A Comprehensive Review on Applications of Polylactic Acid in Novel Drug Delivery System
Department of Pharmaceutics, Modern College of Pharmacy, Affiliated to Savitribai Phule Pune University, Pune, India.
Corresponding Author E-mail: krishnabharti3008@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3456
ABSTRACT:Both natural and synthetic polymers are crucial for dosage forms and packaging materials in the pharmaceutical industry. The use of biodegradable polymers in drug delivery systems has increased recently, and their ease of excretion from the body makes them particularly desirable. The potential for controlled drug release in biomedical applications has been shown by the effective loading and release of a range of pharmaceuticals utilizing polylactic acid (PLA) based polymers, highlighting the need for modification to accept both hydrophobic and hydrophilic medications. PLA is also utilized as a tissue anti-adhesion substance and as a product packaging material. This review covers the manufacture and modification of (PLA) for a range of biomedical uses, such as nanoparticles, copolymers, injectable hydrogels, microspheres, nanofibers, transdermal, liposomes, microfibers, sol gels drug delivery systems and tissue engineering. It investigates surface modification methods such coating, chemical modification, and plasma treatment as well as the copolymerization of lactide with other monomers like malic acid, PEG, and PGA. The FDA has approved PLA-based polymers, such as PLGA, for use in prolonged medication release formulations and bioresorbable sutures. The creation of injectable hydrogels made of sulfobetaine-modified PLA for anti-adhesion and hemostasis, as well as PLA nanoparticles loaded with curcumin for tissue engineering and controlled drug release, are also covered in the paper. Mannan-decorated PLGA nanoparticles for cancer immunotherapy are also investigated; in mouse models, they show improved immune responses and tumor regression. these developments hold great promise for tissue engineering and medical treatments.
KEYWORDS:Applications; Biodegradable polymers; Nanoparticles; Novel drug delivery system; Polylactic acid; Tissue engineering
Introduction
In the 1970s, the FDA approved poly (glycolic acid) (PGA), poly (lactic acid) (PLA), and poly (lactic-co-glycolic acid) (PLGA) for use in sustained medication release formulations and bioresorbable surgical sutures. Since then, injectable controlled release systems especially drug delivery microparticles—have made extensive use of these polymers. There are currently more than 15 PLGA-based microsphere products available on the market, having started with the 1986 approval of Decapeptyl® SR. Due to growing scientific and commercial interest, PLA/PLGA microparticles are being developed again because they are thought to be a dependable drug delivery technology, particularly for therapeutic peptides. pharmacological companies are investigating generic PLA/PLGA-based medicines as a result of the expiration of initial patents; nevertheless, proving pharmacological and bioequivalence is more difficult than with traditional oral dosage forms. As a component of the larger drug delivery device industry, which was valued at over $330 billion in 2016 and is projected to grow to almost $930 billion by 2024, the PLA/PLGA microparticle market is a lucrative one. The main turning points in the development of injectable PLA/PLGA microparticles are described in this article, along with preliminary studies on polymer properties, controlled drug administration, and traditional fabrication techniques. While poly (dl-lactic acid) (PDLLA) is amorphous, polylactic acid (PLA) comes in two semi-crystalline enantiomeric forms: poly (l-lactic acid) (PLLA) and poly (d-lactic acid) (PDLA). Compared to PLA, poly (glycolic acid) (PGA) is a more hydrophilic and highly crystalline polymer. Because of their degrading properties, PLA and PGA, although pioneers in the field of biodegradable polymers, have not found widespread application in injectable microspheres.1
Because PLA degrades slowly, it is utilized in long-term medication formulations such as Sculptra®, a cosmetic filler devoid of active pharmaceutical ingredients (API), and Lupron Depot®, a leuprolide acetate formulation that lasts three to six months. On the other hand, because of its high hydrophilicity, quick degradation, and production of porous particles, PGA has not been marketed in products based on microparticles and is therefore not appropriate for long-term drug release. The predominant copolymer in commercially available microsphere formulations, such as Lupron Depot®, is poly (lactic-co-glycolic acid) (PLGA). The lactic acid/glycolic acid ratio can be changed to modify the hydrophilicity and rate of breakdown of PLGA. Slower disintegration and less hydrophilicity are the results of a higher lactic acid level. PLGA degradation is also influenced by variables such as end-chain type, molecular weight, and polydispersity index. As demonstrated by Parlodel LAR®, a monthly bromocriptine formulation, branched PLGA modifications, such as glucose-linked star-like PLGA, allow for quicker breakdown. Depending on the stereochemistry of lactic acid and its ratio to PGA, PLGA can be either semi-crystalline or amorphous. Because of its superior API dispersion, amorphous d,l-PLGA is frequently chosen for medicinal formulations. One important factor affecting its change from a brittle to a rubbery condition is its glass transition temperature (Tg).
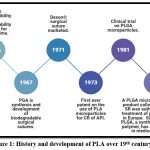 |
Figure 1: History and development of PLA over 19th century.Click here to view Figure |
SYNTESIS
With throughputs of up to 20 kg/hr., PLA, a polymer made from LA monomers, is a commonly used industrial method. However, metal catalyst residues can make it hazardous. Either an AE source or a metal-free catalyst should be utilized to create non-toxic PLA. Research indicates that PLA with low molecular weight and conversion rates can be produced using a non-metal catalyst alone. Computational models and additional research are required to expand the process to an industrial level. Computational chemistry modeling techniques can be used to improve PLA-polymer composites, which could eventually replace traditional polymers in nano-filler-based pharmaceuticals, cosmetics, and food contact packaging materials.2
The equilibrium between free acid, oligomers, and water results in a low molecular weight for direct condensation polymerization, a process that uses high purity lactide to create big molecular weight molecules. However, ring opening polymerization can contaminate the polymer and necessitates catalysts based on heavy metals, which restricts its use in medical and food packaging engineering systems. To get around these obstacles, scientists are looking into safe catalysts, unique treatments, or other polymerization techniques. Moderate conditions and environmental control are provided by enzymatic polymerization. PLA has undergone surface modifications and treatments to enhance its qualities and uses.3
Table 1: General applications of poly lactic acid.
| Category | Details |
| Industries Using PLA | Textiles, Packaging, Medicine (Tissue Engineering, Orthopaedics, Dentistry, Cardiology) |
| Key Benefits | Environmentally friendly, Recyclable, Compostable, Biocompatible, Non-toxic, Simple to manufacture |
| Energy Efficiency | Consumes 25–55% less energy than petroleum-based polymers |
| Challenges | Low hardness (limits orthopaedic use), Hydrophobicity (limits cell ingress, tissue engineering) |
| Thermal Properties | Semi-crystalline, Glass transition temperature (Tg), Melting point (Tm) |
| Blends and Modifications | Mixed with Polystyrene, PET, Polycarbonates, Polyethylene, Chitosan for better thermal stability |
| Copolymers | PEG, Glycolic acid (more hydrophilic), PCL maleation (property modification) |
| Biodegradability Factors | Polymer composition, pH, Geometry, Molecular weight, Crystallinity, Additives, Stress, Sterilization |
| Chemical Properties | Lactic acid in L- and D- forms, optically active, Difficult to modify due to chemical inertness |
| Tissue Engineering | Used in bone grafting, Combined with HA for osteogenesis and improved flexural strength |
| Dental Applications | Used in dental implants, Composite materials, Drug delivery for endodontic procedures |
| Orthopaedic Uses | Resorbable fracture plates, Sutures, ACL reconstruction, Osteogenic implants |
| Antibacterial Properties | Immobilized saccharides (glucosamine, chondroitin sulphate), Mg-reinforced PLA for osteogenesis |
| 3D Printing Applications | Used in additive manufacturing, FDM enhances mechanical properties, Useful for medical devices, PPE |
| Future Potential | Field hospitals, Military hospitals, Space medicine, Disaster relief applications |
PLA in Nanoparticles
The study done by Fiorenza Rancan et al. explores the use of Polylactic Acid (PLA) nanoparticles as targeted dermatotherapy medication delivery vehicles, revealing successful penetration into human skin explants and fluorescent dye release. The findings highlight the importance of understanding nanoparticle toxicological profiles.4-6
The study explores the use of polylactic acid (PLA) nanoparticles as carriers for targeted dermatotherapy. The nanoparticles, stable in aqueous conditions, became unstable when contact with skin, causing dye accumulation. They are suitable for transdermal medication delivery, allowing longer drug release and quicker penetration into the epidermis.6
Here, the author Fernanda Zamboni et al. shown that Curcumin, an anti-inflammatory substance from turmeric, can be synthesized using polylactic acid nanoparticles. The process involves evaporation of curcumin and PLA in a single emulsion fluid, producing nanodroplets with nanometer size 7. The NPs have a spherical morphology with a smooth surface. They have an average size of 160 nm, and show a 5.5-fold increase in bioavailability compared to crude curcumin.8-10
Scanning Electron Microscopy (SEM)analysis of nanoparticles (NPs) from fibroblast cells showed curcumin’s cytotoxicity, and they were crosslinked into hydrogels for 3D printing. These hydrogels showed promise in tissue engineering and inhibiting Tumor Necrosis Factor alpha (TNF-α) generation.
