A Review- Nanoparticle-Mediated Modulation of Tumor Hypoxia: Enhancing Cancer Immunotherapy and Therapeutic Efficacy
1Department of Pharmaceutical Chemistry, KVPS’s Maharani Ahilyabai Holkar College of Pharmacy, Shirpur, India.
2Department of Pharmaceutical Chemistry, Indore institute of pharmacy, Pithampur road, Indore, India.
3Department of Chemistry and Environmental Science, New Jersey Institute of Technology, Newark, USA.
4Department of Chemistry, Lamar University, Beaumont, Texas, USA.
5Exemplify Biopharma Inc. East Windsor, New Jersey, USA.
Corresponding Author:E-mail: rakeshdhole94@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3457
ABSTRACT:Tumor hypoxia, characterized by insufficient oxygen supply within the tumor microenvironment TME plays a key role in helping tumors grow, avoiding the body's immune system, and becoming resistant to treatments like chemotherapy, photodynamic therapy, and radiation. High levels of certain metabolites that are made without oxygen, such as adenosine and lactate, weaken the ability of cytotoxic T lymphocytes (CTLs) to attack cancer cells. These metabolites also reduce the production of interferon-gamma (IFN-γ) and create an environment that supports immune-suppressing cells like regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs). Nanoparticle-based strategies have emerged as promising tools to alleviate hypoxia through various mechanisms, including catalytic oxygen generation, direct oxygen delivery, and targeted modulation of hypoxia-driven pathways. By enhancing oxygen availability and normalizing the aberrant TME, these nanoplatforms provide a promising avenue to overcome hypoxia-induced therapeutic resistance. This review summarizes recent progress in nanoparticle-mediated hypoxia modulation in the TME and highlights perspectives for future clinical translation in cancer immunotherapy and combined treatment approaches.
KEYWORDS:Tumor hypoxia; Tumor microenvironment; Nanoparticles; Oxygen delivery; immunosuppression; Cytotoxic T lymphocytes; Photodynamic therapy; Radiotherapy; therapeutic resistance; Nanomedicine
Introduction
The tumor microenvironment is made up of many different types of cells, like immune cells, fibroblasts, endothelial cells, inflammatory cells, and lymphocytes. It also includes the extracellular matrix, blood vessels, and chemokines. These components work together in a complex way. The immune cells in this environment help control both the body’s natural immune response and the more specialized immune response. Some immune cells, like macrophages and dendritic cells, can either help the tumor grow or fight against it, depending on how they interact with other cells and the signals from chemokines in the microenvironment. The adaptive immune system, which is activated by the innate immune system, can identify and destroy cancer cells. This makes it one of the strongest ways the body can clear tumors.
Cancer-associated fibroblasts (CAFs), derived from activated fibroblasts, contribute substantially to the ECM framework and influence tumor progression and therapeutic outcomes through structural remodeling and secretion of bioactive molecules.¹ Additionally, the TME is characterized by aberrant vasculature with irregular morphology and functionality, leading to hypoxia, which disrupts normal microenvironmental function, fosters tumor progression, and reduces treatment efficacy.² Interactions between tumor cells and TME components directly regulate tumor growth, metastatic potential, drug penetration, and resistance.³ Consequently, modulation of the TME—a new approach—has become a hopeful way to make cancer immunotherapy more effective.⁴⁻⁵
Targeted techniques addressing ligand–receptor communication at the cell surface and downstream signaling pathways within the TME have shown potential in improving therapeutic durability.⁶ TME-modulating agents such as the CTLA-4 antagonist plerixafor,⁷ anti-interleukin-6 monoclonal antibody siltuximab,⁸ anti-CCL2 antibody carlumab,⁹ and integrin α4β1/α4β7 antagonist natalizumab¹⁰ have demonstrated preliminary efficacy. In addition, the USFDA (FDA) granted accelerated approval to pembrolizumab, an anti-PD-1 checkpoint inhibitor, in 2017.¹¹ However, not all patients benefit, and many experience severe adverse events such as gastrointestinal, hematologic, and endocrine disorders, arthritis, rash, neuropathy, and acute kidney injury.¹²⁻¹⁴
Chimeric antigen receptor T-cell (CAR-T)
Chimeric antigen receptor T-cell (CAR-T) therapy targeting CD19 has shown remarkable efficacy in acute lymphoblastic leukemia but limited results in melanoma¹⁵ and non-small-cell lung cancer,¹⁶ alongside substantial toxicities. In contrast, nanoparticles offer advantages such as prolonged retention time, targeted delivery, and reduced systemic toxicity.¹⁷ Specifically, they can remodel the immunosuppressive TME by targeting its major cellular and molecular components. Hypoxia, a consequence of abnormal vasculature and rapid tumor proliferation, fosters immunosuppression by recruiting regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs), and by promoting the secretion of vascular endothelial growth factor (VEGF) and transforming growth factor-β (TGF-β), which inhibit DC function, polarize macrophages toward an M2 phenotype, and drive fibrosis.¹⁸
Nanoparticles engineered with specific functionalities can target these elements to shift the TME toward an immunosupportive phenotype, thereby enhancing immunotherapy efficacy.¹⁹ Passive accumulation in tumors via the enhanced permeability and retention (EPR) effect occurs due to leaky tumor vasculature and impaired lymphatic drainage.²⁰ Structural modifications, such as ligand conjugation, can further improve tumor-specific delivery and TME modulation.²¹ Moreover, multifunctional nanoplatforms can co-deliver multiple therapeutic agents. Several clinically approved nanodrugs, including Doxil²² and Abraxane,²³ have demonstrated clinical success.
Targeting Dendritic Cells (DCs)
Dendritic cells (DCs), which come from the bone marrow, are some of the strongest antigen-presenting cells (APCs) in the immune system.²¹ Normally, they travel through the blood, but when they get activated, they move to nearby lymph nodes. There, they meet with T cells to start and control immune responses. DCs act as a key link between the body’s first line of defense and its adaptive immune system. They take in both proteins made inside the cell and those from outside, break them into smaller pieces called antigenic peptides, and show these pieces on their surface using MHC molecules. This allows T cells to recognize them and start a targeted immune response Fig.1 [(A), (B)].
Among different types of DCs, conventional DCs (cDCs) are very important in fighting cancer. These cells pick up antigens linked to tumors in the tumor environment and take them to lymph nodes near the tumor. There, they activate T cells that can kill cancer cells.²² However, in the tumor environment, cDCs often don’t work well because of substances that stop immune responses, low oxygen levels, and things like lactic acid, which all prevent DCs from maturing and properly showing antigens.²³
Nanoparticle-based strategies offer a promising approach to overcome these inhibitory mechanisms. By functionalizing nanoparticles with DC-targeting ligands, it is possible to enhance antigen delivery, promote DC maturation, and restore their ability to initiate robust antitumor immune responses (see Table 1 [(A), (B)] for representative applications). Such targeted nanocarriers not only improve the specificity of therapeutic delivery but also facilitate synergistic effects when combined with other immunotherapeutic modalities.
