Exploring New Antifungal Agents for the Treatment of Candida Infections in the Era of Resistance
Department of Pharmacology, Acropolis Institute of Pharmaceutical Education and Research, Indore, M.P., India.
Corresponding Author Email: sangeetadwivedi@acropolis.edu.in
DOI : http://dx.doi.org/10.13005/bbra/3448
ABSTRACT:Candida albicans is the most common fungal pathogen responsible for both invasive and mucosal infections. The primary antifungal drug classes used against C. albicans remain azoles, echinocandins, and polyenes. However, resistance to these agents has been steadily increasing, thereby limiting their clinical effectiveness. To overcome these challenges, several novel antifungal agents have been developed and recently approved. This review aims to highlight recent developments in antifungal drug discovery and resistance mechanisms associated with Candida albicans. A comprehensive literature review was conducted using electronic databases including PubMed, ScienceDirect, and Scopus. Rezafungin, a long-acting echinocandin, offers potent fungicidal activity with convenient once-weekly dosing. Ibrexafungerp, the first orally available glucan synthase inhibitor, retains efficacy against echinocandin-resistant isolates. Fosmanogepix, a prodrug targeting the Gwt1 enzyme, disrupts mannoprotein anchoring and compromises fungal cell wall integrity. Oteconazole, a highly selective tetrazole that inhibits fungal CYP51, demonstrates enhanced effectiveness against azole-resistant strains and recurrent vulvovaginal candidiasis. In conclusion, these emerging antifungals expand the antifungal arsenal, addressing limitations of current treatments while offering improved efficacy, safety, and options against drug-resistant Candida albicans infections.
KEYWORDS:Antifungal Resistance; Biofilm; Candida albicans; Fungal; Infection; Novel Antifungal Agents
Introduction
Candida albicans is diploid, polymorphic yeast that constitutes a major component of the normal human microbiome, colonizing the skin, oral cavity, gastrointestinal tract, and genital mucosa. Globally, it is recognized as the most common pathogen responsible for invasive candidiasis and has been classified as a priority pathogen on the global health agenda, given its substantial health burden in immunocompromised and hospitalized populations.1,2 Although C. albicans generally exists as a harmless symbiont, conditions such as immune suppression, prolonged antibiotic use, or mucosal disruption can convert it into an opportunistic pathogen, leading to a wide spectrum of infections ranging from superficial mucosal diseases to life-threatening systemic infections.3–5 One of the key attributes contributing to the adaptability of C. albicans is its highly malleable genome, which enables rapid growth and survival under diverse and often harsh conditions within the human host.6 This genomic plasticity—encompassing aneuploidy, chromosomal rearrangements, and regulation of major signaling pathways—plays a critical role in the acquisition of drug resistance, enhancement of virulence, and evasion of host immune defenses.7 Pathogenicity is further augmented by its ability to transition between yeast and hyphal forms, form robust biofilms on host tissues and medical devices, and secrete hydrolytic enzymes that facilitate tissue invasion and immune evasion.8 Among these, biofilm formation represents a particularly serious clinical challenge, as biofilm-associated cells are intrinsically resistant to antifungal agents and are often linked to chronic, intractable infections.9 While C. albicans remains largely susceptible to most antifungal agents, recent reports have highlighted alarming trends of rising resistance, particularly in clinical care settings and middle-income regions. This growing resistance underscores the urgent need for ongoing surveillance and the development of novel therapeutic strategies.10 Among these adaptive features, morphological plasticity is one of the most studied mechanisms driving pathogenicity.
Morphogenesis and Phenotypic Switching
Morphological adaptations in Candida albicans involve reversible transitions between yeast-like, hyphae-like, and pseudohyphal forms, which are central to its phenotypic plasticity and virulence. These transitions are regulated through both cAMP-dependent and cAMP-independent pathways, with key regulators including basal PKA activity, cyclin-dependent kinases such as Cdc28, and environmental cues like N-acetylglucosamine (GlcNAc), which can induce hyphal growth independent of its metabolic role.11
In addition to signaling pathways, epigenetic processes such as chromatin reorganization and histone modifications (e.g., H3K56 acetylation) play a crucial role in regulating gene expression in response to environmental conditions, thereby influencing virulence traits and immune evasion.12,13
A hallmark example of morphological plasticity is phenotypic switching, particularly the white-opaque transition, which represents a reversible and heritable switch between two distinct cell types. These forms differ in morphology, gene expression, metabolic preferences, and mating capacity.14,15 The transition is orchestrated by a network of at least 14 transcription factors, whose activity is strongly influenced by strain background and environmental factors such as pH, amino acid availability, and zinc levels.16
Epigenetic regulation also underpins this process, with chromatin accessibility controlled by histone-modifying enzymes and chromatin remodelers, supporting an epigenetic rather than genetic basis for the white-opaque switch.14,15 Furthermore, switching frequency and stability are modulated by additional pathways, including the PHO phosphate metabolism pathway, as well as genetic background effects such as SIR2 function and chromosomal imbalances. Together, these findings suggest that multiple regulatory systems converge to govern cell fate decisions in C. albicans.17,18 These morphological transitions not only support virulence but also enable adhesion to host tissues, the next critical step in pathogenesis.
Adhesion and Invasion
Candida albicans possesses a complex, multi-step adhesion and invasion mechanism that is essential for colonization and infection of host tissues. Adhesion is mediated by cell surface proteins (adhesins), most notably Als3, which binds tightly to host epithelial cells through its peptide-binding cavity (PBC). Disruption of PBC activity or deletion of ALS3 significantly impairs adhesion and invasion, particularly the active entry of fungal cells into host tissues.19 The structure and composition of the fungal cell wall also play a critical role in adhesion, with transcription factors such as Cas5 regulating β-glucan exposure, surface hydrophobicity, and expression of cell wall proteins. These features not only enhance adhesion but also support the morphological transitions required for invasion. Following attachment, C. albicans invades host tissues primarily through two mechanisms: (i) active hyphal penetration and (ii) induced endocytosis by host cells. In both cases, hyphal proliferation and the targeted release of the peptide toxin Candidalysin are central to epithelial tissue damage.19,20 During invasion, C. albicans may traverse host membranes or cross epithelial barriers via transcellular tunnels, processes that do not necessarily result in immediate host cell damage.21 Beyond direct tissue invasion, the fungus can also modulate host immune responses. For example, remodeling of neutrophil extracellular trap (NET) proteins on its surface paradoxically enhances its ability to damage epithelial cells.22 These adhesion and invasion pathways represent potential therapeutic targets. Strategies such as inhibiting cell wall biosynthesis, suppressing adhesin expression, or applying antimicrobial peptides that disrupt hyphal development and adhesion-related genes have shown promise in antifungal therapy.23,24 Following adhesion and invasion, C. albicans further strengthens its pathogenic potential by forming biofilms, which represent one of the most clinically challenging aspects of infection.
Candida albicans Biofilm Formation
Candida albicans biofilm formation is a complex, multi-step process consisting of four key stages: adherence, initiation/filamentation, maturation, and dispersal (Figure 1). Each stage is tightly regulated by both genetic and environmental factors.
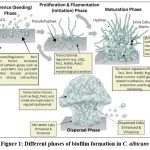 |
Figure 1: Different phases of biofilm formation in C. albicans. |
The consecutive phases of biofilm formation in C. albicans (1). In the adhesion phase, yeast cells adhere promptly to the surface. (2) Initiation phase (early phase), spherical yeast cells replicate and start to secrete extracellular matrix (ECM) and develop Pseudohyphae. (3) Maturation phase (intermediate phase), the mature biofilm develops with hyphal filaments, the extracellular matrix (yellow) accumulates, and drug resistance increases. (4) Dispersal (dispersion phase), yeast cells disperse from the biofilm and diffuse, expanding the infection and starting the cycle again.
