified phytases from Enterobacter and Serratia species 63, B. megaterium 46, B. subtilis BS4664, and Geobacillus sp.65. In contrast, M. jalaludinii has been reported to exhibit a higher optimum temperature of 55–60 °C.61
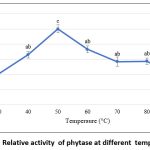 |
Figure 5: Relative activity of phytase at different temperatures |
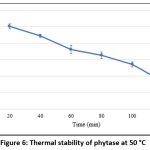 |
Figure 6: Thermal stability of phytase at 50 °C |
Effect of metal ions and additives
The study evaluated the effect of various metal ions (as sulphate and chloride salts) on the activity of purified phytase. Phytase activity was inhibited by Zn²⁺, K⁺, Na⁺, Co²⁺, and Fe³⁺, but enhanced by Ca²⁺, Mg²⁺, and Mn²⁺. Metal ions are known to improve thermal stability and stimulate phytase function, and the presence of zinc, iron, and calcium in food and feed may significantly influence the efficacy of phytase supplements.4 Therefore, it is critical to investigate their inhibitory and synergistic effects when phytase is added.66 Moreover, it is challenging to ascertain whether the inhibitory action of certain metal ions results from direct binding to the enzyme or from the formation of poorly soluble complexes with phytic acid, which reduces the quantity of available active substrate.
Negative regulation may also occur through the direct interaction of metal ions with the substrate or with insoluble metal–phytate complexes, preventing the enzyme from functioning effectively.67 Similarly, studies on the influence of various additives on purified phytase indicate that the presence of Tween 20 and Tween 80 enhances relative activity compared to the control, whereas β-mercaptoethanol (βME), SDS, and EDTA were found to reduce enzymatic activity (Figure 8). These findings are consistent with previous reports, Jain et al. observed that phytase activity increased in the presence of Tween 20, Tween 80, Ca²⁺, and Co²⁺, while Mg²⁺, Al³⁺, and Fe²⁺ inhibited it.68 These results align with the present study, which also found that certain metal ions can either enhance or inhibit phytase activity. Wang and Tong (2010) reported that EDTA had a stimulatory effect,69 while phytase from M. jalaludinii was strongly inhibited by Cu²⁺, Zn²⁺, Fe²⁺, and Fe³⁺, but slightly stimulated by Ca²⁺ and Mn²⁺ 51, in contrast, B. subtilis (natto) N-77, enzyme activity was strongly suppressed by EDTA, Cu²⁺, Zn²⁺, Fe²⁺, Ba²⁺, Cd²⁺, and Al³⁺.52 These results suggest that the impact of metal ions and additives on phytase activity varies depending on the source organism.
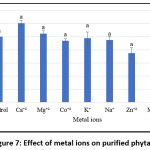 |
Figure 7: Effect of metal ions on purified phytase |
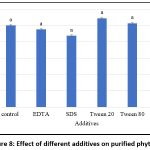 |
Figure 8: Effect of different additives on purified phytase |
Determination of the Michaelis–Menten constant (Kₘ) and Vₘₐₓ values
The phytase assay data for the purified enzyme from B. subtilis BGS3 at varying substrate concentrations (0.05–0.50 mM) indicate that the enzyme follows a typical Michaelis–Menten pattern (Figure 9). By plotting the Lineweaver–Burk reciprocal graph, the Km and Vmax values for B. subtilis BGS3 phytase were determined to be 0.086 mM and 651.83 µM/min, respectively.
Enzyme kinetics for various Bacillus species have been studied and revealed Km values ranging from 0.08 to 10 mM, indicating differing affinities for phosphorylated substrates. Lower Km values correspond to higher substrate affinity, while higher Vmax values indicate greater catalytic efficiency.54 The kinetic parameters of phytase enzymes vary across bacterial strains. For B. tequilensis Dm018, Km and Vmax were reported as 61 μmol and 585 μmol/min, respectively.70 In comparison, the Vmax values for B. subtilis (natto) and Bacillus sp. DS11 were 500 μM and 550 μM, respectively.47,52 For Geobacillus stearothermophilus DM12, the Km and Vmax for sodium phytate were 0.177 mM and 1.126 μmol/min, respectively.54 In B. subtilis subsp. subtilis JJB250, the reported Km and Vmax values were 0.293 mM and 11.49 nmol/s.68 These variations reflect strain-specific differences in substrate affinity and catalytic efficiency.
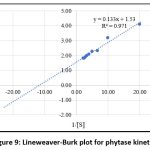 |
Figure 9: Lineweaver-Burk plot for phytase kinetics |
Amelioration of pearl-millet flour nutrition using purified phytase from B. subtilis BGS3
Millets exhibited phytate concentrations ranging from 7.13 to 7.20 mg/g dry matter. Despite their high nutritional value, the formation of insoluble metal–phytate complexes at physiological pH is considered a primary cause of nutritional deficiencies.71 Pearl millet flour is one of the most important staple foods for human consumption. To address this nutritional deficit, purified phytase from B. subtilis BGS3 was employed, and the release of essential nutrient elements was analysed.
