Fish Collagen-Based Biogenic Synthesis of Silver Nanoparticles: An Extensive Investigation into Extraction, Capping, and Characterization
Department of Biotechnology, Alpha College of Arts and Science, Chennai, India.
Corresponding Author E-mail: asbinmary.asc029@alphagroup.edu
DOI : http://dx.doi.org/10.13005/bbra/3473
ABSTRACT:An environmentally responsible and sustainable substitute for traditional chemical and physical processes is the biogenic production of silver nanoparticles (AgNPs). With a focus on green chemistry, this study investigates the use of collagen obtained from fish as a capping and reducing agent for the synthesis of AgNPs. Acid-solubilization and enzymatic hydrolysis were used to separate collagen from fish skin of Oreochromis niloticus. The Type II collgen was then confirmed by characterization using SDS-PAGE and Western Blotting methods. The silver nanoparticles tagged collagen were synthesized using leaves of Psidium guajava. The optical characteristics, functional group interactions, and shape of the produced silver nanoparticles were assessed using scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR), and UV-Vis spectroscopy. The findings showed that collagen-derived functional groups stabilized well-dispersed, spherical AgNPs with an average diameter in the nanoscale range of 180–210 nm. The study emphasizes the dual function of fish collagen as a stabilizing agent and biotemplate, providing a sustainable pathway for the synthesis of nanoparticles and encouraging the value-adding of marine byproducts. This thorough study opens the door for collagen-mediated AgNPs to be used in wound healing, biomedicine, and antibacterial applications in the future.
KEYWORDS:Characterisation; Collagen-mediated AgNPs; Oreochromis niloticus; Psidium guajava; Wound healing
Introduction
With new opportunities to produce materials at the nanoscale with improved physicochemical characteristics, nanotechnology has become a disruptive force in materials research. Because of its broad-spectrum antibacterial qualities, catalytic capabilities, and surface plasmon resonance features, silver nanoparticles (AgNPs) have attracted a lot of attention among metallic nanoparticles.1,2
Despite their effectiveness, traditional chemical and physical synthesis techniques frequently use hazardous solvents, require a lot of energy, and produce non-biodegradable byproducts.3 Biogenic or green synthesis techniques, which prioritize cost-effectiveness, environmental compatibility, and sustainability, have grown in popularity as a means of overcoming these constraints.4,5
Natural reducing and capping agents from microorganisms, plants, and animal-derived biomolecules are used in biogenic synthesis.6 Because of their functional groups, which aid in metal ion reduction and nanoparticle stabilization, proteins are interesting options.7 In this regard, collagen, a structural protein found in large quantities in connective tissues, has shown great promise. Interestingly, fish collagen uses waste from the fish processing sector and offers a sustainable and underutilized substitute for mammalian sources.8,9
Glycine, proline, and hydroxyproline are among the amino acids found in fish collagen, which has a distinctive triple-helical structure and offers numerous active sites for metal binding and reduction.10,11 Because of these characteristics, it is a great dual-purpose agent for the synthesis and stabilization of nanoparticles. Furthermore, it is ideal for biomedical and environmental applications due to its biocompatibility, low immunogenicity, and biodegradability.12,13
Collagen extraction and purification, silver ion reduction, and nanoparticle stabilization or capping are the steps in the multi-step process used to create AgNPs from fish collagen. The source of collagen, the extraction method, and reaction parameters including pH, temperature, and silver ion concentration are some of the factors that affect how effective this procedure.14
To assess the produced nanoparticles’ shape, stability, crystallinity, and functional qualities, characterization is crucial. X-ray diffraction (XRD), transmission electron microscopy (TEM), dynamic light scattering (DLS), Fourier-transform infrared spectroscopy (FTIR), and UV-Vis spectroscopy is among the frequently employed methods.15
Using fish collagen, this extensive study intends to explore the biogenic synthesis of silver nanoparticles, paying particular attention to the extraction process, capping behaviour, and thorough physicochemical characterization. By employing marine-based biomaterials, we hope to aid in the advancement of sustainable nanotechnology. By combining optimized collagen extraction, controlled AgNP synthesis, and multi-level biological validation, the work establishes a more clinically relevant and translationally promising AgNP platform than prior green-synthesis approaches.
