Role of Oxidative Stress and Inflammation in Diabetic Wound Healing: Therapeutic Potential of Medicinal Plants.
Department of Pharmacology, Navsahyadri Institute of Pharmacy, Pune, India.
Corresponding Author E-mail: surajjadhao999@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3481
ABSTRACT:Diabetic wound healing remains a major clinical challenge due to persistent hyperglycaemia-associated metabolic disturbances, oxidative stress, chronic inflammation, impaired angiogenesis, and defective extracellular matrix remodelling. Unlike normal wound repair, diabetic wounds are frequently arrested in a prolonged inflammatory phase, resulting in delayed epithelialisation and chronic non-healing ulcers. Increasing evidence indicates that oxidative stress and inflammation act as interlinked pathological drivers in diabetic wound progression, forming a vicious cycle that amplifies tissue damage and suppresses repair processes. Excessive generation of reactive oxygen species activates redox-sensitive signalling pathways, including NF-κB, leading to sustained release of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β, which impair fibroblast function, collagen synthesis, and neovascularisation. Conventional therapies, including dressings, antibiotics, and growth factors, often show limited success due to high cost, adverse effects, antimicrobial resistance, and failure to address underlying molecular dysfunctions. Medicinal plants have emerged as promising alternatives owing to their multi-target actions, antioxidant and anti-inflammatory properties, cost-effectiveness, and accessibility. Evidence from streptozotocin-induced diabetic animal models demonstrates that plant-based therapies enhance wound contraction, reduce oxidative and inflammatory biomarkers, promote collagen deposition, and improve histopathological outcomes. Moreover, molecular docking studies provide mechanistic support by revealing interactions between phytochemicals and key inflammatory and oxidative targets. However, it is important to note that the majority of available evidence is derived from preclinical animal studies, with limited well-designed clinical trials in humans. This highlights the need for further clinical validation to establish safety, efficacy, and translational applicability of plant-based therapies in diabetic wound management. This review highlights the therapeutic potential of medicinal plants and emphasizes the need for standardized formulations and integrated experimental and molecular approaches for effective diabetic wound management.
KEYWORDS:Diabetic wound healing; Herbal therapy; Inflammation; Inflammatory cytokine; Medicinal plants; Oxidative stress
Introduction
Diabetes mellitus is a chronic metabolic disease that is manifested by persistent hyperglycaemia and is caused by impairments in insulin secretion, insulin action, or both. Diabetes has been a significant health issue in the world in the last several decades, driven by rapid urbanization, sedentary lifestyle, and dietary changes. The International Diabetes Federation reports that over 530 million adults worldwide were living with diabetes in 2021, with this figure expected to reach over 780 million by 2045 unless the current trends are reversed. .[1] Diabetes is connected with several chronic complications of the cardiovascular system, kidneys, nervous system and skin, leading to increased morbidity, mortality, and healthcare expenditure. Impaired wound healing is one of the most debilitating conditions of diabetes among others. Diabetic ulcers are chronic wounds especially diabetic foot ulcers, which occur in about 15 to 25 percent of diabetic patients during their lifetime. They are wounds that take a long time to heal, are highly vulnerable to infection and recur frequently. Diabetic wounds are the primary cause of non-traumatic lower-limb amputations in the global society and they significantly reduce quality of life of the patients and cause a heavy financial burden to the health care systems.2
Although wound care technologies have improved, diabetic wound management has not been effectively done. The traditional treatment interventions involve debridement, pressure off-loading, infection control, growth factor therapy, and the use of advanced wound dressings. Nevertheless, these strategies are proving to be limited efficacy because of ineffective vascularization, neuropathy, compromised immunity and inflammation that persists at the wound site. Moreover, synthetic drugs and antibiotics can also cause long-term consequences with which include adverse effects, antimicrobial resistance, and high costs of treatment. Therefore, safer and multi-target therapeutic approaches are urgently needed. Normal wound healing is a process that is highly controlled as it consists of four overlapping stages in which they are haemostasis, inflammation, proliferation, and remodelling. A coordinated interaction between immune cells, fibroblasts, keratinocytes, endothelial cells, and extracellular matrix components is needed in each of the phases. In diabetes, molecular-level dysregulation of this finely tuned process, which causes chronic inflammation, inhibition of angiogenesis, impaired collagen synthesis, and defective tissue remodelling.3 Each of those changes is associated with underlying molecular processes that can be identified to develop new therapeutic targets and enhance clinical outcomes in healing diabetic wounds.