Curcumin encapsulation using PLA enhanced mechanical performance of alginate-gelatin hydrogels, promoting cell proliferation and maintaining monocyte viability. A 3D printed bioink with improved shape retention was created.11
Millions of people worldwide suffer from osteoporosis, a condition that is on the rise. A strong bone resorption inhibitor called alendronate (AL) is used to treat and prevent osteoporosis.12,13 Oral administration does have drawbacks, too, including as poor absorption and adverse effects. Using biodegradable polymeric microspheres like PLA, researchers have created local delivery methods to enhance drug loading capacity and sustained release efficacy.14,15
This study by Ibrahim SEN focuses on the synthesis of zidovudine-loaded nanoparticles employing polymers poly (lactic acid) (PLA) and poly (ethylene glycol) (PEG). The method of double emulsion solvent evaporation was used to create the nanoparticles.16 Determining the nanoparticles’ size, shape, zeta potential, polydispersity index, and drug entrapment efficiency were all part of their physicochemical characterization. Wistar rats were given azidothymidine (AZT-loaded) nanoparticles intranasally as part of the investigation, and a Varian HPLC system was used to measure AZT in plasma in vivo.17 The study utilized AZT concentration vs. time curve to determine plasma concentration, Least-squares regression to estimate AUC0–∞, and one-way Analysis of Variance (ANOVA) to account for significant differences.
SEM analysis revealed that the nanoparticles had a spherical form. The physicochemical properties of the particles changed when PEG was added to the mixture. A wider size distribution was indicated by the larger and greater polydispersity index of the PLA–PEG blend nanoparticles. To determine whether the PLA/PEG blend might alter the particle surface charge, the zeta potential was examined. The study found that AZT-loaded PLA and PLA-PEG blend nanoparticles enhanced drug bioavailability, prolonged release, and t1/2, with lower zeta potential, suggesting the formation of a PEG coat on the nanoparticle surface.18 The findings suggested that AZT-loaded nanoparticles might be a useful treatment option for gastrointestinal conditions.17
The study by author Juliana Palacio et al. explores the creation of polylactide with tocopheryl polyethylene glycol succinate (PLATPGS) nanoparticles (MPs/NPs) for regulated medication administration, using materials like lactide, stannous octoate, PBS, sodium azide, vitamin E TPGS, and PLGA. The NPs’ structural integrity was assessed and there in vitro cellular absorption was examined.19-21
The study reveals that PLA-TPGS copolymers outperform PLGA NPs in protein EE due to their hydrophilicity and lower molecular weight, making erosion easier. Their degradation depends on molecular weight and TPGS content, and they maintain BSA (Bovine Serum Albumin) activity for at least 35 days.22
PLA-TPGS nanoparticles are a promising solution for regulating protein and peptide delivery, offering superior drug encapsulation efficiency, controlled release of BSA, and enhanced cellular uptake, making them a promising solution for improving medication efficacy.
The goal of this work done by Mira Dhiraj Buhecha et al. is to create a technique for co-encapsulating PLA nanoparticles with the lipophilic medication’s theophylline and budesonide for prolonged drug release. PLA and PLGA are examples of biodegradable polymers that break down into natural metabolites, producing non-toxic byproducts.23 Increased patient adherence and a longer dose interval are the goals. Budesonide and theophylline co-encapsulated PLA nanoparticles were created using the Double emulsification solvent diffusion (DESD) method. Their properties, including size, zeta potential, surface features, morphology, loading efficiency, drug release, and in vitro deposition, were assessed. Additionally, the nanoparticles’ ability to traverse an airway epithelium and cause cellular toxicity were evaluated.24
Theophylline was dissolved in a 2% w/v PVA solution, and co-encapsulated nanoparticles were made by dissolving PLA and budesonide in dichloromethane (DCM) as part of a modified DESD procedure. To create encapsulated PLA nanoparticles, the nanoparticles were subsequently freeze-dried for a full day. A PerkinElmer Spectrum 65 Infra-red spectrophotometer was used to examine surface properties, and Photon correlation spectroscopy (PCS) was used to measure particle size and charge.25,26 The study used a DSC to determine the melting points of medicines, polymers, and nanoparticles, and found that nebulization effectively decreased theophylline and budesonide concentrations, indicating their potential in medication administration.24 With the innovative use of a second water-miscible organic solvent, acetone/acetonitrile/ethyl acetate, the DESD method used in this study successfully produced nanoparticles and assisted in the encapsulation of a hydrophilic medication (theophylline).27,28 The method was modified and optimized in several ways to optimize the loading efficiency of the nanoparticles. According to morphological analysis, the nanoparticles had a broad particle size distribution, a smooth surface, and were spherical. To examine the surface features and attributes of the nanoparticles and identify any drug adsorption, FT-IR spectroscopy was used.29 The study found that nanoparticles, specifically theophylline and budesonide, did not harm the viability of 16HBE14o-cells. Theophylline was transported more quickly when applied in solution, while budesonide transferred less than 1%. When nebulized, a significant percentage of the nanoparticles stayed in the nebulizer’s chamber, making the formulation suitable for inhalation due to its low droplet sizes and high FPF.24
A modified DESD approach successfully co-encapsulated Budesonide and theophylline in PLA nanoparticles, enhancing drug-loading efficiency, reducing inhaler requirements, and improving patient compliance over a 24-hour period.30 Studies on cytotoxicity and permeability revealed that, at therapeutically relevant doses, the nanoparticle formulations were well tolerated by airway cells. Studies on permeability confirmed the nanoparticles’ ability to deliver drugs continuously. Nebulization of nanoparticle suspensions demonstrated good overall recovery and effective drug deposition in stages 3-5 (particle size < 6.8 μm). Despite their small size, these nanoparticles can be used to provide a new treatment technique for pulmonary medication administration, and when administered with a nebulizer, they can target the right lung location.24
The food industry is studying bacteriostasis in food since contaminated food is the main source of foodborne illnesses. Since ancient times, silver nanoparticles (AgNPs) have been employed as broad-spectrum antibiotics; nevertheless, the majority of manufacturing methods are costly and environmentally unfriendly.31,32,33 Plant extract synthesis of AgNPs has become more and more popular because of their affordable synthesis, environmental friendliness, and usefulness in food packaging applications.34,35 By altering its interactions with components that create films, polylactic acid (PLA), a renewable aliphatic thermoplastic polyester, can regulate the release of AgNPs. The goal of this study is to create an easy, affordable, and environmentally friendly method for preparing AgNP.36, 37
This study by Jun Cheng et al. investigates the production and characterization of MPE and Mango Peel Extract/Silver Nanoparticles (MPE/AgNPs) from mango peel waste. Waste is cleansed, dried, and pulverized before dissolving in dichloromethane. MPE/AgNPs are created by adding 20 weight percent AgNO3. Analyses include FT-IR spectroscopy, opacity characteristics, and X-ray diffraction.38 The results showed that PLA films have potential as biomaterials for various applications, including medical devices. The work looks at the synthesis of silver nanoparticles (AgNPs) using aluminum oxide (AgNPs) and metal-polymer (MPE). The silver element in MPE/AgNPs was found to be zero-valent by XPS measurements, and its binding energy was marginally higher than that of bulk silver metal.39 Perhaps as a result of the chemical environment and the small particle size of the synthesized AgNPs, FT-IR analysis verified the synthesis and stability of MPE/AgNPs. AgNP formation was verified by XRD studies to be crystalline in character, with good dispersion at 2.5–6.5 nm.40,41 The composite films of PLA, PLA/MPE, and PLA/MPE/AgNPs were characterized using binding energy spectra and ATR-FTIR analysis. The composite films showed improved AgNP size and dispersion, reduced particle size distribution, and enhanced rigidity and ductility. These films outperformed pure PLA films in preservation.
The study proved that created films for food packaging are safe, and that PLA/MPE film’s modest antibacterial, antioxidant, and anti-ultraviolet properties improve its ability to preserve freshness.
