Development and In-Vitro Characterization of Luteolin-Loaded Nanostructured Lipid Carrier
Department of Pharmacy, NCRD’s Sterling Institute of Pharmacy, Navi Mumbai, India.
Corresponding Author E-mail: bbhairav@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3464
ABSTRACT:This study aimed to enhance the therapeutic potential of luteolin, a natural flavonoid with strong antioxidant, anti-inflammatory, and anti-cancer effects. However, its poor water solubility and limited oral absorption restrict clinical applications. To overcome these limitations, researchers have developed nanostructured lipid carriers (NLCs) loaded with luteolin to improve its solubility, stability, and bioavailability for oral administration. We prepared the NLCs using a hot emulsification–probe sonication method with Precirol ATO 5 (solid lipid) and Capmul MCM (liquid lipid). We employed a Central Composite Design (CCD) to optimize the formulation by varying lipid ratio, lipid concentration, and sonication time. The optimized NLCs showed a mean particle size of 166.0 nm, a zeta potential of –33.6 mV, and an entrapment efficiency of 86.43%. FTIR, DSC, and XRD characterization confirmed the encapsulation of luteolin in an amorphous state, without any chemical interaction with the excipients. Transmission electron microscopy (TEM) revealed spherical, uniform NLCs. In vitro release studies demonstrated that the NLCs provided sustained luteolin release for 36 hours, outperforming pure luteolin. Overall, the optimized NLCs significantly enhanced luteolin’s solubility, stability, and release behaviour, indicating strong potential as an efficient oral delivery platform. These findings suggest that luteolin-loaded NLCs could serve as a promising approach for improving therapeutic outcomes in breast cancer and related diseases.
KEYWORDS:Bioavailability; Central Composite Design (CCD); Entrapment efficiency; Luteolin; Nanostructured lipid carrier (NLC); Sustained release
Introduction
Luteolin, a naturally occurring flavonoid with potent pharmacological activities, faces significant challenges in clinical application due to its poor water solubility and resultant low oral bioavailability.1-3 Despite its promising antioxidant, anti-inflammatory, anticancer, and anti-diabetic properties, the therapeutic potential of luteolin is limited by its low absorption when administered orally, with bioavailability often reported to be less than 30% in relevant animal models.4,5 Enhancing its solubility and bioavailability is therefore crucial to harness its full clinical benefits.
Various strategies have been employed to improve luteolin delivery, including complexation with cyclodextrins,6 phospholipid complexation,7 and incorporation into nanocarrier systems such as liposomes, micelles, and nanoparticles.8–10 Among these, lipid-based nanocarriers have gained significant attention due to their biocompatibility, ability to improve drug solubilization, and potential to protect labile compounds from degradation.11,12
Nanostructured Lipid Carriers (NLCs), as a second-generation lipid nanoparticulate system, comprise a mixture of solid and liquid lipids, stabilized by surfactants to form a stable colloidal dispersion.13,14 NLCs have demonstrated remarkable potential in enhancing the solubility, controlled release, and bioavailability of poorly water-soluble drugs.15,16 Their unique lipid matrix facilitates improved drug loading, stability, and cellular uptake, making them ideal candidates for delivering lipophilic compounds like luteolin.17,18
Recent advances indicate that NLCs can significantly surpass the limitations of traditional solid-lipid nanoparticles by reducing drug expulsion during storage and providing sustained release profiles.19 The ability of NLCs to enhance the pharmacokinetic profile and therapeutic efficacy of flavonoids has been documented in various preliminary studies. 20-22 Although reports on luteolin-loaded NLCs remain scarce.
In this context, the development of luteolin-loaded NLCs offers a promising avenue to overcome the drawbacks associated with its poor water solubility. This study aims to formulate and optimize luteolin-loaded NLCs, evaluate their physicochemical properties, and assess their biological efficacy, thereby contributing to the advancement of effective delivery systems for luteolin and similar nutraceuticals.
 |
Figure 1: Chemical structure of luteolin (3,4,5,7-tetrahydroxy flavone). |
Materials and Methods
Chemicals and Reagents
Luteolin (LTN) was procured from Yarrow Chemical Products, Ghatkopar, Mumbai. The Precirol ATO 5 (solid lipid) & Capmul MCM (liquid lipid) was provided by Gattefosse as a gift sample. The experiment was performed with Milli Q water obtained from the laboratory. All other chemicals and reagents used were of AR grade.
