A Systematic Review on Bovine Serum Albumin Nanoparticle: Preparation and Therapeutic Application
1Department of Pharmacy, Chaitanya Deemed to be University, Warangal, India
2School of Pharmacy, The Neotia University, Parganas (South), West Bengal. India
Corresponding Author: Email: praveenamr18@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3454
ABSTRACT:Nanoparticles made from protein-based materials are showing great potential as drug delivery systems because of their special characteristics, such as biodegradability, biocompatibility, and the capacity to hold different therapeutic agents. Among these substances, bovine serum albumin (BSA), a globular protein with superior drug-binding properties, stands out. The manufacturing methods and medicinal uses of BSA nanoparticles are explored in this comprehensive review. The preparation techniques covered in this discussion include self-assembly, desolvation, emulsification, thermal gelation, Nanoparticle albumin-bound (NAB) technology, and nanospray drying. Each of these techniques has unique benefits in terms of drug loading capacity, controlled release characteristics, and particle stability. The applications of BSA nanoparticles in medication delivery systems, cancer therapy, and medical imaging demonstrate their versatility. In drug delivery, they provide controlled release and targeted delivery, enhancing therapeutic efficacy while minimizing side effects. Their capacity to improve chemotherapeutic drug distribution and expedite gene therapy highlights their significance in cancer therapy. Furthermore, the diagnostic capacities are improved by the use of BSA nanoparticles in imaging modalities, including fluorescence and Magnetic Resonance Imaging (MRI). BSA nanoparticles have many benefits, but they also have drawbacks, such as immunogenicity, a low drug loading capacity, stability problems, and size fluctuation. For its wider clinical use, these drawbacks must be addressed. This review highlights the potential of BSA nanoparticles in revolutionising drug delivery and therapeutic strategies, underscoring the need for further research to address existing challenges and optimise their clinical utility.
KEYWORDS:Biodegradability; Bovine Serum Albumin; Desolvation; Emulsification; Nanoparticles
Introduction
Sub-nanoscale colloidal formations made of synthetic or semi-synthetic polymers are known as nanoparticles. Solid-core spherical nanoparticles with a material adsorbed onto the surface or incorporated into the matrix are called nanospheres. Nanoparticles are tiny particles whose sizes are expressed in nanometres (nm).1 Nanoparticles are usually smaller than 100 nm in at least one dimension. The size range of small particles is 100-2500 nanometres, whereas that of ultrafine particles is 1-100 nanometres. The size-related characteristics of tiny particles may or may not be present in nanoparticles.2 Individual molecules are typically not referred to as nanoparticles, even if they are the size of ultrafine particles.
When compared to conventional materials, they are flexible and can be adjusted to obtain certain characteristics or targeted traits like high surface area due to their ability to demonstrate auto-reactive stability and self-reassembly. Nanoparticles found their way into various fields such as material science, energy, biomedicine, and many more. Drug delivery is one of the most promising application areas of nanoparticles, as drugs can be delivered to particular human regions or cells using nanoparticles acting as carriers.3
It is possible to create nanoparticles with particular surface characteristics that
enable them to target diseased cells specifically while avoiding healthy ones, potentially increasing therapeutic efficacy and lowering side effects. Furthermore, nanoparticles can be engineered to release their contents in a regulated way, enabling long-term, continuous medication administration.4 Considering their versatility, researchers developed nanoparticles from various materials such as proteins, metals, and inorganic and organic polymers, which can be engineered and evaluated based on the characteristics of the drug, target environment, and disease. One such category of nanoparticles that have seen the limelight due to biocompatibility and potential in drug delivery is protein nanoparticles.
Protein nanoparticles have attracted significant attention for their impressive attributes, such as being non-toxic, non-immunogenic, biodegradable, nutritionally valuable, sourced abundantly from renewable sources, having exceptional drug binding capability, increased stability in storage and the body, and being easily scalable for production. Controlling the size, surface area, and properties of protein-based nanoparticles is the primary goal when developing them as a drug delivery vehicle. This guarantees that the drug-carrying nanoparticle system releases the active ingredient and carries the appropriate amount of drug to produce the intended pharmacological activity and part-specific action.5 Six different studies have looked into utilizing protein in the form of nanoparticles for drug delivery purposes since 2008. The synthesis of biocompatible nanoparticles is one of the most significant developments in drug delivery. Proteins and hybrid drug carriers made from proteins and synthetic polymers have shown versatility in the literature, resulting in the success of using nanoparticle formulation for nutraceutical protection and targeted delivery.6,7
Albumin is one protein that fits the bill for this use; its special qualities make it a great option for a drug transporter. Using albumin as a carrier has fewer negative effects compared to other carriers due to its high abundance in the body. Not only does the use of albumin minimize expenses during transportation, but it also lessens toxicity. Albumin and hydrophobic chemicals form a reversible connection that facilitates drug transport and release to the cell surface within the body. Because albumin contains a large number of functional groups, it has a high affinity for drug binding.8 Furthermore, various methods, including electrostatic interactions with negatively charged drugs (such as Ganciclovir), positively charged drugs, amphipathic compounds (like Doxorubicin), and hydrophobic drugs (such as Paclitaxel), facilitate the binding of drugs to nanoparticles via amino acid sequences and albumin structures. Specifically, the albumin protein exhibits a wide range of qualities, such as its non-immunogenicity, high rate of biodegradation that is metabolized in vivo to yield safe breakdown products, and water solubility.9 Moreover, the variety of drug-binding sites found in albumin nanoparticles increases the number of medications that can be incorporated into the matrix.7 The lipid matrix’s ability to reduce encapsulated content leakage during preparation and increase drug-loading efficiency—which in turn leads to the formation of micelles or other aggregates in aqueous media—improves self-assembly.10
Specifically, two tryptophan residues, Trp134 and Trp212, are located in bovine serum albumin (BSA) and reside in a hydrophobic pocket on the molecule’s surface in their native state.9 Due to this fact, polyphenols are more attracted to BSA as protein unfolding leads to the exposure of amine groups, thiol groups, and hydrophobic regions on the molecule’s surface, enhancing intramolecular binding and reducing hydrophobic interactions.10 Some references in academic work recommend using lipids combined with BSA,11 which can form micellar structures and other nanoparticles surrounded by protein shells.