Figure 1. shows Schematic representation role of the dendritic cells (DCs) in tumor immunity and the functions of nanoparticles (NPs) in modulating DC-mediated immune responses. Upon encountering endogenous or exogenous antigens, immature DCs undergo activation and maturation.²¹ They process these antigens into peptide pieces of cancer cells shown on the surface of MHC molecules to new T cells. When these T cells interact with the cancer pieces, they become activated. This activation causes the T cells to turn into cytotoxic T lymphocytes (CTLs). These CTLs can kill cancer cells directly or by using interferon-gamma (IFN-γ) to trigger other immune responses.²²
NP-based strategies enhance this process through multiple mechanisms: NPs functionalized with tumor antigens and immune adjuvants facilitate targeted antigen delivery to DCs. These NPs promote DC maturation and CTL activation by enhancing antigen presentation and providing adjuvant signals.²³ Upon antigen presentation, CD4⁺ helper T cells and CD8⁺ CTLs undergo clonal expansion and acquire effector functions, including IFN-γ secretion and direct tumor cell lysis. Incorporating Toll-like receptor (TLR) ligands within NP formulations induces potent innate and adaptive immune responses.²⁴ In addition to adjuvant supplementation, certain nanomaterials—such as Fe₃O₄ magnetic nanoparticles—can act as intrinsic nano-immunopotentiators, directly promoting DC maturation and enhancing antitumor immunity.²⁵
Table 1: Summary of the application of nanoparticles.
| Nanoparticle Type | Target in TME | Mechanism of Action | Example / Drug | Therapeutic Outcome |
| Liposomes | Tumor vasculature, CAFs | Encapsulation of chemotherapeutics; passive targeting via EPR effect | Doxil(PEGylated liposomal doxorubicin) | Prolonged circulation, reduced cardiotoxicity, improved tumor accumulation 26 |
| Albumin-bound nanoparticles | Tumor cells, ECM | Exploits albumin transport pathways; enhanced penetration in stroma-rich tumors | Abraxane (albumin-bound paclitaxel) | Higher intratumoral drug concentration, improved response in breast and lung cancer 27 |
| Polymeric nanoparticles | DCs, macrophages | Controlled release of immune stimulants; TME immune reprogramming | PLGA nanoparticles carrying CpG oligonucleotides | Activation of antigen-presenting cells, enhanced anti-tumor immunity 28 |
| Gold nanoparticles | Tumor vasculature, hypoxia | Photothermal ablation, vascular normalization | PEGylated AuNPs with targeting ligands | Localized tumor heating, enhanced chemotherapy efficacy 29 |
| Iron oxide nanoparticles | tdLNs, macrophages | Magnetic hyperthermia; modulation of TAM polarization | Ferumoxytol-based formulations | Polarization of TAMs to M1 phenotype, suppression of tumor growth 30 |
| Mesoporous silica nanoparticles | Hypoxia, CAFs | Co-delivery of drugs and oxygen carriers | MSNs loaded with doxorubicin + perfluorocarbon | Alleviation of hypoxia, reduced fibrosis, improved drug penetration 31 |
| Exosome-mimetic nanoparticles | Immune cells | Immune checkpoint blockade delivery | PD-1 siRNA-loaded exosome-like vesicles | Reversal of T cell exhaustion, enhanced anti-tumor immunity 32 |
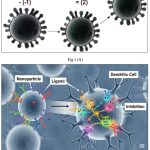 |
Figure 1: [(A), (B)]. Nanoparticles conjugated with specific ligands could target DCs and modulate their activation and maturation. |
Nanoparticles engineered to selectively target dendritic cell (DC) surface receptors—such as CD40, CD11c, mannose receptor, Fc receptor, or CD205—have emerged as a promising strategy to enhance antigen delivery and modulate immune responses.³³ These approaches have consistently demonstrated stronger immune activation compared with non-targeted formulations.³⁴⁻³⁶ To systematically assess which receptor offers the most effective targeting,³⁷ scientists made special tiny particles called pegylated poly(lactic-co-glycolic acid) or PLGA nanoparticles. These particles contain a protein called ovalbumin, along with substances that trigger certain receptors in the immune system, specifically TLR3 and TLR7. They also attached antibodies to these nanoparticles that target CD40, DEC-205, or CD11c, which are proteins on certain immune cells. The team compared binding efficiency, uptake, and immune stimulation in DCs. In vitro results revealed that CD40-targeted nanoparticles achieved the highest receptor binding, internalization, and interleukin-12 (IL-12) secretion. However, in vivo experiments showed no statistically significant differences in antigen-specific T cell proliferation among the various targeting strategies. These findings suggest that while receptor-specific binding can modestly influence nanoparticle uptake, the incorporation of potent immunostimulatory cues—such as TLR ligands—plays a far more dominant role in driving robust T cell responses.
Multiple studies have demonstrated that nanoparticle-based delivery systems incorporating dendritic cell (DC)-activating molecules can effectively promote DC activation and maturation.³⁸ For example, researchers created lipid-coated calcium phosphate nanoparticles to deliver a specific cancer-related peptide, BRAFV600E, to dendritic cells through antigen presentation. The lipid layer helped the nanoparticles pass through body barriers and kept the peptide safe from breaking down or clumping together. This method improved how well T cells responded to the antigen and increased interferon-gamma production, leading to a 20% chance of survival without tumors in treated mice.³⁹
Similarly, developed an erythrocyte membrane–coated nanoplatform functionalized with the antigenic peptide hgp10025-33, a Toll-like receptor 4 (TLR4) agonist, and mannose. The erythrocyte membrane was chosen for its biocompatibility, ease of preparation, and intrinsic adjuvant-like properties, creating a “depot effect” that prolonged antigen exposure at the injection site. Targeting through mannose–mannose receptor binding enabled efficient DC activation and yielded substantial tumor growth inhibition.⁴⁰
In addition to conventional adjuvant incorporation, certain nanoparticles inherently exhibit immunomodulatory effects. For instance, ultra-small Fe₃O₄ nanoparticles, when combined with ovalbumin, were shown to directly promote DC maturation and augment subsequent immune responses.³⁷ Notably, this study was the first to report Fe₃O₄ nanoparticles functioning dually as antigen carriers and active immunomodulators, suggesting their unique potential in cancer immunotherapy.
Beyond single-antigen strategies, introduced antigen-capturing nanoparticles (AC-NPs) capable of sequestering a wide range of tumor-derived protein antigens (TDPAs) through a combination of ionic, covalent, and hydrophobic interactions. These AC-NPs efficiently targeted dendritic cells (DCs) and elicited robust CD8⁺ T cell responses, resulting in a 20% cure rate in melanoma-bearing mice, compared with 0% in control groups. By capturing a diverse repertoire of tumor antigens in situ, AC-NPs help overcome the challenges of tumor heterogeneity and expand therapeutic efficacy beyond that of conventional single-antigen vaccines.³⁸
Targeting Tumor-Associated Macrophages
Macrophages, as terminally differentiated cells of the mononuclear phagocyte system, play essential roles in maintaining homeostasis, promoting wound healing, facilitating tissue regeneration, and orchestrating immune defense.[42-44] Their primary function, phagocytosis, enables them to engulf and degrade pathogens, cellular debris, and other particles unrecognized as “self.”
Upon stimulation by different environmental cues, macrophages exhibit distinct polarization states. Exposure to interferon-γ (IFN-γ) and lipopolysaccharide (LPS) drives classical (M1) activation, leading to the secretion of pro-inflammatory cytokines such as interleukin-12 (IL-12), which promote anti-tumor immunity.[45-48] In contrast, alternative (M2) activation—induced by interleukin-4 (IL-4) or interleukin-13 (IL-13)—drives the production of interleukin-10 (IL-10), thereby promoting tissue repair and wound healing, but also contributing to tumor progression.[49-50]
Owing to this functional dichotomy, macrophages are often described as a “double-edged sword,” exhibiting both pro-tumorigenic and anti-tumorigenic activities. Tumor-associated macrophages (TAMs), which represent a predominant immune cell population within the tumor microenvironment (TME), initially support anti-tumor immunity during early stages of tumorigenesis. However, as the tumor advances, TAMs frequently adopt an M2-like phenotype, thereby driving angiogenesis, immunosuppression, tumor invasion, and metastasis.⁴⁹ This phenotypic shift is largely attributed to macrophage plasticity—where changes in the TME, such as hypoxia, trigger polarization from the M1 to the M2 state.⁵⁰
Reprogramming TAMs toward an M1-like phenotype has emerged as a promising therapeutic approach in oncology. Nanoparticles designed to selectively deliver therapeutic agents to TAMs and modulate their polarization states offer substantial potential for enhancing anti-tumor immunity. (Table 1).