The work focuses on developing a unique multifunctional thin-film material using AgNPs, which were created by reducing silver nitrate with MPE and combining with PLA. For uniform dispersion, PLA was added, and the diameter of the film ranged from 2.5 to 6.5 nm. Compared to PLA/MPE and pure PLA films, the film’s Water Vapor Transmission Rate and Oxygen Transmission Rate (WVTR and OTR) were superior.38 It also had strong antibacterial properties, exhibited decreased cytotoxicity, and complied with European Commission No. 10/2011 regulations. The exceptional freshness-keeping properties of the PLA/MPE/AgNPs film may help strawberries last longer on the shelf. The study’s conclusions indicate that PLA/MPE/AgNPs film is a promising food packaging material with low cytotoxicity and strong antibacterial activity, which may increase its use in the biomedical industry.38
PLA in co-Aspartic acid Copolymers
Author Nita Tudorachi et al. discussed the synthesis and characterization of poly(lactic acid)-co-aspartic acid copolymers (PLA-co-Asp) and their potential as biodegradable carriers in drug delivery systems.42 Using solution polycondensation, different molar ratios of PLA and L-aspartic acid were tested. Characterization techniques included Fourier Transform Infrared Spectroscopy (FT-IR), Proton Nuclear Magnetic Resonance (1H-NMR), Gel Permeation Chromatography(GPC), Differential Scanning Calorimetry (DSC) analyses.43 The study specifically explored the loading of diclofenac sodium, a widely used anti-inflammatory drug, into the synthesized copolymers and evaluated the in vitro drug release in a phosphate buffer solution at physiological conditions. The results suggest that these copolymers demonstrate favorable properties for use in controlled drug delivery applications, showing a balance of hydrophobicity and hydrophilicity, along with good biodegradability.44
PLA-co-Asp copolymers are being developed for biodegradable drug delivery systems, with properties influenced by molar ratios. In vitro studies show release of diclofenac sodium depends on copolymer composition and structure.45 Characterization techniques provide insights into thermal stability, molecular weight, and release kinetics. These copolymers offer a promising alternative to conventional drug carriers.46
PLA-PEG Nanoparticles in protein corona formation and brain delivery property
Although it is essential for preserving the internal environment of the brain, the blood-brain barrier (BBB) restricts the absorption of the majority of medications. Brain illnesses are being treated with nanoparticulate drug delivery devices, albeit only a small portion of nanomedicine can get through the blood-brain barrier. The safety and clinical translation capabilities of PEG-PLA nanoparticles have led to their widespread use in medication delivery.47,48 A study done by Yuyun Tanga et al. reveals that drug-loaded PEGPLA nanoparticles can penetrate the blood-brain barrier and reach microglia. The study found that drug loading and dosage frequency can alter the protein corona on PEG-PLA nanoparticles, affecting their ability to carry drugs and accumulate in microglia. Multiple-dose or aM-loaded nanoparticles showed greater microglial accumulation and brain distribution.49
PLA Hydrogels in used in Injectables
Scientists named Xinran Yang a, b, c et al. have created injectable hydrogels with anti-adhesive qualities using gelatin and polylactic acid treated with sulfobetaine.50,51 Although these hydrogels can rapidly seal wounds and conceal uneven bleeding sites, their viscosity may lead to issues. For incompressible wounds, they have shown tissue adherence, quick hydrophobic interface reaction, and quick hemostasis.52 The hydrogels could be used as prehospital emergency treatments for surgical recovery and incompressible hemorrhage.53
For quick hemostasis and tissue adhesion prevention, the study created injectable PLA-gelatin hydrogels modified with sulfobetaine. Through water treatment and the anti-protein properties of sulfobetaine, the hydrogels PMP-30 and PLA-MPS-PEG (often referred to as PMP) create an anti-adhesive surface.54 According to in vitro studies, it performed better than commercial hydrogels and sponges, establishing blood coagulation in 45 seconds with lower BCI and superior clot adsorption. The hydrogel is perfect for additional animal research because of its improved hydrophilicity and electrostatic interactions, which speed up hemostasis. The biological uses of zwitterionic sulfonate betaine PLA are advanced by this work, especially for hemostatic purposes.55
PMP-30, a new hemostatic substance, has been created for in vivo anti-adhesion and incompressible bleeding. Hydrophilic PEG and anti-protein zwitterionic sulfobetaine have been added to PMP-30, a hydrophobic PLA hain.56 It has better anti-adhesion qualities than commercially available hydrogel materials and forms micelles in watery conditions. PMP-30 works well in several hemorrhage models and is useful in the pre-hospital management of incompressible hemorrhage.57,58
PLA in Microspheres
The author Shunyu Chen et al. investigates the production of EPLA nanofibrous microspheres and EPLA/nHAp composite microspheres using poly-L-lactic acid (PLLA) as a model medication. The loading efficiency of alendronate was assessed using UV-V spectroscopy. The study used murine pre-osteoblast MC3T3-E1 cells cultured in α-MEM and CCK-8 assay to assess cell viability. Statistical analysis was performed using GraphPad Prism 6.0.59
After being digested, the cells were ready for usage. The Cell Counting Kit-8 (CCK-8) assay, which gauged cell activity and proliferation on microspheres, was used to assess cell viability. GraphPad Prism 6.0 was used to do the statistical analysis, and the findings were displayed as mean ± standard deviation. When the p-value was 0.05, statistical significance was taken into account.60
The EPLA/nHAp (Aminated modified polylactic acid/nanohydroxyapatite composite microsphere), a novel drug delivery system, shows improved drug loading capacity and sustained release performance, with alendronate as a model drug, suggesting its potential for bone tissue repair.59
Gold coated PLA Nanofiber based electrochemical Aptasensor
Composed of polylactic acid and gold, the conductive nanofiber has potential applications in wearable biosensors, e-textiles, and tissue engineering. It was used to develop an electrochemical aptamer-based biosensor for cortisol monitoring. Cortisol is a hormone that is necessary for metabolism, immune response, and stress response. Adrenal insufficiency is indicated by low cortisol levels, while obesity, diabetes, heart disease, and cognitive decline are caused by excessive levels. Accurate cortisol monitoring can provide early warning indicators of health concerns and help treat stress-related disorders. Currently, the most reliable method for identifying cortisol in biofluids is Enzyme-Linked Immunoassay (ELISA) although it has drawbacks such as lengthy wait periods, expensive expenses, and the need for specialized equipment. The sensor’s sensitivity, operability, and biofluid adaptability can all be enhanced by the conductive Au-PLA nanofiber sheet.
The author Mkliwa Koumbial et al. represents a novel method for producing highly conductive and flexible nanofibers using electrospinning and electroless deposition.61 The nanofiber sheet, coated with Au, monitors cortisol, a stress biomarker, suitable for wearable technologies and with a low detection limit.62 The Aptasensor, a gold-coated PLA nanofiber sensor, detects cortisol in artificial sweat and saliva, offering fast, wide detection range, and stretchability, making it ideal for real-time stress monitoring.63
Three concentrations of PLA solution were electrospun to produce microfibers and nanofibers, with uneven, bead-free nanofibers. PLA 8 wt.% solution produced homogenous, bead-free microfibers, suitable for further testing.64
Using an aptamer, a conductive nanofiber (CN) sensor was used to detect cortisol in artificial biofluids selectively, with a detection limit of 1 pg./ml.
Transdermal iontophoresis of flufenamic acid (FFA) loaded PLGA nanoparticles
Because transdermal administration of pharmacologically active drugs has the potential to address problems including fluctuating bioavailability and poor gastrointestinal absorption, it has been a focus of attention for decades. However, the skin barrier’s characteristics have restricted its clinical application. To get over these restrictions, methods like iontophoresis and drug-loaded nanosized carrier systems have been created.65 The study aimed to create FFA-loaded nanoparticles, investigate transdermal transport of free and nano-encapsulated FFA, determine iontophoretic current’s impact on FFA penetration, and visualize skin distribution after iontophoretic treatment.65
The study by K. Malinovskaja- Gomez et al. explores the synthesis of flufenamic acid-loaded nanoparticles for transdermal iontophoretic tests, using various compounds and techniques, and examining their shape, colloidal properties, and drug release through skin.66
The benefits of iontophoretic treatment for transdermal drug delivery were two-fold.67 When an electric current is applied, the nanoparticles are forced into hair follicles, which serve as a drug depot. The drug is then released from the nanoparticles and transported via the skin to the bloodstream.68 Because of its free acid end functionality and the quickest degradation period, Resomer RG 503H grade PLGA was selected for this investigation. The average size of the almost spherical, negatively charged FFA-loaded FA-PLGA nanoparticles was 174.2 ± 1.8 nm.69 The drug-free reference nanoparticles had a similar size but slightly lower negative zeta potential.66
The study examined the release kinetics of FFA from FA-PLGA nanoparticles in static Franz diffusion cells. Results showed that iontophoresis treatment significantly improved FFA penetration into the skin, while alternate current was ineffective. The study also examined the impact of current type on transdermal medication penetration.70
PLA in liposomal drug delivery system
Traditional medicine makes use of glycyrrhizin (GL) and glycyrrhetinic acid (GA), which are present in liquor ice root (Glycyrrhiza glabra).71 GA has higher antibacterial, antiviral, and antihepatotoxic properties.72 The negative effects include pseudo-aldosteronism, salt retention, hypertension, and hypokalemia. A hyper mineralocorticoid effect can result from excessive quantities in the kidney because they inhibit renal 11-hydroxy-steroid-dehydrogenase and increase cortisol levels.73,74
A modified GA liposome (PL-GA) was developed by Juan Li et al. to enhance liver therapeutic benefits. A high-performance liquid chromatographic approach was used to assess the effectiveness of GA delivery to the liver. Three GA formulations were administered to ICR mice, showing high drug loading and entrapment efficiency. The mPEGylated PL-GA formulation showed enhanced efficacy, reduced renal toxicity, and improved liver absorption.75
Hemostasis, inflammation, proliferation, and tissue remodeling are all interrelated components of the intricate physiological process that is wound healing. Stress from movement puts wounds at higher risk of infection and delayed healing, particularly those in movable areas like the neck.76,77 This stress can lead to prolonged discomfort, inflammation, and, in severe cases, disability. To address these challenges, wound dressings must possess mechanical properties that align with the tissue’s needs and include antibacterial and anti-inflammatory features.78,79 Enhancing the toughness of biodegradable PLA wound dressings, such as hollow porous microfibers (HPMFs), is crucial for efficient healing in movable body areas, promoting cell proliferation and wound healing.