Adherence Phase (Seeding)
The process begins with the attachment of yeast cells to either biotic surfaces, such as mucosal tissues, or abiotic surfaces, including medical devices like catheters, pacemakers, heart valves, and dentures. This initial adherence is mediated by adhesin proteins that facilitate stable binding and subsequent biofilm development.25
To initiate biofilm formation, Candida albicans begins its life cycle by adhering free-floating, round-shaped yeast cells to a surface, whether biological tissue or an abiotic substrate such as silicone or plastic. This initial attachment is a relatively slow process, requiring approximately 60–90 minutes, yet it provides the critical foundation for subsequent biofilm development. Adhesion is mediated by cell surface adhesins, including members of the ALS (agglutinin-like sequence) protein family and Hwp1 (hyphal wall protein 1). These molecules function like molecular “glue,” promoting firm attachment of cells both to surfaces and to each other. Cells unable to adhere are washed away or removed by shear forces, leaving behind a stable basal layer of yeast cells. This early binding step is essential for progression to later stages of biofilm formation.26
Proliferation and Filamentation Phase (Initiation)
Once adhesion is established, yeast cells proliferate and spread across the surface. At this stage, a critical morphogenetic transition occurs, whereby yeast cells differentiate into filamentous forms, including hyphae and pseudohyphae. Hyphae are elongated, cylindrical cells, while pseudohyphae consist of chains of slightly elongated, ellipsoidal cells joined end-to-end. This yeast-to-filament transition is unique to Candida albicans and is essential for biofilm structural complexity. The filamentous forms act as a scaffold, providing mechanical stability and enabling the development of multilayered biofilm structures. This morphological switch is tightly regulated by several transcriptional regulators, such as Efg1, Tec1, Ndt80, and Rob1.27,28
Key signaling pathways involved include
Ras1/cAMP/PKA pathway
Ras1, a GTPase, activates adenylate cyclase, leading to increased cAMP levels. Elevated cAMP activates protein kinase A (PKA), which in turn stimulates transcription factors such as Efg1 and Tec1. These factors regulate hypha-specific genes, including ALS3 and HWP1. Efg1 plays a central role in morphogenesis, as mutants lacking this regulator are defective in filamentation and biofilm formation.
MAPK pathways (Cek1, Hog1)
Mitogen-activated protein kinases (MAPKs) relay environmental signals to promote filamentous growth and enhance virulence. Both Cek1 and Hog1 contribute to filamentation and the overall architecture of the biofilm.29
Quorum sensing
Small signaling molecules such as farnesol and other autoinducers regulate the yeast-to-hypha transition, thereby modulating biofilm density and dispersal.30
Maturation Phase
As biofilm development progresses (typically requiring around 24 hours in laboratory models), Candida albicans forms a thick, complex, multi-layered community composed of yeast cells, pseudohyphae, and hyphae. Mature biofilms appear experimentally as a translucent, hazy structure covering the surface, with a highly organized cellular arrangement visible microscopically. During this stage, the biofilm produces an extracellular matrix (ECM) that encapsulates the community, conferring both chemical and mechanical protection. Biofilm maturation is accompanied by extensive metabolic and transcriptional adaptations that reflect its specialized lifestyle and environmental responses.
Key signaling pathways and regulatory networks include
PKC–MAPK cell wall integrity pathway
The Sdd3 protein activates Rho1 GTPase, which initiates the PKC–MAPK cascade. This pathway regulates the synthesis of chitin (via CHS2 and CHS8), a critical component for maintaining strong biofilm architecture. MAPKs such as Mkc1 and Cek1 further contribute to cell wall maintenance and ECM synthesis, ensuring biofilm structural integrity and survival.31
Transcriptional regulatory network
Approximately 1,000 genes involved in adhesion, ECM production, and antifungal resistance are controlled by six master regulators—Efg1, Tec1, Bcr1, Ndt80, Brg1, and Rob1. These regulators form an interconnected transcriptional network that coordinates biofilm development in a temporally regulated manner, ensuring the transition from early adherence to maturation.
Extracellular Matrix (ECM)
The extracellular matrix (ECM) is a critical component of Candida albicans biofilms, produced predominantly during the maturation phase. It is a sticky, complex substance composed of approximately 55% glycoproteins, 25% carbohydrates (mainly mannan–glucan complexes), 15% lipids, and 5% nucleic acids. The ECM serves multiple functions: it provides physical protection against host immune defenses and antifungal agents, offers structural support to maintain biofilm architecture and integrity, and may exert enzymatic activities that degrade molecules for protection or nutritional purposes. In addition, ECM composition can incorporate host-derived proteins and cellular debris, with variations depending on the infection context.32,27
Dispersal Phase
Dispersal is the least well-characterized stage of Candida albicans biofilm development, during which round yeast cells are released from the mature biofilm into the surrounding environment. These dispersed cells are phenotypically distinct from planktonic yeast, exhibiting enhanced adherence, increased biofilm-forming capacity, and greater virulence. Key transcriptional regulators implicated in this process include Nrg1, Pes1, and Ume6, which together govern the control of dispersal. Clinically, this stage is highly significant, as dispersal enables colonization of new host sites and predisposes to disseminated infections, including candidemia. Environmental factors, as well as the balance between yeast and filamentous cell forms, also play critical roles in regulating dispersal.33
The interplay of genetic regulators, signaling pathways, and extracellular matrix production not only drives biofilm development but also enhances the pathogen’s resilience against host immune defenses and antifungal agents. Consequently, biofilm-associated infections are particularly challenging to treat, underscoring the need to understand the molecular mechanisms of antifungal resistance within these structures.
Genetic Transcription Regulators of Candida Albicans
Biofilms of Candida albicans are structured microbial colonies adhered to surfaces, which exhibit great susceptibility to antifungal medications and host immunity. They are orchestrated by a complex genetic network, in the middle of which are a few masters transcriptional regulators. They engage with them and regulate hundreds of genes to coordinate various stages of biofilm development: adhesion, proliferation, matrix production, and dispersal. (Figure 2).
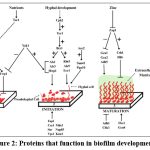 |
Figure 2: Proteins that function in biofilm development. |
In the adherence step, yeast-form cells adhere to the substrate. At the initiation, cells multiply to produce microcolonies with the formation of germ tubes that produce hyphae. During the maturation step, a growth of the biofilm biomass takes place, the extracellular matrix (green) is deposited and drug resistance enhanced. During the dispersal step, the cells in the yeast form are discharged to settle on the surrounding environment. Some of the known pathway relationships are illustrated on the upper half of the diagram. Proteins located in the lower half are involved in a particular step, but may not be part of a recognized pathway. Proteins are not displayed more than once when their functions are in more than one step of biofilm development. T-shaped bars with dashes show that repression is done by an indirect mechanism. m. Plus and minus symbols indicate that the upstream gene or signal stimulates (+) or inhibits (–) the expression of the downstream target.
Antifungal Resistance
Mechanisms of Azole Resistance
Target Modification and Overexpression
Mutations in the ERG11 (or CYP51A) gene alter the structure of the lanosterol 14α-demethylase enzyme, the primary target of azoles, thereby reducing drug affinity and efficacy. In addition, ERG11 overexpression or genomic amplification increases enzyme levels, further diminishing the effectiveness of azole therapy. Notably, mutations such as Y132F, S154F, A395T, and L98H are strongly associated with resistance.34
Efflux Pump Upregulation
Resistance can also arise through the upregulation of efflux pumps (e.g., CDR1, CDR2, MDR1, PDR10), which actively export azole drugs out of the fungal cell. This process lowers the intracellular concentration of the drug, thereby reducing its antifungal activity.34,35
Alternative Pathways and Additional Mechanisms
Other components of the ergosterol biosynthetic pathway (e.g., ERG3, ERG1) or regulatory elements (e.g., cyp51A promoter tandem repeats such as TR34/L98H and TR46/Y121F/T289A) may also undergo mutations. These alterations can either confer resistance to azole toxicity or drive overexpression of target genes. Importantly, such mechanisms have been observed in both clinical and environmental isolates, highlighting the cross-sectoral and global nature of azole resistance.36,35 Together, these mechanisms underscore the multifaceted nature of azole resistance in Candida albicans, necessitating careful surveillance and encouraging the exploration of alternative antifungal strategies, including echinocandins and polyenes.