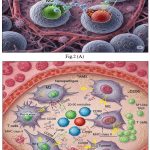 |
Figure 2: [(A), (B)]. Nanoparticles capable of selectively delivering drugs to TAMs and modulating their polarization states may represent an effective strategy in cancer immunotherapy. |
Fig.2 [(A), (B)] illustrates interferon-γ (IFN-γ) and lipopolysaccharide (LPS), tumor-associated macrophages (TAMs) polarize toward an M1-like (TAM1) phenotype, characterized by elevated secretion of interleukin-12 (IL-12), thereby promoting anti-tumor immunity.51 Conversely, stimulation with interleukin-4 (IL-4) or interleukin-13 (IL-13) promotes the M2-like (TAM2) phenotype, which produces interleukin-10 (IL-10) and facilitates tumor progression (73,74). In the hypoxic tumor microenvironment (TME), TAM1 cells can repolarize to TAM2, reinforcing immunosuppression.52
TAM reprogramming using nanoparticles: NPs functionalized with hyaluronic acid (HA), iron oxide nanoparticles, or regorafenib have been shown to reprogram TAM2 cells toward TAM1 polarization. HA interacts with CD44 on TAMs and activates Refined Version:
Activation of toll-like receptor 4 (TLR4) pathways enhances M1-associated markers, including CD86 and tumor necrosis factor-α (TNF-α), while concurrently reducing M2-associated markers such as interleukin-10 (IL-10) and CD206.53 Iron oxide NPs preferentially accumulate in macrophages, promoting recruitment and polarization to M1. They also induce a Fenton reaction, generating reactive oxygen species (ROS) that trigger tumor cell apoptosis; apoptotic tumor cells in turn reinforce M1 polarization in a positive feedback loop. Regorafenib, an oral multi-kinase inhibitor, inhibits angiopoietin-2 (ANG2)/TIE2 signaling, reducing TAM accumulation and shifting polarization toward M1.55
Direct inhibition of TAM2 survival: NPs can also suppress TAM2 activity by delivering small interfering RNA (siRNA) or ibrutinib (IBR). siRNA against colony-stimulating factor-1 receptor (CSF-1R) blocks survival pathways specific to M2 macrophages.⁵⁴ IBR, an irreversible Bruton’s tyrosine kinase (BTK) inhibitor, disrupts TAM-mediated tumorigenesis and reverses TME-associated immunosuppression.⁵⁵
Abbreviations: ANG2, angiopoietin-2; HA, hyaluronic acid; IFN-γ, interferon-γ; IBR, ibrutinib; IL, interleukin; LPS, lipopolysaccharide; NPs, nanoparticles; TAMs, tumor-associated macrophages; TIE2, tyrosine kinase with immunoglobulin-like and epidermal growth factor–like domains 2; TLR, toll-like receptor; TME, tumor microenvironment.
Several studies have investigated the potential of nanoparticles to modulate TAM polarization in cancer immunotherapy. Notably, iron oxide nanoparticles—already FDA-approved for treating iron deficiency—have shown promising off-label immunomodulatory effects.⁵⁶ Ferumoxytol has been reported to reprogram M2-polarized TAMs toward the M1 phenotype, thereby inhibiting tumor growth. Mechanistically, iron oxide nanoparticles promote M2-to-M1 repolarization and trigger the Fenton reaction, generating reactive oxygen species (ROS) that induce tumor cell apoptosis. These apoptotic cells further reinforce M1 polarization via a feedback loop involving nitric oxide (NO) and tumor necrosis factor-α (TNF-α) production. In murine models, ferumoxytol administration led to an 11-fold increase in hydrogen peroxide, a 16-fold increase in hydroxyl radicals, elevated expression of M1 markers (TNF-α, CD86), and reduced expression of M2 markers (CD206, IL-10), ultimately inhibiting tumor progression in a TAM-dependent manner.⁵⁷
In another study, developed a multifunctional nanoplatform composed of nano-Fe₃O₄ CpG-loaded liposomes (Lipo) combined with cancer cell membrane-derived antigenic microparticles (MPs).⁵⁸ The Fe₃O₄/MPs-CpG/Lipo construct combines immunomodulators, tumor antigens, and adjuvants into a single delivery platform. MPs displaying a diverse array of tumor antigens enhanced antigen-presenting cell (APC) maturation and T cell activation, while Fe₃O₄ nanoparticles promoted M1 polarization via the Fenton reaction, effectively converting an immunosuppressive (“cold”) tumor microenvironment (TME) into an immunostimulatory (“hot”) one. In vivo, this system led to a 2.55-fold increase in CD4⁺ T cell proliferation and a 3.84-fold increase in CD8⁺ T cell proliferation, ultimately extending median survival in murine models of mammary and lung cancer.⁵⁹
Addressing the delivery challenges posed by the blood–brain barrier (BBB),⁶⁰ designed a “three-bird-one-stone” nanoparticle system capable of targeting transferrin receptors (TfRs) and albumin-binding receptors such as SPARC, expressed on the BBB, glioma cells, and TAMs. This platform co-encapsulated disulfiram/copper (DSF/Cu), a chemotherapeutic complex, and regorafenib, a multi-kinase inhibitor. After crossing the blood–brain barrier (BBB) via transferrin receptors (TfRs) and SPARC, the nanoparticles selectively targeted SPARC and mannose receptors on M2 TAMs. Regorafenib modulated TAM polarization, while DSF/Cu induced tumor cell apoptosis and released tumor-associated antigens, thereby enhancing cytotoxic T lymphocyte (CTL) activity and suppressing regulatory T cells (Tregs). This dual-targeting approach markedly prolonged survival in glioma-bearing mice.[61-62]
Further improving TAM-targeted delivery, researchers developed dual-targeting nanoparticles functionalized with mannosylated carboxymethyl chitosan and hyaluronan, demonstrating enhanced delivery efficiency and stronger immune activation compared with single-targeting systems.[63-64] In a complementary approach, employed natural killer (NK) cell membrane-coated, photosensitizer-loaded nanoparticles.⁶⁵ The NK membrane coating not only facilitated M1 polarization but also extended circulation time and reduced nanoparticle toxicity, highlighting its potential as a safe and effective cancer immunotherapy strategy.⁶⁶
Beyond modulating TAM polarization, an alternative strategy involves directly inhibiting TAM survival. Qian et al. designed a dual-targeting nanoparticle (M2NP) functionalized with scavenger receptor B type 1 ligands and an M2-specific binding peptide (M2pep), encapsulating small interfering RNA (siRNA) against colony-stimulating factor-1 receptor (CSF-1R) to modulate the survival and function of TAMs⁶⁷. This nanoplatform demonstrated significantly higher affinity for M2-like TAMs compared with other macrophage subtypes and effectively inhibited M2 survival signaling. In vivo administration led to the elimination of approximately 52% of M2 macrophages, accompanied by tumor regression and improved overall survival. Additionally, levels of pro-inflammatory cytokines, including interferon-γ (IFN-γ) and interleukin-12 (IL-12), were elevated, while immunosuppressive cytokines such as transforming growth factor-β (TGF-β) and interleukin-10 (IL-10) were markedly reduced, highlighting its potential as a molecularly targeted cancer immunotherapy.⁶⁸
In addition to nucleic acid delivery, nanoparticles can be employed to deliver small-molecule inhibitors that disrupt TAM function. Bruton’s tyrosine kinase (BTK), which is overexpressed in TAMs, plays a key role in tumor progression, angiogenesis, and immunosuppression. Ibrutinib (IBR), an irreversible BTK inhibitor, has demonstrated potential in reversing TAM-mediated immunosuppression; however, its rapid renal clearance limits therapeutic efficacy. To overcome this limitation,⁶⁹ developed a sialic acid-modified, amphiphilic egg phosphatidylglycerol-based nanoplatform (SA/IBR/EPG). The amphiphilic structure provided high drug-loading efficiency and prolonged circulation time, while sialic acid enabled targeted delivery to Siglec-1-expressing TAMs via ligand–receptor binding. In vivo, SA/IBR/EPG nanoparticles enhanced immunostimulatory cytokine production and significantly reduced tumor volume.⁷⁰
Nanoparticle-based TAM targeting can also be combined with other therapeutic modalities to maximize efficacy and reduce systemic toxicity. As demonstrated by,⁶⁹ a multifunctional delivery system achieved selective cytotoxic drug delivery to TAMs, effectively integrating immunotherapy with chemotherapy. Similarly, nanoparticles have been employed to augment photodynamic therapy (PDT). PDT efficacy is often limited by hypoxia in the tumor microenvironment (TME), as it relies on photosensitizers (PS) and oxygen to generate reactive oxygen species (ROS).⁷⁰ engineered a TAM-targeted nanoplatform consisting of hyaluronic acid (HA)-modified manganese dioxide (MnO₂) nanosheets loaded with PS. MnO₂ reacted with excessive hydrogen peroxide in the TME to generate oxygen, thereby alleviating hypoxia and enhancing PDT efficacy. Concurrently, HA mediated TAM-specific delivery and facilitated M2-to-M1 repolarization, reducing tumor recurrence post-PDT.