Hollow porous PLA/PBAT composite microfibers
The study by author Yufei Liu et al. utilized coaxial electrospinning to create a PLA/PBAT-based HPMF composite dressing, enhancing toughness and biodegradability. The composite promoted wound healing, cell culture, and effectively regulated CRT-15 release, with in vivo wound healing experiments showing it can stimulate healing on the thirteenth day.80 Anticancer drug combinations are increasingly being researched, with the primary goals being synergy, non-overlapping toxicity, and overcoming drug resistance.81,82 The FDA has recognized the importance of emerging and innovative anticancer combinations and has drafted guidance for co-development.83 However, opportunities in multi-drug delivery have been overlooked. Block copolymers based on poly(ethylene glycol) and poly(β-hydroxy acid) are being studied for drug delivery due to their biocompatibility, controlled biodegradability, ease of polymer synthesis, and potential for localized cancers. These block copolymers have been tested extensively in humans and have proven safety profiles .84,85
PEG-b-PLA micelles and PLGA-b-PEG-b-PLGA sol-gels for drug delivery
Polymeric micelles, including polyethylene glycol-block-poly (lactic acid) PEG-b-PLA micelles, are used for drug delivery and clinical trials targeting key cancer targets. These micelles are smaller, less stable, and faster to release anticancer agents in blood, similar to free paclitaxel.86 PEG-b-PLA micelles have been shown to slow drug release for drug targeting by the EPR effect. Genexol-PM®, an injectable formulation of paclitaxel based on PEG-b-PLA micelles, has gained approval in several Asian countries and has high response rates in clinical trials with patients with non-small cell lung, gastric, and breast cancers.87 Triolimus, a novel 3-drug nanotherapeutic containing paclitaxel, 17-AAG, and rapamycin, has been investigated for its combined anticancer action on microtubules, Hsp90, and mTOR.88 PLGA-b-PEG-b-PLGA sol-gels have shown significant clinical progress in localized cancer control as an adjunct to surgery and radiation.89
Regel®, a sol-gel containing PLGA-b-PEG-b-PLGA, is being studied by Hyunah Cho et al. for systemic and local drug delivery and is entering clinical trials for esophageal cancer treatment. It can be synthesized with poly(ethylene glycol) and incorporated with hydrophilic and hydrophobic drugs .90 Oncogel™, a drug containing paclitaxel, is a potent radiosensitizer that clears slowly over a 6-week period with a half-life of 21 days, with minimal recovery in major organs. It entered phase 2 clinical trials for the treatment of esophageal cancer due to its ease of access to tumors through endoscopic ultrasound-guided injection and potential synergy with radiation treatment. Oncogel™ has been evaluated in an intracranial 9L gliosarcoma rat model as an adjunct to radiation, with a combination of Oncogel™ and oral temozolomide significantly increasing survival times of rats. Trio gel, a sol-gel similar to PEG-b-PLA micelles, has a multi-drug capacity for paclitaxel, 17-AAG, and rapamycin, enabling concurrent local multi-drug delivery for cancer treatment.91 Genexol-PM®/Cynviloq™ and Oncogel™ are advancements in drug delivery, advancing paclitaxel into clinical trials. PEG-b-PLA and PLGA-b-PEG-b-PLGA have good safety records, offering potential for systemic and local drug delivery. Challenges in chemistry, manufacturing, and control remain.92
Table 2: Marketed formulation of PLA in different dosage forms
| Sr. No. | Poly Lactic Acid used in different dosage forms | Marketed formulations | Manufacturer |
| 1. | Nanoparticles | Budesonide | Metrochem API Private Limited (HYDERABAD, INDIA) |
| Theophylline | Manus Aktteva Biopharma LLP (GUJRAT, INDIA) | ||
| 2. | Biodegradable drug delivery system | Lupron Depot® | AbbVie Inc (CHICAGO, USA) |
| Sandostatin LAR® | Novartis Pharmaceuticals Corporation (BASEL, SWITZERLAND) | ||
| 3. | Brain delivery system | Levodopa | Allmpus Laboratories Pvt. Ltd. (MAHARASHTRA, INDIA) |
| Rivastigmine | Sun Pharmaceutical Industries Ltd. (MAHARASHTRA, INDIA) | ||
| 4. | Injectables | Histrelin Acetate Implant (Supprelin LA) | Endo Pharmaceuticals |
| Histrelin Acetate Implant (Supprelin LA) | |||
| 5. | Microspheres | Leuprolide Acetate (Lupron Depot) | AbbVie Inc. (CHICAGO, USA) |
| Histrelin Acetate (Vantas, Supprelin LA) | Endo Pharmaceuticals (Rochester, Michigan, USA) | ||
| 6. | Nanofibers | Paclitaxel (Taxol) | Pfizer (New York, USA) |
| Doxorubicin (Adriamycin) | Hikma pharmaceuticals (Ohio, USA) | ||
| 7. | Anticancer drug delivery system | Doxorubicin (Adriamycin) | Hikma pharmaceuticals (New Jersey, USA) |
| MediChip | BioSapien Inc (New York City, United States) | ||
| 8. | Food Packaging | Probiotics | Vitaquest (New Jersey, USA) |
| Omega-3 Fatty Acids (e.g., Fish Oil) | Kerry Group Plc (County Kerry, Ireland) | ||
| 9. | Transdermal drug delivery system | Fentanyl | Johnson and Johnson (New Brunswick, USA) |
| Nicotine | Alchem International Pvt. Ltd (New Delhi, INDIA) | ||
| 10. | Pulmonary drug delivery system | Pulmospheres™ | Alliance Pharmaceuticals (Wiltshire, UK) |
| Albuterol (Salbutamol) | Orex Pharma (MAHARASHTRA, INDIA) | ||
| 11. | Liposomes | Doxorubicin (Doxil) | Sun Pharmaceutical Industries (GUJRAT, INDIA) |
| Amphotericin B (AmBisome) | Astellas Pharma US, Inc (Northbrook, US) | ||
| 12. | Sol- Gels | Dexamethasone | Amneal Pharmaceuticals (GUJRAT, INDIA) |
| Methotrexate | Teva Pharmaceuticals (MAHARASHTRA, INDIA) |
Table 3: Patents of Poly lactic acid granted to inventors
| TYPES | APPLICATION | DESCRIPTION | ISSUE DATE | PATENT NO | INVENTORS |
| Microparticles for Drug Delivery | The technology is widely applied in vaccines, peptide and protein delivery, as well as cancer treatments. | This patent involves the development of PLA microparticles to encapsulate therapeutic agents. The microparticles are designed to slowly release drugs over time, ensuring a controlled and sustained delivery. | March 18, 1997 | US5612345A | Joseph F. McCall, Alexander J. McHugh |
| PLA Nanoparticles for Targeted Drug Delivery | PLA nanoparticles are used in cancer therapy, where targeted drug delivery to tumor sites reduces side effects. | This patent focuses on PLA-based nanoparticles designed for targeted drug delivery, particularly for delivering poorly soluble drugs. These nanoparticles can improve the solubility of hydrophobic drugs and provide controlled release over extended periods | February 24, 2009 | US7494602B2 | Jong Y. Park, Kyu H. Kim, M. J. Hee, Soo Y. Kim |
| PLA and PEG-PLA Copolymer Systems | This type of formulation is especially useful for delivering biologic drugs, such as proteins or vaccines. | This patent describes the use of PLA and PEG (polyethylene glycol) copolymers in drug delivery systems to enhance solubility, stability, and prolong the release profile of drugs. The PEG-PLA copolymer is commonly used in biologic drug delivery, improving circulation time and reducing immune system recognition. | August 6, 2002 | US6428909B1 | |
| PLA-based Injectable Drug Delivery Systems | Such formulations are used for long-acting injectable drugs for chronic diseases, hormone therapies, or oncology treatments | This patent pertains to injectable PLA-based drug delivery systems, focusing on sustained-release formulations. The injectable formulations are used for chronic treatments, such as hormone replacement therapies or long-acting injectable medications. | January 1, 2002 | US6334974B1 | Robert Langer, Jeffrey M. Kohn |
| PLA-PLGA [Poly(lactic-co-glycolic acid)] Nanoparticles | Used for sustained release of chemotherapeutic agents, pain medications, and gene delivery. | Though more commonly associated with PLGA, this patent covers polymer blends that involve PLA and PLGA for drug encapsulation and controlled release. These copolymers are popular in drug delivery systems because they offer control over degradation rates and drug release kinetics. | October 19, 1999 | US5968922A | Susan P. Packhauser, John W. Hall, David S. Jayne |
Discussion
PLA Nanoparticles: Characteristics and Methods Made from natural resources, PLA is a biocompatible and biodegradable polymer. Several methods are used to create its nanoparticles, including: The most used technique, emulsification-solvent evaporation, guarantees high drug encapsulation efficiency. Nanoprecipitation: The process of creating stable, homogeneous nanoparticles. Spray drying: Provides scalability for production on a big scale.