The results indicated that phytase efficiently released inorganic phosphate (Pi) from millet flour, with an estimated value of 2143 ± 10 µg after 18 hours of incubation. Beyond 18 hours, no further significant increase in Pi release was observed (Figure 10a). Phytase treatment also released 315 ± 15 µg/mL of free amino acids after 18 hours compared to the control (Figure 10b). A slow decline in amino acid levels was noted thereafter, likely due to enzyme instability at elevated temperatures. Total soluble protein content increased to 2612 ± 110 µg/mL after 24 hours of treatment at 50 °C (Figure 10c). While nutritional components increased over time, phytic acid content declined to 0.62 ± 0.19 µg/mL, resulting in the release of free phosphorus (Figure 10d).
Superscript letters (a–d) above the columns indicate significant differences among the values. Statistical significance was determined using Tukey’s post-hoc test.
Previous studies support these findings, Shivanna and Venkateswaran demonstrated the ameliorative efficacy of lactic-acid-derived phytase in various cereals and pulses, in agreement with the current study.72 Similarly, Humicola nigrescens has been reported to release inorganic phosphate from wheat flour and gram flour at 50 °C, with a positive correlation observed between soluble protein and inorganic phosphate release over time.73 Phytase from the thermophilic mould Sporotrichum thermophile has been shown to effectively dephytinize soymilk, wheat flour, and sesame oil cake.74 Additionally, phytase from Aspergillus oryzae dephytinized wheat bran, releasing inorganic phosphate while simultaneously reducing sugars and soluble protein.75
In the present study, the efficacy of purified phytase in releasing metal ions from pearl millet flour was further validated using ICP-MS. Phytase treatment resulted in the release of metal ions in the following order: highest for Fe, followed by Ca and Zn, with the least release observed for Mg (Figure 11).
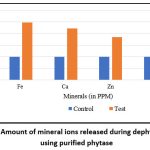 |
Figure 11: Amount of mineral ions released during dephytinization using purified phytase |
Dephytinization of food and feed product using purified phytase from B. subtilis BGS3
The effect of purified phytase was evaluated on various food and feed products, including white bread, brown bread, standard diet, and an experimental phytic-acid-rich diet prepared in the laboratory. The experimental diet used in the current study was based on Indian food and differed from the standard diet in terms of caloric content, proteins, lipids, and carbohydrates, with the exception of a high fibre content primarily composed of phytic acid.76,77 The efficacy of phytase purified from B. subtilis BGS3 was assessed by analysing the phytate content before and after treatment, as well as the amount of inorganic phosphate released in each product (Figure 12). Among all products tested, the highest dephosphorylation was observed in the experimental diet (94.24%), followed by brown bread (68.00%), standard diet (43.45%), and the lowest in white bread (41.67%) (Figure 13).
Treatment with purified phytase significantly enhanced the release of free phosphorus in the products compared to the control, demonstrating the efficacy of the purified enzyme and underscoring its potential as a feed additive for poultry and as a functional ingredient in human food products. Previous studies have reported that increased in vitro dephytinization results in enhanced phosphorus availability and a concomitant reduction in phytic acid content in food and feed.78 Other studies have shown that pretreatment with phytase from E. coli and Citrobacter braakii enhances calcium availability in soybean-based basal diets.79
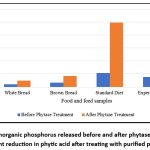 |
Figure 12: Inorganic phosphorus released before and after phytase treatment, per cent reduction in phytic acid after treating with purified phytase |
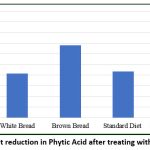 |
Figure 13: Per cent reduction in Phytic Acid after treating with purified phytase |
Since phytase hydrolyses phytic acid, it consequently improves the bioavailability of chelated cations, including calcium, zinc, magnesium, sodium, potassium, and iron. This process can also reduce the need for inorganic phosphate supplements, thereby preventing environmental pollution and limiting phytate excretion.80 Currently, recombinant or in vitro-produced phytases are used to hydrolyse phytic acid, however due to the high cost, bioengineered strains could be preferred as bioreactors for phytase production.81,82 Moreover, recombinant phytases are expensive and involve legal complexities, making alternative food supplements a viable option.
Monogastric animals fed on plant-based diets often exhibit limited phosphorus assimilation, and inorganic phosphorus is frequently added to their diets to meet nutritional requirements. Interestingly, exogenous phytase supplementation has been shown to increase body mass, as dietary phytase effectively dephytinises phytic acid, reducing phytic acid content and decreasing phosphorus excretion in faeces.83,84 Studies have also demonstrated that endogenous wheat phytase increases phosphorus levels and nearly completely degrades phytic acid in fonio porridge. Additionally, weight gain has been reported in broilers, chicks, catfish, rainbow trout, and Labeo rohita fed plant, microbial, or commercial phytase, or recombinant phytase-producing Lactobacillus gasseri TDCC65.85–87 Furthermore, researchers have also shown that enzymatic, physical, or chemical degradation of phytic acid enhances the bioavailability of phosphorus, iron, zinc, and calcium in broiler chickens, fish, and poultry.88
Discussion
The present study successfully purified, characterised, and evaluated the nutritional enhancement potential of phytase derived from Bacillus subtilis BGS3. The multistep purification protocol, comprising ammonium sulphate precipitation, ion-exchange chromatography, and gel filtration, resulted in a 46.04-fold purification with a final specific activity of 64.12 U/mg and a recovery of 32.22%. This purification efficiency is comparatively higher than values reported for several other microbial phytases, such as those from B. licheniformis PFBL-03 (39-fold; 10% yield) and B. cereus isolates (12.8-fold; 1.6% yield).40,58 The elevated purification fold and yield obtained in the present study suggest that the purification steps were efficient and well-optimized for B. subtilis BGS3 phytase.