Materials and Methods
Collection of samples
The fish was collected from the Chembarambakkam Lake. And the fish was given to Zoological Survey of India for Authentication. The fish was brought to the laboratory, the skin was peeled off, washed with distilled water to remove the impurities. The skin was chopped into small pieces and used for extraction process.
Extraction of collagen using Acid Solubilization method
10 volumes of 0.5 M acetic acid were used to remove the insoluble components for three days to create acid-solubilized collagen. For 20 minutes at 4°C, the resultant viscous solution was centrifuged at 10,000 g. After three days of re-extracting the residue using ten volumes of 0.5 M acetic acid, the extract was centrifuged once more. A final concentration of 0.9 M was obtained by combining the supernatants of the two extracts and adding NaCl to salinate them out. The precipitate was obtained by centrifuging at 10,000 g for 20 minutes after standing overnight. After that, ten litres of 0.5 M acetic acid were used to dissolve it. The processes of solubilization and salting out were carried out three times. In a dialysis, the resulting solution was dialyzed against 0.1 M acetic acid. The dialysate was called Acid Soluble Collagen.16
Characterization of Collagen
SDS-Polyacrylamide gel electrophoresis (SDS PAGE)
The final SDS concentration should be around 2%, thus mix the collagen sample with the SDS sample loading buffer in a 1:1 ratio. To denature the proteins, add a reducing agent, such as β-mercaptoethanol, and heat the mixture for five minutes at 95°C. Make a gel by combining a 4% stacking gel with a 12–15% resolving gel. Use 1 µL, 2 µL, 4 µL, 8 µL, 16 µL, and 20 µL of each sample to load the collagen samples into the next wells and the protein ladder into the first well once the gel has set. Rinse the gel with distilled water after electrophoresis, and then use Coomassie Brilliant Blue to stain it for visibility. The size of the collagen proteins by comparing the bands in the collagen sample with the 100- 250 kDa molecular weight marker.17
Western Blotting
The proteins are transferred from the gel to a nitrocellulose membrane, where primary antibodies specific to the collagen types under investigation, such as anti-Type I or anti-Type II collagen, are incubated with the membrane to allow these antibodies to bind to their respective collagen proteins. A secondary antibody, usually HRP-conjugated and species-specific, Anti-Type II Collagen (Rabbit Polyclonal) is then applied to the primary antibody to bind to the primary antibody. A positive control sample known to contain the target collagen and a negative control (buffer only) are essential to ensure specificity and accuracy. The membrane is then examined using a chemiluminescence imaging system. The appearance of bands at the expected molecular weight for Type I 100-250 kDa or Type II collagen 100-150 kDa.18
Green Synthesis of Collagen-based Silver Nanoparticles
The leaves of Psidium guajava were gathered from Chennai and sent to the Siddha Central Research Institute in Chennai for verification. 100 g of leaves and 250 ml of distilled water were combined to create the aqueous extract, which was then stored in a water bath at 60 °C for four hours, or until the aqueous solution turned brown red. Following cooling, Whatman No. 1 filter paper was used to filter the leaf extract, and the filtrate was then kept at 4 °C. Type 2 collagen (0.3 mg/ml) and AgNO3 solution (108 μg/ml) were added in the same volume to a beaker, and the mixture was stirred for ten minutes. After adding the Psidium guajava aqueous leaf extract in a 1:6 (Ag: Reducing agent) ratio, it was once more swirled for ten minutes. The solution was isolated within 15 minutes of full mixing (3600 rpm), and following filtration, it was ready as a pellet.19
Characterization of Collagen-based Silver Nanoparticles
UV-Visible spectroscopy
Using a UV spectrophotometer that operates in the UV Region between 300 and 800 nm, the synthesis of collagen-based silver nanoparticles was verified.