Mechanisms of Echinocandin Resistance
FKS Gene Mutations (Target Modification)
Echinocandins inhibit β-1,3-glucan synthase, a key enzyme required for fungal cell wall synthesis. Resistance most commonly arises from point mutations in hotspot regions of the FKS genes (FKS1, FKS2), which encode subunits of this enzyme. Mutations such as S643P, S645P, S639F, F635Y, R1354H, and S656P alter enzyme structure, reducing drug binding affinity, increasing minimum inhibitory concentrations (MICs), and contributing to clinical treatment failure.37,38 Clinically, FKS mutations represent the primary mechanism of echinocandin resistance in Candida glabrata, C. auris, C. parapsilosis, and C. albicans, and are strongly associated with prior echinocandin therapy.39
Cell Wall Adaptation and Stress Response
Fungi can compensate for echinocandin-induced cell wall damage by enhancing chitin synthesis, thereby reinforcing the cell wall and reducing drug susceptibility. This mechanism is particularly evident in C. glabrata and C. haemulonii, where resistant strains exhibit increased chitin content and altered cell wall architecture.40
Additional Factors
Genes involved in cell wall integrity (e.g., CHS1, CHS2, CHS3, YPS1, YPK2, SLT2) and stress tolerance pathways (e.g., PKC, calcineurin) further contribute to echinocandin tolerance and the development of resistance.
Other/Emerging Mechanisms
Additional FKS mutations may promote drug tolerance, serving as a reservoir for the emergence of resistance. Mitochondrial function and calcium homeostasis, involving factors such as Cdc50 and Crm1 (in Cryptococcus), also play supporting roles in echinocandin resistance.41
Mechanisms of Polyene Resistance
Polyenes exert their antifungal activity by targeting ergosterol in the fungal cell membrane, forming pores that compromise membrane integrity. The most common mechanism of resistance involves alterations in membrane sterol composition, typically through a decrease in ergosterol levels or its replacement with alternative sterols. Genetic changes in the ergosterol biosynthesis pathway (ERG2, ERG3, ERG5, ERG6, ERG11)—including mutations or loss-of-function events—lead to the accumulation of sterols such as lanosterol, eburicol, or 4,14-dimethyl-zymosterol, which reduce polyene binding and activity. Clinically, in Candida albicans, resistance can arise from loss-of-function mutations in ERG11 or ERG3, or mutations in ERG2, ERG5, or ERG6. Notably, ERG2 mutations have also been reported as a resistance mechanism in Cryptococcus neoformans. Polyene resistance can be either intrinsic, as in species that naturally lack ergosterol or possess alternative sterols, or acquired, developing during therapy as a consequence of genetic mutations induced by drug exposure.42 Collectively, these mechanisms underscore the challenges of treating polyene-resistant infections and highlight the importance of understanding sterol-mediated adaptations in fungal pathogens. (Figure 3).
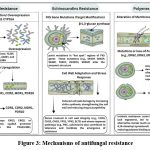 |
Figure 3: Mechanisms of antifungal resistance |
The diagram summarizes the main resistance strategies employed by fungi against three major antifungal classes. (A) Azole resistance involves target modification (ERG11/CYP51A overexpression or mutation) and/or upregulation of efflux pumps (CDR1, CDR2, MDR1, PDR10). (B) Echinocandin resistanceis the major cause of Echinocandin resistance, which is due to the occurrence of hot spot mutations in the FKS gene, which affects the 2-1,3-glucan synthase target, frequently with cell wall adaptation. (C) Polyene resistance is mediated by altered membrane sterol composition due to ERG gene mutations or loss-of-function, leading to reduced ergosterol. Both acquired and intrinsic resistance mechanisms are observed for polyenes.
FDA-approved antifungal
The FDA has granted approval for several novel antifungal agents- Ibrexafungerp, Rezafungin, Fosmanogepix, Oteseconazole, Olorofim, and Opelconazole- which provide improved therapeutic options for severe, recurrent, and drug-resistant fungal infections. These agents combine innovative mechanisms, improved pharmacokinetics, and enhanced tissue penetration, offering clinical advantages over conventional therapies. A summary of these new antifungals is provided in Table 1.
Table 1: Summary of New Antifungal Agents
| Field | Rezafungin | Ibrexafungerp | Olorofim | Opelconazole | Fosmanogepix | Oteseconazole |
| Mechanism 43 | Echinocandin; inhibits 1,3-β-D-glucan synthase | Triterpenoid glucan synthase inhibitor | Orotomide class; inhibits dihydroorotate dehydrogenase (pyrimidine synthesis) | Inhaled triazole (lanosterol 14α-demethylase inhibitor) | Gwt1 inhibitor (GPI-anchor biosynthesis) | Tetrazole azole; inhibits fungal CYP51 (14α-demethylase) |
| Discovery/ Development History 44 |
Developed ~2010s; FDA approved 2023–2024; next-gen echinocandin | Discovered from natural product (enfumafungin); developed 2010s; FDA approved 2021–2022 | First-in-class, developed mid-2010s; in advanced phase 2/3 studies | Synthetic analog, designed for inhalation; development since the mid-2010s | First-in-class; discovered 2010s (prodrug of manogepix); phase 2/3 | Rational design, FDA approved 2022; Mycovia Pharmaceuticals |
| Formulations 43 | IV only | Oral | Oral | Inhaled (nebulized) | IV, oral | Oral |
| Clinical Status & Trials 45 |
Approved for candidemia/invasive candidiasis; ReSTORE Phase 3 | Approved for VVC; SCYNERGIA (VVC), FURI, and CARES (C. auris, invasive molds) trials | Phase 2/3 for invasive mold infections in rare and resistant fungi | Phase 2/3 for refractory pulmonary aspergillosis; expanded special-use UK program | Phase 2 open-label (AEGIS; molds), phase 2 (C. auris/candidemia), ongoing pivotal studies | Approved for RVVC; pivotal and global phase 3 (ultraVIOLET, VIOLET) studies |
| Spectrum/ Resistance 46 |
Broad activity vs. Candida (including azole-resistant), some Aspergillus; not active in CNS/urine | Candida spp. (including azole/echinocandin resistant), some activity vs. Aspergillus | Potent vs. Aspergillus, Scedosporium, Fusarium, rare molds; not active vs. Candida or Mucorales | Highly active vs. Aspergillus spp., good local activity in the lung; some Candida, limited data on resistance | Broad: Candida (exc. C. krusei/kefyr), Aspergillus, Fusarium, Scedosporium, some Mucorales; active vs. many resistant strains | Potent against Candida spp. (esp. fluconazole-resistant and C. glabrata) High selectivity reduces off-target effects |
| Safety Highlights 47 | Good tolerability; hypokalemia, fever, diarrhea; no major new safety signals | GI side effects are the most common; contraindicated in pregnancy; there are a few drug-drug interactions. | Generally well tolerated, favorable PK, limited data on long-term safety | Very low systemic toxicity, high lung retention; no major AEs in early trials | Well tolerated, low DDI, good oral bioavailability; more data in larger studies pending | Excellent selectivity (>2000-fold for fungal vs. mammalian CYP); GI effects, minimal DDI; not for women of reproductive potential |
| Ongoing Research/Future Directions 43 | Prophylactic use, expansion to other mold infections, and resistance monitoring | Trials in invasive candidiasis and aspergillosis, combinations, and global surveillance | Pivotal trials for salvage/refractory/rare infections, resistance evolution | Combined regimens, prevention in lung transplant, and global regulatory review | Phase 3 for invasive molds/yeasts, resistance mapping, and real-world use |
New indications (prophylaxis, recalcitrant VVC), regional approvals, resistance surveillance |
Rezafungin (Rezzayo)
Rezafungin (CD101, Brand Name: Rezzayo)
Rezafungin is a second-generation echinocandin developed to overcome limitations of earlier echinocandins, focusing on improved dosing convenience, drug stability, and resistance prevention. It exhibits a novel pharmacokinetic profile that allows once-weekly intravenous administration for invasive fungal infections, particularly candidemia and invasive candidiasis.48
Drug Discovery and Development
The design of rezafungin was guided by structure-activity relationship (SAR) studies and pharmacokinetic optimization to enhance stability, tissue penetration, solubility, and half-life compared to first-generation echinocandins like caspofungin. These modifications enable potent antifungal activity with once-weekly dosing, a regimen not previously achievable. Rezafungin was initially patented and preclinically tested by Seachaid Pharmaceuticals, later acquired by Cidara Therapeutics, which advanced it through clinical development.49
Clinical Trials
Two major randomized studies, Phase 2 STRIVE and Phase 3 ReSTORE, evaluated rezafungin. In STRIVE, rezafungin achieved a 76.1% overall cure rate, compared to 67.2% with daily caspofungin, with faster negative blood culture conversion (average 3.3 hours) and lower 30-day all-cause mortality (4.4% vs. 13.1%). ReSTORE confirmed noninferior efficacy, with day-14 global cure rates of 59% for rezafungin versus 61% for caspofungin, and comparable 30-day mortality (24% vs. 21%).50
Mechanism of Action and Pharmacology
Rezafungin is a potent 1,3-β-D-glucan synthase inhibitor, disrupting the synthesis of β-1,3-glucan, a key polymer of the fungal cell wall, leading to rapid fungal cell death. It selectively targets fungal cells, sparing mammalian cells, and demonstrates fungicidal, dose-dependent activity against Candida and Aspergillus species, including some echinocandin-resistant isolates.51 Its long plasma half-life allows front-loaded, once-weekly dosing that maintains drug levels above the minimum inhibitory concentration (MIC), enhancing tissue penetration, accelerating fungal clearance, and potentially reducing the emergence of resistance.48,52,53
Safety and Tolerability
Clinical evidence indicates that rezafungin has a safety and tolerability profile comparable to caspofungin. Most adverse effects are mild, including fever, gastrointestinal symptoms, and hypokalemia, with rare treatment-limiting toxicity even during extended courses of up to 39 weeks.