Oxidative stress and chronic inflammation are also considered to be the key factors that lead to the slow healing of wounds in diabetes. Oxidative stress and inflammation are closely interconnected, contributing to impaired wound healing, as discussed in later sections.
This article is a narrative review aimed at summarizing the role of oxidative stress and inflammation in diabetic wound healing and the therapeutic potential of medicinal plants. A comprehensive literature search was conducted using electronic databases including PubMed, Scopus, Web of Science, and Google Scholar. Relevant articles published between 2000 and 2025 were considered.
Keywords used for the search included “diabetic wound healing,” “oxidative stress,” “inflammation,” “medicinal plants,” “phytochemicals,” “Nrf2,” and “NF-κB.” Both original research articles and review papers were included to provide a broad understanding of the topic.
Studies were selected based on their relevance to the mechanisms of diabetic wound healing and plant-based therapeutic interventions, including in vitro, in vivo (animal), and in silico studies, as well as available clinical evidence. This review does not follow a systematic review protocol but aims to provide a comprehensive and integrative overview of current knowledge in this field.
Normal Process of Wound Healing
Wound healing is a complex and highly controlled biological phenomenon that reestablishes tissue integrity following damage that are regulated by interactions among cells, growth factors, cytokines, and the extracellular matrix. Any disruption in these tightly regulated processes, as observed in diabetes, leads to delayed or impaired wound healing.
Phases of Woundhealing
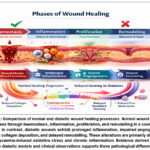 |
Figure 1: Comparison of normal and diabetic wound healing processes. Normal wound healing progresses through haemostasis, inflammation, proliferation, and remodelling in a coordinated manner.
|
Haemostasis
Haemostasis is the initial stage of wound healing and occurs immediately after tissue injury. It entails the constriction of the blood vessels and combining platelets to avoid blood loss. Activated platelets secrete clotting factors and growth factors including platelet-derived growth factor (PDGF) and transforming growth factor-b (TGF-β), which form a provisional fibrin matrix and initiate subsequent repair responses.4
Inflammation
Unlike haemostasis, the inflammatory phase follows injury and normally lasts several days. At this stage, neutrophils and Macrophages are recruited to the wound site to eliminate debris, pathogens, and necrotic tissue. Macrophages also play a major role by releasing cytokines (TNF-α, IL-1β, IL-6) and growth factors that control fibroblast activation, angiogenesis and re-epithelialisation. Normal healing requires the modulated inflammatory response, but persistent inflammation may slow down the process of tissue healing.3,4
Proliferation
It is a rapid stage in which tissue growth and wound bed healing take place. The fibroblasts grow and develop extracellular matrix proteins, especially collagen. Keratinocytes migrate from the wound margins to re-epithelialize the surface as endothelial cells create new blood vessels by angiogenesis. A characteristic of this stage is the formation of granulation tissue, which is an assembly of fibroblasts, new capillaries, and provisional matrix.5
Remodelling
Remodelling is often referred to as the maturation period and may take weeks or months. At this stage, collagen type III formed during proliferation is gradually replaced by the stronger collagen type I, the number of fibroblasts is reduced due to apoptosis and newly formed blood vessels regress as the tissue gains strength. An appropriate remodelling enhances tensile strength and normal tissue structure, but healed tissue is seldom as strong as uninjured skin.5(As shown in fig 1).