Central Composite Design (CCD)
Central Composite Design (CCD) is a widely employed experimental design for optimization purposes due to its efficiency in requiring fewer experimental trials compared to a full factorial design with three levels for each factor. 23 A typical CCD comprises three different types of points: (a) the design points (black dots), (b) the axial or star points (grey dots), and (c) the center points (white dots). 24,25 For this study, a CCD was generated using Design Expert Software (Version 7.1.6, Stat-Ease Inc., MN) to facilitate the optimization of nanostructured lipid carriers (NLCs), aiming to reduce the number of experiments while maintaining thorough exploration of the formulation space.
A total of 20 NLC batches were designed and formulated, with the goal of investigating the influence of critical formulation variables on key properties of the NLCs. Based on an extensive review of the literature, three independent variables were selected: (X₁) surfactant to lipid ratio, (X₂) total lipid concentration, and (X₃) sonication time. These parameters were chosen because the ratio of surfactant and lipid can significantly impact the characteristics of the NLCs, such as particle size, surface charge (zeta potential), and drug entrapment efficiency (%EE). The use of ethanol as a solvent was justified, given that both cholesterol and Span 60 are highly soluble in ethanol, facilitating consistent formulation. The dependent variables evaluated in this study included particle size, ζ-potential, and entrapment efficiency (%). 26 These parameters are critical for assessing the stability, bioavailability, and therapeutic efficacy of the NLC formulations.
The study also focuses on optimization and validation of a reliable model for the desired response by using Design Expert, 12.03.0 [Stat-Ease.Inc.]. According to the central composite design generated by Design-Expert software, a total of 20 experiments were performed
Table 1: Central composite design parameters and experimental conditions
| Independent Variables | Levels | ||
| Low(-1) | Medium(0) | High(+1) | |
| X1: Precirol ATO 5: Capmul MCM ratio | 0.5 | 1 | 1.5 |
| X2: Lipid conc. (mg) | 200 | 300 | 400 |
| X3: Sonication Time (min) | 2 | 4 | 6 |
| Dependent Variables | Constraints | ||
| Y1: Vesicle Size(nm) | Minimum | ||
| Y2: zeta potential(mV) | Maximum | ||
| Y3: EE (%) | Maximum | ||
Preparation and Optimization of Luteolin- loaded NLC
Preparation of Luteolin- loaded NLC by Hot emulsification- probe sonication method
Luteolin-loaded NLCs were prepared by dissolving 10 mg of Luteolin along with the specified amounts of Precirol® ATO 5 and Capmul MCM in 5 mL of methanol. The organic phase was sonicated for 5 to 10 minutes to ensure complete dissolution of both the API and excipients. This organic solution was then injected into an aqueous phase, which was heated to 60 °C under continuous magnetic stirring to facilitate the hot emulsification process. During injection, the formation of NLCs was initiated in the aqueous medium. To achieve the desired particle size, the formulation was subjected to probe sonication.
Following emulsification, ethanol was removed by evaporating it using a rotary evaporator (Model 600, Switzerland) at 40 °C. The resulting NLC dispersion was then characterized for various parameters such as particle size, entrapment efficiency, and stability. 27,28
 |
Figure 2: Hot emulsification- probe sonication method for preparation of NLC |
Characterization of Lut-loaded NLC
Particle size, size distribution, and Zeta-potential
The mean particle size (MPS), polydispersity index (PDI), and ζ-potential of NLC formulations were determined using Photon Correlation Spectroscopy (PCS) with a Malvern Zetasizer Nano ZS90 (Malvern Instruments Ltd., UK). Samples (0.1 mL) were diluted 100-fold with double-distilled water before analysis. Measurements were carried out at 25 °C, using a 90° scattering angle for particle size/PDI and a dip cell with an applied electric field strength of 20 V cm⁻¹ for ζ-potential determination. The Z-average particle size, PDI, and ζ-potential values were automatically calculated by the instrument software from multiple successive runs to ensure reproducibility.