Targeting Cancer-Associated Fibroblasts
Fibroblasts are essential for maintaining tissue homeostasis, as they synthesize extracellular matrix (ECM) components and facilitate tissue repair.71 In the tumor microenvironment (TME), however, fibroblasts remain persistently activated and are termed cancer-associated fibroblasts (CAFs). As key components of the tumor stroma, CAFs promote tumor progression by secreting angiogenic factors such as vascular endothelial growth factor (VEGF) and expressing α-smooth muscle actin (α-SMA), along with other pro-tumorigenic molecules.[72-74] These molecules are frequently overexpressed in most solid tumors, while their expression is largely absent in normal tissues.72 Additionally, CAFs secrete extracellular matrix (ECM) proteins—including type I, III, and V collagens and fibronectin—forming a dense stromal barrier that restricts the penetration and efficacy of conventional chemotherapeutic and immunotherapeutic agents. Consequently, CAFs have emerged as promising targets for nanoparticle-mediated tumor microenvironment remodeling. (Table 1).
Regulating CAF formation
One therapeutic approach is to prevent fibroblast activation and myofibroblast differentiation. Myofibroblasts, which promote tumor invasion by contracting collagen fibers and producing pro-invasive molecules, are driven primarily by TGF-β1 signaling, which triggers ROS-dependent α-SMA expression[20-24] developed cerium oxide nanoparticles (nanoceria) that exert pro-oxidant effects via Ce⁴⁺, selectively increasing ROS in tumor cells while sparing normal stromal cells. At non-toxic concentrations, nanoceria inhibited TGF-β1-induced myofibroblast formation, reduced α-SMA expression, and decreased tumor cell invasiveness by approximately 70%. Similarly, Fe₃O₄ nanoparticles were tested due to their reported cytotoxic effects,²⁵ but despite reducing α-SMA levels, they paradoxically enhanced squamous cell invasiveness, making them less suitable for therapy.
In pancreatic ductal adenocarcinoma, pancreatic stellate cells (PSCs) act as precursors to CAFs.²⁶ designed superparamagnetic iron oxide nanoparticles (SPIONs) conjugated with relaxin-2, a peptide known to suppress TGF-β-induced PSC differentiation via pSmad2 pathway inhibition. This targeted delivery system reduced CAF formation and demonstrated significant anti-tumor activity in vivo.
Selective CAF eradication
Directly targeting CAFs for elimination can be achieved using enzyme-activated nanoparticles.⁵⁰ developed a cleavable amphiphilic peptide (CAP)-based nanoparticle, which self-assembles in solution but disassembles upon cleavage by fibroblast activation protein (FAP)—an enzyme highly expressed on CAFs.⁵² This triggered the release of chemotherapeutic payloads directly within CAF populations, enhancing drug penetration into tumors. Another example is a CAF-targeted nanoliposome carrying the pro-apoptotic drug navitoclax, conjugated with a tenascin-C binding peptide (FH).⁵⁴ This system selectively induced CAF apoptosis, reducing their proportion from 77% in controls to 18% post-treatment.⁵⁶ However, since some CAF subtypes may have anti-tumor functions, complete CAF depletion could be counterproductive.⁵⁸
Combining CAF-targeted therapy with photodynamic therapy (PDT)
FAP has been widely explored as a CAF marker for PDT-based strategies.⁶⁰ Researchers developed ferritin nanoparticles modified with a FAP-specific single-chain variable fragment (scFv) and a photosensitizer (PS).⁶² Upon photo-irradiation, these nanoparticles selectively ablated CAFs, enhanced T cell infiltration, and improved nanoparticle penetration into the tumor core. Compared to conventional PDT using non-targeted PS, CAF-targeted nanomedicine enables simultaneous delivery of multiple therapeutic agents, thereby amplifying both immunotherapeutic and photodynamic effects.⁶⁴
Precision targeting over complete depletion
While CAFs often act as a physical and biochemical barrier to anti-tumor therapy, emerging evidence indicates that certain subpopulations may have tumor-restraining functions.⁶⁶ Recent studies suggest that CAFs can be classified into cancer-promoting (F2) and cancer-restraining (F1) subtypes, with their functional role dependent on tumor stage and context.[67-68] This duality implies that precise modulation of CAF activity, rather than wholesale elimination, may be the optimal therapeutic approach.⁷⁰
Tumor Vascular Remodeling
Tumor vasculature is both structurally abnormal and functionally compromised, playing a critical role in shaping the tumor microenvironment (TME).⁵⁵ Hypoxia within the TME stimulates sustained production of pro-angiogenic factors, including vascular endothelial growth factor (VEGF) and transforming growth factor-β (TGF-β).[56-58] An imbalance between pro-angiogenic and anti-angiogenic signals leads to rapid but abnormal blood vessel formation. These tumor vessels are often tortuous, irregularly distributed, and highly permeable, primarily due to pericyte detachment from the endothelial cells.[59-60] The heightened vascular permeability leads to protein leakage, which elevates interstitial fluid pressure (IFP) within the TME.⁶¹ Elevated interstitial fluid pressure (IFP) compresses blood vessels, worsening hypoxia and perpetuating a vicious cycle of vascular and microenvironmental abnormalities. Moreover, dysfunctional vasculature restricts immune cell infiltration and impedes the effective delivery of nanoparticles and therapeutic agents.⁶²
Given these challenges, vascular normalization has emerged as a promising therapeutic strategy. Anti-angiogenic therapies, particularly anti-VEGF agents, can transiently reverse vascular abnormalities; however, endothelial cells often develop adaptive resistance by upregulating alternative pro-angiogenic factors.⁶³ Rakesh K. Jain proposed the concept of a transient “normalization window” during which tumor vessels exhibit improved structure and function, thereby enhancing the efficacy of concomitant anti-tumor therapies.⁶⁴ This window varies across tumor types and disease stages, making its precise timing challenging in clinical practice.