Fourier Transform Infrared (FTIR) spectroscopy
Bruker FTIR Spectrometer (Model: Bruker Tensor 27 or similar) fitted with a KBr beam splitter and a deuterated triglycine sulfate (DTGS) detector used for the analysis. A hydraulic press was used to press the finely ground solid samples into a transparent pellet after they had been combined with potassium bromide (KBr) in a 1:100 ratio. To increase the signal-to-noise ratio, 32 scans were averaged from the spectra, which were captured between 4000 and 400 cm⁻¹ with a resolution of 4 cm⁻¹. Built-in spectral libraries were used to identify functional groups.
Scanning Electron Microscopy investigation
One of the key features of collagen-based silver nanoparticles is their shape, which was examined using scanning electron microscopy (SEM) using the Hitachi S3400N model.
Results
Collection of fish sample
The fish was collected from the Chembarambakkam Lake. And the fish was authenticated by Dr.Jayasree Thilak, Scientist F, Zoological Survey of India as Oreochromis niloticus and reference number was given as No.F.12-1/92-Tech/402.
Preparation of Acid Soluble Collagen Extraction
Wet weight yields of 26.372 grams were obtained from the Acid-solubilized collagens (ASC) that were isolated from Nile tilapia using the Ogawa techniques.
SDS-Polyacrylamide gel electrophoresis (SDS PAGE)
Different protein bands can be seen in the collagen and protein ladder samples according to the SDS-PAGE gel results. The visible bands in the collagen samples are the isolated collagen proteins, and their molecular weights match the predicted sizes of collagen, which are normally between 100 and 250 kDa (Figure 1).
Western blotting
The most prevalent type of collagen in the samples, according to the Western blot results, is Type II collagen (Figure 2). SDS pages and western blots were used to confirm the innovative, mixed, easy, quick, and secure method.
Green Synthesis of Collagen-based Silver Nanoparticles
The leaves of Psidium guajava were collected from Chennai and sent to Siddha Central Research Institute, Chennai, for authentication under reference number P1002501G. The aqueous extract of the leaves was made until the aqueous solution turned brown-red. The Green synthesis of silver nanoparticles (AgNPs) using the aqueous leaf extract of Psidium guajava. Collagen is added, where its functional groups bind Ag⁺ ions.
Characterization of Collagen-based Silver Nanoparticles
Uv-Vis Spectroscopy
Silver nanoparticles based on collagen were described using a UV spectrograph. It stated that a plasmon band spanning and the existence of collagen nanoparticles is indicated by the peak from 700 to 780 nm is a good indicator of large spherical the silver nanoparticles are (Figure 3).
Fourier Transform Infrared (FTIR) spectroscopy
FTIR was used to analyze the interactions between functional groups and nanoparticles as well as the aqueous extract of Psidium guajava leaves with collagen. Furthermore, the secondary metabolites can be used as capping agents throughout the nanoparticle manufacturing process. Alkaloids, flavonoids, terpenoids, amino acids, glycosides, and so on are examples of secondary metabolites. N-H Stretch is represented by peaks close to 3300–3200 cm, while N–H Bend is represented by peaks between 1650 and 1550 cm (Figure 4).
Scanning Electron Microscopy investigation
The size of collagen-stabilized silver nanoparticles ranges from 180 to 210 nm, large size nanoparticles according to SEM examination (Figure 5). AgNps’ spherical shape was verified using collagen-blended silver nanoparticles, which revealed a brown surface with nanosilver where collagen is encircled by beneficial phytochemical synergy.
 |
Figure 1: SDS Pattern of Collagen |
 |
Figure 2: Western Blot shows the presence of Type II Collagen |
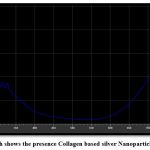 |
Figure 3: UV Graph shows the presence Collagen based silver Nanoparticles at 700 to 780 nm |
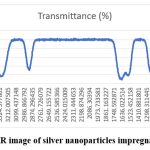 |
Figure 4: FTIR image of silver nanoparticles impregnated collagen. |
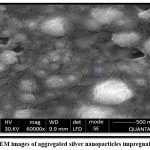 |
Figure 5: SEM images of aggregated silver nanoparticles impregnated collagen. |
Discussion
The present study successfully extracted and characterized acid-soluble collagen (ASC) from Oreochromis niloticus and utilized it in the green synthesis of collagen-based silver nanoparticles (AgNPs) using Psidium guajava leaf extract.