Entrapment efficiency (EE)
Entrapment efficiency (EE%) refers to the proportion of drug entrapped within the NLCs compared to the total amount of drug initially used in the formulation. To evaluate EE%, the NLC suspension was transferred into a dialysis bag (molecular weight cut-off 12 kDa, Thermo Scientific, Waltham, MA, USA) and immersed in the corresponding hydration medium. Dialysis was carried out for 4 h, with the duration optimized based on the drug characteristics. The adequacy of dialysis time was validated using control solutions of the drug, where equilibrium was confirmed once >99.8% of the drug diffused into the external medium. At predetermined time intervals, aliquots were withdrawn from the external medium, and the concentration of free drug was quantified using a UV–visible spectrophotometer (UV 1700, Shimadzu, Japan). Finally, the entrapment efficiency (%) of Luteolin-loaded NLCs was calculated using the following equation:
![]()
Surface morphology
The morphology of the optimized Luteolin-loaded NLC vesicles was examined using a transmission electron microscope (TEM) (Tecnai G2, F30, Netherlands) operating at an accelerating voltage of 100 kV. A small volume of the NLC dispersion was diluted 50-fold with double-distilled water. A few drops of this diluted dispersion were mounted onto a 400-mesh carbon-coated copper grid. Before imaging, the sample on the grid was negatively stained with 1% phosphotungstic acid for 10 seconds, then air-dried. The TEM analysis provided a detailed visualization of the shape, size, and surface characteristics of the NLC vesicles.
Differential Scanning Calorimetry (DSC) Studies
DSC analysis was performed using a Mettler-Toledo DSC 1 STAR system (Switzerland) to investigate the thermal properties of the formulations. For each measurement, approximately 2 mg of Luteolin, freeze-dried Luteolin-loaded NLCs, and the optimized NLC batch were accurately weighed and placed in aluminium pans, which were then crimped and sealed. A blank aluminium pan served as the reference. The samples were heated from 30°C to 400°C at a rate of 10°C/min under a continuous nitrogen flow of 50 mL/min. The DSC thermograms were analysed to assess the physical state, melting behaviour, and potential interactions between Luteolin and the NLC components.
Fourier Transform-Infrared spectroscopy (FTIR) study
The freeze-dried Luteolin-loaded NLC (LUT-NLC) powder was prepared as a pellet using potassium bromide (KBr), an IR-grade spectroscopic grade. The sample was homogenized with KBr in a ratio of 1:100 (sample: KBr). The mixture was then pressed into a uniform disc with a diameter of 13 mm using a hydraulic pellet press (Kimaya Engineers, India) under a pressure of 10 tons. The prepared pellet was scanned over a wave number range of 4000–400 cm⁻¹ using an FTIR spectrometer (Shimadzu 8400S, Japan). The resulting spectra were analysed to identify characteristic functional groups and to assess potential interactions between Luteolin and the NLC components.
X-Ray Diffraction (XRD)
The crystalline structures of the optimized Luteolin-loaded NLCs and the freeze-dried Luteolin-loaded NLC powder were examined using a Bruker D2 Phaser 2nd Generation X-ray diffractometer (Germany). Samples were prepared by mounting on the sample holder, and their diffraction patterns were recorded over a 2θ range of 5° to 60°. The scans were conducted at a chart speed of 5° per minute. For comparison, pure Luteolin and the freeze-dried LNC formulations were also analysed. The XRD patterns were interpreted to assess the physical state, crystalline or amorphous nature, of Luteolin.29
Results
Formulation and optimization of Lut-loaded NLC
The Luteolin-loaded NLCs were statistically optimized using a central composite design (CCD). The influence of the independent variables, namely Precirol: Capmul ratio (X1), lipid concentration (X2), and sonication time (X3), was evaluated on the dependent responses: particle size (Y1), zeta potential (Y2), and entrapment efficiency (Y3). The effect of individual variables and their interactions was visualized through three-dimensional response surface plots and contour plots (Figure 1). Polynomial equations were derived to quantify the relationship between formulation factors and responses. Among the tested models, the quadratic model was found to be most appropriate for accurately describing the experimental data.
The polynomial equation representing the relationship between factors and particle size in terms of coded variables was:
Particle Size (Y1) = +199.99 – 81.34X1 + 210.64X2 + 78.21X3 – 113.87X1X2 – 132.68X1X3 + 124.40X2X3 – 7.13X1² + 196.14X2² – 8.58X3²
Lipid concentration (X2) exerted the most pronounced effect on particle size, with higher lipid content leading to significantly larger particles. This could be attributed to reduced emulsification efficiency and particle aggregation at elevated lipid levels. Conversely, sonication time (X3) reduced particle size, as extended ultrasonication provided sufficient energy to break lipid droplets into smaller nanocarriers. Interaction effects (X1X2 and X1X3) also influenced particle size, indicating the need for fine-tuning both lipid composition and processing time to achieve nanoscale dimensions.