Ongoing Studies and Future Directions
Rezafungin is under preclinical investigation for prophylaxis in patients with hematological malignancies or transplants and for treatment of Pneumocystis pneumonia in HIV-positive adults, as well as other systemic fungal infections. Its positive clinical safety profile, broad-spectrum activity, and pharmacokinetic advantages underscore how SAR-based design and pharmacometric strategies can yield clinically significant advances in antifungal therapy.52
Ibrexafungerp
Ibrexafungerp (SCY-078, MK-3118; Brand Name: Brexafemme)
Ibrexafungerp is an orally bioavailable, semi-synthetic triterpenoid antifungal that belongs to the glucan synthase inhibitor class. Similar to rezafungin, its development employed structure-activity relationship (SAR) optimization and innovative chemistry to create a compound with a novel mechanism of action, convenient oral dosing, and potent efficacy against resistant fungal pathogens.54
Drug Discovery and Development
Ibrexafungerp was derived through systematic SAR-based modification of the naturally occurring enfumafungin to enhance antifungal activity, oral bioavailability, and stability. The resulting triterpenoid glucan synthase inhibitor binds to a site distinct from that of echinocandins and azoles, enabling activity against echinocandin-resistant strains. The drug was developed to provide a broad-spectrum oral option capable of treating Candida and Aspergillus infections, including multidrug-resistant isolates.55
Clinical Trials and Efficacy
Ibrexafungerp has been evaluated in both mucosal and systemic Candida infections. Phase 3 VANISH trials (VANISH 303 and VANISH 306) demonstrated high clinical cure rates and long-term symptom resolution in vulvovaginal candidiasis (VVC), including azole-resistant cases, establishing ibrexafungerp as an effective oral alternative to fluconazole. It has also shown high microbiological eradication rates in severe or resistant candidemia, either as first-line or salvage therapy, consistently demonstrating noninferiority or superiority compared with existing treatments.56
Mechanism of Action and Pharmacology
Ibrexafungerp inhibits 1,3-β-D-glucan synthase, an enzyme critical for fungal cell wall biosynthesis. Its unique binding site allows it to retain activity against echinocandin-resistant isolates, including those with FKS mutations, while sparing mammalian cells, which lack β-glucan synthesis. Oral administration achieves high tissue penetration, including vaginal mucosa, blood, and deep tissues, and dosing regimens maintain drug concentrations above the minimum inhibitory concentration (MIC) for both susceptible and resistant strains. These pharmacokinetic and pharmacodynamic properties support its use in both outpatient mucosal infections and severe invasive candidiasis.44 (Figure 4)
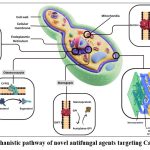 |
Figure 4: Mechanistic pathway of novel antifungal agents targeting Candida species |
The mechanism by which novel antifungal agents target Candida species is as follows: Rezafungin and Ibrexafungerp target the cell-wall enzyme complex known as (1,3) ‐β‐d‐glucan synthase, but interact with different subunits of this complex. The FKS genes are Responsible for encoding (1,3) ‐β‐d‐glucan synthase. The active analog of fosmanogepix, manogepix, targets the fungal acetyltransferase enzyme (Gwt1) present in the endoplasmic reticulum to prevent inositol acetylation and the production of glycosylphosphatidylinositol: this interferes with the operation of mannoproteins. Oteseconazole inhibits the fungal cyp51, which is the fungal enzyme that prevents lanosterol- to the ergosterol conversion, and olorofim is a reversible inhibitor of dihydroorotate dehydrogenase (DHODH), a fungal oxidoreductase enzyme that mediates the fourth step in pyrimidine de novo synthesis.
Safety and Tolerability
Ibrexafungerp is generally well tolerated, with mild gastrointestinal side effects, such as nausea and diarrhea, being the most common. Serious adverse events and treatment discontinuations are uncommon. It exhibits a low risk of hepatotoxicity and clinically significant drug-drug interactions. Importantly, its distinct mechanism provides activity against echinocandin-, fluconazole-, and multidrug-resistant Candida species, including Candida auris, with a high barrier to resistance; clinical failures are rare and typically associated with stepwise mutations.46
Ongoing Studies and Future Directions
Current investigations are expanding ibrexafungerp’s indications to include prophylaxis in immunocompromised patients, salvage therapy in refractory mycoses, and use in combination regimens for highly resistant infections. Its oral route, favorable tolerability, and efficacy against non-albicans Candida and molds make it a promising candidate for broader clinical application.57 Ibrexafungerp exemplifies the successful application of rational drug design, SAR-based optimization, and novel chemistry to deliver a clinically meaningful, orally available, broad-spectrum antifungal with activity against resistant species.
Fosmanogepix
Fosmanogepix (APX001)
Fosmanogepix is a first-in-class antifungal with a novel mechanism of action and broad-spectrum activity against both yeast and molds, including drug-resistant pathogens. It is a prodrug of manogepix (MGX) and is rapidly converted in vivo by systemic phosphatases into its active form. Fosmanogepix was developed through targeted drug discovery against the fungal enzyme Gwt1, which is essential for glycosylphosphatidylinositol (GPI) anchor biosynthesis, a key process in attaching mannoproteins to the fungal cell wall and membrane.58
Drug Discovery and Development
The discovery of fosmanogepix involved structure-activity relationship (SAR) optimization to identify compounds that effectively inhibit Gwt1. By blocking the inositol acylation step of GPI-anchor biosynthesis, fosmanogepix disrupts fungal cell wall integrity, adhesion, virulence, and immune evasion. The drug exhibits high oral bioavailability (>90%) and can also be administered intravenously, providing versatility in managing invasive fungal infections such as candidemia, including multidrug-resistant strains like Candida auris. Phase 2 trials in non-neutropenic adults with candidemia demonstrated an 80% treatment success rate and 85% survival at 30 days. The drug was well tolerated, and many patients successfully transitioned from intravenous to oral therapy. Early evidence also suggests efficacy against molds such as Aspergillus species and difficult-to-treat fungi including Fusarium and Scedosporium.59,60
Mechanism of Action
Fosmanogepix inhibits the fungal-specific enzyme Gwt1, preventing GPI-anchor biosynthesis necessary for mannoprotein attachment to the cell wall. This disruption compromises cell wall integrity, biofilm formation, adhesion, and virulence, ultimately leading to fungal cell death. The mammalian homologue, PIG-W, shares low sequence homology, minimizing off-target effects and toxicity 61. Fosmanogepix demonstrates extensive tissue distribution, including lungs, brain, liver, kidneys, and eyes, and is primarily eliminated via biliary and fecal routes. Pharmacodynamic studies indicate strong correlations between drug exposure (AUC/MIC and Cmax/MIC) and antifungal activity, supporting dosing regimens that maintain therapeutic drug levels.62
Safety, Tolerability, and Resistance
Clinical trials indicate that fosmanogepix is generally well tolerated, with most adverse events being mild to moderate and no significant treatment-related toxicity. There are no major hepatotoxicity or nephrotoxicity concerns, and drug-drug interactions are minimal, making it suitable for patients with complex medical conditions or polypharmacy. Its novel mechanism provides a high barrier to resistance, requiring multiple genetic mutations for the development of resistance. The drug has shown efficacy against multidrug-resistant fungi, including Candida auris, with treatment success rates of 80–89% and 30-day survival rates up to 85% in clinical studies.63,64
Ongoing Studies and Future Directions
Fosmanogepix is currently undergoing Phase 3 trials (FAST-IC and FORWARD-IM) to evaluate efficacy and safety in larger populations and against a broader range of invasive fungal infections, including molds. Future applications under investigation include prophylaxis in immunocompromised patients, salvage therapy for refractory infections, and combination therapy in difficult-to-treat cases. The dual oral/IV administration, broad-spectrum activity, and unique mechanism make fosmanogepix a promising next-generation antifungal capable of addressing unmet clinical needs in resistant and challenging fungal infections.45 Fosmanogepix exemplifies the successful application of rational drug design and fungal-specific targeting to meet the urgent clinical demand for effective antifungal therapy.