Key Cellular and Molecular Events
Fibroblasts, keratinocytes and macrophages
Fibroblasts play a vital role in repairing the wound because they are involved in producing collagen, fibronectin and proteoglycan which constitute the extracellular matrix. They also differentiate into myofibroblasts, which contribute to wound contraction by migrating and proliferating, keratinocytes are important in the process of re-epithelialisation in order to cover the wound surface. Macrophages serve as regulatory cells and organize healing by removing debris and releasing cytokines and growth factors that regulate inflammation, angiogenesis, and extracellular matrix deposition.6,7
Collagen Synthesis and Angiogenesis
Collagen production is also the key to tissue integrity and strength. Fibroblast activity and an adequate oxygen supply are essential for proper collagen deposition Angiogenesis is necessary to supply oxygen, nutrients, and immune cells to the wound site. Some important mediators of angiogenesis are vascular endothelial growth factor (VEGF) and fibroblast growth factor (FGF). Development of weak granulation tissue and slow wound repair is caused by impaired collagen synthesis or decreased angiogenesis.3,7
Pathophysiology of Diabetic Wound Healing
Metabolic and molecular perturbations caused by chronic hyperglycaemia significantly hinders diabetic wound healing. One difference between the normal and diabetic wound repair is that diabetic wounds have a prolonged inflammatory response, poor angiogenesis, defective collagen production, and impaired immune responses which cause the development of chronic non-healing wounds.
Effect of Hyperglycaemia on Tissue Repair
The chronic hyperglycaemia is the major cause of impairment of wound healing in diabetes. High glucose levels enhance the development of advanced glycation end products (AGEs), polyol pathway activation, and oxidative stress, each of which contributes to protein, lipid, and DNA damage at the cellular level. Hyperglycaemia also changes signalling mechanisms of cell migration and proliferation leading to fibroblast and keratinocyte dysfunction. These changes disrupt the normal wound healing process at multiple stages and postpone healing of tissues.8
Impaired Angiogenesis
In diabetes, angiogenesis fails to meet the needs of the healing tissue to provide sufficient oxygen and nutrients to the healing tissue. Hyperglycaemia inhibits endothelial cell proliferation and migration and downregulates the expression of pro-angiogenic factors like vascular endothelial growth factor (VEGF). Moreover, oxidative stress causes excessive damage to endothelial cells and inhibits the availability of nitric oxide further reducing the ability of blood vessels to form. Effects of reduced angiogenesis include tissue hypoxia and insufficient granulation tissue development which causes delayed wound healing.8,9
Delayed Collagen Deposition
Collagen synthesis and remodelling play a key role in the restoration of wound strength and integrity. Fibroblast dysfunction induces a reduction in collagen synthesis and extracellular matrix structure in diabetic wounds. Excessive activity of the matrix metalloproteinases (MMPs) that break down newly synthesized collagen is also encouraged by hyperglycaemia-induced oxidative stress. This results in weak granulation tissue and reduced tensile strength and delays wound remodelling.9
Reduced Immune Response
Diabetes has been linked to the loss of both innate and adaptive immune responses. The neutrophil chemotaxis, phagocytosis, and bactericidal activity is impaired making the wound more prone to infection. Due to macrophage dysfunction, the release of cytokines and growth factors is disrupted and which interferes with the transition from the inflammatory to the proliferative phase. This defective immune response that is associated with chronic inflammation, delayed pathogen clearance and impaired wound healing in diabetic patients.9,10
Role of Oxidative Stress in Diabetic Wounds
Hyperglycaemia-induced ROS
Oxidative stress is a key factor in the pathogenesis of impaired wound healing in diabetes. It occurs when the rate of generation of reactive oxygen species (ROS) is too high to be countered by natural antioxidant defence mechanisms, resulting in cellular and molecular damage within wound tissue. Continuous oxidative stress in diabetic wounds disturbs the normal healing process due to its effects on cellular function, extracellular matrix remodelling and inflammatory responses.
Mitochondrial Dysfunction
Mitochondria are a major source of ROS in diabetic conditions. Hyperglycaemia increases flux through the mitochondrial electron transport chain, resulting in electron leakage and excessive superoxide production. Mitochondrial dysfunction impairs cellular energy metabolism and promotes oxidative damage to mitochondrial DNA, proteins, and lipids. In wound tissue, mitochondrial dysfunction leads to reduced cell viability, impaired migration of fibroblasts and keratinocytes, and delayed tissue regeneration.10 (As shown in fig 2).