Table 1: A compilation of clinically approved formulations related to albumin
| Brand Name | Company | Molecular Type | Clinical Application |
| Albiglutide | GlaxoSmithKline | Peptide HSA conjugate | Diabetes mellitus, Type 212 |
| Abraxane | Abraxis BioScience | Paclitaxel HSA-boundnanoparticles | Metastatic breast cancer, non-small cell lung cancer,adenocarcinoma of the pancreas13 |
| Nanocoll | Nycomed Amersham | 99mTc labelled HSA | SPECT scan for sentinel node localization in breastCancer14 |
| Optison | GE Healthcare | Perflutren Protein-type AMicrosphere Injectable Suspension | Contrast agent for ultrasound imaging15 |
| Jeanatope | IsoTex | Albumin iodinated I-125 serum | Determination of total blood and plasma volume16 |
| Megatope | IsoTex | Albumin iodinated I-131 serum | Determination of total blood and plasma volumes,cardiac output, cardiac and pulmonary blood volumes
and circulation times; protein turnover studies; heart and large vessel delineation; localization of placenta and celebral neospasms17 |
| Alburex | CSL Behring | Human Albumin | Restoration and maintenance of the circulating bloodVolume18 |
| Aralast NP | Baxter Healthcare Corporation | Human albumin | Emphysema19 |
| Pegintron | Merck | Human albumin | Cancers (leukaemia, melanoma, AIDS-related Kaposi’s sarcoma) and virus infections (e.g. chronic hepatitis B, chronic hepatitis C, condylomata acuminata)19 |
| Avonex | Biogen | Human albumin | Multiple sclerosis20 |
Type of Albumin
Animal, vegetable, or human sources are the main sources of albumin, which is easily supplied commercially. Egg white (OVA), BSA, and HSA are a few common types of albumin. These three forms of albumin have been widely used in research as well as in the food sector. Because of its inexpensive cost and simple purification procedure, BSA is frequently employed in research. Bovine spongiform encephalopathy is one of the losses from animal serum albumin that is commonly prevented using HSA. OVA is commonly utilized in the food sector for its ability to create foam and gel networks.[12] BSA and HSA, with a 76% sequence identity, share similar structures, conformation, and traits. The key difference lies in the placement of the tryptophan (Trp) residue. HSA has one Trp at position 214, while BSA has Trp-212 in subdomain IIA and an exposed Trp-134 in subdomain IB.21
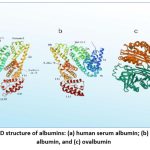 |
Figure 1: The 3D structure of albumins: (a)human serum albumin; (b)bovine serum albumin, and (c) ovalbumin.Click here to view Figure |
Human Serum Albumin (HSA)
Human serum contains the globular protein monomer known as HSA, which has a heart-shaped structure.14 HSA is a plasma protein responsible for transporting a range of substances, including fatty acids, steroids, thyroid hormones, endogenous ligands, metabolites, and drugs. The molecular weight of the substance is approximately 67 kDa, and it remains in the body for a period of 14 days. Additional features include the ability to withstand temperatures of up to 60°C for 10 hours, remaining stable in pH levels ranging from 4 to 9, and ensuring the appropriate volume of extracellular fluid is maintained. HSA, produced in the liver, consists of a lone polypeptide chain comprising 585 amino acids. It is rich in cysteine and low in tryptophan. HSA has gained significant interest as a drug carrier lately due to its numerous advantages, such as long half-life, excellent stability, easy purification, high drug delivery capacity, targeting ability, and good biocompatibility.22
Bovine Serum Albumin (BSA)
BSA is a spherical protein derived from cow serum, with an isoelectric point of 4.7 and a molecular weight of around 66.43 kDa. BSA, a water-soluble monomeric protein, contains 583 amino acid residues, 17 disulfide bonds, 9 bridge-created loops, 1 cysteine, and 8 pairs of disulfide bonds. BSA is the most commonly used protein in the pharmaceutical field, mainly employed as a protein model to examine protein aggregation and fibrillation.16 Nanofibers made from beta-amyloid (BSA) are used to create a crucial drug delivery system for treating diseases, such as cancer. This naturally existing biomolecule is also commonly employed in making gene medicines, antibodies, and drug delivery systems. Advantages compared to other carrier materials consist of a lack of toxicity, a lack of immunogenicity, and the ability to degrade naturally. Furthermore, BSA can pinpoint tumor cells in two ways: through interaction with secreted protein acidic and rich in cysteine (SPARC)proteins overexpressed on various tumor cells, and by attaching to the albumin gp60 receptor on vascular endothelial cells. This effective binding to albumin encourages drug-loaded nanoparticles to aggregate in the tumor cells’ stroma (Figure 2).23
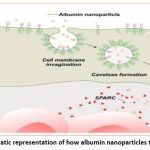 |
Figure 2: Diagrammatic representation of how albumin nanoparticles target tumor tissue.Click here to view Figure |
Ovalbumin (OVA)
The main spherical protein found in the whites of chicken eggs, called dietary protein OVA or egg albumin, is commonly used in creating food products. OVA weighs approximately 47 kDa and has a PI of 4.8. Additionally, it is a single-unit phosphoglycoprotein made up of 385 amino acids with an internal disulfide bond and four unbound sulfhydryl groups. OVA is used in drug delivery systems due to its cost-effectiveness and availability compared to other proteins, as well as its capacity to create gels, foams, and emulsions, and its responsiveness to pH and temperature.24
Rat Serum Albumin (RSA)
Rat Serum Albumin (RSA) is the primary plasma protein present in rats and functions similarly to other mammalian albumins by maintaining colloid osmotic pressure and transporting various biomolecules. With an approximate molecular weight of 67 kDa and an isoelectric point around 4.7–5.0, RSA is a single-chain globular protein composed of about 585 amino acids, stabilized by multiple intramolecular disulfide bonds that contribute to its structural integrity. It binds a wide range of endogenous and exogenous substances—such as fatty acids, hormones, dyes, and pharmaceutical agents—making it highly relevant for biomedical applications. RSA is widely used in preclinical research, particularly in pharmacokinetic, pharmacodynamic, and toxicology studies, because rats are one of the most common animal models used to predict drug behavior before human trials. Its structural similarity to human serum albumin and strong drug-binding capacity make it an important tool in investigating drug–protein interactions, nanoparticle formulations, drug release behavior, and toxicity profiles. Its availability, reliability, and biochemical characteristics make RSA a valuable protein for early-stage drug development and experimental drug delivery studies.25