SDS–PAGE and MALDI-TOF MS analyses revealed that the purified enzyme is monomeric, with a molecular mass of ~47.25 kDa by SDS–PAGE and 51 kDa via MALDI-TOF MS. The slight variation in molecular mass is expected, as SDS–PAGE provides only approximate values due to migration anomalies, whereas MALDI-TOF MS allows precise mass determination.89 The mass of BGS3 phytase aligns well with the typical molecular weight range (37–48 kDa) reported for most Bacillus phytases. Similar molecular masses have been reported for phytases from B. subtilis P6 (40 kDa), B. nealsonii ZJ0702 (43 kDa), and Bacillus sp. MD (47.5 kDa).42,44,49 The consistency in molecular mass further supports the classification of this enzyme within the β-propeller phytase family commonly found in Bacillus species.90
The biochemical characterisation of the purified enzyme demonstrates that B. subtilis BGS3 phytase is optimally active at pH 6 and retains substantial activity across a moderately acidic range (pH 5–7). This pH profile is consistent with those of other Bacillus phytases, including B. subtilis P6 and B. subtilis natto N-77, which also show maximum activity near pH 6.42,44,49,52 The stability of the enzyme at this pH for up to 100 minutes suggests potential compatibility with food processing applications, as many cereal-based foods fall within this pH range.2
Temperature profiling revealed that the enzyme exhibits maximum activity at 50 °C, and retains 50% of its activity for up to 100 minutes at this temperature. This is consistent with the thermostability profile of phytases from B. megaterium, B. subtilis BS46, and Geobacillus species.47,54,64 The ability of the enzyme to function efficiently at moderately high temperatures is particularly advantageous for industrial applications such as baking, fermentation, and pellet feed processing, where elevated temperature conditions are commonly encountered.66
The purified phytase exhibited varied responses to metal ions and additives. Activity was stimulated by Ca²⁺, Mg²⁺, and Mn²⁺, whereas ions such as Zn²⁺, Co²⁺, Na⁺, K⁺, and Fe³⁺ were inhibitory. These observations are consistent with earlier reports indicating that divalent cations, especially Ca²⁺, often play a stabilizing role in phytase activity.58 The positive influence of non-ionic surfactants (Tween 20 and Tween 80) and inhibitory effects of β-mercaptoethanol, SDS, and EDTA further reflect the enzyme’s structural dependence on disulfide bonds and hydrophobic interactions. Differences in metal ion sensitivity across species underscore the strain-specific catalytic properties of microbial phytases.58,91
Kinetic analyses demonstrated that BGS3 phytase follows Michaelis–Menten kinetics, with a Km value of 0.086 mM and Vmax of 651.83 µM/min. The low Km value indicates high affinity toward sodium phytate, suggesting effective substrate utilization. These kinetic parameters compare favourably with those reported for several Bacillus phytases, including B. tequilensis DM018 and Geobacillus stearothermophilus DM12.70,54 The high catalytic efficiency highlights the potential of BGS3 phytase for applications requiring rapid phytic acid degradation.
The functional efficacy of the purified enzyme was assessed using pearl millet flour, a nutrient-rich but phytate-dense staple. The enzyme effectively released phosphorus, amino acids, and soluble proteins while significantly reducing phytic acid levels. These findings align with earlier reports on phytases from Humicola, Sporotrichum thermophile, and A. oryzae, which demonstrated similar nutrient-releasing capabilities in cereal-based substrates.73-75 The release of essential minerals such as Fe, Ca, Zn, and Mg, confirmed by ICP-MS, underscores the enzyme’s potential to ameliorate mineral bioavailability in plant-based foods—a critical factor for populations reliant on cereal-based diets.
In food and feed products, BGS3 phytase exhibited variable but significant dephytinization efficiency. The highest reduction was observed in the phytic-acid-rich experimental diet, followed by brown bread, standard diet, and white bread. This variation may be attributed to differences in matrix composition, phytate distribution, and intrinsic inhibitors.92 Nonetheless, the substantial release of inorganic phosphorus across all products demonstrates the applicability of BGS3 phytase in both human food processing and animal nutrition. Increased mineral release through phytase supplementation has been linked to improved growth performance in poultry, fish, and mammals, reduced environmental phosphorus loading, and enhanced nutrient digestibility—all of which reinforce the utility of microbial phytases as sustainable nutritional enhancers.84,85,87
Conclusion
In summary, the application or pre-treatment of non-ruminant diets with purified microbial phytase may serve as an alternative to biofortification strategies, not only to address phosphorus deficiency but also to alleviate hidden hunger caused by micronutrient and trace element deficiencies. This is primarily achieved by modulating metabolism and physiology, leading to increased body weight, enhanced phosphate levels, and improved concentrations of micro- and trace minerals, particularly in populations consuming plant-based diets. Focusing the future studies on enzyme immobilization to enhance stability and efficacy, comparative evaluation with commercial phytases, and in vivo assessment will be critical to validate these results and to explore the commercial potential of this isolate.