Impact on Wound Healing
Damage to Proteins, Lipids, and DNA
Excess ROS can cause oxidative damage to proteins, lipid peroxidation of cell membranes, and DNA strand breakages. These molecular alterations compromise cell integrity and cellular function, impairing cellular signalling, reducing growth factor responsiveness, and increasing cell death. These damages in diabetic wound undermine the process of granulation tissue formation and delay the transition from inflammation to proliferation.7-10
Inhibition of Fibroblast Migration
Fibroblasts play a vital role in the production of collagen and the development of extracellular matrix. Oxidative stress suppresses fibroblast proliferation and migration disrupting cytoskeletal organization and growth factor signalling. Reduced fibroblast activity results in decreased collagen deposition and reduced tensile strength of the wound and this fact is associated with delayed healing of diabetic wound.11
Delayed Epithelialisation
Keratinocyte movement and increase are essential to the re-epithelialisation of the wound surface. Oxidative DNA damage and alterations in cell–cell adhesion indicate dysfunctional keratinocyte activity under conditions of elevated ROS levels. This results in delayed healing of the epithelial lining and the extension of the wound bed, which leads to a higher risk of infection and the development of chronic ulcers.12
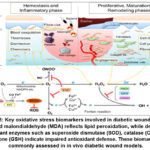
Oxidative Stress Biomarkers
malondialdehyde (MDA)
Malondialdehyde has been widely used as a biomarker of lipid peroxidation and oxidative stress. High levels of MDA in diabetic tissue demonstrate that there is more damage to the membranes and oxidative injury associated with delayed wound healing and poor tissue regeneration.13,41
Superoxide Dismutase (SOD)
Superoxide dismutase is a key antioxidant enzyme, which converts superoxide radicals into hydrogen peroxide and oxygen. Low SOD activity in diabetes is an indication of a hampered antioxidant defence resulting in the accumulation of ROS and subsequent oxidative damage within the wound site.13
reduced glutathione (GSH)
Reduced glutathione is a significant intracellular antioxidant that helps to shield cells against the adverse effects of oxidation and ensures a redox potential factor. Reduced GSH levels in diabetic conditions undermine cellular defence systems and contribute to impaired wound healing mediated by oxidative stress–induced damage contributes to impaired wound healing. Improved wound healing has been linked to restoration of GSH levels.14
Role of Inflammation in Diabetic Wound Healing
Inflammation is an important early inflammation process in normal wound healing; but in diabetes becomes dysregulated and prolonged. Inflammation in diabetes mellitus is chronic and low-grade in nature, which is the key factor in the wound healing delay. This results from persistent inflammatory signalling that disrupts cell migration, angiogenesis, and remodelling of the extra cellular matrix ultimately resulting in persistent non-healing wounds.
Chronic Inflammatory State in Diabetes
Prolonged Inflammatory Phase
In normal wound healing, the inflammatory condition is temporary and goes away as the wound reveals into the proliferative phase. In contrast, diabetic wounds remain in a sustained inflammatory state Hyperglycaemia promotes continuous recruitment of inflammatory cells of neutrophils and macrophages that leads to the persistent generation of pro-inflammatory cytokines. This ongoing inflammation delays the transition of the wound into the tissue formation and remodelling phases thus hampering wound closure.3,14
Dysregulated Immune Response
Diabetes is linked to the defect in both innate and adaptive immune responses. This causes dysfunction of neutrophils resulting in a loss of chemotaxis and phagocytosis and an aberrant phenotype of the macrophage that oversecretes inflammatory mediators and underproduces growth factors to facilitate healing. Such an unregulated immune response result in a reduced clearance debris and pathogens, increased susceptibility to infection and prolonged wound inflammation.15
Key Inflammatory Mediators
Tumour necrosis factor-a (TNF-α)
TNF-α is one of the major pro-inflammatory cytokines that is significantly increased in diabetic wounds Overexpression of TNF-α inhibits fibroblast proliferation and collagen synthesis and stimulates apoptosis of endothelial cells and keratinocyte. Increased TNF- α is closely linked to the delayed wound healing processes and failure to develop the granulation tissue.4
Interleukin-6 (IL-6)
IL-6 has a dual role in regulating the immune responses and tissue restoration; nevertheless, chronic overexpression of IL-6 in diabetes is causing extensive inflammation. High IL-6 concentrations also disrupt keratinocyte migration and angiogenesis and are associated with delayed keratinization and prolonged wound persistence.5