Porcine Serum Albumin (PSA)
Porcine Serum Albumin (PSA) is the major serum protein derived from pigs and shares structural and functional characteristics similar to other mammalian albumins. With a molecular weight of approximately 66–67 kDa, PSA is a single-chain globular protein composed of multiple α-helical domains stabilized by intramolecular disulfide bonds, providing both flexibility and structural stability. Its primary physiological roles include maintaining osmotic balance, regulating fluid distribution, and binding a wide variety of biomolecules such as fatty acids, hormones, metabolites, and pharmaceutical agents. Due to its biochemical stability, strong ligand-binding capacity, and structural resemblance to human serum albumin, PSA is widely utilized in immunology for antibody generation, assay development, and immune response studies. In vaccine stabilization, PSA functions as an effective excipient to protect antigens during storage and transportation by preventing denaturation and aggregation, thereby improving vaccine shelf life and potency. Additionally, PSA plays a significant role in biomedical research, where it serves as a model protein for studying protein–drug interactions, carrier systems, nanoparticle formulations, and diagnostic assay development. Its availability, cost-effectiveness, and compatibility with biological systems make PSA an important tool in both experimental and applied biomedical sciences.26
Canine Serum Albumin (CSA)
Canine Serum Albumin (CSA) is the principal plasma protein found in dogs and plays essential physiological roles similar to other mammalian serum albumins. With an approximate molecular weight of around 66–67 kDa, CSA is a single-chain globular protein composed of multiple α-helical domains stabilized by several intramolecular disulfide bonds, giving it structural strength and high binding versatility. Its primary biological functions include maintaining colloid osmotic pressure, regulating fluid balance, and transporting a wide range of endogenous and exogenous substances such as hormones, fatty acids, metabolites, and therapeutic drugs. Due to its species-specific nature, CSA is widely utilized in veterinary medicine, particularly for formulating diagnostic kits, studying drug–protein interactions in dogs, and developing species-appropriate therapeutic formulations. It also serves as an important reference protein in diagnostic assays, including immunoassays, serological tests, and biochemical investigations aimed at detecting diseases, evaluating organ function, and monitoring systemic health in canines. Additionally, CSA is valuable in veterinary pharmacology and biomedical research as it aids in understanding pharmacokinetics, drug distribution, and potential toxicological effects specific to canine physiology. Its availability and relevance to dog-specific health applications make CSA a widely used protein in veterinary diagnostics and research.27
Equine Serum Albumin (ESA)
Equine Serum Albumin (ESA) is the major plasma protein found in horses and performs vital physiological functions similar to other mammalian serum albumins. With a molecular weight of approximately 66–67 kDa, ESA is a single-chain, globular protein composed of several α-helical domains that are stabilized by multiple intramolecular disulfide bonds, giving it strong structural stability and a broad binding capacity. ESA plays key biological roles in maintaining colloid osmotic pressure, regulating fluid balance, and transporting an array of molecules such as hormones, fatty acids, metabolites, and therapeutic agents. Its species-specific characteristics make ESA particularly useful in immunology, where it is employed in producing antibodies, developing immunoassays, and studying immune responses in equines. Additionally, ESA is widely used in disease-specific research, especially for conditions that commonly affect horses, such as equine infectious anaemia, equine influenza, and autoimmune disorders. It serves as a reliable model protein for assessing antigen–antibody interactions, evaluating vaccine performance, and understanding protein stability under various pathological conditions. Due to its biological relevance, high stability, and compatibility with equine physiology, ESA is an important tool in veterinary immunology, diagnostic test development, and research focused on equine health and disease mechanisms.28
Techniques used for the Preparation of BSA Nanoparticles
Desolvation Method
In the desolvation process, an aqueous solution of BSA(bovine serum albumin) is continuously stirred while organic solvents such as ethanol or methanol are added dropwise until the solution turns turbid. BSA has a high-water solubility; therefore, when an organic solvent is added, its phases will separate. Once the nanoparticles have adequately formed, they will stabilize and subsequently dissolve again in the aqueous solution.29 The process of coalescing and solidifying through a condensation reaction involves the aldehyde part of glutaraldehyde, the amino components in lysine residues, and the arginine parts in the guanidino side chain of albumin, resulting in the formation of the cross-linking. In 1978, Marty and his coworkers presented a new method. This technique entails gradually introducing an organic solvent, like methanol, ethanol, or regular salts, to the protein structure to produce a desolvation effect. This component will cause a change in the third protein structure. Once we reach a particular degree of desolvation, a protein cluster forms. This polymerization cluster cross-linked with a glutaraldehyde manufacturing factor will result in nanoparticles in the next stage. We should halt the framework before particles start to accumulate with the particular purpose of obtaining scattered nanoparticles, not in a bulk frame. This desolvation factor will lead to an expansion in framework turbidity estimation. Aggregation of particles will form independently with increasing background turbidity. We should use a resolving agent to prevent this kind of accumulation and create an ideal nano dispersion.30 The process of creating albumin nanoparticles employing a desolvating chemical is depicted in Figure 3.