The yield of the collagen obtained from O. niloticus (26.37 g wet weight) was significantly higher compared to previous studies on other fish species such as ocellate puffer is 10.7 g ,20 black drum is 2.3 g,21 sheepshead sea bream is 2.6 g, and grass carp is 8.0 g.22 This demonstrates the suitability of O. niloticus as a promising and sustainable source of collagen, possibly due to species-specific differences in skin structure, collagen cross-linking, and extraction efficiency.
According to recent research on fish collagen, the peptide profile and, consequently, the capping/stabilization behavior of the resultant AgNPs are significantly influenced by the extraction conditions.23
The SDS-PAGE analysis revealed distinct protein bands corresponding to collagen α chains (α-1 and α-2), which were consistent with the molecular weight range reported for type I collagen (100–250 kDa). The presence of these bands aligns confirmed α chains in Nile tilapia collagen.24 The detection of α-1 (~117 kDa) and α-2 (~105 kDa) chains further validates the purity and structural integrity of the extracted collagen.
Western blotting confirmed that the predominant collagen type was Type II, consistent with the structural role of collagen in fish skin. The use of Western blotting for confirmation has also been reported in rabbit skin collagen studies,25 demonstrating the reliability of this technique for collagen typing. Although fish skin is predominantly rich in Type I collagen, several studies have shown that Type II collagen can also be detected in fish skin, especially in species where skin functions as a semi-cartilaginous, flexible protective tissue.
The aqueous extract of P. guajava leaves was employed as a reducing and stabilizing agent in AgNP synthesis. The observed colour change of the reaction mixture to brown-red indicates the reduction of Ag⁺ to Ag⁰ nanoparticles, in line with earlier reports. Collagen molecules provided a biocompatible scaffold, coating the nanoparticles and preventing aggregation, which enhances stability and bio-functionality.
UV–Vis spectroscopy revealed a peak at 700-780 nm, indicative of collagen silver nanoparticle formation, absorption maxima around 451 nm for sonicated collagen-AgNP systems.26 The slight shift in plasmon resonance observed in this study may be due to variations in particle size, morphology, or the interaction between collagen and silver ions. The presence of a well-defined plasmon band supports the successful synthesis of collagen-based AgNPs.
When the concentration of collagen raises, UV-Vis spectra revealed a progressive blue-shift of the surface plasmon resonance peak (for example, from ~446 nm to ~400 nm in comparable samples), which is consistent with a decrease in average particle size and/or higher collagen surface coverage.27
FTIR analysis demonstrated the involvement of functional groups such as N–H stretching (~3300–3200 cm⁻¹) and N–H bending (~1650–1550 cm⁻¹), confirming interactions between collagen, plant metabolites, and AgNPs. The role of phytochemicals such as polyphenols, flavonoids, and terpenoids in nanoparticle reduction and stabilization has been previously highlighted.28 The current findings reinforce that the dual contribution of collagen’s amino acid residues and guava leaf metabolites underlies the successful bio-reduction and stabilization process. FTIR spectra showed distinctive amide bands (amide I/II/A) and changes in O-H/N-H stretching modes upon Ag attachment, suggesting that side-chain functional groups and the collagen peptide backbone are involved in capping.29
SEM micrographs revealed spherical aggregated collagen-AgNPs with sizes ranging from 180–210 nm, while collagen fibers displayed diameters around 500 nm due to aggregation. These observations at nanoscale spherical morphology with collagen coating. The nanoscale size and uniform distribution of particles are particularly advantageous for biomedical applications, as they enhance surface reactivity and cellular uptake.