Acknowledgment
The authors would like to thank Ganpat University for allowing the research laboratory space and facilities to work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Madhvi: Conceptualization, Methodology, Writing Original Draft, Data Collection, Analysis, Writing, Review & Editing.
- Hardik Shah: Supervision.
References
- Ojo MA. Phytic acid in legumes: A review of nutritional importance and hydrothermal processing effect on underutilised species. Food Res. 2021;5(3):22-28. doi:10.26656/fr.2017.5(3).325
CrossRef - Oatway L, Vasanthan T, Helm JH. Phytic acid. Food Rev Int. 2001;17(4):419-431. http://dx.doi.org/10.1081/FRI-100108531
CrossRef - Chondrou T, Adamidi N, Lygouras D, Hirota SA, Androutsos O, Svolos VS. Dietary phytic acid, dephytinization, and phytase supplementation alter trace element bioavailability: a narrative review of human interventions. Nutrients. 2024;16(23):4069. doi:10.3390/nu16234069
CrossRef - Valente Junior DT, Genova JL, Kim SW, Saraiva A, Rocha GC. Carbohydrases and phytase in poultry and pig nutrition: a review beyond the nutrients and energy matrix. Animals (Basel). 2024;14(2):226. doi:10.3390/ani14020226
CrossRef - Rizwanuddin S, Kumar V, Naik B, et al. Microbial phytase: Their sources, production, and role in the enhancement of nutritional aspects of food and feed additives. J Agric Food Res. 2023;12:100559. doi:10.1016/j.jafr.2023.100559
CrossRef - Wang R, Guo S. Phytic acid and its interactions: Contributions to protein functionality, food processing, and safety. Comprehensive Reviews in Food Science and Food Safety. 2021; 20(2):2081-105.
CrossRef - López-Moreno M, Castillo-García A, Roldán-Ruiz A, Viña I, Bertotti G, et al. Plant-based diet and risk of iron-deficiency anemia: a review of the current evidence and implications for preventive strategies. Curr Nutr Rep. 2025;14:81. doi:10.1007/s13668-025-00671-Y
CrossRef - Swaminathan S, Hemalatha R, Pandey A, et al. The burden of child and maternal malnutrition and trends in its indicators in the states of India: the Global Burden of Disease Study 1990–2017. Lancet Child Adolesc Heal. 2019;3(12):855-870. doi:10.1016/S2352-4642(19)30273-1
CrossRef - Iglesias-Puig E, Monedero V, Haros M. Bread with whole quinoa flour and bifidobacterial phytases increases dietary mineral intake and bioavailability. LWT – Food Sci Technol. 2015;60(1):71-77. doi:10.1016/J.LWT.2014.09.045
CrossRef - Sanz-Penella JM, Laparra JM, Sanz Y, Haros M. Assessment of Iron Bioavailability in Whole Wheat Bread by Addition of Phytase-Producing Bifidobacteria. J Agric Food Chem. 2012;60(12):3190-3195. doi:10.1021/JF205048R
CrossRef - Sanz-Penella JM, Laparra JM, Sanz Y, Haros M. Bread Supplemented with Amaranth (Amaranthus cruentus): Effect of Phytates on In Vitro Iron Absorption. Plant Foods Hum Nutr. 2012;67(1):50-56. doi:10.1007/S11130-011-0269-6
CrossRef - Wang H, Chen Y, Hua Y, Kong X, Zhang C. Effects of phytase-assisted processing method on physicochemical and functional properties of soy protein isolate. J Agric Food Chem. 2014;62(45):10989-10997. doi:10.1021/jf503952s
CrossRef - Kruger J, Taylor JRN, Du X, De Moura FF, Lönnerdal B, Oelofse A. Effect of phytate reduction of sorghum, through genetic modification, on iron and zinc availability as assessed by an in vitro dialysability bioaccessibility assay, Caco-2 cell uptake assay, and suckling rat pup absorption model. Food Chem. 2013;141(2):1019-1025. doi:10.1016/j.foodchem.2013.01.105
CrossRef - Yehuala TF, Karavoltsos S, Aslam MFS, et al. Effect of fermentation on the proximate composition, antinutrients, and mineral bioaccessibility of pearl millet-based injera: implications for iron and zinc availability. Int J Food Sci Technol. 2025;60(2). : org/10.1093/ijfood/vvaf132
CrossRef - Joudaki H, Aria N, Moravej R, Rezaei Yazdi M, Emami-Karvani Z, Hamblin MR. Microbial Phytases: Properties and Applications in the Food Industry. Curr Microbiol. 2023;80(12). doi:10.1007/s00284-023-03471-1
CrossRef - Sharma B, Shukla G. Isolation, identification, and characterization of phytase producing probiotic lactic acid bacteria from neonatal fecal samples having dephytinization activity. Food Biotechnol. 2020;34(2):151-171. doi:10.1080/08905436.2020.1746332