Uses for local Dermatotherapy
Treatment for Acne: PLA nanoparticles containing antibiotics (such as erythromycin and clindamycin) have shown enhanced drug penetration and extended drug retention in sebaceous glands. Management of Psoriasis: PLA-based nanoparticles that deliver corticosteroids have demonstrated improved therapeutic outcomes with less systemic adverse effects. Wound Healing: Growth factors encapsulated in PLA nanoparticles have improved tissue regeneration, enabled controlled release, and decreased the danger of infection.
Skin Cancer Therapy: Localized medication delivery is made possible by PLA nanoparticles coated with chemotherapy drugs, which reduce toxicity to nearby healthy tissue.
Analysis of a Case Study
The potential of poly (Lactic Acid)-co-aspartic acid copolymers to improve drug encapsulation effectiveness and regulate release rates in drug delivery systems has been investigated. PEG-PLA nanoparticles have been investigated for their ability to load drugs and the impacts of repeated administrations, especially with regard to protein corona development and how it affects the delivery of drugs to the brain. While curcumin-encapsulated PLA nanoparticles in bioinks have demonstrated promise for in situ immunoregulation and tissue engineering, water-triggered injectable PLA hydrogels have been produced for hemostatic applications and tissue anti-adhesion. A novel drug delivery method for treating osteoporosis has been studied: biomimetic mineralization on PLA microspheres. Through the use of electrochemical sensors and gold-coated PLA nanofibers, non-invasive cortisol monitoring in saliva and perspiration is now possible. The delivery of anticancer drugs has advanced thanks to PLGA-based nanofibers, with a focus on fiber-based PLA drug carriers. Boric acid and levite have been added to PLA films in the food packaging industry for their mechanical benefits and antibacterial qualities. The administration of zidovudine intranasally via PLA nanoparticles has demonstrated encouraging outcomes in terms of increasing drug bioavailability. For oral drug administration applications, PEGylation of PLA has been investigated because it enhances colloidal stability and mucus penetration. Enhancing transdermal drug delivery has also been studied using transdermal iontophoresis of PLGA nanoparticles loaded with flufenamic acid. PLA-PBSA eco-composite films have shown increases in toughness and flexibility for use in packaging. The use of PLA nanoparticles in pulmonary drug administration has also been explored, with an emphasis on co-encapsulation techniques for both lipophilic and hydrophilic medications.
In packaging, the mechanical and biodegradable qualities of combining PLA with tapioca starch have been investigated. PLA-based catalytic microflow reactors have been investigated for their potential for preparation, use, and recycling. In order to promote prolonged medication distribution and excretion in vivo, mPEG-PLA liposomes have been studied for liver drug delivery. For better wound healing applications, hollow porous PLA/PBAT composite microfibers have been developed. For environmentally friendly antimicrobial packaging, silver nanoparticles have been created and integrated into PLA food packaging films using extract from mango peels. PLGA-b-PEG-b-PLGA sol-gels and PEG-b-PLA micelles have been studied as multipurpose nanocarriers in drug delivery systems. Lastly, SERS-based contamination detection has been used using silver/PLA-coated papers, demonstrating their promise for analytical uses. created an innovative sensor platform to identify environmental contaminants.
PLA Nanoparticle Benefits for Dermatotherapy:
Improved Drug Stability: Guards against environmental deterioration of active components. Controlled and Sustained Release: Improves patient adherence and lowers dosage frequency.
Drug diffusion across the stratum corneum is facilitated by improved skin penetration. Safety and Biodegradability: PLA breaks down into non-toxic metabolites, lowering the possibility of buildup over time.
Challenges and Limitations
Complexity of Formulation: Needs to optimize medication loading efficiency, charge, and nanoparticle size. Production Cost: Using advanced production techniques might be costly.
Skin Irritation Risk: Hypersensitivity reactions could be brought on by some formulations.
Prospects for the Future
Optimizing PLA nanoparticle compositions, improving their targeting capabilities, and securing regulatory approval for therapeutic uses all require more research. Functionalized PLA nanoparticles and hybrid systems are two examples of nanotechnology advancements that could lead to more successful dermatological treatments.
Conclusion
PLA nanoparticles offer better treatment results, increased penetration, and regulated drug distribution, making them a novel technique in dermatotherapy. Notwithstanding obstacles, it is anticipated that continued research and technical developments will get past these restrictions and open the door for their extensive clinical application. The knowledge gathered from a variety of case studies highlights PLA’s adaptability in biomedical engineering and medication delivery.
Acknowledgement
The authors express their sincere gratitude to Modern College of Pharmacy, Pune, affiliated to Savitribai Phule Pune University, for providing the necessary facilities, guidance, and constant encouragement throughout the completion of this review work. The authors are also thankful to the teaching and non-teaching staff of the Department of Pharmaceutics for their valuable support and cooperation.
The authors extend their appreciation to all researchers and scholars whose previous studies and publications have contributed significantly to the understanding of polylactic acid and its applications in novel drug delivery systems.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contribution Statement
Krishna Bhagwan Bharti – Conceptualization, Methodology, Writing – Original Draft.
Pranoti Sunil Mohod – Data Collection, Analysis, Writing – Review & Editing.
Raksha Laxman Mhetre – Visualization, Supervision, Project Administration.
Nilesh Shrikant Kulkarni – Resources, Supervision.
Shashikant Nivrutti Dhole – Final approval of Manuscript.