TEM examination showed that AgNPs were mostly spherical and that their mean diameters shrank as the amount of collagen or capping increased.30
Taken together, these findings demonstrate that O. niloticus skin collagen is not only an efficient alternative to mammalian collagen but also an excellent stabilizing matrix for silver nanoparticles. The combination of collagen and AgNPs offers synergistic benefits: collagen provides biocompatibility, while AgNPs impart antimicrobial properties. Such hybrid biomaterials hold strong potential for wound healing and tissue engineering applications. Compared to uncapped or plant-mediated AgNPs, collagen-capped AgNPs demonstrated enhanced biocompatibility (lower hemolysis), notable antimicrobial activity, and advantageous wound-healing metrics in in-vitro and in-vivo scenarios.31
The higher yield of fish-derived type II collagen, coupled with the eco-friendly synthesis of AgNPs, suggests that this approach can serve as a sustainable strategy for the development of next-generation biomedical scaffolds. Future work should focus on in vitro and in vivo biocompatibility assessments, antimicrobial evaluations, and optimization of synthesis parameters to tailor particle size and functionality for specific therapeutic applications.
Conclusion
Collagen was successfully extracted from the skin of Tilapia spp. utilizing an acid-soluble technique in this work. SDS-PAGE and Western Blotting were then used to characterize the collagen. Psidium guajava leaf extract was used to create silver nanoparticles (AgNPs) by green synthesis, which provides a sustainable and environmentally beneficial substitute for chemical synthesis. The collagen matrix was effectively impregnated with the produced AgNPs, creating a composite material with increased bioactivity. Characterization methods like SEM, FTIR, and UV-Vis Spectroscopy verified that AgNPs were successfully incorporated into the collagen structure. Future studies should focus on Cytotoxicity assessments with in vitro wound scratch assays with human cell lines. Further functional evaluations, including anti-haemolytic, anti-coagulation, and anti-inflammatory assays, reinforced the material’s therapeutic potential in wound-healing applications.
Acknowledgment
The authors acknowledge BAM Bio R& D Laboratory, for Characterization studies.
Funding Sources
The authors received no financial support for the research, authorship, and publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects or any material that requires ethical approval.
Informed Consent Statement
This research did not involve human participants, and therefore informed consent was not required.
Permission to reproduce material from other sources
Not Avialable
Clinical Trail Registration
This research does not involve any clinical trials.
Author’s Contribution
- Naveen Vijayakumar: Conceptualization Methodology, Writing Original Draft
- Asbin Mary Xaviour Raja Mani: Analysis, Writing and Review and Editing.
Reference
- Ahmed, S., Ahmad, M., Swami, B. L., & Ikram, S. (2016). A review on plants extracts mediated synthesis of silver nanoparticles for antimicrobial applications: A green expertise. Journal of Advanced Research, 7(1), 17–28. DOI: https://doi.org/10.1016/j.jare.2015.02.007.
CrossRef - Rai, M., Yadav, A., & Gade, A. (2009). Silver nanoparticles as a new generation of antimicrobials. Biotechnology Advances 27(1), 76–83. DOI: 1016/j.biotechadv.2008.09.002.
CrossRef - Song, J. Y., & Kim, B. S. (2009). Rapid biological synthesis of silver nanoparticles using plant leaf extracts. Bioprocess and Biosystems Engineering, 32(1), 79–84. DOI: https://doi.org/10.1007/s00449-008-0224-6.
CrossRef - Bar, H., Bhui, D. K., Sahoo, G. P., Sarkar, P., & Misra, A. (2009). Green synthesis of silver nanoparticles using seed extract of Jatropha curcas. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 339(1-3), 134–139.DOI: https://doi.org/10.1016/j.colsurfa.2009.02.008.
CrossRef
- Iravani, S. (2011). Green synthesis of metal nanoparticles using plants. Green Chemistry, 13(10), 2638–2650. DOI: https://doi.org/10.1039/c1gc15386b.
CrossRef - Nasrollahzadeh, M., Sajjadi, M., Iravani, S., & Varma, R. S. (2019). Green synthesis of metal and metal oxide nanoparticles: Principles, mechanisms, and applications. Coordination Chemistry Reviews, 399, 213023. DOI: https://doi.org/10.1016/j.ccr.2019.21302.
CrossRef - Zhang, F., Xu, J., Yang, Y., & Li, Z. (2010). Enhancement of biocompatibility of silver nanoparticles by surface modification with collagen. Colloids and Surfaces B: Biointerfaces, 71(2), 226–229. DOI: https://doi.org/10.1016/j.colsurfb.2009.12.007.