CrossRef - de Sousa JPL, Albino LFT, Vaz RGMV, et al. The effect of dietary phytase on broiler performance and digestive, bone, and blood biochemistry characteristics. Brazilian J Poult Sci. 2015;17(1):69-76. doi:10.1590/1516-635X170169-76
CrossRef - Urgessa OE, Koyamo R, Dinka H, Tefese K, Gemeda MT. Review on Desirable Microbial Phytases as a Poultry Feed Additive: Their Sources, Production, Enzymatic Evaluation, Market Size, and Regulation. Int J Microbiol.2024;1:9400374. doi:https://doi.org/10.1155/2024/9400374
CrossRef - Sacakli P, Ramay MS, Ceylan A, et al. Effects of supplemental hybrid bacterial 6-phytase in low-energy, inorganic phosphorus-free hen diets on laying performance, egg quality, and bone strength. PLoS One. 2025;20(4):e0322135. doi:10.1371/journal.pone.0322135
CrossRef - Habibollahi M, Abousadi MA, Nakhaee P. The Effect of Phytase on Production Performance, Egg Quality, Calcium and Phosphorus Excretion, and Fatty Acids and Cholesterol Concentration in Hy-Line Layers Fed Diets Containing Rice Bran. J Appl Poult Res. 2019;28(3):688-698. doi:10.3382/japr/pfz020
CrossRef - Öztürk S, Çalik P, Özdamar TH. Fed-Batch Biomolecule Production by Bacillus subtilis: A State of the Art Review. Trends Biotechnol. 2016;34(4):329-345. doi:10.1016/j.tibtech.2015.12.008
CrossRef - Chatterjee A, Mathew AM, George A, et al. Purification Strategies for Microbial Phytase. Int J Pharm Sci Res. 2020;11(1):25. http://dx.doi.org/10.13040/IJPSR.0975-8232.11
- Jain J, Sapna, Singh B. Characteristics and biotechnological applications of bacterial phytases. Process Biochem. 2016;51(2):159-169. doi:10.1016/J.procbio.2015.12.004
CrossRef - Bandari NM, Abootaleb M, Nikokar I, Karimli M. Biologically engineered probiotic supplement production containing phytase enzyme for livestock, poultry, and aquaculture consumption. J Basic Appl Zoo. 2024;85(1):41. https://doi.org/10.1186/s41936-024-00361-1
CrossRef - Negm El-Dein A, Ezzat A, Aboseif AM, Flefil NS. Enhancing micronutrients bioavailability for tilapia reared in green water system using rice bran fermented with newly isolated phytase-producer, Pediococcus pentosacious, maintained both economic and environmental sustainability. Discov Food. 2025;5(1):155. https://doi.org/10.1007/s44187-025-00434-2
CrossRef - Terrey DE, Braidi DA, Serwata R. Effect of a microbial phytase on the growth performance, digestibility and retention in a high plant meal inclusion diet for Atlantic salmon (Salmo salar). Aquac Int. 2024 ;32(3):2757-72. doi: 1007/s10499-023-01295-1
CrossRef - Lagos LV, Bedford MR, Stein HH. Amino acid and mineral digestibility, bone ash, and plasma inositol is increased by including microbial phytase in diets for growing pigs. J Anim Sci Biotechnol. 2023; 14(1):152. doi: 10.1186/s40104-023-00953-x.
CrossRef - Singh B, Kunze G, Satyanarayana T. Developments in biochemical aspects and biotechnological applications of microbial phytases. Biotechnol Mol Biol Rev. 2011;63(3):69-87.
- Madhvi, Dhavni P and Hardik S. Optimization of physical parameters for the production of Phytase from isolate BGS3 (Bacillus ) isolated from North Gujarat region. Adv Biores. 2024;15(6): 416-421. doi: 10.15515/abr.0976-4585.15.6.416421
- Madhvi, Shah H. Screening and molecular characterization of phytase producer from north Gujarat region. J of Appl Biol Biotechnol. 2024;12(3):146-152. doi: 10.7324/JABB.2024.172285
CrossRef - Lowry O, Schagger H, Cramer WA, Vonjagow G. Protein Measurement With the Folin Phenol Reagent. Anal Biochem. 1994;217(2):220-230. doi:10.1016/S0021-9258(19)52451-6
CrossRef - Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227(5259):680-685. doi:10.1038/227680a0
CrossRef - Wieser A, Schneider L, Jung J, Schubert S. MALDI-TOF MS in microbiological diagnostics-identification of microorganisms and beyond (mini review). Appl Microbiol Biotechnol. 2012;93(3):965-974. doi:10.1007/s00253-011-3783-4
CrossRef - Powar VK, Jagannathan V. Purification and properties of phytate-specific phosphatase from Bacillus subtilis. J Bacteriol. 1982;151(3):1102. doi:10.1128/jb.151.3.1102-1108.1982
CrossRef - Fiske CH, Subbarow Y. the Colorimetric Determination of Phosphorus. J Biol Chem. 1925;66(2):375-400. doi:10.1016/s0021-9258(18)84756-1