Refrences
- Blasi P. Poly (lactic acid)/poly (lactic-co-glycolic acid)-based microparticles: An overview. J Pharm Investig. 2019;49:337-346. https://doi.org/10.1007/s40005-019-00453-z
CrossRef - Cheng Y, Deng S, Chen P, Ruan R. Polylactic acid (PLA) synthesis and modifications: a review. Front Chem China. 2009;4:259-264.DOI 10.1007/s11458-009-0092-x
CrossRef - Blasi P. Poly(lactic acid)/poly(lactic-co-glycolic acid)-based microparticles: an overview. J Pharm Investig. 2019;49:337-346. https://doi.org/10.1007/s40005-019-00453-z
CrossRef - Li L, Hoffman RM. Topical liposome delivery of molecules to hair follicles in mice. J Dermatol Sci. 1997;14(2):101-108.https://doi.org/10.1016/S0923-1811(96)00557-9
CrossRef - Alvarez-Román R, Naik A, Kalia YN, Guy RH, Fessi H. Skin penetration and distribution of polymeric nanoparticles. J Control Release. 2004;99(1):53-62. https://doi.org/10.1016/j.jconrel.2004.06.015
CrossRef - Vogt A, Mandt N, Lademann J, Schaefer H, Blume-Peytavi U. Follicular targeting – a promising tool in selective dermatotherapy. J Invest Dermatol Symp Proc. 2005;10(3):252-255. https://doi.org/10.1111/j.1087-0024.2005.10124.x
CrossRef - Loibl M, Wuertz-Kozak K, Vadala G, Lang S, Fairbank J, Urban JP. Controversies in regenerative medicine: should intervertebral disc degeneration be treated with mesenchymal stem cells? JOR Spine. 2019;2(1):e1043. https://doi.org/10.1002/jsp2.1043
CrossRef - Rachmawati H, Yanda YL, Rahma A, Mase N. Curcumin-loaded PLA nanoparticles: formulation and physical evaluation. Sci Pharm. 2016;84(1):191.http://dx.doi.org/10.3797/scipharm.ISP.2015.10
CrossRef - Liang H, Friedman JM, Nacharaju P. Fabrication of biodegradable PEG–PLA nanospheres for solubility, stabilization, and delivery of curcumin. Artif Cells Nanomed Biotechnol. 2017;45(2):297-304. https://doi.org/10.3109/21691401.2016.1146736
CrossRef - Xie X, Tao Q, Zou Y, et al. PLGA nanoparticles improve the oral bioavailability of curcumin in rats: characterizations and mechanisms. J Agric Food Chem. 2011;59(17):9280-9289. https://pubs.acs.org/doi/10.1021/jf202135j
CrossRef - Wang Y, Che M, Xin J, Zheng Z, Li J, Zhang S. The role of IL-1β and TNF-α in intervertebral disc degeneration. Biomed Pharmacother. 2020;131:110660. https://doi.org/10.1016/j.biopha. 2020.110660
CrossRef - Leslie WD, Seeman E, Morin SN, Lix LM, Majumdar SR. The diagnostic threshold for osteoporosis impedes fracture prevention in women at high risk for fracture: a registry-based cohort study. 2018;114:298-303. https://doi.org/10.1016/j.bone.2018.07.004
CrossRef - Chen Q, Zheng C, Li Y, et al. Bone targeted delivery of SDF-1 via alendronate functionalized nanoparticles in guiding stem cell migration. ACS Appl Mater Interfaces. 2018;10(28):23700-23710. https://pubs.acs.org/doi/abs/10.1021/acsami.8b08606
CrossRef - Liu YF, Liu R, Li XY, Song Z, Zhao XH. Development of docetaxel and alendronate-loaded chitosan-conjugated polylactide-co-glycolide nanoparticles: in vitro characterization in osteosarcoma cells. Trop J Pharm Res. 2016;15(7):1353-1360. http://dx.doi.org/10.4314/tjpr.v15i7.1
CrossRef - Yang H, Gao H, Wang Y. Hollow hydroxyapatite microsphere: a promising carrier for bone tissue engineering. J Microencapsul. 2016;33(5):421-426. https://doi.org/10.1080/02652048.2016.1202347
CrossRef - Alonso MJ. Nanomedicines for overcoming biological barriers. Biomed Pharmacother. 2004;58(3):168-172. https://doi.org/10.1016/j.biopha.2004.01.007
CrossRef - Mainardes RM, Gremião PD. Reversed phase HPLC determination of zidovudine in rat plasma and its pharmacokinetics after a single intranasal dose administration. Biol Res. 2009;42(3):357-364. http://dx.doi.org/10.4067/S0716-97602009000300010
CrossRef - Blum MR, Liao SH, Good SS, De Miranda P. Pharmacokinetics and bioavailability of zidovudine in humans. Am J Med. 1988;85(2A):189-194. https://europepmc.org/article/med/3165603
- Plapied L, Duhem N, des Rieux A, Préat V. Fate of polymeric nanocarriers for oral drug delivery. Curr Opin Colloid Interface Sci. 2011;16(3):228-237. https://doi.org/10.1016/j.cocis.2010.12.005
CrossRef - Ogier J, Arnauld T, Doris E. Recent advances in the field of nanometric drug carriers. Future Med Chem. 2009;1(4):693-711. https://doi.org/10.4155/fmc.09.48
CrossRef - Felice B, Prabhakaran MP, Rodriguez AP, Ramakrishna S. Drug delivery vehicles on a nano-engineering perspective. Mater Sci Eng C. 2014;41:178-195. https://doi.org/10.1016/j.msec.2014. 04.049
CrossRef - Diab R, Jaafar-Maalej C, Fessi H, Maincent P. Engineered nanoparticulate drug delivery systems: the next frontier for oral administration? AAPS J. 2012;14:688-702. DOI: 10.1208/s12248-012-9377-y
CrossRef - Merkli A, Tabatabay C, Gurny R, Heller J. Biodegradable polymers for the controlled release of ocular drugs. Prog Polym Sci. 1998;23(3):563-580. https://doi.org/10.1016/S0079-6700(97)00048-8
CrossRef - Buhecha MD, Lansley AB, Somavarapu S, Pannala AS. Development and characterization of PLA nanoparticles for pulmonary drug delivery: co-encapsulation of theophylline and budesonide, a hydrophilic and lipophilic drug. J Drug Deliv Sci Technol. 2019;53:101128. https://doi.org/10.1016/ j.jddst.2019.101128
CrossRef - Reis CP, Neufeld RJ, Ribeiro AJ, Veiga F. Nanoencapsulation I. Methods for preparation of drug-loaded polymeric nanoparticles. Nanomed Nanotechnol Biol Med. 2006;2(1):8-21.https://doi.org/ 10.1016/j.nano.2005.12.003
CrossRef - Li Z, Liu K, Sun P, et al. Poly(D,L-lactide-co-glycolide)/montmorillonite nanoparticles for improved oral delivery of exemestane. J Microencapsul. 2013;30(5):432-440. https://doi.org/10.3109/ 02652048.2012.746749
CrossRef - Lai P, Daear W, Löbenberg R, Prenner EJ. Overview of the preparation of organic polymeric nanoparticles for drug delivery based on gelatin, chitosan, poly(D,L-lactide-co-glycolic acid) and polyalkylcyanoacrylate. Colloids Surf B Biointerfaces. 2014;118:154-163. https://doi.org/10.1016/ j.colsurfb.2014.03.017
CrossRef - Zambaux MF, Bonneaux F, Gref R, et al. Influence of experimental parameters on the characteristics of poly(lactic acid) nanoparticles prepared by a double emulsion method. J Control Release. 1998;50(1-3):31-40. https://doi.org/10.1016/S0168-3659(97)00106-5
CrossRef - Sinha B, Mukherjee B, Pattnaik G. Poly-lactide-co-glycolide nanoparticles containing voriconazole for pulmonary delivery: in vitro and in vivo study. Nanomed Nanotechnol Biol Med. 2013;9(1):94-104. https://doi.org/10.1016/j.nano.2012.04.005
CrossRef - Forbes B, Shah A, Martin GP, Lansley AB. The human bronchial epithelial cell line 16HBE14o− as a model system of the airways for studying drug transport. Int J Pharm. 2003;257(1-2):161-167. https://doi.org/10.1016/S0378-5173(03)00129-7
CrossRef - Chen J, Ying GG, Deng WJ. Antibiotic residues in food: extraction, analysis, and human health concerns. J Agric Food Chem. 2019;67(27):7569-7586. https://pubs.acs.org/doi/abs/ 10.1021/acs.jafc.9b01334
CrossRef - Pooresmaeil M, Namazi H. Developments on carboxymethyl starch-based smart systems as promising drug carriers: a review. Carbohydr Polym. 2021;258:117654. https://doi.org/10.1016/ j.carbpol.2021.117654
CrossRef - Pooresmaeil M, Namazi H. Advances in development of the dendrimers having natural saccharides in their structure for efficient and controlled drug delivery applications. Eur Polym J. 2021;148:110356. https://doi.org/10.1016/j.eurpolymj.2021.110356
CrossRef - Nasiriboroumand M, Montazer M, Barani H. Preparation and characterization of biocompatible silver nanoparticles using pomegranate peel extract. J Photochem Photobiol B. 2018;179:98-104. https://doi.org/10.1016/j.jphotobiol.2018.01.006
CrossRef - López-Miranda JL, Vázquez M, Fletes N, Esparza R, Rosas G. Biosynthesis of silver nanoparticles using a Tamarix gallica leaf extract and their antibacterial activity. Mater Lett. 2016;176:285-289. https://doi.org/10.1016/j.matlet.2016.04.126