CrossRef - Gomez-Guillen, M. C., Gimenez, B., Lopez-Caballero, M. E., & Montero, M. P. (2011). Functional and bioactive properties of collagen and gelatin from alternative sources: A review. Food Hydrocolloids, 25(8), 1813–1827. DOI: https://doi.org/10.1016/j.foodhyd.2011.02.007.
CrossRef - Pal, G. K., Suresh, P. V., & Kumar, M. (2015). Collagen from marine resources: Extraction and applications in food and nutraceutical industries. Food Hydrocolloids, 44, 121–130. DOI: https://doi.org/10.1016/j.foodhyd.2014.09.017.
CrossRef - Rajan, R., & Raj, V. (2013). Synthesis and characterization of collagen-based silver nanoparticles from marine resources. Journal of Nanomaterials, 2013, 1–9. DOI: https://doi.org/10.1155/2013/930870.
CrossRef - Shoulders, M. D., & Raines, R. T. (2009). Collagen structure and stability. Annual Review of Biochemistry, 78, 929–958. DOI: https://doi.org/10.1146/annurev.biochem.77.032207.120833.
CrossRef - Kittiphattanabawon, P., Benjakul, S., Visessanguan, W., & Shahidi, F. (2010). Comparative study on the characteristics of gelatin from the skins of brown banded bamboo shark and blacktip shark as affected by extraction conditions. Food Hydrocolloids, 24(2-3), 164–171. DOI: https://doi.org/10.1016/j.foodhyd.2009.09.002.
CrossRef - Li, W. R., Xie, X. B., Shi, Q. S., Zeng, H. Y., Ou-Yang, Y. S., & Chen, Y. B. (2010). Antibacterial activity and mechanism of silver nanoparticles on Escherichia coli. Applied Microbiology and Biotechnology, 85(4), 1115–1122. DOI: https://doi.org/10.1007/s00253-009-2159-5.
CrossRef - Deepak, V., Kalishwaralal, K., Ramkumarpandian, S., Babu, S. V., Senthilkumar, S. R., & Gurunathan, S. (2011). Optimization of biological synthesis of silver nanoparticles using response surface methodology and their antimicrobial activity against multi-drug resistant clinical isolates. Colloids and Surfaces B: Biointerfaces, 86(2), 228–233. DOI: https://doi.org/10.1016/j.colsurfb.2011.03.009.
CrossRef - Raveendran, P., Fu, J., & Wallen, S. L. (2003). Completely “green” synthesis and stabilization of metal nanoparticles. Green Chemistry, 5(1), 763–765. DOI: https://doi.org/10.1039/b304701f.
- Ogawa, M., Portier, R.J., Moody, M.W., Bell, J., Schexnayder, M.A., & Losso, J.N. (2004). Biochemical properties of bone and scale collagens isolated from the subtropical fish black drum (Pogonia cromis) and sheepshead seabream (Archosargus probatocephalus). Food Chemistry 88: 495–501. DOI: 1016/j.foodchem.2004.02.006.
CrossRef - Liu, W., Zhang, Y., Cui, N., (2019). Extraction and characterization of pepsin-solubilized collagen from snakehead (Channa argus) skin: effects of hydrogen peroxide pretreatments and pepsin hydrolysis strategies. Process Biochemistry, 76: 194–202. DOI: 10.1016/j.procbio.2018.10.017.
CrossRef
- Seiichiro Ishihara, Takeomi Mizutani, Kazushige Kawabata, Hisashi Haga, (2014). An improved method for western blotting when extracting proteins from mammalian cells cultured on a collagen gel under serum-free conditions. Cytotechnology;68(1):25–32. DOI: 1007/s10616-014-9766-4.
CrossRef - Sougandhi PR, Reddeppaand M, Harini SS, Shobha Rani T, Gangadhara R, (2018). Green synthesis and characterization of silver nanoparticles by using Psidium guajava leaf extract, Journal of Drug Delivery and Therapeutics.8(5-s):301-305. DOI: 22270/jddt.v8i5-s.2025.