Oteseconazole
Oteseconazole (Vivjoa, formerly VT-1161)
Oteseconazole is a next-generation oral antifungal, commonly referred to as a tetrazole, specifically developed for the prevention and treatment of recurrent vulvovaginal candidiasis (RVVC), particularly in women not of reproductive age. In April 2022, Mycovia Pharmaceuticals developed oteseconazole as the first highly selective, orally bioavailable inhibitor of fungal lanosterol 14α-demethylase (CYP51), a critical enzyme in ergosterol biosynthesis. Rational drug design strategies focused on maximizing selectivity for fungal CYP51 while minimizing inhibition of human cytochrome P450 enzymes, thereby reducing drug–drug interactions and adverse effects associated with older azoles.65
Clinical Trials and Efficacy
Oteseconazole has been extensively evaluated in Phase 2 and 3 clinical trials. In women with severe vulvovaginal candidiasis, it demonstrated statistically and clinically superior outcomes compared to fluconazole, achieving higher mycological and clinical cure rates at day 28, with similar rates of mild or moderate adverse events. For RVVC prevention, oteseconazole was noninferior to fluconazole for acute episode resolution and significantly more effective than placebo in preventing recurrence over nearly one year of follow-up (recurrence 5% with oteseconazole vs. >40% with placebo). The drug exhibits broad-spectrum antifungal activity in vitro and in vivo, including efficacy against fluconazole-resistant Candida species, with particularly potent activity against Candida glabrata and other common vaginal pathogens.66
Mechanism of Action
Oteseconazole exerts its antifungal effect by selectively binding to the heme iron of fungal CYP51, inhibiting lanosterol demethylation, thereby disrupting ergosterol production and compromising fungal cell membrane integrity, which leads to cell death. Its tetrazole core and tailored side chains confer over 2,000-fold selectivity for fungal versus human CYP enzymes, improving specificity and safety compared to triazoles. This high selectivity, combined with favorable pharmacodynamics, results in enhanced potency, minimal drug–drug interactions, and reduced interference with human hormone or hepatic metabolism.67
Pharmacokinetics and Pharmacodynamics
Oteseconazole is readily absorbed orally, with dose-proportional exposure between 20–320 mg. Peak plasma concentrations are achieved within 5–10 hours, with a long elimination half-life exceeding 140 hours, high protein binding, and extensive tissue distribution, allowing for both acute dosing and long-term prophylaxis. The drug is primarily excreted unchanged in feces, with minimal renal elimination and negligible metabolism, further reducing drug–drug interaction potential. Its pharmacodynamic efficacy is determined by the ratio of minimum plasma concentration (Cmin) to minimum inhibitory concentration (MIC) against target Candida species, maintaining therapeutic trough levels for both susceptible and resistant isolates. Stability studies confirm that oteseconazole is chemically stable in solid form under recommended storage conditions, supporting long-term clinical use.68,69
Resistance and Future Directions
Oteseconazole remains effective against most fluconazole- and multi-azole-resistant Candida species. Potential resistance mechanisms include upregulation of efflux pumps (e.g., CDR1, MDR1) or CYP51 mutations; however, current clinical surveillance indicates low rates of resistance. Its potent efficacy, oral administration, safety profile, and broad spectrum make it a valuable option for the management of difficult-to-treat, chronic, or recurrent fungal infections.70
Olorofim
Olorofim (F901318)
Olorofim is a first-in-class, orally administered antifungal that inhibits fungal dihydroorotate dehydrogenase (DHODH), disrupting pyrimidine synthesis and consequently fungal growth and survival. It shows particular activity against azole-resistant Aspergillus species and rare pathogenic molds. Developed through structure-activity relationship (SAR) optimization by F2G, olorofim addresses clinical gaps in resistant invasive fungal infections with a novel mechanism not shared by existing antifungal classes.71
Drug Discovery Process
Olorofim was identified via a systematic SAR-driven process targeting essential fungal-specific proteins, leading to the discovery of DHODH as a critical enzyme. Preclinical studies demonstrated broad activity against Aspergillus species—including cryptic and azole-resistant strains—as well as rare pathogens such as Lomentospora prolificans, Scedosporium spp., and Scopulariopsis spp. Its selective inhibition of fungal DHODH minimizes mammalian toxicity, supporting its clinical development.72
Mechanism of Action
Olorofim selectively and reversibly binds fungal DHODH, inhibiting de novo pyrimidine biosynthesis and thereby blocking DNA, RNA, and protein synthesis. This results in fungal cell cycle arrest and eventual cell death. Its mechanism is independent of ergosterol or cell wall synthesis pathways targeted by azoles, echinocandins, and polyenes, allowing efficacy against fungi resistant to these classes. Olorofim exhibits fungistatic activity initially, progressing to fungicidal effects with prolonged exposure, particularly against Aspergillus spp.71,73
Pharmacokinetics and Pharmacodynamics
Olorofim shows rapid tissue distribution via both oral and intravenous administration, with a high volume of distribution (~3 L/kg) and oral bioavailability exceeding 45%. Its half-life of 20–30 hours allow convenient dosing, achieving steady-state concentrations within three days orally. It is primarily metabolized by cytochrome P450 isoenzymes (mainly CYP3A4) but is only a weak inhibitor, minimizing clinically significant drug interactions. Less than 0.2% is excreted unchanged in urine, and protein binding exceeds 99%. The C_min/MIC ratio is the primary PK/PD determinant of efficacy. Olorofim’s pharmacokinetic and pharmacodynamic profiles support both outpatient therapy and long-term treatment, with stability over extended oral administration and a low likelihood of resistance due to the conserved nature of DHODH.74–76.