CrossRef - Kanmani P, Rhim JW. Antimicrobial and physical–mechanical properties of agar-based films incorporated with grapefruit seed extract. Carbohydr Polym. 2014;102:708-716. https://doi.org/10.1016/j.carbpol.2013.10.099
CrossRef - Pooresmaeil M, Namazi H. Preparation and characterization of polyvinyl alcohol/β-cyclodextrin/GO–Ag nanocomposite with improved antibacterial and strength properties. Polym Adv Technol. 2019;30(2):447-456. https://doi.org/10.1002/pat.4484
CrossRef - Cheng J, Lin X, Wu X, Liu Q, Wan S, Zhang Y. Preparation of a multifunctional silver nanoparticles polylactic acid food packaging film using mango peel extract. Int J Biol Macromol. 2021;188:678-688. https://doi.org/10.1016/j.ijbiomac.2021.07.161
CrossRef - Zhang Q, Wang Y, Zhang W, et al. In situ assembly of well-dispersed Ag nanoparticles on the surface of polylactic acid–Au@polydopamine nanofibers for antimicrobial applications. Colloids Surf B Biointerfaces. 2019;184:110506. https://doi.org/10.1016/j.colsurfb.2019.110506
CrossRef - Meng Y, Sun Y. Development of biogenic silver nanoparticle using Rosa chinensis flower extract and its antibacterial property. J Nanosci Nanotechnol. 2016;16(4):3969-3972. https://doi.org/10.1166/jnn.2016.11899
CrossRef - Meng F, Zhang Y, Xiong Z, Wang G, Li F, Zhang L. Mechanical, hydrophobic and thermal properties of an organic–inorganic hybrid carrageenan–polyvinyl alcohol composite film. Compos Part B Eng. 2018;143:1-8. https://doi.org/10.1016/j.compositesb.2017.12.009
CrossRef - Gad HA, El-Nabarawi MA, Abd El-Hady SS. Formulation and evaluation of PLA and PLGA in situ implants containing secnidazole and/or doxycycline for treatment of periodontitis. AAPS PharmSciTech. 2008;9:878-884. DOI: 10.1208/s12249-008-9126-9
CrossRef - Tomida M, Nakato T, Matsunami S, Kakuchi T. Convenient synthesis of high molecular weight poly(succinimide) by acid-catalysed polycondensation of L-aspartic acid. Polymer (Guildf). 1997;38(18):4733-4736. https://doi.org/10.1016/S0032-3861(96)01079-8
CrossRef - Shinoda H, Asou Y, Suetsugu A, Tanaka K. Synthesis and characterization of amphiphilic biodegradable copolymer, poly(aspartic acid-co-lactic acid). Macromol Biosci. 2003;3(1):34-43. https://doi.org/10.1002/mabi.200390007
CrossRef - Coats AW, Redfern JP. Kinetic parameters from thermogravimetric data. 1964;201(4914):68-69. https://doi.org/10.1002/pol.1965.110031106
CrossRef - Lipsa, R., Tudorachi, N., Vasile, C., Chiriac, A., & Grigoras, A. (2013). Novel environmentally friendly copolymers carboxymethyl starch grafted poly (lactic acid). Journal of Polymers and the Environment, 21(2), 461–471. DOI 10.1007/s10924-012-0470-1
CrossRef - Dong, X. (2018). Current strategies for brain drug delivery. Theranostics, 8(6), 1481–1493. doi: 10.7150/thno.21254
CrossRef - Zhou, Y., Peng, Z., Seven, E. S., & Leblanc, R. M. (2018). Crossing the blood–brain barrier with nanoparticles. Journal of Controlled Release, 270, 290–303. https://doi.org/10.1016/j.jconrel. 2017.12.015
CrossRef - Schöttler, S., Becker, G., Winzen, S., Steinbach, T., Mohr, K., Landfester, K., Mailänder, V., & Wurm, F. R. (2016). Protein adsorption is required for stealth effect of poly (ethylene glycol)- and poly (phosphoester)-coated nanocarriers. Nature Nanotechnology, 11(4), 372–377. https://www.nature.com/articles/nnano.2015.330
CrossRef - Chen, S., Zheng, J., Li, L., & Jiang, S. (2005). Strong resistance of phosphorylcholine self-assembled monolayers to protein adsorption: insights into nonfouling properties of zwitterionic materials. Journal of the American Chemical Society, 127(41), 14473–14478. https://pubs.acs.org/doi/abs/ 10.1021/ja054169u
CrossRef - Jiang, S., & Cao, Z. (2010). Ultralow-fouling, functionalizable, and hydrolyzable zwitterionic materials and their derivatives for biological applications. Advanced Materials, 22(9), 920–932. https://doi.org/10.1002/adma.200901407
CrossRef - Yang, G., Xiao, Z., Long, H., Ma, K., Zhang, J., Ren, X., & Zhang, J. (2018). Assessment of the characteristics and biocompatibility of gelatin sponge scaffolds prepared by various crosslinking methods. Scientific Reports, 8(1), 1616. https://www.nature.com/articles/s41598-018-20006-y.pdf
CrossRef - Su, X., Xie, W., Wang, P., Tian, Z., Wang, H., Yuan, Z., Liu, X., & Huang, J. (2021). Strong underwater adhesion of injectable hydrogels triggered by diffusion of small molecules. Materials Horizons, 8(8), 2199–2207. https://doi.org/10.1039/D1MH00533B
CrossRef - Narayanan, A., Xu, Y., Dhinojwala, A., & Joy, A. (2020). Advances in photoreactive tissue adhesives derived from natural polymers. ChemEngineering, 4(2), 32. https://doi.org/10.3390/chemengineering 4020032
CrossRef - Park, J., Kim, T. Y., Kim, Y., An, S., Kim, K. S., Kang, M., Kim, S. A., Kim, J., Lee, J., Cho, S. W., & Seo, J. (2023). A mechanically resilient and tissue-conformable hydrogel with hemostatic and antibacterial capabilities for wound care. Advanced Science, 10(30), 2303651. https://doi.org/10.1002/advs. 202303651
CrossRef - Ishikawa, S., Matsukuma, D., Iijima, K., Iijima, M., Osawa, S., & Otsuka, H. (2019). N-hydroxysuccinimide-bifunctionalized triblock cross-linker having hydrolysis property for a biodegradable and injectable hydrogel system. ACS Biomaterials Science & Engineering, 5(11), 5759–5769. https://pubs.acs.org/doi/abs/10.1021/acsbiomaterials.9b00218
CrossRef - Huang, Y., Zhao, X., Zhang, Z., Liang, Y., Yin, Z., Chen, B., Bai, L., Han, Y., & Guo, B. (2020). Degradable gelatin-based IPN cryogel hemostat for rapidly stopping deep noncompressible hemorrhage and simultaneously improving wound healing. Chemistry of Materials, 32(15), 6595–6610. https://pubs.acs.org/doi/abs/10.1021/acs.chemmater.0c02030
CrossRef - Zhu, J., Li, F., Wang, X., Yu, J., & Wu, D. (2018). Hyaluronic acid and polyethylene glycol hybrid hydrogel encapsulating nanogel with hemostasis and sustainable antibacterial property for wound healing. ACS Applied Materials & Interfaces, 10(16), 13304–13316. https://pubs.acs.org/doi/abs/10.1021/acsami.7b18927
CrossRef - Chen, S., Guo, R., Xie, C., Liang, Q., & Xiao, X. (2020). Biomimetic mineralization of nanocrystalline hydroxyapatites on aminated modified polylactic acid microspheres to develop a novel drug delivery system for alendronate. Materials Science and Engineering: C, 110, 110655. https://doi.org/10.1016/j.msec.2020.110655
CrossRef - Kuljanin, J., Janković, I., Nedeljković, J., Prstojević, D., & Marinković, V. (2002). Spectrophotometric determination of alendronate in pharmaceutical formulations via complex formation with Fe (III) ions. Journal of Pharmaceutical and Biomedical Analysis, 28(6), 1215–1220. https://doi.org/10.1016/S0731-7085(02)00021-3
CrossRef - Ji, D., Lin, Y., Guo, X., Ramasubramanian, B., Wang, R., Radacsi, N., Jose, R., Qin, X., & Ramakrishna, S. (2024). Electrospinning of nanofibres. Nature Reviews Methods Primers, 4(1), 1. https://www.nature.com/articles/s43586-023-00278-z
CrossRef - Horiuchi, S., & Nakao, Y. (2010). Platinum colloid catalyzed etchingless gold electroless plating with strong adhesion to polymers. Surface and Coatings Technology, 204(23), 3811–3817. https://doi.org/10.1016/j.surfcoat.2010.04.064
CrossRef - Koumbia, M., & Madoka, T. (2025). Conductive and flexible gold-coated polylactic acid nanofiber-based electrochemical aptasensor for monitoring cortisol level in sweat and saliva. Bioelectrochemistry, 163, 108908. https://doi.org/10.1016/j.bioelechem.2025.108908
CrossRef - Hoy, C. F., Kushiro, K., & Takai, M. (2018). Fabrication and assessment of an electrospun polymeric microfiber-based platform under bulk flow conditions with rapid and efficient antigen capture. Analyst, 143(4), 865–873. https://pubs.rsc.org/en/content/ articlelanding/2018/an/c7an01366c/ unauth