Nanoparticles (NPs) offer a solution by enabling co-delivery of anti-angiogenic compounds and cytotoxic therapeutics, bypassing the need for precise therapeutic window targeting. For example a team developed polymeric nanoparticles co-encapsulating the epidermal growth factor receptor (EGFR) inhibitor erlotinib combined with doxorubicin,⁶⁵ leveraging the enhanced permeability and retention (EPR) effect for targeted tumor delivery. Erlotinib’s anti-angiogenic action complemented doxorubicin’s sustained release, resulting in superior chemotherapeutic outcomes.⁶⁶
Similarly, lipid-based nanoparticles incorporating low-molecular-weight heparin (LMWH), gemcitabine, and paclitaxel. LMWH inhibited VEGF–VEGFR interactions, while low-dose, high-frequency gemcitabine promoted metronomic vessel normalization. ⁶⁷ In acidic TME conditions, the nanoparticles rapidly degraded, releasing both gemcitabine and paclitaxel, leading to enhanced perfusion and more regular vessel architecture during the normalization phase. Subsequent vascular regression reduced vessel density, thereby impairing tumor nutrient supply. Notably, combining vascular normalization with extracellular matrix (ECM) modulation further improved nanoparticle distribution in tumors compared with anti-angiogenesis alone.⁶⁸
In addition to VEGF inhibition, vascular disrupting agents (VDAs) can collapse existing tumor vasculature, inducing necrosis in the tumor core. However, peripheral tumor cells often escape this effect, necessitating combination strategies.⁶⁹ PLGA nanoparticles were formulated to co-deliver the vascular disrupting agent (VDA) 5,6-dimethylxanthenone-4-acetic acid (DMXAA) and the Toll-like receptor (TLR) 7/8 agonist gardiquimod. DMXAA promoted endothelial apoptosis through TNF-α–mediated pathways, while gardiquimod stimulated immune responses via MyD88 and NF-κB signaling. In a melanoma model, this combination achieved a 63.6% survival rate, significantly outperforming monotherapies with PLGA (18.1%) or DMXAA (9%).⁷⁰
Copper, an essential trace element, also facilitates angiogenesis by enhancing endothelial cell migration and invasion.⁷¹ Leveraging this,⁷² copper-chelating polymer nanoparticles engineered to deliver the TLR7/8 agonist resiquimod integrated anti-angiogenic and immune-activating mechanisms. In a breast cancer mouse model, this system achieved synergistic tumor suppression with reduced tumor size and slower growth compared to single-modality treatments.⁷³
While promising, anti-angiogenic nanomedicine faces two major challenges: (i) a deeper mechanistic understanding of tumor angiogenesis across cancer types and progression stages is needed to optimize therapy design, and (ii) achieving uniform nanoparticle distribution within solid tumors remains a significant barrier. Nonetheless, given that nanomedicine itself benefits from vascular normalization, the combination of nanoparticles, anti-angiogenic drugs, and cytotoxic agents represents a compelling therapeutic strategy for overcoming TME-mediated drug resistance. [74-75]
Targeting Tumor-Draining Lymph Nodes (TdLNs)
Tumor-draining lymph nodes (TdLNs) are often the first sites of metastasis and are enriched with immunosuppressive cell populations such as regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs).[74-75] These lymph nodes exhibit an immuno-privileged status, where antigen-presenting cells (APCs) and T cells undergo tolerization, thereby contributing to tumor immune evasion.⁷⁴ Despite their immunosuppressive environment, TdLNs are also sites of antigen priming due to lymphatic drainage of tumor-associated antigens (TAAs) from the primary tumor.⁷⁵
The cellular composition of TdLNs includes both tumor-promoting suppressor cells and tumor-fighting effector immune cells, positioning them as critical hubs for initiating and regulating tumor-specific immune responses.⁷⁴ Historically, management of TdLNs has relied on surgical resection, particularly sentinel lymph node removal. However, recent advances in tumor immunology have shifted attention toward functional modulation of TdLNs to either reverse immunosuppression or enhance anti-tumor immunity.⁷⁵
Nanomedicine offers unique opportunities for targeted intervention within TdLNs. Specifically engineered nanoparticles can be designed for preferential lymphatic uptake and accumulation in TdLNs, enabling localized delivery of immunostimulatory agents or therapeutic payloads. This targeted approach can enhance antigen presentation, activate cytotoxic T cells, and reduce the suppressive influence of Tregs and MDSCs. [74-75] Several nanoparticle systems have been developed for this purpose. showing promising preclinical results in boosting systemic anti-tumor immune responses while minimizing off-target effects (Table 1).
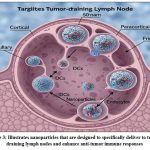 |
Figure 3: Illustrates nanoparticles that are designed to specifically deliver to tumor-draining lymph nodes and enhance anti-tumor immune responses. |
As shown in Fig. 3. Tumor-draining lymph nodes (TdLNs) are enriched with both immunostimulatory cells, such as dendritic cells (DCs), T cells, and B cells, and immunosuppressive populations, including regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs). [40-41] Tumor-associated antigens (TAAs) are transported to tumor-draining lymph nodes (TdLNs) via lymphatic vessels, where dendritic cells (DCs) process and present them to T cells. Activated T cells secrete interferon-gamma (IFN-γ) and stimulate B cells to produce antibodies, together orchestrating a robust anti-tumor immune response.⁴² However, myeloid-derived suppressor cells (MDSCs) and regulatory T cells (Tregs) within tumor-draining lymph nodes (TdLNs) inhibit T cell activation and function, facilitating tumor immune evasion.⁴³
Nanoparticles (NPs) offer a targeted strategy for modulating TdLN immunity. Medium-sized NPs (10–100 nm) can efficiently drain into TdLNs, maximizing delivery efficiency.⁴⁴ Once inside, NPs encapsulating TAAs activate DCs, enhance antigen presentation, and enhance T cell responses. The incorporation of Toll-like receptor (TLR) ligands, such as CpG oligodeoxynucleotides (TLR9 agonists), further augments immune activation by inducing pro-inflammatory cytokines, promoting MDSC differentiation, and relieving T-cell suppression.⁴⁵
Nanoparticle size and immune efficacy
Particle size strongly influences lymphatic trafficking and immune activation. Large particles are often retained in the extracellular matrix, restricting lymph node entry, whereas very small particles rapidly exit lymph nodes and have limited immunological impact. Morikwal S et al. demonstrated that 40–50 nm antigen-conjugated beads elicited 2–10-fold stronger IFN-γ and antibody responses compared to smaller or larger counterparts. [46-47] Similarly, Maeda et al., reported that a 60 nm cholesteryl pullulan hydrogel NP delivering synthetic long peptide antigens preferentially localized to TdLNs, was taken up by medullary macrophages, and effectively activated CD8⁺ T cells in the context of TLR stimulation.⁴8 Team of Banco E found that 10–22 nm gold NPs were optimal for eliciting robust immune responses. ⁴9 Collectively, these studies suggest that nanoparticles (NPs) in the 10–100 nm range achieve an optimal balance between lymph node retention and immune stimulation, whereas particles <10 nm undergo rapid renal clearance and those >100 nm are rapidly removed by the reticuloendothelial system.⁵⁰
Targeting TdLNs with NP-based vaccines