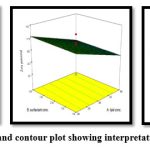 |
Figure 3: Response surface and contour plot showing interpretation of independent variables |
 |
Figure 4: Overlay plot of formulation composition |
The polynomial equation for zeta potential in coded factors was
Zeta Potential (Y2) = –37.74 – 0.47X1 + 0.38X2 + 10.44X3 + 0.86X1X2 + 1.39X1X3 + 1.09X2X3 – 1.34X1² – 2.89X2² + 11.16X3²
The effect of the formulation variables on zeta potential is illustrated in Figure 1B. ANOVA confirmed the statistical significance of the model (F = 3.12, p = 0.0453). Sonication time (X3, p = 0.0067) and its quadratic term (X3², p = 0.0039) were the major contributors, suggesting that longer sonication enhanced the negative surface charge of NLCs. A higher negative zeta potential reflects improved colloidal stability due to electrostatic repulsion between particles. Other factors, such as lipid concentration and Precirol: Capmul ratio, showed comparatively minor effects. However, a significant lack of fit (F = 22.16, p = 0.0020) indicated that model refinement may be required for better predictive accuracy.
Particle size, size distribution, and Zeta-potential
The average particle size of Luteolin-loaded NLCs was obtained in the nanometric range, with the Z-average particle size measured at 166.0 nm. The polydispersity index (PDI) was found to be 0.177, indicating a narrow particle size distribution and good homogeneity of the formulation. The relatively small particle size provides a larger interfacial surface area, which may facilitate faster drug release, enhanced absorption, and improved bioavailability.
The ζ-potential values of Luteolin-loaded NLCs prepared using Capmul and Precirol were observed in the range of −42.3 to −27.8 mV, with the optimized batch showing a value of −33.6 mV. These values suggest that the NLCs possess sufficient surface charge to maintain electrostatic repulsion between particles, thereby preventing aggregation and ensuring dispersion stability. The higher negative ζ-potential values can be attributed to the lipid components used in the formulation, which contribute to the overall colloidal stability of the system.
Surface morphology
To examine the morphological characteristics of the formulated NLCs, Transmission Electron Microscopy (TEM) was performed. The NLCs exhibited a roughly spherical, ovoidal shape with a particle size ranging from approximately 20 to 200 nm. This indicates that the particle size of the NLCs is within a moderate and desirable range. The surfaces of the NLC particles appeared smooth under the TEM analysis.
 |
Figure 5: Morphology analysis by TEM |
In-Vitro Drug Release
The drug release profiles of the formulation, pure Luteolin, and Luteolin-loaded NLCs are depicted in Figure 6. The NLC formulation demonstrated a higher and prolonged release behaviour compared to pure Luteolin. Incorporation of Luteolin into the NLCs resulted in a significantly slower and sustained release, achieving 100% release over 36 hours. This controlled release is attributed to the presence of Capmul within the NLC matrix, which stabilizes the lipid bilayer, reduces drug leakage, and enhances vesicle strength, thereby preventing an initial burst release. In contrast, pure Luteolin exhibited a rapid initial release, with approximately 76.88% released within the first 6 hours, reaching 100% by 36 hours. The sustained release behaviour of Luteolin from the NLCs is primarily due to the structural properties of the lipid bilayer, which modulate the drug diffusion rate.30
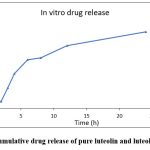 |
Graph 1: % Cumulative drug release of pure luteolin and luteolin-loaded NLC |
Discussion
The present study successfully demonstrated that nanostructured lipid carriers can serve as an efficient delivery platform to overcome the limitations of luteolin, including poor aqueous solubility and low oral bioavailability. The optimized formulation, prepared using Precirol ATO 5 and Capmul MCM via hot emulsification–probe sonication, achieved desirable physicochemical properties with nanosized particles, high entrapment efficiency, and a stable negative zeta potential, which together contribute to enhanced stability and drug loading. Characterization confirmed successful incorporation of luteolin in an amorphous form without undesirable drug–excipient interactions, while TEM images revealed uniformly spherical NLCs. Importantly, the in vitro elease profile indicated a sustained and complete release of luteolin over 36 hours compared to the rapid release of pure drug, suggesting improved therapeutic potential and reduced dosing frequency. These findings collectively highlight the ability of NLCs to significantly improve the solubility, stability, and controlled release of luteolin, supporting their promise as an oral livery system for effective cancer therapy.