CrossRef - Luengo, J., Weiss, B., Schneider, M., Ehlers, A., Stracke, F., König, K., Kostka, K. H., Lehr, C. M., & Schaefer, U. F. (2006). Influence of nanoencapsulation on human skin transport of flufenamic acid. Skin Pharmacology and Physiology, 19(4), 190–197. https://doi.org/10.1159/000093114
CrossRef - Malinovskaja-Gomez, K., Labouta, H. I., Schneider, M., Hirvonen, J., & Laaksonen, T. (2016). Transdermal iontophoresis of flufenamic acid loaded PLGA nanoparticles. European Journal of Pharmaceutical Sciences, 89, 154–162. https://doi.org/10.1016/j.ejps.2016.04.034
CrossRef - Miller, R. A., Brady, J. M., & Cutright, D. E. (1977). Degradation rates of oral resorbable implants (polylactates and polyglycolates): rate modification with changes in PLA/PGA copolymer ratios. Journal of Biomedical Materials Research, 11(5), 711–719. https://doi.org/10.1002/jbm.820110507
CrossRef - Houchin, M. L., & Topp, E. M. (2008). Chemical degradation of peptides and proteins in PLGA: A review of reactions and mechanisms. Journal of Pharmaceutical Sciences, 97(7), 2395–2404. https://doi.org/10.1002/jps.21176
CrossRef - Stolnik, S., Garnett, M. C., Davies, M. C., Illum, L., Bousta, M., Vert, M., & Davis, S. S. (1995). The colloidal properties of surfactant-free biodegradable nanospheres from poly (β-malic acid-co-benzyl malate)s and poly (lactic acid-co-glycolide). Colloids and Surfaces A: Physicochemical and Engineering Aspects, 97(3), 235–245. https://doi.org/10.1016/0927-7757(95)03081-N
CrossRef - Luengo, J., Weiss, B., Schneider, M., Ehlers, A., Stracke, F., König, K., Kostka, K. H., Lehr, C. M., & Schaefer, U. F. (2006). Influence of nanoencapsulation on human skin transport of flufenamic acid. Skin Pharmacology and Physiology, 19(4), 190–197. https://doi.org/10.1159/000093114
CrossRef - Fiore, G., Nencini, C., Cavallo, F., Capasso, A., Bader, A., Giorgi, G., & Micheli, L. (2006). In vitro antiproliferative effect of six Salvia species on human tumor cell lines. Phytotherapy Research, 20(8), 701–703. https://doi.org/10.1002/ptr.1911
CrossRef - Armanini, D., Fiore, C., Bielenberg, J., Sabbadin, C., & Bordin, L. (2020). Coronavirus-19: Possible therapeutic implications of spironolactone and dry extract of Glycyrrhiza glabra (licorice). Frontiers in Pharmacology, 11, 558418. https://clinicaltrials.gov/
CrossRef - Ram, A., Mabalirajan, U., Das, M., Bhattacharya, I., Dinda, A. K., Gangal, S. V., & Ghosh, B. (2006). Glycyrrhizin alleviates experimental allergic asthma in mice. International Immunopharmacology, 6(9), 1468–1477. https://doi.org/10.1016/j.intimp.2006.04.020
CrossRef - Yamamura, Y., Kawakami, J., Santa, T., Kotaki, H., Uchino, K., Sawada, Y., Tanaka, N., & Iga, T. (1992). Pharmacokinetic profile of glycyrrhizin in healthy volunteers by a new high-performance liquid chromatographic method. Journal of Pharmaceutical Sciences, 81(10), 1042–1046. https://doi.org/10.1002/jps.2600811018
CrossRef - Li, J., Yu, H., Li, S., & Wang, G. J. (2010). Enhanced distribution and extended elimination of glycyrrhetinic acid in mice liver by mPEG-PLA modified (mPEGylated) liposome. Journal of Pharmaceutical and Biomedical Analysis, 51(5), 1147–1153. https://doi.org/10.1016/ j.jpba.2009.11.005
CrossRef - Ionescu, O. M., Mignon, A., Minsart, M., Caruntu, I. D., Giusca, S. E., Gardikiotis, I., Van Vlierberghe, S., & Profire, L. (2021). Acrylate-endcapped urethane-based hydrogels: An in vivo study on wound healing potential. Materials Science and Engineering: C, 130, 112436. https://doi.org/10.1016/ j.msec.2021.112436
CrossRef - Hu, W., Wang, Z., Zha, Y., Gu, X., You, W., Xiao, Y., Wang, X., Zhang, S., & Wang, J. (2020). High flexible and broad antibacterial nanodressing induces complete skin repair with angiogenic and follicle regeneration. Advanced Healthcare Materials, 9(23), 2000035. https://doi.org/10.1002/ adhm.202000035
CrossRef - Blacklow, S. O., Li, J., Freedman, B. R., Zeidi, M., Chen, C., & Mooney, D. J. (2019). Bioinspired mechanically active adhesive dressings to accelerate wound closure. Science Advances, 5(7), eaaw3963. https://doi.org/10.1126/sciadv.aaw3963
CrossRef - Lan, G., Zhu, S., Chen, D., Zhang, H., Zou, L., & Zeng, Y. (2021). Highly adhesive antibacterial bioactive composite hydrogels with controllable flexibility and swelling as wound dressing for full-thickness skin healing. Frontiers in Bioengineering and Biotechnology, 9, 785302. https://doi.org/10.3389/ fbioe.2021.785302
CrossRef - Liu, Y., Wei, Y., Xiong, Q., Xie, C., An, T., He, M., Xia, L., Wei, S., Yu, J., Cheng, S., & Xu, B. (2024). Hollow porous PLA/PBAT composite microfibers with enhanced toughness and prolonged drug release for wound healing. Materials & Design, 237, 112624. https://doi.org/10.1016/j.matdes. 2023.112624
CrossRef - Al-Lazikani, B., Banerji, U., & Workman, P. (2012). Combinatorial drug therapy for cancer in the post-genomic era. Nature Biotechnology, 30(7), 679–692. https://www.nature.com/articles/nbt.2284
CrossRef - Vanneman, M., & Dranoff, G. (2012). Combining immunotherapy and targeted therapies in cancer treatment. Nature Reviews Cancer, 12(4), 237–251. 12(4): 237–251. doi:10.1038/nrc3237
CrossRef - Woodcock, J., Griffin, J. P., & Behrman, R. E. (2011). Development of novel combination therapies. New England Journal of Medicine, 364(11), 985–987. DOI: 10.1056/NEJMp1101548
CrossRef - Gaucher, G., Marchessault, R. H., & Leroux, J. C. (2010). Polyester-based micelles and nanoparticles for the parenteral delivery of taxanes. Journal of Controlled Release, 143(1), 2–12. https://doi.org/10.1016/ j.jconrel.2009.11.012
CrossRef - Peer, D., Karp, J. M., Hong, S., Farokhzad, O. C., Margalit, R., & Langer, R. (2020). Nanocarriers as an emerging platform for cancer therapy. In Nano-Enabled Medical Applications (pp. 61–91). http://www.nature.com/naturenanotechnology
CrossRef - Ramalingam, S. S., Egorin, M. J., Ramanathan, R. K., Remick, S. C., Sikorski, R. P., Lagattuta, T. F., Chatta, G. S., Friedland, D. M., Stoller, R. G., Potter, D. M., & Ivy, S. P. (2008). A phase I study of 17-allylamino-17-demethoxygeldanamycin combined with paclitaxel in patients with advanced solid malignancies. Clinical Cancer Research, 14(11), 3456–3461.https://doi.org/10.1158/1078-0432.CCR-07-5088
CrossRef - Kim, S. C., Kim, D. W., Shim, Y. H., Bang, J. S., Oh, H. S., Kim, S. W., & Seo, M. H. (2001). In vivo evaluation of polymeric micellar paclitaxel formulation: Toxicity and efficacy. Journal of Controlled Release, 72(1–3), 191–202. https://doi.org/10.1016/S0168-3659(01)00275-9
CrossRef - Shin, H. C., Alani, A. W., Cho, H., Bae, Y., Kolesar, J. M., & Kwon, G. S. (2011). A 3-in-1 polymeric micelle nanocontainer for poorly water-soluble drugs. Molecular Pharmaceutics, 8(4), 1257–1265. Mol Pharm. 2011 August 1; 8(4): 1257–1265. doi:10.1021/mp2000549.
CrossRef - Wolinsky, J. B., Colson, Y. L., & Grinstaff, M. W. (2012). Local drug delivery strategies for cancer treatment: Gels, nanoparticles, polymeric films, rods, and wafers. Journal of Controlled Release, 159(1), 14–26. https://doi.org/10.1016/j.jconrel.2011.11.031
CrossRef - Zentner, G. M., Rathi, R., Shih, C., McRea, J. C., Seo, M. H., Oh, H., Rhee, B. G., Mestecky, J., Moldoveanu, Z., Morgan, M., & Weitman, S. (2001). Biodegradable block copolymers for delivery of proteins and water-insoluble drugs. Journal of Controlled Release, 72(1–3), 203–215. https://doi.org/10.1016/S0168-3659(01)00276-0
CrossRef - Cho, H., & Kwon, G. S. (2014). Thermosensitive poly-(D,L-lactide-co-glycolide)-block-poly (ethylene glycol)-block-poly-(D,L-lactide-co-glycolide) hydrogels for multi-drug delivery. Journal of Drug Targeting, 22(7), 669–677. https://doi.org/10.3109/1061186X.2014.931406
CrossRef - Cho, H., Gao, J., & Kwon, G. S. (2016). PEG-b-PLA micelles and PLGA-b-PEG-b-PLGA sol–gels for drug delivery. Journal of Controlled Release, 240, 191–201. https://doi.org/10.1016/j.jconrel. 2015.12.015
CrossRef
Accepted on: 20-11-2025
Second Review by: Dr. Vikas Guleriaand Dr. Afreen Khan (BNCP)
Final Approval by: Dr. Wagih Ghannam