CrossRef - Nagai, T., Y. Araki and N. Suzuki. (2002). Collagen of the skin of ocellate puffer fish (Takifugu rubripes). Food Chem. 78: 173-177. DOI: 1016/S0308-8146(01)00396-X.
CrossRef - Ogawa, M., M.W. Moody, R.J. Portier, J. Bell, M.A. Schexnayder and J.N. Losso. (2003). Biochemical properties of black drum and sheepshead seabream skin collagen. Agri. Food Chem. 51: 8088-8092. DOI: 10.1021/jf034350r.
CrossRef - Y, W. Liu, G. Li, B. Shi, Y. Miao and X. Wu, “Isolation and Partial Characterization of Pepsin-Soluble Collagen from the Skin of Grass Carp (Ctenopharyngodon idella),” Food Chemistry, Vol. 103, No. 3, 2007, pp. 906-912. DOI: 10.1016/j.foodchem.2006.09.053.
CrossRef - Mudhafar, M., Zainol, I., Ameer, A. J., Abd, M. Y., Alsailawi, H. A., Ghazaly, N. M.,Zorah, M. (2024). The effect of fish collagen on the silver nanoparticles sizes and shapes using modified microwave-assisted green synthesis method and their antibacterial activities. Heliyon, 10, e32837. DOI: https://doi.org/10.1016/j.heliyon.2024.e32837.
CrossRef - Treesin Potaros, Nongnuch Raksakulthai, Jiraporn Runglerdkreangkrai and Wanchai Worawattanamateekul. (2009). Characteristics of Collagen from Nile Tilapia (Oreochromis niloticus) Skin Isolated by Two Different Methods. Kasetsart Journal – Natural Science. 43 : 584 – 593.
- Martínez-Ortiz MA, Hernández-Fuentes AD, Pimentel-González DJ, Campos-Montiel RG, Vargas-Torres A, Aguirre-Álvarez G. (2015). Extraction, and characterization of collagen from rabbit skin: partial characterization. Cyta-J Food. 13:253–258.
CrossRef - Subha V, Kirubanandan S, Ilangovan R, Renganathan S (2021) Silver Nanoparticles Impregnated Nano collagen as Scaffold for Soft Tissue Repair-Synthesis, Characterization, and In vitro International Journal of Medical Nano Research. 8:034. DOI: doi.org/10.23937/2378-3664.1410034.
CrossRef - Vijayakumar, S., Chen, J., & co-authors. (2024). Biomedical and eco safety assessment of marine fish collagen-capped silver nanoparticles. International Journal of Biological Macromolecules, 260, 129324. DOI: https://doi.org/10.1016/j.ijbiomac.2024.129324.
CrossRef - Cen, L., Liu, W., Cui, L., Zhang, W., & Cao, Y. (2008). Collagen tissue engineering: development of novel biomaterials and applications. Pediatric research, 63(5), 492–496. https://doi.org/10.1203/PDR.0b013e31816c5bc3.
CrossRef - Mandal, A., et al. (2023). Collagen biocomposites derived from fish waste: doping and nanoparticle functionalization. ACS Omega, 8, (article). DOI: https://doi.org/10.1021/acsomega.3c01106.
CrossRef - El-Sawah, A. A., et al. (2024). Bionanofactory for green synthesis of collagen-based nanoparticles: characterization and biological evaluation. Scientific Reports, (article). DOI: https://doi.org/10.1038/s41598-024-56064-8.
CrossRef - Murugan, K., Subramaniam, J., Rajaganesh, R., Panneerselvam, C., Amuthavalli, P., Vasanthakumaran. (2021). Efficacy and side effects of bio-fabricated sardine fish scale silver nanoparticles against malarial vector Anopheles stephensi. Scientific Reports, 11, 98899. DOI: https://doi.org/10.1038/s41598-021-98899-5.
CrossRef
Abbreviations List
Silver nanoparticles: AgNP
Scanning electron microscopy: SEM
Fourier-transform infrared spectroscopy: FTIR
Accepted on: 11-12-2025
Second Review by: Dr. Deepali Bansode
Final Approval by: Dr. Wagih Ghannam