The concept of targeting TdLNs for vaccination is supported by studies showing that, despite their immunosuppressive milieu, TdLNs remain sites of potent antigen priming.⁵¹ In murine lymphoma and melanoma models, vaccines delivering TAAs specifically to TdLNs generated stronger CD8⁺ T-cell responses, reduced MDSC and Treg populations, and suppressed tumor growth more effectively than vaccines targeting non-tumor-draining lymph nodes.⁵² demonstrated that PLGA NPs loaded with TLR7/8 agonists accumulated in TdLNs, expanded DC populations threefold, increased CD8⁺ T cells 4.5-fold, and inhibited both primary and metastatic tumor growth.⁵³
Expanding antigen diversity for vaccine efficacy
While TAA-based vaccines hold promise, the limited availability of immunogenic TAAs for many cancers poses a challenge.⁵⁴ To address this, Pelaz B and scientists developed a biomimetic “artificial necroptotic cancer cell” system comprising cancer cell membrane proteins, a heat shock protein 70 (HSP70) peptide, and a calcium phosphate core.55 This formulation presented a broad spectrum of TAAs, enhanced DC maturation, activated natural killer (NK) cells, and reduced lung metastases by 77% in melanoma-bearing mice. When combined with anti-PD-1 checkpoint inhibitors, metastasis reduction reached 89%.⁵⁶
Whole-cell antigen vaccines offer another strategy to overcome TAA limitations. Zhao et al., designed a melittin–lipid NP that lyses tumor cells to release the full repertoire for tumor antigens.57 With an optimal particle size of 20 nm, these NPs efficiently reached TdLNs, increased CD8⁺ T-cell responses by 3.6-fold, and achieved 95% and 92% inhibition of primary and distant tumor growth, respectively.⁵⁸ These findings collectively highlight the dual role of TdLNs as both immunosuppressive and immunostimulatory hubs and underscore the potential of nanoparticle-mediated, lymph node–targeted vaccines to strengthen anti-tumor immunity.⁵⁹–⁶⁰
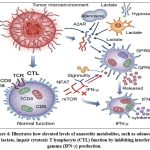 |
Figure 4: Illustrates how elevated levels of anaerobic metabolites, such as adenosine and lactate, impair cytotoxic T lymphocyte (CTL) function by inhibiting interferon-gamma (IFN-γ) production. |
As detailed in Fig. 4. Impact of Hypoxia and the Role of Nanoparticles in Modulating Tumor Immunity, Hypoxia within the tumor microenvironment (TME) suppresses anti-tumor immunity through multiple mechanisms. Increased levels of anaerobic metabolites, such as adenosine and lactate, compromise cytotoxic T lymphocyte (CTL) function by reducing interferon-γ (IFN-γ) production.[⁷⁵–⁷⁶] Hypoxia also promotes the recruitment of immunosuppressive cell populations, including myeloid-derived suppressor cells (MDSCs) and regulatory T cells (Tregs), and drives the polarization of tumor-associated macrophages (TAMs) from the anti-tumorigenic M1 phenotype toward the pro-tumorigenic M2 phenotype.⁷⁷
Furthermore, hypoxia enhances secretion of immunosuppressive factors, notably transforming growth factor-β (TGF-β) and vascular endothelial growth factor (VEGF), with TGF-β promoting fibroblast-to–cancer-associated fibroblast (CAF) transition,⁷⁸ while VEGF stimulates tumor angiogenesis. Excessive VEGF alters the balance between pro- and anti-angiogenic factors, leading to structurally abnormal vasculature that further exacerbates hypoxia.⁷⁹
NP-based strategies to modulate hypoxia and its downstream effects include:
Oxygen generation – MnO₂-modified NPs react with hydrogen peroxide (H₂O₂) in acidic TME to produce oxygen, mitigating hypoxia and improving therapy efficacy.⁷⁵
Oxygen delivery – Perfluorocarbon (PFC)-loaded NPs exploit PFC’s high oxygen solubility to store and release oxygen in situ.⁷⁶
CAF inhibition – Ce⁴⁺-based nanoceria prevent TGF-β1–initiated, ROS-dependent myofibroblast formation,⁷⁷ while relaxin-2 (RLX)-functionalized NPs inhibit pancreatic stellate cell differentiation by suppressing pSmad2 signaling.⁷⁸
CAF depletion – NP delivery of cytotoxic agents, such as the Bcl-2 inhibitor navitoclax, directly induces CAF apoptosis.⁷⁹
Vascular modulation – VEGF inhibitor–loaded NPs block VEGF receptor binding and signaling in endothelial cells, while vasculature-disrupting agents (VDAs) delivered via NPs induce endothelial apoptosis through TNF-α–mediated pathways.⁸⁰
Conclusions and Perspectives
Tumor hypoxia is a critical factor driving tumor progression, promoting immune evasion, and contributing to resistance against conventional therapies such as chemotherapy.”, photodynamic therapy, and radiotherapy. The hypoxic tumor microenvironment (TME) promotes immunosuppression through accumulation of regulatory cells like MDSCs and Tregs, the polarization of macrophages toward the pro-tumorigenic M2 phenotype and the upregulation of immunosuppressive cytokines, including TGF-β and VEGF.. These changes not only impair cytotoxic T lymphocyte (CTL) functions, particularly reducing IFN-γ production, but also exacerbate abnormal tumor vasculature and stromal remodeling, creating a vicious cycle that sustains hypoxia.
Nanoparticles have emerged as a promising platform for overcoming tumor hypoxia and enhancing therapeutic efficacy. Several strategies have been developed…”, including in situ oxygen generation via MnO₂-based nanomaterials reacting with tumor H₂O₂, direct oxygen delivery through perfluorocarbon (PFC)-loaded nanoparticles, and combinational delivery of chemotherapeutic agents with oxygen-generating nanoplatforms. These approaches have been shown to significantly alleviate hypoxia, enhance CTL infiltration, and potentiate the effects of chemotherapy, radiotherapy, and photodynamic therapy in preclinical models.
Furthermore, nanoparticle-mediated delivery systems targeting the hypoxic TME can modulate the immunosuppressive environment by repolarizing macrophages, reducing pro-tumorigenic fibroblast activation, and normalizing aberrant vasculature. Advanced nanostructures, such as hypoxia-responsive mesoporous silica and nanoscale metal-organic frameworks (nMOFs), provide controlled release of therapeutic payloads and enhanced photodynamic effects. Additionally, pH-sensitive and charge-convertible nanoparticles capitalize on the acidic TME to achieve site-specific drug release and improved cellular uptake.
Despite these advances, however, significant challenges remain in translating these nanotechnologies into clinical practice.. The heterogeneity of tumor hypoxia across cancer types and stages requires tailored nanomedicine designs and comprehensive understanding of TME dynamics. Optimizing nanoparticle size, surface properties, and biocompatibility is essential to balance effective tumor penetration with prolonged circulation and minimal off-target effects.
“Future research should prioritize the development of multifunctional nanoparticles that can simultaneously alleviate hypoxia, deliver immunomodulatory agents, and synergize with immune checkpoint blockade therapies.” to achieve synergistic anti-tumor responses. Real-time imaging and biomarker development are also crucial to monitor hypoxia modulation and guide treatment scheduling. Ultimately, rationally designed nanoparticle-based strategies hold great promise “to overcome hypoxia-driven therapeutic resistance and enhance clinical outcomes in cancer patients.”
Acknowledgement
The authors express their sincere gratitude to KVPS Maharani Ahilyabai Holkar College of Pharmacy, Shirpur, for providing valuable support.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not include human participants, and informed consent was not required. The work does not constitute a clinical trial.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable.
Author Contributions
Rakesh Dhole.: Conceptualization, Visualization, Writing- Original Draft Project Administration;
Mahesh Pawar.: Supervision, Writing- Review and Editing;
Bhagyashri Ahirrao: Data Analysis;
Gurmeet Singh Chhabra and Nalla Seshadri: Data Analysis, Editing and Supervision;
Praneeth Ivan Joel FNU: Data Analysis;
Pravinkumar Darji and Binit Patel: Writing- Review and Editing.