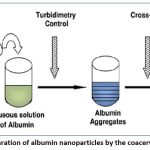 |
Figure 3: Preparation of albumin nanoparticles by the coacervation method.Click here to view Figure |
Emulsification
Several macromolecules are typically utilized to create nanoparticles; these primarily include proteins, such as gelatin, albumin, legumin, and vicillin, and polysaccharides, such as alginate or agarose. Due to their inherent qualities—such as biodegradability and biocompatibility—these materials are widely used in the arrangement of biomaterials. Of the macromolecules mentioned, gelatin and albumin have been used. For the creation of nanoparticles, there are two fundamental methods [31]. After rapidly mixing the oil phase (like cottonseed oil) with the albumin beads, the albumin nanospheres (0.3–1 μm) were created. The oil was heated to 175 to 180 °C for 10 minutes to achieve thermal balance. This blend was cooled and thinned with ethyl ether to help with centrifugation-induced separation. On the other hand, egg whites were mixed with cottonseed oil at 25 °C to create an emulsion in compound adjustment, before being denatured by being recombined with ether containing formaldehyde or 2,3-butadiene. Cefamandole nafate-stacked BSA nanoparticles were arranged by Crisante et al. using glutaraldehyde crosslinking without the use of a single emulsion agent. A constant phase consisting of cyclohexane with glutaraldehyde was mixed with the BSA fluid stage dropwise and homogenized quickly thereafter. Yang et al. created BSA nanoparticles to encapsulate the poorly soluble 10 Hydroxycamptothecin (HCPT) to enhance its stability, particularly in its active lactone form. They accomplished this through the implementation of a corrective process involving warm emulsion reformulation. The warm adjustment was utilized instead of concoction crosslinking in a study where the w/o single emulsion was drip-fed into castor oil at 140±5 °C.32 The potential of merging w/o/w multiple emulsion-based ovalbumin nanoparticles was examined in mucosal immunizations. The presence of light indicators in the nanoparticles indicated their absorption and subsequent internal movement within macrophage cell populations.
Thermal Gelation Method
Thermal gelation involves a sequence of processes, including unfolding due to heat, as well as interactions between proteins such as hydrophobic associations, disulfide-sulfhydryl exchange, and hydrogen bonding.33 In a study conducted by Yu et al., a method known as warm gelation was utilized to produce nanogels with a spherical core-shell structure, measuring approximately 100 nm in size. When ovalbumin and lysozyme were mixed at pH 5.3, the pH of the mixture rose to 10.3, leading to the mixture being settled and heated as a result. Additional information has been offered about how BSA gels when heated. BSA-dextran-chitosan nanoparticles were formed by heating a blend of chitosan and BSA-dextran conjugates, which are biocompatible. Due to the electrostatic attraction between chitosan and BSA, BSA molecules agglomerate and form the core of the nanoparticles with the help of chitosan chains, resulting in partial entrapment. The dextran and chitosan left over continue across the entire nanoparticle shell. Once the pH of the mixture is increased to 7.4, the nanoparticles can effectively encapsulate doxorubicin through dispersion, enabled by the inherent electrostatic and hydrophobic interactions between them.34
Nab-technology
One tactic used to plan albumin NPs is to seize innovation, which may also be utilized to incorporate the best lipophilic drugs into the NPs. The drug and BSA combined to form a thin dissolvable. Drug albumin NPs were added by passing the mixture through a fly spot while it was heavily weighted. The administered NPs have a size range of around 100–200 nm.35 A recent article described the creation of an LHNP structure by combining lapatinib chloroform, and ethanol with BSA. Next, the procedure underwent intense shear forces to create a rough emulsion. To finish the last step of emulsification, the emulsion was passed through a smaller fluidiser, then the NPs suspension was sifted and disappeared. Following that, the NPs were preserved, freeze-dried, and hardened. The drug may combine with the ethanol and chloroform before the emulsification process. Using this method, paclitaxel—the first drug based on nanotechnology to receive FDA approval—was created and is now used to treat metastatic breast tumours as well as other types of growths.36 According to preclinical and clinical experiments, this product, which has particles roughly 130 nm wide, exhibits more drug tumour accumulation and superior antitumor viability when compared to standard treatments.37
Self-Assembly
A novel technique for creating albumin nanoparticles is the self-assembly method. By employing this method, the primary amine group located on the protein’s outer surface is eliminated; alternatively, albumin’s hydrophobic properties are enhanced through the breaking of disulfide bonds. The self-assembly of molecules with lipophilic substances can create albumin nanoparticles. The addition of hydrophobic medications or reducing agents during the process helps the hydrophobically modified albumin build up in the core. Dithiothreitol, cysteine, and β-mercaptoethanol are often utilized as reducing agents. The self-assembly method and the desolvation process share several phases. Through the use of this self-assembly technique, albumin nanoparticles with active targeting qualities can more effectively preserve protein function.38, 39
Nanoparticle Albumin-Bound (NAB) Technology
The approval of the Nab-Paclitaxel (AbraxaneTM) by the US FDA resulted in the extensive use of the NAB Technology method in creating anticancer drug delivery systems. This method is effective for distributing hydrophobic anticancer drugs in a protein matrix for optimal efficiency. The NAB drug is formed by mixing a chloroform/acetone+acetone organic solution with an albumin solution containing a hydrophobic medication with low rotation to create a rough emulsion. A high-pressure homogenization process is used to produce a nanoemulsion. The organic solvent in the colloid is removed using rotary evaporation in a vacuum. After being produced, the nanoparticles are either filtered (using a syringe filter) or centrifuged, and the resulting liquid is freeze-dried to form powdered nanoparticles.40
Nanospray Drying
The process of turning liquid ingredients into powder nanoparticles is called “nanospray drying” in the pharmaceutical business. Because of the albumin nanoparticles’ long shelf life, high EE, and narrow size distribution, this approach has various advantages. This method reduces the risk of decay by being effective at low temperatures and relying less on the solubility of drugs and albumin. The spray drying process consists of four stages: atomization of material, drying with gas, formation of particles, and collection of particles.41 In this method, a liquid sample is sprayed into a chamber, where heated nitrogen and carbon dioxide gases are directed towards the spray from a nozzle to finish the process. Surfactant is included in the liquid albumin sample to stabilize the polymer particles. The electrodes positioned at the chamber’s bottom gather nanoparticles. These electrodes provide an electrostatic charge to the sprayed droplets, which causes them to move towards the bottom of the chamber.42 This method produces small-scale protein particles quickly and economically.