Clinical Trials and Efficacy
In phase 2b clinical trials, olorofim demonstrated high efficacy in invasive fungal infections with limited treatment options, including CNS, bone, and disseminated infections caused by azole-resistant Aspergillus and rare molds. Clinical responses were observed at day 42, with sustained responses at day 84 and overall improvement in nearly 29% of patients. The safety profile was favorable, with primarily mild adverse events and rare, reversible liver enzyme elevations; no treatment-related deaths were reported.77
Spectrum and Resistance
Olorofim is highly active in vitro and in vivo against Aspergillus spp., including azole-resistant strains, and rare pathogenic molds. However, it has limited activity against yeasts such as Candida and Mucorales, emphasizing the importance of accurate diagnosis before treatment. Resistance development is expected to be slow due to the conserved nature of DHODH, with current surveillance indicating robust activity.78
Ongoing Studies and Future Directions
Olorofim is currently undergoing phase 3 trials comparing its efficacy and safety to standard therapies such as liposomal amphotericin B (AmBisome) for invasive aspergillosis and other difficult mold infections, particularly in immunocompromised and multidrug-resistant populations. Its oral bioavailability and long tissue persistence support outpatient management of chronic, deep-seated infections. Investigational uses include salvage therapy, combination regimens, and refractory mycoses.79
Opelconazole
Opelconazole (PC945)
Opelconazole is a novel inhalable antifungal agent belonging to the broad-spectrum triazole class, developed by Pulmocide Ltd., London, UK. Its mechanism of action mirrors that of other azoles: the triazole moiety inhibits lanosterol 14α-demethylase (CYP51A1), an enzyme essential for converting lanosterol to ergosterol. This inhibition reduces ergosterol synthesis, disrupting fungal cell membrane integrity and preventing fungal growth.80
Conclusion
Candida albicans is the most common opportunistic fungal pathogen, with its pathogenicity driven by morphogenetic switching, adhesion, and biofilm formation. Resistance to traditional antifungals- such as azoles, polyenes, and echinocandins- arises from genetic changes, including FKS gene mutations, overexpression of efflux pumps, and alterations in the sterol biosynthesis pathway. To address these challenges, several novel antifungal agents have been developed with enhanced efficacy against resistant strains. The long-acting echinocandin rezafungin exhibits fungicidal activity for up to one week, while the oral glucan synthase inhibitor ibrexafungerp remains effective against echinocandin-resistant isolates. The Gwt1 inhibitor fosmanogepix disrupts fungal cell wall integrity, and the innovative tetrazole oteseconazole is active against azole-resistant infections. Investigational agents such as olorofim and opelconazole further expand therapeutic options. However, to comprehensively manage Candida infections, integrating alternative approaches—such as combination therapies, natural product-based antifungals, immune modulation, and resistance monitoring is essential. Continued clinical evaluation, molecular research, and surveillance of resistance patterns will be vital to ensure long-term efficacy and sustainability of these novel treatments.
Acknowledgement
The author would like to thank Acropolis Institute of Pharmaceutical Education and Research (AIPER), Indore, M.P., for providing the academic environment and support for this review work. The author also expresses gratitude to the Department of Pharmacology for their guidance and access to essential resources. Sincere thanks are extended to all faculty members and mentors for their valuable insights and encouragement.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Jyoti Bhatol: Conceptualization, Literature Review, Writing – Original Draft.
Sangeeta Dwivedi: Review & Editing, Literature organization, Supervision.
Vikas Kumar Jain: Assistance in figure and table design, Supervision.
Neha Sisodiya: Antifungal resistance section, manuscript refinement.
Gajanan Darwhekar: Validation, supervision.
References
- Parambath S, Dao A, Kim HY, et al. Candida albicans—A systematic review to inform the World Health Organization Fungal Priority Pathogens List. Med Mycol. 2024;62(6): myae045. doi:10.1093/mmy/myae045
CrossRef - Katsipoulaki M, Stappers MHT, Malavia-Jones D, Brunke S, Hube B, Gow NAR. Candida albicans and Candida glabrata: global priority pathogens. Heitman J, ed. Microbiol Mol Biol Rev. 2024;88(2):e00021-23. doi:10.1128/mmbr.00021-23
CrossRef - Lopes JP, Lionakis MS. Pathogenesis and virulence of Candida albicans. Virulence. 2022;13(1):89-121. doi:10.1080/21505594.2021.2019950
CrossRef - Dadar M, Tiwari R, Karthik K, Chakraborty S, Shahali Y, Dhama K. Candida albicans – Biology, molecular characterization, pathogenicity, and advances in diagnosis and control – An update. Microb Pathog. 2018; 117:128-138. doi: 10.1016/j.micpath.2018.02.028
CrossRef - Talapko J, Juzbašić M, Matijević T, et al. Candida albicans—The Virulence Factors and Clinical Manifestations of Infection. J Fungi. 2021;7(2):79. doi:10.3390/jof7020079
CrossRef - Schikora-Tamarit MÀ, Gabaldón T. Recent gene selection and drug resistance underscore clinical adaptation across Candida species. Nat Microbiol. 2024;9(1):284-307. doi:10.1038/s41564-023-01547-z
CrossRef - Wang W, Wang C, Dong Y, Yang F, Xu Y. Aneuploidy enables adaptation to brefeldin A in Candida albicans. Front Cell Infect Microbiol. 2025; 15:1562726. doi:10.3389/fcimb.2025.1562726
CrossRef - El Hachem S, Fattouh N, Chedraoui C, Finianos M, Bitar I, Khalaf RA. Sequential Induction of Drug Resistance and Characterization of an Initial Candida albicans Drug-Sensitive Isolate. J Fungi. 2024;10(5):347. doi:10.3390/jof10050347
CrossRef - Pereira R, Santos Fontenelle RO, Brito EHS, Morais SM. Biofilm of Candida albicans: formation, regulation and resistance. J Appl Microbiol. 2021;131(1):11-22. doi:10.1111/jam.14949
CrossRef - Lee Y, Puumala E, Robbins N, Cowen LE. Antifungal Drug Resistance: Molecular Mechanisms in Candida albicans and Beyond. Chem Rev. 2021;121(6):3390-3411. doi: 10.1021/acs.chemrev.0c00199
CrossRef - Min K, Park A. Shape-Shifting Mechanisms: Integrative Multi-Omics Insights Into Candida albicans Morphogenesis. Mycobiology. 2025;53(2):250-257. doi:10.1080/12298093.2025.2460304
CrossRef - Iracane E, Vega-Estévez S, Buscaino A. On and Off: Epigenetic Regulation of C. albicans Morphological Switches. Pathogens. 2021;10(11):1463. doi:10.3390/pathogens10111463
CrossRef - Conte M, Eletto D, Pannetta M, et al. H3K56 acetylation affects Candida albicans morphology and secreted soluble factors interacting with the host. Biochim Biophys Acta BBA – Gene Regul Mech. 2024;1867(3):195048. doi: 10.1016/j.bbagrm.2024.195048
CrossRef - Qasim MN, Valle Arevalo A, Nobile CJ, Hernday AD. The Roles of Chromatin Accessibility in Regulating the Candida albicans White-Opaque Phenotypic Switch. J Fungi. 2021;7(1):37. doi:10.3390/jof7010037
CrossRef - Beekman CN, Cuomo CA, Bennett RJ, Ene IV. Comparative genomics of white and opaque cell states supports an epigenetic mechanism of phenotypic switching in Candida albicans. Rokas A, ed. G3 GenesGenomesGenetics. 2021;11(2):jkab001. doi:10.1093/g3journal/jkab001