References
- Hanahan D, Coussens LM. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell. 2012;21(3):309-322. https://doi.org/10.1016/j.ccr.2012.02.022
CrossRef - Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19(11):1423-1437. https://doi.org/10.1038/nm.3394
CrossRef - Hinshaw DC, Shevde LA. The tumor microenvironment innately modulates cancer progression. Cancer Res. 2019;79(18):4557-4566. https://doi.org/10.1158/0008-5472.CAN-18-1367
CrossRef - Gajewski TF, Schreiber H, Fu YX. Innate and adaptive immune cells in the tumor microenvironment. Nat Immunol. 2013;14(10):1014-1022. https://doi.org/10.1038/ni.2703
CrossRef - Chen DS, Mellman I. Elements of cancer immunity and the cancer–immune set point. Nature. 2017;541(7637):321-330. https://doi.org/10.1038/nature21349
CrossRef - Junttila MR, de Sauvage FJ. Influence of tumour micro-environment heterogeneity on therapeutic response. Nature. 2013;501(7467):346-354. https://doi.org/10.1038/nature12626
CrossRef - Jain RK. Anti-angiogenesis strategies revisited: from starving tumors to alleviating hypoxia. Cancer Cell. 2014;26(5):605-622. https://doi.org/10.1016/j.ccell.2014.10.006
CrossRef - Goel S, Duda DG, Xu L, et al. Normalization of the vasculature for treatment of cancer and other diseases. Physiol Rev. 2011;91(3):1071-1121. https://doi.org/10.1152/physrev.00038.2010
CrossRef - Ferrara N, Kerbel RS. Angiogenesis as a therapeutic target. Nature. 2005;438(7070):967-974. https://doi.org/10.1038/nature04483
CrossRef - Semenza GL. Hypoxia-inducible factors in physiology and medicine. Cell. 2012;148(3):399-408. https://doi.org/10.1016/j.cell.2012.01.021
CrossRef - Binnewies M, et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med. 2018;24(5):541-550. https://doi.org/10.1038/s41591-018-0014-x
CrossRef - Salmon H, et al. Matrix architecture defines the preferential localization and migration of T cells into the stroma of human lung tumors. J Clin Invest. 2012;122(3):899-910. https://doi.org/10.1172/JCI45817
CrossRef - Joyce JA, Fearon DT. T cell exclusion, immune privilege, and the tumor microenvironment. Science. 2015;348(6230):74-80. https://doi.org/10.1126/science.aaa6204
CrossRef - Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med. 2018;378(2):158-168. https://doi.org/10.1056/NEJMra1703481
CrossRef - Wang DY, et al. Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol. 2018;4(12):1721-1728. https://doi.org/10.1001/jamaoncol.2018.3923
CrossRef - Spain L, Diem S, Larkin J. Management of toxicities of immune checkpoint inhibitors. Cancer Treat Rev. 2016;44:51-60. https://doi.org/10.1016/j.ctrv.2016.02.001
CrossRef - Beatty GL, O’Hara M. Chimeric antigen receptor–modified T cells for the treatment of solid tumors. Pharmacol Ther. 2016;166:30-39. https://doi.org/10.1016/j.pharmthera.2016.06.010
CrossRef - Sterner RC, Sterner RM. CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J. 2021;11(4):69. https://doi.org/10.1038/s41408-021-00472-9
CrossRef - Shi J, Kantoff PW, Wooster R, Farokhzad OC. Cancer nanomedicine: progress, challenges and opportunities. Nat Rev Cancer. 2017;17(1):20-37. https://doi.org/10.1038/nrc.2016.108
CrossRef - Noman MZ, et al. Hypoxia: a key player in antitumor immune response. Aging (Albany NY). 2015;7(10):849-862. https://doi.org/10.18632/aging.100859
CrossRef - Miao L, Lin CM, Huang L. Nanoparticle modulation of the tumor immune microenvironment. Cell Mol Immunol. 2019;16(4):375-385. https://doi.org/10.1038/s41423-019-0210-3
- Maeda H, Nakamura H, Fang J. The EPR effect for macromolecular drug delivery to solid tumors. J Control Release. 2013;164(2):138-144. https://doi.org/10.1016/j.jconrel.2012.10.017
CrossRef - Torchilin VP. Multifunctional, stimuli-sensitive nanoparticulate systems for drug delivery. Nat Rev Drug Discov. 2014;13(11):813-827. https://doi.org/10.1038/nrd4333
CrossRef - Blanco E, Shen H, Ferrari M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat Biotechnol. 2015;33(9):941-951. https://doi.org/10.1038/nbt.3330
CrossRef - Barenholz Y. Doxil®—the first FDA-approved nano-drug: lessons learned. J Control Release. 2012;160(2):117-134. https://doi.org/10.1016/j.jconrel.2012.04.046
CrossRef - Desai N, et al. Increased antitumor activity and intratumor paclitaxel concentrations of cremophor-free albumin-bound paclitaxel (ABI-007). Clin Cancer Res. 2006;12(4):1317-1324. https://doi.org/10.1158/1078-0432.CCR-05-1766
CrossRef - Steinman RM, Banchereau J. Taking dendritic cells into medicine. Nature. 2007;449(7161):419-426. https://doi.org/10.1038/nature06175
CrossRef - Miller JC, et al. Deciphering the transcriptional network of the dendritic cell lineage. Nat Immunol. 2012;13(9):888-899. https://doi.org/10.1038/ni.2370
CrossRef - Gardner A, Ruffell B. Dendritic cells and cancer immunity. Trends Immunol. 2016;37(12):855-865. https://doi.org/10.1016/j.it.2016.09.006
CrossRef - Colegio OR, et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature. 2014;513(7519):559-563. https://doi.org/10.1038/nature13490
CrossRef - Koppolu B, Zaharoff DA. The effect of antigen encapsulation in chitosan particles on antigen-presenting cells. Biomaterials. 2013;34(9):2359-2369. https://doi.org/10.1016/j.biomaterials. 2012.12.011
CrossRef - Chen G, Roy I, Yang C, Prasad PN. Nanochemistry and nanomedicine for nanoparticle-based diagnostics and therapy. Chem Rev. 2016;116(5):2826-2885. https://doi.org/10.1021/acs.chemrev.5b00148
CrossRef - Tacken PJ, et al. Effective induction of naïve and recall T-cell responses by targeting antigen to human dendritic cells via DC-SIGN. Blood. 2005;106(4):1278-1285. https://doi.org/10.1182/blood-2005-03-1169
CrossRef - Cruz LJ, et al. Targeted PLGA nanoparticles deliver antigen to human dendritic cells via DC-SIGN in vitro. J Control Release. 2010;144(2):118-126. https://doi.org/10.1016/j.jconrel.2010.01.002
CrossRef - Liu X, et al. Lipid-coated calcium phosphate nanoparticles as an adjuvant-delivery system for cancer vaccines. Biomaterials. 2018;178:1-15. https://doi.org/10.1016/j.biomaterials.2018.06.024
CrossRef - Guo Y, et al. Erythrocyte membrane-enveloped polymeric nanoparticles as nanovaccine for anti-tumor immunity. ACS Nano. 2020;14(5):6316-6325. https://doi.org/10.1021/acsnano.0c00696
CrossRef - Zhang P, Chiu YC, Tostanoski LH, Jewell CM. Polyelectrolyte multilayers on gold nanoparticles promote antigen-specific T-cell responses. ACS Nano. 2019;13(4):4600-4611. https://doi.org/10.1021/acsnano.8b09651
CrossRef - Min Y, et al. Antigen-capturing nanoparticles improve the abscopal effect and cancer immunotherapy. Nat Nanotechnol. 2017;12(9):877-882. https://doi.org/10.1038/nnano.2017.113
CrossRef - Murray PJ, Wynn TA. Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol. 2011;11(11):723-737. https://doi.org/10.1038/nri3073
CrossRef - Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010;141(1):39-51. https://doi.org/10.1016/j.cell.2010.03.014
CrossRef - Noy R, Pollard JW. Tumor-associated macrophages: from mechanisms to therapy. Immunity. 2014;41(1):49-61. https://doi.org/10.1016/j.immuni.2014.06.010
CrossRef - Mosser DM, Edwards JP. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008;8(12):958-969. https://doi.org/10.1038/nri2448
CrossRef - Martinez FO, Gordon S. The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep. 2014;6:13. https://doi.org/10.12703/P6-13
CrossRef - Sica A, Mantovani A. Macrophage plasticity and polarization: in vivo veritas. J Clin Invest. 2012;122(3):787-795. https://doi.org/10.1172/JCI59643