Applications of BSA Nanoparticles
In the world of medicine, nanoparticles have become a game-changing technological advancement, providing fresh approaches to age-old problems with therapeutic applications and drug delivery. Of them, Bovine Serum Albumin (BSA) nanoparticles have drawn a lot of interest because of their biodegradability, biocompatibility, and range of uses.43 Because of its advantageous characteristics, bovine serum albumin—a protein that is frequently present in blood plasma—makes a great option for the creation of nanoparticles. This paper investigates the medical uses of BSA nanoparticles, emphasizing their function in medication administration, cancer treatment, and imaging.
Drug Delivery Systems
The use of BSA nanoparticles in drug delivery systems is growing as they can trap different therapeutic agents.44 The key benefits of employing BSA nanoparticles for drug delivery are their strong stability, controlled release characteristics, and ability to target specific cells or tissues.
Controlled Drug Release
BSA nanoparticles can be engineered to release drugs in a controlled manner. The protein’s structure allows for the formation of a nanocarrier that can encapsulate drugs through non-covalent interactions. This encapsulation can protect drugs from degradation and provide a sustained release profile. This feature is particularly beneficial for drugs that require prolonged exposure to achieve therapeutic efficacy, such as in chronic diseases.44
Targeted Delivery
Targeted drug delivery is another significant advantage of BSA nanoparticles. By modifying the surface of BSA nanoparticles with specific ligands, researchers can direct these nanoparticles to target cells or tissues.45 For instance, conjugating BSA nanoparticles with antibodies or peptides that bind specifically to cancer cells enables the delivery of anticancer drugs directly to tumour sites, minimizing side effects and improving therapeutic outcomes.
Cancer Therapy
The application of BSA nanoparticles in cancer therapy represents one of the most promising areas of research. These nanoparticles offer several mechanisms to enhance the efficacy of cancer treatments.46
Chemotherapy Enhancement
BSA nanoparticles can be loaded with chemotherapeutic agents and used to deliver these drugs more efficiently to cancer cells.47 The enhanced permeability and retention effect of nanoparticles can result in increased drug accumulation in tumor tissues compared to conventional chemotherapy, leading to improved therapeutic responses and reduced systemic toxicity.
Gene Therapy
Gene therapy is another innovative application of BSA nanoparticles in cancer treatment. By encapsulating nucleic acids such as plasmid DNA or small interfering RNA (siRNA), BSA nanoparticles can deliver genetic material into cancer cells to induce cell death or inhibit tumor growth.48 This approach can be combined with conventional therapies to enhance overall treatment efficacy.
Imaging and Diagnostic Applications
BSA nanoparticles are also making strides in the field of medical imaging and diagnostics. Their ability to be functionalized with imaging agents allows for enhanced imaging capabilities.
Magnetic Resonance Imaging (MRI)
BSA nanoparticles can be conjugated with contrast agents to improve MRI imaging. The high surface area and biocompatibility of BSA nanoparticles make them ideal carriers for contrast agents, leading to improved image contrast and resolution.49 This enhanced imaging capability facilitates better diagnosis and monitoring of diseases.