CrossRef - Yemm EW, Cocking EC, Ricketts RE. The determination of amino-acids with ninhydrin. Analyst. 1955;80(948):209-214. doi:10.1039/AN9558000209
CrossRef - McKie VA, MccleAry Ba V. Food composition and additives:A Novel and Rapid Colorimetric Method for Measuring Total Phosphorus and Phytic Acid in Foods and Animal Feeds. J aoac Int. 2016;99(3). doi:10.5740/jaoacint.16-0029
CrossRef - Kumari N, Bansal S. Statistical modeling and optimization of microbial phytase production towards utilization as a feed supplement. Biomass Convers Biorefinery. 2023;13(9):8339-8349. doi:10.1007/s13399-021-01672-x
CrossRef - Yaver E, Bilgiçli N. Effects of different dephytinisation methods on chemical properties of commercial and traditional breads prepared from composite flour. Food Chem. 2019;276:77-83. doi:10.1016/J.foodchem.2018.09.169
CrossRef - Fasimoye FO, Olajuyigbe FM, Sanni MD. Purification and characterization of a thermostable extracellular phytase from Bacillus licheniformis PFBL-03. Prep Biochem Biotechnol. 2014;44(2):193-205. doi:10.1080/10826068.2013.812565
CrossRef - Sanni DM, Jimoh MB, Lawal OT, Bamidele SO. Purification and biochemical characterization of phytase from Bacillus cereus isolated from gastrointestinal tract of African giant snail (Achatina fulica). Int Microbiol. 2023;26(4):961-972. doi: 10.1007/s10123-023-00350-4
CrossRef - Yu P, Chen Y. Purification and characterization of a novel neutral and heat-tolerant phytase from a newly isolated strain Bacillus nealsonii BMC biotechnology. 2013;13:1-7.
CrossRef - Luo HY, Shi PJ, Li J, Wang YR, Yao B. Purification and properties of Citrobacter freundii Wei Sheng Wu Xue Bao. 2006; 46(1):139-42.
- Trivedi S, Husain I, Sharma A. Purification and characterization of phytase from Bacillus subtilis P6 : Evaluation for probiotic potential for possible application in animal feed. 2022;3:194-205. doi:10.1002/fft2.118
CrossRef - Dan SK, Nandi A, Banerjee G, Ghosh P, Ray AK. Purification and Characterization of Extracellular Phytase from Bacillus licheniformis Isolated from Fish Gut. Proc Natl Acad Sci India Sect B – Biol Sci. 2015;85(3):751-758. doi:10.1007/s40011-015-0571-4
CrossRef - Demirkan E, Sevgi T, Akcakoca D, Ersoy F. Partial purification, characterization and wheat bran degradation studies of a new phytase from the Bacillus megaterium EBD 9-1 strain. Turkish Journal of Biochemistry. 2017; 42(3):329-37. doi:10.1515/tjb-2016-0265
CrossRef - Kim YO, Kim HK, Bae KS, Yu JH, Oh TK. Purification and properties of a thermostable phytase from Bacillus DS11. Enzyme Microb Technol. 1998;22(1):2-7. doi:https://doi.org/10.1016/S0141-0229(97)00096-3
CrossRef - Choi YM, Suh HJ, Kim JM. Purification and properties of extracellular phytase from Bacillus KHU-10. J Protein Chem. 2001;20(4):287-292. doi:10.1023/A:1010945416862
CrossRef - Tran TT, Hashim SO, Gaber Y, Mamo G, Mattiasson B, Hatti-Kaul R. Thermostable alkaline phytase from Bacillus MD2: Effect of divalent metals on activity and stability. J Inorg Biochem. 2011;105(7):1000-1007. doi:10.1016/J.Jinorgbio.2011.04.005
CrossRef - Hong SW, Chu IH, Chung KS. Purification and biochemical characterization of thermostable phytase from newly isolated Bacillus subtilis J Appl Biol Chem. 2011;54(1):89-94. doi:10.3839/jksabc.2011.012
CrossRef - Escobin-Mopera L, Ohtani M, Sekiguchi S, et al. Purification and characterization of phytase from Klebsiella pneumoniae 9-3B. J Biosci Bioeng. 2012;113(5):562-567. doi:10.1016/j.jbiosc.2011.12.010
CrossRef - Shimizu M. Purification and Characterization of Phytase from Bacillus subtilis ( natto ) N – 77. Biosc Biotechnol and Biochem.1992; 56(8):1266–1269. https://doi.org/10.1271/bbb.56.126
- El-toukhy NMK, Youssef AS, Mikhail MGM. Isolation , purification and characterization of phytase from Bacillus subtilis Afr J Biotechnol. 2013;12(20). doi:10.5897/AJB2013.12304
- Parhamfar M, Badoei-dalfard A, Khaleghi M, Hassanshahian M. Purification and characterization of an acidic , thermophilic phytase from a newly isolated Geobacillus stearothermophilus strain DM12. Prog Biol Sci. 2015;5(1):61-73.
- Patki JM, Singh S, Mehta S. Partial purification and characterization of phytase from bacteria inhabiting the mangroves of the western coast of India. Inter J of Curr Microbiol and Appl Sci. 2015; 4(9):156-69.