CrossRef - Sharma A, Homayoon A, Weyler M, et al. Transcriptional control of C. albicans white-opaque switching and modulation by environmental cues and strain background. Heitman J, ed. mBio. 2025;16(5):e00581-25. doi:10.1128/mbio.00581-25
CrossRef - Woodruff AL, Berman J, Anderson M. Strain background of Candida albicans interacts with SIR2 to alter phenotypic switching. Microbiology. 2024;170(3). doi:10.1099/mic.0.001444
CrossRef - Zheng Q, Guan G, Cao C, Li Q, Huang G. The PHO pathway regulates white–opaque switching and sexual mating in the human fungal pathogen Candida albicans. Curr Genet. 2020;66(6):1155-1162. doi:10.1007/s00294-020-01100-z
CrossRef - Oh SH, Hoyer LL. Assessing Als3 Peptide-Binding Cavity and Amyloid-Forming Region Contributions to Candida albicans Invasion of Human Oropharyngeal Epithelial Cells. Front Cell Infect Microbiol. 2022; 12:890839. doi:10.3389/fcimb.2022.890839
CrossRef - Mogavero S, Sauer FM, Brunke S, et al. Candidalysin delivery to the invasion pocket is critical for host epithelial damage induced by Candida albicans. Cell Microbiol. 2021;23(10). doi:10.1111/cmi.13378
CrossRef - Lachat J, Pascault A, Thibaut D, Le Borgne R, Verbavatz JM, Weiner A. Trans-cellular tunnels induced by the fungal pathogen Candida albicans facilitate invasion through successive epithelial cells without host damage. Nat Commun. 2022;13(1):3781. doi:10.1038/s41467-022-31237-z
CrossRef - Karkowska-Kuleta J, Smolarz M, Seweryn-Ozog K, et al. Proteinous Components of Neutrophil Extracellular Traps Are Arrested by the Cell Wall Proteins of Candida albicans during Fungal Infection, and Can Be Used in the Host Invasion. Cells. 2021;10(10):2736. doi:10.3390/cells10102736
CrossRef - Martin H, Kavanagh K, Velasco-Torrijos T. Targeting Adhesion in Fungal Pathogen Candida Albicans. Future Med Chem. 2021;13(3):313-334. doi:10.4155/fmc-2020-0052
CrossRef - Chen M, Huang WK, Yao Y, et al. Heterologous expression of the insect SVWC peptide WHIS1 inhibits Candida albicans invasion into A549 and HeLa epithelial cells. Front Microbiol. 2024; 15:1358752. doi:10.3389/fmicb.2024.1358752
CrossRef - Finkel JS, Mitchell AP. Genetic control of Candida albicans biofilm development. Nat Rev Microbiol. 2011;9(2):109-118. doi:10.1038/nrmicro2475
CrossRef - Desai JV, Mitchell AP. Candida albicans Biofilm Development and Its Genetic Control. Ghannoum M, Parsek M, Whiteley M, Mukherjee P, eds. Microbiol Spectr. 2015;3(3):3.3.04. doi:10.1128/microbiolspec. MB-0005-2014
CrossRef - Gulati M, Nobile CJ. Candida albicans biofilms: development, regulation, and molecular mechanisms. Microbes Infect. 2016;18(5):310-321. doi: 10.1016/j.micinf.2016.01.002
CrossRef - Mayer FL, Wilson D, Hube B. Candida albicans pathogenicity mechanisms. Virulence. 2013;4(2):119-128. doi:10.4161/viru.22913
CrossRef - Wang D, Zeng N, Li C, Li Z, Zhang N, Li B. Fungal biofilm formation and its regulatory mechanism. Heliyon. 2024;10(12): e32766. doi: 10.1016/j.heliyon. 2024.e32766
CrossRef - Uppuluri P, Pierce CG, López-Ribot JL. Candida albicans biofilm formation and its clinical consequences. Future Microbiol. 2009;4(10):1235-1237. doi:10.2217/fmb.09.85
CrossRef - Pang LM, Zeng G, Chow EWL, et al. Sdd3 regulates the biofilm formation of Candida albicans via the Rho1-PKC-MAPK pathway. Berman J, ed. mBio. 2025;16(2): e03283-24. doi:10.1128/mbio.03283-24
CrossRef - Nobile CJ, Johnson AD. Candida albicans Biofilms and Human Disease. Annu Rev Microbiol. 2015;69(1):71-92. doi:10.1146/annurev-micro-091014-104330
CrossRef - Wall G, Montelongo-Jauregui D, Vidal Bonifacio B, Lopez-Ribot JL, Uppuluri P. Candida albicans biofilm growth and dispersal: contributions to pathogenesis. Curr Opin Microbiol. 2019; 52:1-6. doi: 10.1016/j.mib.2019.04.001
CrossRef - Paul S, Shaw D, Joshi H, et al. Mechanisms of azole antifungal resistance in clinical isolates of Candida tropicalis. Sturtevant J, ed. PLOS ONE. 2022;17(7):e0269721. doi: 10.1371/journal.pone.0269721
CrossRef - Hagiwara D, Watanabe A, Kamei K, Goldman GH. Epidemiological and Genomic Landscape of Azole Resistance Mechanisms in Aspergillus Fungi. Front Microbiol. 2016;7. doi:10.3389/fmicb.2016.01382
CrossRef - Sewell TR, Zhu J, Rhodes J, et al. Nonrandom Distribution of Azole Resistance across the Global Population of Aspergillus fumigatus. Chowdhary A, ed. mBio. 2019;10(3):e00392-19. doi:10.1128/mBio.00392-19
CrossRef - Coste AT, Kritikos A, Li J, et al. Emerging echinocandin-resistant Candida albicans and glabrata in Switzerland. Infection. 2020;48(5):761-766. doi:10.1007/s15010-020-01475-8
CrossRef - Silva LN, Ramos LS, Oliveira SSC, et al. Development of Echinocandin Resistance in Candida haemulonii: An Emergent, Widespread, and Opportunistic Fungal Pathogen. J Fungi. 2023;9(8):859. doi:10.3390/jof9080859
CrossRef - Ning Y, Xiao M, Perlin DS, et al. Decreased echinocandin susceptibility in Candida parapsilosis causing candidemia and emergence of a pan-echinocandin resistant case in China. Emerg Microbes Infect. 2023;12(1):2153086. doi:10.1080/22221751.2022.2153086
CrossRef - Garcia-Rubio R, Hernandez RY, Clear A, Healey KR, Shor E, Perlin DS. Critical Assessment of Cell Wall Integrity Factors Contributing to in vivo Echinocandin Tolerance and Resistance in Candida glabrata. Front Microbiol. 2021; 12:702779. doi:10.3389/fmicb.2021.702779
CrossRef - Cao C, Xue C. More than flipping the lid: Cdc50 contributes to echinocandin resistance by regulating calcium homeostasis in Cryptococcus neoformans. Microb Cell. 2020;7(4):115-118. doi:10.15698/mic2020.04.714
CrossRef - Carolus H, Pierson S, Lagrou K, Van Dijck P. Amphotericin B and Other Polyenes—Discovery, Clinical Use, Mode of Action and Drug Resistance. J Fungi. 2020;6(4):321. doi:10.3390/jof6040321
CrossRef - Kriegl L, Egger M, Boyer J, Hoenigl M, Krause R. New treatment options for critically important WHO fungal priority pathogens. Clin Microbiol Infect. 2025;31(6):922-930. doi: 10.1016/j.cmi.2024.03.006
CrossRef - Ordaya EE, Clement J, Vergidis P. The Role of Novel Antifungals in the Management of Candidiasis: A Clinical Perspective. Mycopathologia. 2023;188(6):937-948. doi:10.1007/s11046-023-00759-5
CrossRef - Jacobs SE, Zagaliotis P, Walsh TJ. Novel antifungal agents in clinical trials. F1000Research. 2022; 10:507. doi:10.12688/f1000research.28327.2
CrossRef - Hoenigl M, Sprute R, Egger M, et al. The Antifungal Pipeline: Fosmanogepix, Ibrexafungerp, Olorofim, Opelconazole, and Rezafungin. Drugs. 2021;81(15):1703-1729. doi:10.1007/s40265-021-01611-0
CrossRef - Logan A, Wolfe A, Williamson JC. Antifungal Resistance and the Role of New Therapeutic Agents. Curr Infect Dis Rep. 2022;24(9):105-116. doi:10.1007/s11908-022-00782-5
CrossRef - Garcia-Effron G. Rezafungin—Mechanisms of Action, Susceptibility and Resistance: Similarities and Differences with the Other Echinocandins. J Fungi. 2020;6(4):262. doi:10.3390/jof6040262
CrossRef - Fung S, Shirley M. Rezafungin: A Review in Invasive Candidiasis. Drugs. 2025;85(3):415-423. doi:10.1007/s40265-024-02134-0
CrossRef - Thompson GR, Soriano A, Skoutelis A, et al. Rezafungin Versus Caspofungin in a Phase 2, Randomized, Double-blind Study for the Treatment of Candidemia and Invasive Candidiasis: The STRIVE Trial. Clin Infect Dis. 2021;73(11):e3647-e3655. doi:10.1093/cid/ciaa1380