Fluorescence Imaging
For fluorescence imaging, BSA nanoparticles can be labeled with fluorescent dyes. These fluorescent BSA nanoparticles can be used to visualize and track biological processes at the cellular or tissue level, aiding in early disease detection and therapeutic monitoring.50
Salient Features of BSA Nanoparticles
Biocompatibility: BSA is a natural protein found in blood plasma, making it biocompatible and well-tolerated by the body. When used as a carrier, BSA nanoparticles are less likely to cause adverse reactions.51
Abundance and Availability: BSA is readily available and abundant, which simplifies its production for drug delivery applications. This availability contributes to its widespread use.52
Non-Toxicity: BSA is non-toxic, making it safe for use in pharmaceutical applications. This is crucial for minimizing harm to patients.53
Non-Immunogenicity: BSA nanoparticles do not trigger significant immune responses, reducing the risk of rejection or adverse effects. This property is essential for long-term drug delivery.54
Biodegradability: BSA degrades naturally over time, ensuring that it doesn’t accumulate in the body. This feature aligns well with sustainable drug delivery systems.55
High Water Solubility: BSA dissolves readily in water, allowing efficient drug loading and release from the nanoparticles. Water solubility is crucial for effective drug delivery.56
Selective Conduction Ability: BSA nanoparticles can target specific cells or tissues due to their ability to bind to certain receptors. This selectivity enhances drug delivery precision.57
Current Research on BSA
In this study, researchers encapsulated a flavonoid (morin hydrate) in BSA NPs using the desolvation method. The resulting BSA-MH nanoparticles had ~90 nm size, good stability, improved solubility and bioavailability of the drug, and enhanced antioxidant and anticancer activity compared to free morin. (Singh S.K et al 2025)
This work combined computational modelling (molecular docking, DFT) with experimental synthesis to create BSA-coated magnetic nanoparticles loaded with quercetin (a flavonoid). The BSA coating improved drug loading (~27.5%), and the system showed controlled, pH-sensitive release (much higher release in acidic conditions like tumour microenvironment), demonstrating a promising platform for targeted cancer therapy. (Hashemi N et al 2025)
Conclusion
Bovine serum albumin (BSA) nanoparticles have numerous benefits for drug delivery systems, such as excellent drug-binding capacity, biodegradability, and biocompatibility. Different fabrication methods, including self-assembly, emulsification, thermal gelation, NAB technology, desolvation, and nanospray drying, enable these nanoparticles to be tailored for certain medicinal applications. BSA nanoparticles, which offer targeted delivery, controlled release, and improved diagnostic capabilities, have potential use in cancer treatment, drug delivery, and medical imaging. Despite their potential, issues like immunogenicity, restricted drug loading capacity, stability concerns, and size variations need to be dealt with. To overcome these obstacles and fulfil the full clinical potential of BSA nanoparticles, ongoing research and innovation are essential. In conclusion, BSA nanoparticles have a lot of potential for use in medicine down the road. To make these nanoparticles suitable for clinical application, more research and development are needed. This could result in better patient outcomes, more effective therapies, and fewer side effects in the field of nanomedicine.
Acknowledgment
The authors are thankful to the Institutional Animal Ethical Committee of Chaitanya Deemed to be University, Hanamkonda, Telangana, for providing facilities to carry out the research-related literature review in the University library and providing me with e-Journal access. The authors acknowledge the chairman and principal of Anwarul Uloom College of Pharmacy in Hyderabad, Telangana, for obtaining permission to conduct the research. The author is also thankful to The Neotia University, Kolkata, for allowing us to carry out some evaluations for the research process.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Sameena Begum contributed to Conceptualisation, investigation, data curation, and writing the original draft.
Niranjan Panda contributed to writing, review & editing.
Ch. Praveena acted as supervisor.
References:
- Anand KV, Mallakpour S, Hussain CM, editors. Antimicrobial Nanosystems: Fabrication and Development. Elsevier; 2023.
- Khan KA, Rasel SR. The present scenario of nanoparticles in the world. Int J Adv Res Innov Ideas Educ. 2019;5(2):462-471.
- Zahin N, Anwar R, Tewari D, Kabir MT, Sajid A, Mathew B, Uddin MS, Aleya L, Abdel-Daim MM. Nanoparticles and its biomedical applications in health and diseases: special focus on drug delivery. Environmental Science and Pollution Research. 2020 Jun;27(16):19151-68.
CrossRef - Yusuf A, Almotairy AR, Henidi H, Alshehri OY, Aldughaim MS. Nanoparticles as drug delivery systems: a review of the implication of nanoparticles’ physicochemical properties on responses in biological systems. 2023;15(7):1596. doi:10.3390/polym15071596
CrossRef - Petrak K. Essential properties of drug-targeting delivery systems. Drug discovery today. 2005 Dec 1;10(23-24):1667-73.
CrossRef - Pan K, Chen H, Baek SJ, Zhong Q. Self-assembled curcumin–soluble soybean polysaccharide nanoparticles: physicochemical properties and in vitro anti-proliferation activity against cancer cells. Food Chem. 2018;246:82-89. doi:10.1016/j.foodchem.2017.11.066
CrossRef - Ribeiro LNM, Alcantara ACS, Franz-Montan M, Couto VM, Nista SVG, de Paula E. Nanostructured organic-organic bio-hybrid delivery systems. Biomed Appl Nanopart. 2019;341-374. doi:10.1016/B978-0-12-814003-1.00017-4
CrossRef - Sleep D. Albumin and its application in drug delivery. Expert opinion on drug delivery. 2015 May 4;12(5):793-812.
CrossRef - Elblbesy MA. Hemocompatibility of albumin nanoparticles as a drug delivery system—an in vitro study. J Biomater Nanobiotechnol. 2016;7:64-71. doi:10.4236/jbnb.2016.72007
CrossRef - Sadeghi R, Moosavi-Movahedi AA, Emam-Jomeh Z, Kalbasi A, Razavi SH, Karimi M, Kokini J. The effect of different desolvating agents on BSA nanoparticle properties and encapsulation of curcumin. J Nanopart Res. 2014;16:2565. doi:10.1007/s11051-014-2565-8
CrossRef - Gupta R, Jain V, Nagar JC, Ansari A, Sharma K, Sarkar A, Khan MS. Bioavailability enhancement techniques for poorly soluble drugs: a review. Asian J Pharm Res Dev. 2020;8:75-78. doi:10.22270/ajprd.v8i2.875
CrossRef - Sharma AK, Thanikachalam PV, Rajput SK. Albiglutide: Is a better hope against diabetes mellitus?. Biomedicine & Pharmacotherapy. 2016 Feb 1;77:120-8.
CrossRef - Cho H, Jeon SI, Ahn CH, Shim MK, Kim K. Emerging albumin-binding anticancer drugs for tumor-targeted drug delivery: current understandings and clinical translation. Pharmaceutics. 2022 Mar 28;14(4):728.