- Abdolshahi A, Yancheshmeh BS, Arabameri M, Marvdashti LM. Phytase From Bacillus strain LA12: Isolation, Purification and Characterization. J Microbiol Biotechnol Food Sci. 2021;10(4):572-576. doi:10.15414/jmbfs.2021.10.4.572-576
CrossRef - Neira-Vielma AA, Iliná A, Álvarez GM, et al. Recovery and purification of Aspergillus niger phytase from crude extract using AOT / isooctane reversed micelles. Biotechnol Reports. 2020;26. doi:10.1016/j.btre.2020.e00471
CrossRef - Greiner R, Konietzny U, Jany KD. Purification and Characterization of Two Phytases from Escherichia coli. Arch Biochem Biophys. 1993;303(1):107-113. doi:10.1006/ABBI.1993.1261
CrossRef - Suryani AE, Istiqomah L, Anggraeni AS, Windarsih AW. Phytase Production by Lactobacillus plantarum A1-E in Submerged and Solid-State Fermentation. agriTECH. 2023; 43(4):335-43. doi: 22146/agritech.74761
CrossRef - Danial EN, Alkhalf MI. Purification and characterization of phytase from novel slated Bacillus cereus eme 48 and study its kinetic properties. J Pure Appl Microbiol. 2016;10(4):2521-2529. doi:10.22207/JPAM.10.4.06
CrossRef - Lan GQ, Abdullah N, Jalaludin S, Ho YW. Purification and characterization of a phytase from Mitsuokella jalaludinii, a bovine rumen bacterium. African J Biotechnol. 2011;10(59):12766-12776. doi:10.5897/ajb11.294
CrossRef - Yanke LJ, Selinger LB, Cheng KJ. Phytase activity of Selenomonas ruminantium: a preliminary characterization. Lett in Appl Microbiol. 1999; 29(1):20-5. doi:10.1046/j.1365-2672.1999.00568.x
CrossRef - Kalsi HK, Singh R, Dhaliwal HS, Kumar V. Phytases from Enterobacter and Serratia species with desirable characteristics for food and feed applications. 3 Biotech. 2016;6(1):1-13. doi:10.1007/s13205-016-0378-x
CrossRef - Rocky-Salimi K, Hashemi M, Safari M, Mousivand M. A novel phytase characterized by thermostability and high pH tolerance from rice phyllosphere isolated Bacillus subtilis S.46. J Adv Res. 2016;7(3):381-390. doi:10.1016/j.jare.2016.02.003
CrossRef - Jorquera MA, Gabler S, Inostroza NG, et al. Screening and Characterization of Phytases from Bacteria Isolated from Chilean Hydrothermal Environments. Microb Ecol. 2018;75(2):387-399. doi:10.1007/s00248-017-1057-0
CrossRef - Vats P, Banerjee UC. Biochemical characterisation of extracellular phytase (myo-inositol hexakisphosphate phosphohydrolase) from a hyper-producing strain of Aspergillus niger van Teighem. J Ind Microbiol Biotechnol. 2005;32(4):141-147. doi:10.1007/s10295-005-0214-5
CrossRef - Wang R, Guo S. Phytic acid and its interactions: Contributions to protein functionality, food processing, and safety. Compr Rev Food Sci Food Saf. 2021;20(2):2081-2105. doi:10.1111/1541-4337.12714
CrossRef - Jain J, Kumar A, Singh D, Singh B. Purification and kinetics of a protease-resistant, neutral, and thermostable phytase from Bacillus subtilis subtilis JJBS250 ameliorating food nutrition. Prep Biochem Biotechnol. 2018;48(8):718-724. doi:10.1080/10826068.2018.1487848
CrossRef - Zhang GQ, Dong XF, Wang ZH, Zhang Q, Wang HX, Tong JM. Purification, characterization, and cloning of a novel phytase with low pH optimum and strong proteolysis resistance from Aspergillus ficuum NTG-23. Bioresour Technol. 2010;101(11):4125-4131. doi:10.1016/j.biortech.2010.01.001
CrossRef - Badoei-Dalfard A, Parhamfar M, Karami Z. Characterization of a thermostable, acidic-phytase from Bacillus tequilensis Dm018; medium optimization by response surface methodology. Catal Letters. 2019;149:2961-2972. Doi: 10.1007/s10562-019-02881-w
CrossRef - Rao BD, Bhaskarachary K, Christina GD, Devi SG, Vilas AT, Tonapi A. Nutritional and health benefits of millets. ICAR_ Indian Institute of Millets Research (IIMR) Rajendranagar, Hyderabad. 2017;2.