Conclusion
The current study successfully developed Luteolin-loaded nanostructured lipid carriers (NLCs) for oral administration to improve the bioavailability and therapeutic potential of this bioactive compound for breast cancer treatment. Preformulation studies characterized the physicochemical properties of Luteolin, confirming its suitability for incorporation into the NLC formulation. The Luteolin-loaded NLCs were prepared using a solvent diffusion method optimized through a central composite design. Particle size analysis by TEM showed the NLCs had an average size of 150-200 nm, indicating successful nanoparticle formation. Zeta potential measurements demonstrated a highly negative surface charge of around -35 mV, conferring good colloidal stability to the NLC dispersion. XRD analysis confirmed the encapsulation of Luteolin in the lipid matrix, with the drug existing in an amorphous state within the NLCs. The DoE approach allowed optimization of the formulation, with the final NLC formulation achieving a high drug entrapment efficiency of over 90%. In vitro studies further demonstrated the enhanced solubility and dissolution profile of Luteolin from the NLC formulation compared to the free drug. This suggests improved oral bioavailability and better therapeutic performance of the Luteolin-loaded NLCs.
Overall, the comprehensive characterization and optimization of the Luteolin-loaded NLCs developed in this study hold great promise for improving the oral delivery and anticancer efficacy of this bioactive compound in the treatment of breast cancer. Further in vivo evaluations are warranted to validate the enhanced pharmacokinetic profile and therapeutic potential of the optimized NLC formulation.
Acknowledgement
The authors are thankful to NCRD’s Sterling Institute of Pharmacy, Navi Mumbai, for providing the necessary facility for the compilation of this research paper.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Manisha S. Chavan: Conceptualization, Methodology, Experimentation, Data Collection, Writing Original Draft.
Bhushan A. Bhairav: Guidance, Methodology Support, Supervision, and Assistance in Experimentation.
Rupesh Pingale: Provision of Facilities, Resources, and Data Collection
References
- Sahoo L, Jena GK, Patro CS. Impact of parameter on nanostructured lipid carrier formulation and approach of the carrier for cancer treatment: a brief study. Drug Dev Regist. 2022;11(4):95-107. doi:10.33380/2305-2066-2022-11-4-95-107
CrossRef - Mendes M, Sousa J, Pais A, Vitorino C. Designing ultra-small nanostructured lipid carriers: critical process parameters. 1st Int Electron Conf Pharm. 2020;50. doi:10.3390/iecp2020-08691
CrossRef - Anwar W, Dawaba HM, Afouna MI, Samy AM. Screening study for formulation variables in preparation and characterization of candesartan cilexetil-loaded nanostructured lipid carriers. Univ J Pharm Res. 2020. doi:10.22270/ujpr.v4i6.330
CrossRef - Chauhan I, Yasir M, Verma M, Singh AP. Nanostructured lipid carriers: a groundbreaking approach for transdermal drug delivery. Adv Pharm Bull. 2020;10(2):150-165. doi:10.34172/apb.2020.021
CrossRef - Rajendra PKM, Jawahar N, Raman R, Shivakumar H, Puthusserik A. An overview of dual targeting nanostructured lipid carriers for the treatment of ovarian cancer. Indian J Pharm Educ Res. 2021;55(2):330-335. doi:10.5530/ijper.55.2.70
CrossRef - Viegas CM, Patrício AB, Prata JN, et al. Solid lipid nanoparticles vs nanostructured lipid carriers: a comparative review. Pharmaceutics. 2023;15(6):1593. doi:10.3390/pharmaceutics15061593
CrossRef - Sailaja AK, Amareshwar P, Chakravarty P. Formulation of solid lipid nanoparticles and their applications. J Curr Pharma Res. 2011;1(2):197-203. doi:10.33786/jcpr.2011.v01i02.019