CrossRef - Gommans GM, Gommans E, Van der Zant FM, Teule GJ, Van der Schors TG, De Waard JW. 99mTc Nanocoll: A radiopharmaceutical for sentinel node localisation in breast cancer—In vitro and in vivo results. Applied radiation and isotopes. 2009 Sep 1;67(9):1550-8.
CrossRef - Hawley JJ, Allen SL, Thompson DM, Schwarz AJ, Tranquart FJ. Commercially available ultrasound contrast agents: factors contributing to favorable outcomes with ultrasound-mediated drug delivery and ultrasound localization microscopy imaging. Investigative Radiology. 2024 Dec 13:10-97.
- Valeri CR, Cooper AG, Pivacek LE. Limitations of measuring blood volume with iodinated I 125 serum albumin. Archives of Internal Medicine. 1973 Oct 1;132(4):534-8.
CrossRef - Lee J. Electronic Poster Viewing–80 selected posters EP1.
- Simon T, Schumann P, Bieri M, Schirner K, Widmer E. Hyperoncotic human albumin solutions for intravenous fluid therapy: Effectiveness of pathogen safety and purification methods, and clinical safety. Biosafety and Health. 2023 Feb 25;5(01):21-9.
CrossRef - PROFILE AR. Alpha-Antitrypsin− Alpha Therapeutic Corporation.
- Rudick RA, Cookfair DL, Simonian NA, Ransohoff RM, Richert JR, Jacobs LD, Herndon RM, Salazar AM, Fischer JS, Granger CV, Goodkin DE. Cerebrospinal fluid abnormalities in a phase III trial of Avonex®(IFNβ-1a) for relapsing multiple sclerosis. Journal of neuroimmunology. 1999 Jan 1;93(1-2):8-14.
CrossRef - Bourassa P, Kanakis CD, Tarantilis P, Pollissiou MG, Tajmir-Riahi HA. Resveratrol, genistein, and curcumin bind bovine serum albumin. J Phys Chem B. 2010;114:3348-3354. doi:10.1021/jp9108303
CrossRef - Clark AH, Judge FJ, Richards JB, Stubbs JM, Suggett A. Electron microscopy of network structures in thermally-induced globular protein gels. Int J Pept Protein Res. 2009;17:380-392. doi:10.1111/j.1399-3011.1981.tb00461.x
CrossRef - Xie S, Wang S, Zhao B, Han C, Wang M, Zhou W. Effect of PLGA as a polymeric emulsifier on preparation of hydrophilic protein-loaded solid lipid nanoparticles. Colloids Surf B Biointerfaces. 2008;67:199-204. doi:10.1016/j.colsurfb.2008.02.012
CrossRef - Elzoghby AO, Samy WM, Elgindy NA. Albumin-based nanoparticles as potential controlled release drug delivery systems. J Control Release. 2012;157(2):168-182. doi:10.1016/j.jconrel.2011.07.031
CrossRef - Belinskaia DA, Voronina PA, Goncharov NV. Integrative role of albumin: evolutionary, biochemical and pathophysiological aspects. Journal of evolutionary biochemistry and physiology. 2021 Nov;57(6):1419-48.
CrossRef - Gull N, Sen P, Khan RH. Interaction of bovine (BSA), rabbit (RSA), and porcine (PSA) serum albumins with cationic single-chain/gemini surfactants: a comparative study. Langmuir. 2009 Oct 6;25(19):11686-91.
CrossRef - Ketrat S, Japrung D, Pongprayoon P. Exploring how structural and dynamic properties of bovine and canine serum albumins differ from human serum albumin. Journal of Molecular Graphics and Modelling. 2020 Jul 1;98:107601.
CrossRef - Boneschans M. Purification strategies for equine Chorionic Gonadotropin.
- Bolaños K, Kogan MJ, Araya E. Capping gold nanoparticles with albumin to improve their biomedical properties. Int J Nanomed. 2019;14:6387-6406. doi:10.2147/IJN.S220073
CrossRef - Kudarha RR, Sawant KK. Albumin-based versatile multifunctional nanocarriers for cancer therapy: fabrication, surface modification, multimodal therapeutics and imaging approaches. Mater Sci Eng C. 2017;81:607-626. doi:10.1016/j.msec.2017.06.018
CrossRef - Tao YH, Wang QR, Sheng W, Zhen Y. The development of human serum albumin–based drugs and relevant fusion proteins for cancer therapy. Int J Biol Macromol. 2021;187:24-34. doi:10.1016/j.ijbiomac.2021.06.107
CrossRef - Raoufinia R, Mota A, Keyhanvar N, Safari F, Shamekhi S, Abdolalizadeh J. Overview of albumin and its purification methods. Adv Pharm Bull. 2016;6(4):495-507. doi:10.15171/apb.2016.063
CrossRef - Meng R, Zhu H, Wang Z, Hao S, Wang B. Preparation of drug-loaded albumin nanoparticles and its application in cancer therapy. J Nanomater. 2022;2022:1-12. doi:10.1155/2022/1234567
CrossRef - Karimi M, Bahrami S, Ravari SB, et al. Albumin nanostructures as advanced drug delivery systems. Expert Opin Drug Deliv. 2017;13(11):1609-1623. doi:10.1080/17425247.2017.1380678
CrossRef - Li Q, Lu JH, Yao H, Zhang WG, F. Ciprofloxacin-loaded bovine serum albumin microspheres: preparation and drug-release in vitro. J Microencapsul. 2001;18(6):825-829. doi:10.1080/02652040110050117
CrossRef - Kim TH, et al. Preparation and characterization of water-soluble albumin-bound curcumin nanoparticles with improved antitumor activity. Int J Pharm. 2011;403(1-2):285-291. doi:10.1016/j.ijpharm.2010.11.048