- Shivanna GB, Venkateswaran G. Dephytinization of Cereals and Pulses by Phytase Producing Lactic Acid Bacteria. Int J Curr Res Acad Rev. 2015;3(12):61-69
- Bala A, Sapna, Jain J, Kumari A, Singh B. Production of an extracellular phytase from a thermophilic mould Humicola nigrescens in solid state fermentation and its application in dephytinization. Biocatal Agric Biotechnol. 2014;3(4):259-264. doi:10.1016/j.bcab.2014.07.002
CrossRef - Soni SK, Sarkar S, Selvakannan PR, Sarkar D, Bhargava SK. Intrinsic therapeutic and biocatalytic roles of ionic liquid mediated self-assembled platinum–phytase nanospheres. RSC Adv. 2015;5(77):62871-62881. doi:10.1039/C5RA11273G
CrossRef - Ghaly MF, Ezzat SM, Othman AM. Purification and characterization of phytase enzyme extracted from Aspergillus niger SF58 and its application in cereal dephytinization. Trop J Nat Prod Res. 2021;5(2):265-271. doi:10.26538/tjnpr/v5i2.9
CrossRef - Gupta RK, Gangoliya SS, Singh NK. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J Food Sci Technol. 2015;52(2):676-684. doi:10.1007/s13197-013-0978-y
CrossRef - Chondrou T, Adamidi N, Lygouras D, Hirota SA, Androutsos O, Svolos V. Dietary Phytic Acid, Dephytinization, and Phytase Supplementation Alter Trace Element Bioavailability—A Narrative Review of Human Interventions. Nutr . 2024;16(23):1-14. doi:10.3390/nu16234069
CrossRef - Olukosi OA, Fru-Nji F. The interplay of dietary nutrient specification and varying calcium to total phosphorus ratio on efficacy of a bacterial phytase: Growth performance and tibia mineralization. Poult Sci. 2014;93(12):3037-3043. doi:10.3382/ps.2014-03978
CrossRef - Ribeiro V, Salguero SC, Gomes G, et al. Efficacy and phosphorus equivalency values of two bacterial phytases (Escherichia coli and Citrobacter braakii) allow the partial reduction of dicalcium phosphate added to the diets of broiler chickens from 1 to 21 days of age. Anim Feed Sci Technol. 2016;221:226-233.
CrossRef - Sharma R, Kumar P, Kaushal V, Das R, Kumar Navani N. A novel protein tyrosine phosphatase like phytase from Lactobacillus fermentum NKN51: Cloning, characterization and application in mineral release for food technology applications. Bioresour Technol. 2018;249:1000-1008. doi:10.1016/j.biortech.2017.10.106
CrossRef - Wu G, Bazer FW. Application of new biotechnologies for improvements in swine nutrition and pork production. J Anim Sci Biotechnol. 2019;10(1):1-16. doi:10.1186/s40104-019-0337-6
CrossRef - Sun W, Ali I, Liu J, et al. Isolation, identification, and characterization of diesel-oil-degrading bacterial strains indigenous to Changqing oil field, China. J Basic Microbiol. 2019;59(7):723-734. doi:10.1002/jobm.201800674
CrossRef - Valente Junior DT, Genova JL, Kim SW, Saraiva A, Rocha GC. Carbohydrases and phytase in poultry and pig nutrition: a review beyond the nutrients and energy matrix. 2024;14(2):226. doi:10.3390/ani14020226
CrossRef - Koréissi-Dembélé Y, Fanou-Fogny N, Moretti D, et al. Dephytinisation with Intrinsic Wheat Phytase and Iron Fortification Significantly Increase Iron Absorption from Fonio (Digitaria exilis) Meals in West African Women. PLoS One. 2013;8(10). doi:10.1371/journal.pone.0070613
CrossRef - Alçiçek A, Bozkurt M, Çabuk M. The effect of a mixture of herbal essential oils, an organic acid or a probiotic on broiler performance. S Afr J Anim Sci. 2004;34(4):217-222.
- Ahmad T, Rasool S, Sarwar M, Haq AU, Hasan ZU. Effect of microbial phytase produced from a fungus Aspergillus niger on bioavailability of phosphorus and calcium in broiler chickens. Anim Feed Sci Technol. 2000;83(2):103-114. doi:10.1016/S0377-8401(99)00122-4
CrossRef - Baruah K, Sahu NP, Pal AK, Debnath D, Yengkokpam S, Mukherjee SC. Interactions of dietary microbial phytase, citric acid and crude protein level on mineral utilization by Rohu, Labeo rohita (Hamilton), juveniles. J World Aquac Soc. 2007;38(2):238-249. doi:10.1111/j.1749-7345.2007.00092.x
CrossRef - Jin L, Zhang Y, Chen Q, et al. Phytic acid inhibits tumor growth and invasion by suppressing key oncogenic pathways in experimental models. Food Biosci. 2024;60: 104194. doi:10.1016/j.fbio.2024.104194
CrossRef - Takemori N, Takemori A. In-depth structural proteomics integrating mass spectrometry and polyacrylamide gel electrophoresis. Front Anal Sci. 2023;2:1107183. doi:10.3389/frans.2022.1107183
CrossRef - Mishra U, Singh P, Verma A, et al. Classification and functional diversity of phytase enzymes: acid and alkaline phytases with distinct catalytic mechanisms. Int J Curr Microbiol Appl Sci. 2024;13(4).
- Ahmad R, Kumar V. Microbial phytases: properties, production methods, and industrial applications. Extremophiles. 2023;27(6):1–15. doi:10.1007/s00792-023-01374-
- Żyła K, Duda A. Towards Improved Bioavailability of Cereal Inositol Phosphates, Myo-Inositol and Phenolic Acids. Molecules. 2025; 30(3):652. https://doi.org/10.3390/molecules30030652
CrossRef
Accepted on: 18-12-2025
Second Review by: Dr. Dereba Workineh
Final Approval by: Dr. Eugene A. Silow