CrossRef - Izza N, Watanabe N, Okamoto Y, et al. Dependence of the core–shell structure on the lipid composition of nanostructured lipid carriers. ACS Appl Nano Mater. 2022;5(7):9958-9969. doi:10.1021/acsanm.2c02214
CrossRef - Padhi S, Mazumder R, Bisht S. Preformulation screening of lipids using the solubility parameter concept… Braz J Pharm Sci. 2023;59. doi:10.1590/s2175-97902023e21308
CrossRef - Wang J, Wang H, Xia Q. Ubidecarenone-loaded nanostructured lipid carrier: percutaneous penetration and protective effects. Int J Mol Sci. 2018;19(7):1865. doi:10.3390/ijms19071865
CrossRef - Li R, Wang S, Sun L, et al. A novel cationic nanostructured lipid carrier for ocular bioavailability improvement. J Drug Deliv Sci Technol. 2016;33:28-36. doi:10.1016/j.jddst.2016.03.009
CrossRef - Baig MS, Siddiqui A. Cationic nanostructured lipid carriers: optimization of zeta potential and evaluation. Int J Appl Pharm. 2020;12(4):77-84. doi:10.22159/ijap.2020v12i4.37531
CrossRef - Taléns-Visconti R, de Julián-Ortiz JV, Vila-Busó O, et al. Intranasal drug administration in Alzheimer-type dementia. Pharmaceutics. 2023;15(5):1399. doi:10.3390/pharmaceutics15051399
CrossRef - Rincón MLE, Calpena AC, Fábrega M, et al. Pranoprofen-loaded nanostructured lipid carriers for skin inflammatory disorders. Nanomaterials. 2018;8(12):1022. doi:10.3390/nano8121022
CrossRef - Azmi NAN, Hasham R, Ariffin FD, et al. Virgin coconut oil-based nanostructured lipid carrier loaded with Ficus deltoidea extract. Cosmetics. 2020;7(4):83. doi:10.3390/cosmetics7040083
CrossRef - Patra CN. Asian J Pharm. 2018;12(4). doi:10.22377/ajp.v12i04.2902
CrossRef - Higuchi CH, Sales CC, Andréo-Filho N, et al. Citronella oil insect repellent nanotechnology matrix. Life. 2023;13(1):141. doi:10.3390/life13010141
CrossRef - Development and characterization of ofloxacin & prednisolone loaded nanostructured lipid carriers. J Pharm Res. 2019;4(1). doi:10.33140/jpr.04.01.03
CrossRef - Naseri N, Valizadeh H, Zakeri-Milani P. Solid lipid nanoparticles and nanostructured lipid carriers. Adv Pharm Bull. 2015;5(3):305-313. doi:10.15171/apb.2015.043
CrossRef - Hassan DM, El-Kamel AH, Allam EA, et al. Chitosan-coated nanostructured lipid carriers for brain delivery. Drug Deliv Transl Res. 2023;14(2):400-417. doi:10.1007/s13346-023-01407-7
CrossRef - Sarifuddin N, Soerarti W, Rosita N. NLC coenzyme Q10 with hyaluronic acid. Health Notions. 2019;3(1):32-36. doi:10.33846/hn.v3i1.250
CrossRef - Politis SN, Colombo P, Colombo G, Rekkas DM. DoE in pharmaceutical development. Drug Dev Ind Pharm. 2017;43(6):889-901. doi:10.1080/03639045.2017.1291672
CrossRef - Fukuda IM, Pinto CFF, Moreira CDS, Saviano AM, Lourenço FR. DoE applied to QbD. Braz J Pharm Sci. 2018;54: e01006. doi:10.1590/s2175-97902018000001006
CrossRef - García-Manrique P, Machado ND, Fernández MA, et al. Effect of drug molecular weight on niosomes. Colloids Surf B Biointerfaces. 2020;186:110711. doi:10.1016/j.colsurfb.2019.110711
CrossRef - Gutiérrez G, Matos M, Barrero P, et al. Iron-entrapped niosomes for yogurt fortification. LWT Food Sci Technol. 2016;74:550-556. doi:10.1016/j.lwt.2016.08.025
CrossRef - Keshavshetti GG, Shirsand SB. Stability of topical niosomal gel. RGUHS J Pharm Sci. 2021;10(1):19-24. doi:10.26463/rjps.10_1_4
CrossRef - Mohanty D, Sahoo AK, Biswas A, et al. Transdermal naproxen niosomes. J Liposome Res. 2020;30(4):377-387. doi:10.1080/08982104.2019.1652646
CrossRef - Tejas PB, Tushar PR, Mehul PN, Suhagia BN. Ethosomes for transdermal delivery of thiocolchicoside. Der Pharm Lett. 2015;7(5):58-68.
- Ghafelehbashi R, Akbarzadeh I, Tavakkoli Yaraki M, et al. Cephalexin-loaded niosomes. Int J Pharm. 2019;569:118580. doi:10.1016/j.ijpharm.2019.118580
CrossRef
Accepted on: 14-11-2025
Second Review by: Dr. Saeed Kewedar
Final Approval by: Dr. Ali Elshafei