CrossRef - Jahanshahi M, Babaei Z. Protein nanoparticle: a unique system as drug delivery vehicles. Afr J Biotechnol. 2008;7(25):4951-4957. doi:10.5897/AJB08.1661
- Yang L, et al. Preparation, characterization, and biodistribution of the lactone form of 10-hydroxycamptothecin (HCPT)-loaded bovine serum albumin (BSA) nanoparticles. Int J Pharm. 2007;340(1-2):163-172. doi:10.1016/j.ijpharm.2007.03.056
CrossRef - Bronich TK, et al. Polymer micelle with cross linked ionic core. J Am Chem Soc. 2005;127(23):8236-8237. doi:10.1021/ja0512421
CrossRef - Qi J, et al. Nanoparticles with dextran/chitosan shell and BSA/chitosan core—doxorubicin loading and delivery. Int J Pharm. 2010;393(1-2):177-185. doi:10.1016/j.ijpharm.2010.03.023
CrossRef - Elzoghby AO, Samy WM, Elgindy NA. Albumin-based nanoparticles as potential controlled release drug delivery systems. J Control Release. 2012;157(2):168-182. doi:10.1016/j.jconrel.2011.07.031
CrossRef - Battogtokh G, Kang JH, Ko YT. Long circulating self-assembled cholesteryl albumin nanoparticles enhance tumor accumulation of hydrophobic anticancer drug. Eur J Pharm Biopharm. 2015;96:96-105. doi:10.1016/j.ejpb.2015.08.003
CrossRef - Kratz F. Albumin as a drug carrier: design of prodrugs, drug conjugates, and nanoparticles. J Control Release. 2008;132(3):171-183. doi:10.1016/j.jconrel.2008.05.006
CrossRef - Safavi MS, Shojaosadati SA, Yang HG, et al. Reducing agent-free synthesis of curcumin-loaded albumin nanoparticles by self-assembly at room temperature. Elsevier B.V.
CrossRef
- Wang Y, Chen S, Yang X, Zhang S, Cui C. Preparation optimization of bovine serum albumin nanoparticles and its application for siRNA delivery. Drug Des Devel Ther. 2021;15:1531-1547. doi:10.2147/DDDT.S303113
CrossRef - Thao LQ, Byeon HJ, Lee C, et al. Pharmaceutical potential of tacrolimus-loaded albumin nanoparticles having targetability to rheumatoid arthritis tissues. Int J Pharm. 2016;497(1-2):268-276. doi:10.1016/j.ijpharm.2015.12.043
CrossRef - Loureiro A, Azoia G, Gomes A, Cavaco-Paulo A. Albumin-based nanodevices as drug carriers. Curr Pharm Des. 2016;22(10):1371-1390. doi:10.2174/1381612822666160204153045
CrossRef - Mohanty D, Alsaidan OA, Zafar A, et al. Development of atomoxetine-loaded NLC in situ gel for nose-to-brain delivery: optimization, in vitro, and preclinical evaluation. 2023;15(7):1985. doi:10.3390/pharmaceutics15071985
CrossRef - Xu X, Hu J, Xue H, et al. Applications of human and bovine serum albumins in biomedical engineering: a review. Int J Biol Macromol. 2023;242:126914. doi:10.1016/j.ijbiomac.2023.126914
CrossRef - Panda N, Charan Panda K, Reddy AV, Reddy GVS. Process optimization, formulation, and evaluation of hydrogel {guar gum-g-poly(acrylamide)}-based doxofylline microbeads. Asian J Pharm Clin Res. 2014;7(3):60-65.
- Prashanth GK, Sathyananda HM, Prashanth PA, et al. Controlled synthesis of Ag/CuO nanocomposites: evaluation of their antimycobacterial, antioxidant, and anticancer activities. Appl Phys A. 2022;128(7):614. doi:10.1007/s00339-022-05783-0
CrossRef - Solanki R, Rostamabadi H, Patel S, Jafari SM. Anticancer nano-delivery systems based on bovine serum albumin nanoparticles: a critical review. Int J Biol Macromol. 2021;193:528-540. doi:10.1016/j.ijbiomac.2021.10.056
CrossRef - Esim O, Gedik ME, Dogan AL, Gunaydin G, Hascicek C. Development of carboplatin-loaded bovine serum albumin nanoparticles and evaluation of its effect on an ovarian cancer cell line. J Drug Deliv Sci Technol. 2021;64:102655. doi:10.1016/j.jddst.2021.102655
CrossRef - Prajapati R, Somoza A. Albumin nanostructures for nucleic acid delivery in cancer: current trend, emerging issues, and possible solutions. Cancers (Basel). 2021;13(14):3454. doi:10.3390/cancers13143454
CrossRef - Li H, Yan K, Shang Y, et al. Folate–bovine serum albumin functionalized polymeric micelles loaded with superparamagnetic iron oxide nanoparticles for tumor targeting and magnetic resonance imaging. Acta Biomater. 2015;15:117-126. doi:10.1016/j.actbio.2014.12.003
CrossRef - Begum S., Panda N., Praveena C., Design Characterisation Optimisation of Lenalidomide-Loaded Bovine Serum Albumin Nanoparticles for Myeloma Therapy. International Journal of Applied Pharmaceutics,2025;18(1). https://doi.org/10.22159/ijap.2026v18i1.55623.
- Zhang Y, Yue X, Kim B, Yao S, Bondar MV, Belfield KD. Bovine serum albumin nanoparticles with fluorogenic near-IR-emitting squaraine dyes. ACS Appl Mater Interfaces. 2013;5(17):8710-8717. doi:10.1021/am402361w
CrossRef
Accepted on: 05-12-2025
Second Review by: Dr. Ahmed Y Radeef
Final Approval by: Dr. Eugene A. Silow