CrossRef - Johnson MD. Antifungals in Clinical Use and the Pipeline. Infect Dis Clin North Am. 2021;35(2):341-371. doi: 10.1016/j.idc.2021.03.005
CrossRef - Andes D, Brüggemann RJ, Flanagan S, et al. The distinctive pharmacokinetic profile of rezafungin, a long-acting echinocandin developed in the era of modern pharmacometrics. J Antimicrob Chemother. 2025;80(1):18-28. doi:10.1093/jac/dkae415
CrossRef - Soriano A, Honore PM, Cornely OA, et al. Treatment Outcomes Among Patients With a Positive Candida Culture Close to Randomization Receiving Rezafungin or Caspofungin in the ReSTORE Study. Clin Infect Dis. 2024;79(3):672-681. doi:10.1093/cid/ciae363
CrossRef - Sofjan AK, Mitchell A, Shah DN, et al. Rezafungin (CD101), a next-generation echinocandin: A systematic literature review and assessment of possible place in therapy. J Glob Antimicrob Resist. 2018; 14:58-64. doi: 10.1016/j.jgar.2018.02.013
CrossRef - El Ayoubi LW, Allaw F, Moussa E, Kanj SS. Ibrexafungerp: A narrative overview. Curr Res Microb Sci. 2024; 6:100245. doi: 10.1016/j.crmicr.2024.100245
CrossRef - Goje O, Sobel R, Nyirjesy P, et al. Oral Ibrexafungerp for Vulvovaginal Candidiasis Treatment: An Analysis of VANISH 303 and VANISH 306. J Womens Health. 2023;32(2):178-186. doi:10.1089/jwh.2022.0132
CrossRef - Daraskevicius J, Petraitis V, Davainis L, Zucenka A. The Feasibility of Ibrexafungerp for the Treatment of Fungal Infections in Patients with Hematological Malignancies. J Fungi. 2022;8(5):440. doi:10.3390/jof8050440
CrossRef - Almajid A, Bazroon A, Al-awami HM, et al. Fosmanogepix: The Novel Anti-Fungal Agent’s Comprehensive Review of in Vitro, in Vivo, and Current Insights from Advancing Clinical Trials. Cureus. Published online April 28, 2024. doi:10.7759/cureus.59210
CrossRef - Shaw KJ, Ibrahim AS. Fosmanogepix: A Review of the First-in-Class Broad Spectrum Agent for the Treatment of Invasive Fungal Infections. J Fungi. 2020;6(4):239. doi:10.3390/jof6040239
CrossRef - Vazquez JA, Pappas PG, Boffard K, et al. Clinical Efficacy and Safety of a Novel Antifungal, Fosmanogepix, in Patients with Candidemia Caused by Candida auris: Results from a Phase 2 Trial. Antimicrob Agents Chemother. 2023;67(5): e01419-22. doi:10.1128/aac.01419-22
CrossRef - Dai X, Liu X, Li J, et al. Structural insights into the inhibition mechanism of fungal GWT1 by manogepix. Nat Commun. 2024;15(1):9194. doi:10.1038/s41467-024-53512-x
CrossRef - Hodges MR, Ople E, Evans P, et al. A phase 1 open label study to assess the human mass balance and metabolite profile of14 C-fosmanogepix, a novel Gwt-1 inhibitor in healthy male participants. Krysan DJ, ed. Antimicrob Agents Chemother. 2024;68(8): e00273-24. doi:10.1128/aac.00273-24
CrossRef - Pappas PG, Vazquez JA, Oren I, et al. Clinical safety and efficacy of novel antifungal, fosmanogepix, for the treatment of candidaemia: results from a Phase 2 trial. J Antimicrob Chemother. 2023;78(10):2471-2480. doi:10.1093/jac/dkad256
CrossRef - Espinel-Ingroff A, Wiederhold NP. A Mini-Review of In Vitro Data for Candida Species, Including C. auris, Isolated during Clinical Trials of Three New Antifungals: Fosmanogepix, Ibrexafungerp, and Rezafungin. J Fungi. 2024;10(5):362. doi:10.3390/jof10050362
CrossRef - Vanreppelen G, Nysten J, Baldewijns S, Sillen M, Donders G, Van Dijck P. Oteseconazole (VIVOJA) for prevention of recurrent vulvovaginal candidiasis. Trends Pharmacol Sci. 2023;44(1):64-65. doi: 10.1016/j.tips.2022.10.004
CrossRef - Wang X, Chen L, Ruan H, et al. Oteseconazole versus fluconazole for the treatment of severe vulvovaginal candidiasis: a multicenter, randomized, double-blinded, phase 3 trial. Krysan DJ, ed. Antimicrob Agents Chemother. 2024;68(1):e00778-23. doi:10.1128/aac.00778-23
CrossRef - Hoy SM. Oteseconazole: First Approval. Drugs. 2022;82(9):1017-1023. doi:10.1007/s40265-022-01734-y
CrossRef - Susararla KPC, Shelke O, Shorgar N. Quantification of oteseconazole in rat plasma using LC-MS/MS and its application to pharmacokinetic study. J Appl Pharm Res. 2024;12(4):54-65. doi:10.69857/joapr. v12i4.485
CrossRef - Martens MG, Maximos B, Degenhardt T, et al. Phase 3 study evaluating the safety and efficacy of oteseconazole in the treatment of recurrent vulvovaginal candidiasis and acute vulvovaginal candidiasis infections. Am J Obstet Gynecol. 2022;227(6):880.e1-880.e11. doi: 10.1016/j.ajog.2022.07.023
CrossRef - Ghannoum M, Degenhardt T, Person K, Brand S. 719. Susceptibility Testing of Oteseconazole (VT-1161) Against Clinical Isolates from Phase 3 Clinical Studies in Subjects with Recurrent Vulvovaginal Candidiasis. Open Forum Infect Dis. 2021;8(Supplement_1): S459-S459. doi:10.1093/ofid/ofab466.916
CrossRef - Wiederhold NP. Review of the Novel Investigational Antifungal Olorofim. J Fungi. 2020;6(3):122. doi:10.3390/jof6030122
CrossRef - Wiederhold NP. Pharmacodynamics, Mechanisms of Action and Resistance, and Spectrum of Activity of New Antifungal Agents. J Fungi. 2022;8(8):857. doi:10.3390/jof8080857
CrossRef - Kühbacher A, Birch M, Oliver JD, Gsaller F. Anti- Aspergillus activities of olorofim at sub-MIC levels during early-stage growth. Carvalho A, ed. Microbiol Spectr. 2024;12(3): e03304-23. doi:10.1128/spectrum.03304-23
CrossRef - Vanbiervliet Y, Van Nieuwenhuyse T, Aerts R, Lagrou K, Spriet I, Maertens J. Review of the novel antifungal drug olorofim (F901318). BMC Infect Dis. 2024;24(1):1256. doi:10.1186/s12879-024-10143-3
CrossRef - Cornelissen K, Newell PA, Rex JH, et al. 594. An Open-Label Study in Healthy Volunteers to Determine the Absolute Bioavailability of, the Effect of Food and Dosing by Nasogastric Tube upon the Pharmacokinetics of a Single Oral Dose of Olorofim (OLO). Open Forum Infect Dis. 2022;9(Supplement_2): ofac492.646. doi:10.1093/ofid/ofac492.646
CrossRef - Neoh CF, Jeong W, Kong DC, Slavin MA. The antifungal pipeline for invasive fungal diseases: what does the future hold? Expert Rev Anti Infect Ther. 2023;21(6):577-594. doi:10.1080/14787210.2023.2203383
CrossRef - Maertens JA, Thompson GR, Spec A, et al. 754. Olorofim for treatment of invasive fungal infections (IFI) due to moulds in patients with limited or no treatment options: Interim results from a Phase 2b open-label study (NCT03583164, Study 32). Open Forum Infect Dis. 2022;9(Supplement_2): ofac492.039. doi:10.1093/ofid/ofac492.039
CrossRef - Maertens JA, Thompson GR, Spec A, et al. Olorofim for the treatment of invasive fungal diseases in patients with few or no therapeutic options: a single-arm, open-label, phase 2b study. Lancet Infect Dis. Published online June 2025: S1473309925002245. doi:10.1016/S1473-3099(25)00224-5
CrossRef - Feuss A, Bougnoux ME, Dannaoui E. The Role of Olorofim in the Treatment of Filamentous Fungal Infections: A Review of In Vitro and In Vivo Studies. J Fungi. 2024;10(5):345. doi:10.3390/jof10050345
CrossRef - Colley T, Alanio A, Kelly SL, et al. In Vitro and In Vivo Antifungal Profile of a Novel and Long-Acting Inhaled Azole, PC945, on Aspergillus fumigatus Infection. Antimicrob Agents Chemother. 2017;61(5): e02280-16. doi:10.1128/AAC.02280-16
CrossRef
Accepted on: 13-11-2025
Second Review by: Dr. Mytham Jabouri Abdul Hussein
Final Approval by: Dr. Ali Elshafei