CrossRef - Heldin CH, Rubin K, Pietras K, Östman A. High interstitial fluid pressure—an obstacle in cancer therapy. Nat Rev Cancer. 2004;4(10):806-813. https://doi.org/10.1038/nrc1456
CrossRef - Hashizume H, Baluk P, Morikawa S, et al. Openings between defective endothelial cells explain tumor vessel leakiness. Am J Pathol. 2000;156(4):1363-1380. https://doi.org/10.1016/S0002-9440(10)64948-6
CrossRef - Morikawa S, Baluk P, Kaidoh T, Haskell A, Jain RK, McDonald DM. Abnormalities in pericytes on blood vessels and endothelial sprouts in tumors. Am J Pathol. 2002;160(3):985-1000. https://doi.org/10.1016/S0002-9440(10)64917-6
CrossRef - Maeda H, Wu J, Sawa T, Matsumura Y, Hori K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release. 2000;65(1-2):271-284. https://doi.org/10.1016/S0168-3659(99)00248-5
CrossRef - Blanco E, Shen H, Ferrari M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat Biotechnol. 2015;33(9):941-951. https://doi.org/10.1038/nbt.3330
CrossRef - Parveen S, Misra R, Sahoo SK. Nanoparticles: a boon to drug delivery, therapeutics, diagnostics and imaging. Nanomedicine: NBM. 2012;8(2):147-166. https://doi.org/10.1016/j.nano.2011.05.016
CrossRef - Singh R, Lillard JW. Nanoparticle-based targeted drug delivery. Exp Mol Pathol. 2009;86(3):215-223. https://doi.org/10.1016/j.yexmp.2008.12.004
CrossRef - Anselmo AC, Mitragotri S. Nanoparticles in the clinic: an update. Bioeng Transl Med. 2016;1(1):10-29. https://doi.org/10.1002/btm2.10003
CrossRef - Farokhzad OC, Langer R. Impact of nanotechnology on drug delivery. ACS Nano. 2009;3(1):16-20. https://doi.org/10.1021/nn900002m
CrossRef - Singh P, Kim YJ, Zhang D, Yang DC. Biological synthesis of nanoparticles from plants and microorganisms. Trends Biotechnol. 2016;34(7):588-599. https://doi.org/10.1016/j.tibtech. 2016.02.006
CrossRef - Pelaz B, Alexiou C, Alvarez-Puebla RA, et al. Diverse applications of nanomedicine. ACS Nano. 2017;11(3):2313-2381. https://doi.org/10.1021/acsnano.6b06040
CrossRef - Gupta A, Mumtaz S, Li CH, Hussain I, Rotello VM. Combatting antibiotic-resistant bacteria using nanomaterials. Chem Soc Rev. 2019;48(2):415-427. https://doi.org/10.1039/C7CS00748E
CrossRef - Zhao X, Tang Q, Mao G, Zhao Z. Nanotechnology for cancer therapy based on nanomaterials and nanocarriers. Front Bioeng Biotechnol. 2021;9:720192. https://doi.org/10.3389/fbioe.2021.720192
- Bhattacharya R, Mukherjee P. Biological properties of “naked” metal nanoparticles. Adv Drug Deliv Rev. 2008;60(11):1289-1306. https://doi.org/10.1016/j.addr.2008.03.013
CrossRef - Salata OV. Applications of nanoparticles in biology and medicine. J Nanobiotechnology. 2004;2(1):3. https://doi.org/10.1186/1477-3155-2-3
CrossRef - Miao L, Lin CM, Huang L. Nanoparticle modulation of the tumor immune microenvironment. Cell Mol Immunol. 2019;16(4):375-385. https://doi.org/10.1038/s41423-019-0210-3
- Spain L, Diem S, Larkin J. Management of toxicities of immune checkpoint inhibitors. Cancer Treat Rev. 2016;44:51-60. https://doi.org/10.1016/j.ctrv.2016.02.001
CrossRef - Jain RK. Normalizing tumor vasculature with anti-angiogenic therapy: a new paradigm for combination therapy. Nat Med. 2001;7(9):987-989. https://doi.org/10.1038/90925
CrossRef - Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005;307(5706):58-62. https://doi.org/10.1126/science.1104819
CrossRef - Zhao Y, Adjei AA. Targeting angiogenesis in cancer therapy: moving beyond VEGF. Cancer Sci. 2015;106(9):1087-1096. https://doi.org/10.1111/cas.12717
CrossRef - Murray PJ, Allen JE, Biswas SK, et al. Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity. 2014;41(1):14-20. https://doi.org/10.1016/j.immuni.2014.06.008
CrossRef - Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010;11(10):889-896. https://doi.org/10.1038/ni.1937
CrossRef - Noman MZ, Desantis G, Janji B, et al. Hypoxia and immune checkpoint regulation. Cells. 2021;10(5):1006. https://doi.org/10.3390/cells10051006
CrossRef - Doedens AL, Phan AT, Stradner MH, et al. Hypoxia-inducible factors enhance the effector responses of CD8+ T cells to persistent antigen. Nat Immunol. 2010;11(10):1028-1037. https://doi.org/10.1038/ni.1938
- Barsoum IB, Smallwood CA, Siemens DR, Graham CH. A mechanism of hypoxia-mediated escape from adaptive immunity in cancer cells. Cancer Res. 2014;74(3):665-674. https://doi.org/10.1158/0008-5472.CAN-13-0992
CrossRef - Fischer K, Hoffmann P, Voelkl S, et al. Inhibitory effect of tumor cell–derived lactic acid on human T cells. Blood. 2007;109(9):3812-3819. https://doi.org/10.1182/blood-2006-07-036711
CrossRef - Vaupel P, Mayer A. Hypoxia in tumors: pathogenesis-related classification, characterization of hypoxia subtypes, and implications. Adv Exp Med Biol. 2014;812:19-24. https://doi.org/10.1007/978-1-4939-0620-8_3
CrossRef - Rankin EB, Giaccia AJ. Hypoxia: signaling in the metastatic cascade. Trends Cancer. 2016;2(6):295-304. https://doi.org/10.1016/j.trecan.2016.05.006
CrossRef - Jayaprakash P, Ai M, Liu A, et al. Targeted hypoxia reduction restores T cell infiltration and sensitizes prostate cancer to immunotherapy. J Clin Invest. 2018;128(11):5137-5149. https://doi.org/10.1172/JCI99095
CrossRef - Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006;6(5):392-401. https://doi.org/10.1038/nrc1877
CrossRef - Özdemir BC, Pentcheva-Hoang T, Carstens JL, et al. Depletion of carcinoma-associated fibroblasts and fibrosis induces immunosuppression and accelerates pancreas cancer. Cancer Cell. 2014;25(6):719-734. https://doi.org/10.1016/j.ccr.2014.04.005
CrossRef - Monteran L, Erez N. The dark side of fibroblasts: cancer-associated fibroblasts as mediators of immunosuppression in the tumor microenvironment. Front Immunol. 2019;10:1835. https://doi.org/10.3389/fimmu.2019.01835
CrossRef - Goel S, Duda DG, Xu L, et al. Normalization of the tumor vasculature through oncogenic inhibition. J Exp Med. 2012;209(3):529-542. https://doi.org/10.1084/jem.20111712
- Stylianopoulos T, Jain RK. Combining two strategies to improve perfusion and drug delivery in solid tumors. Proc Natl Acad Sci U S A. 2013;110(46):18632-18637. https://doi.org/10.1073/pnas.1318415110
CrossRef - Zhou Q, Zhang F, He Z, et al. Copper-chelator nanoparticles co-loaded with resiquimod improve anti-tumor immunity and inhibit angiogenesis in breast cancer. ACS Nano. 2021;15(2):3058-3072. https://doi.org/10.1021/acsnano.0c08353
- Roberts ZJ, Goutagny N, Perera PY, et al. The chemotherapeutic agent DMXAA activates a murine STING-dependent type I interferon response. Science. 2007;318(5853):1682-1685. https://doi.org/10.1126/science.1144958
CrossRef - Begh MZ, Zehravi M, Gupta JK, Balaji P, Gangireddy R, Singamaneni VR, Jahnavi P, Kumar A, Somasundaram I, Vodeti R, Gupta PS. Mechanistic signaling pathways of flavonoid-induced oral squamous cell carcinoma therapy: Clinical evidence and therapeutic application. Pathology-Research and Practice. 2025 Sep 9:156227. https://doi.org/10.1016/j.prp.2025.156227
CrossRef
Abbreviations List
Bcl-2, B-cell lymphoma 2;
CAFs, cancer-associated fibroblasts;
CTLs, cytotoxic T lymphocytes;
MDSCs, myeloid-derived suppressor cells;
NPs, nanoparticles;
PFCs, perfluorocarbons;
RLX, relaxin-2;
TAMs, tumor-associated macrophages;
TGF-β, transforming growth factor-β;
TME, tumor microenvironment;
TNF-α, tumor necrosis factor-α;
Tregs, regulatory T cells;
VEGF, vascular endothelial growth factor;
VDAs, vascular-disrupting agents.
Accepted on: 26-11-2025
Second Review by: Dr. Aisha Belal
Final Approval by: Dr. Wagih Ghannam