Interleukin-1β (IL-1β)
IL-1β is a strong inflammatory mediator that participates in the recruitment and activation of leukocytes. In diabetic wounds, hypersecretion of IL-1β promotes degradation of the matrix through activation of matrix metalloproteinases and inhibits the activity of the fibroblast, further delayed the tissue regenerative process.6
NF-κB Signalling Pathway
Nuclear factor-κB (NF-κB) refers to a key transcription factor that controls the expression of genes involved in inflammatory processes. Oxidative stress triggered by hyperglycaemia activates NF-κB signalling, which results in the upregulation of TNF-α, IL-6, and IL-1β, among inflammatory mediators. Persistent NF-κB activation promotes a chronic inflammatory state that disrupts angiogenesis, collagen production, and wound remodelling.7
Consequences of Excessive Inflammation
Impaired Granulation Tissue Formation
Granulation tissue formation required a balanced inflammatory signalling and fibroblast activity. The excessive inflammation inhibits fibroblast proliferation and extracellular matrix deposition and produces dispersed and disordered granulation tissue in the diabetic wound.12
reduced Angiogenesis
Chronic inflammation has an adverse impact on angiogenesis by preventing endothelial cell migration, as well as lowering the expression of pro-angiogenic factors including vascular endothelial growth factor (VEGF). Decreased angiogenesis causes hypoxia of the tissues and lack of sufficient nutrients, which further delays the healing process of the wounds.16
Chronic Non-Healing Ulcers
Impaired angiogenesis, together with dysfunctional matrix remodelling and chronic inflammation, contributes to the development of chronic non-healing ulcers. These are very prone to infection, recur often and cut across the board of morbidity and amputation of the limb among diabetic patients.17
Interactions between Oxidative Stress and Inflammation in Diabetic Wound Healing
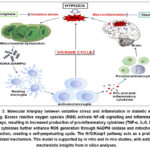
There is a tight interrelationship between oxidative stress and inflammation, which act synergistically to undermine wound repair in diabetes. Instead of working separately, excessive production of reactive oxygen species (ROS) and pro-inflammatory mediators and pro-inflammatory mediators support each other to establish a self-sustaining pathogenic loop that maintains wounds in a chronic, non-healing state.
ROS-Induced Activation of Inflammatory Pathways
Activation of various redox-regulated inflammatory pathways by hyperglycaemia induced ROS over production. ROS activate transcription factors as nuclear factor -κB (NF-κB) and activator protein-1 (AP-1); which cause pro-inflammatory cytokines, chemokines and adhesions to be transcribed more. Prolonged activation of NF-κB in diabetic wounds increases the levels of TNF-α, IL-6, and IL-1β and extends leukocyte recruitment and maintain an inflammatory microenvironment which suppresses transition into the proliferative phase of healing.18 Also, ROS increase inflammasome activations, which further boost the release of cytokines and tissue damage.
Oxidative Damage Mediated by Cytokines
Cytokines that are produced in chronic inflammation also contribute to further oxidative stress. TNF-α and IL-1β trigger NADPH oxidases and mitochondrial dysfunction, enhance intracellular ROS production in fibroblasts, keratinocytes, and endothelial cells. These cytokines also diminish intracellular antioxidant defences e.g. superoxide dismutase (SOD) and reduced glutathione (GSH) thereby reducing the ability of the tissue to counteract ROS. The oxidative stress leads to the destruction of cell migration, angiogenesis and remodelling of the extracellular matrix, critical processes involved in wound repair.7,19
Delaying Wound Repair through a Vicious Cycle
A bidirectional relationship forms a vicious cycle in diabetic wounds. excess ROS cause inflammatory signalling, whereas chronic inflammation produces more ROS, both of which sustain tissue damage and cannot resolve the inflammatory stage. This loop inhibits fibroblast proliferation, inhibits keratinocyte re-epithelialisation, angiogenic responses, and enhances matrix degradation mediated by matrix metalloproteinases. Therefore, the wounds fail to progressed to the proliferative and remodelling stages, which leads to chronic non-healing ulcers.20
It would therefore be logical to focus on inhibiting oxidative stress and inflammation simultaneously to break this cycle. The use of single agent that is both an antioxidant and anti-inflammatory, and agent such as extracts from medicinal plants, hold the most potential in the re-establishment of redox homeostasis, prevention of excessive inflammation, and the enhancement of a coordinated wound repair process in diabetic patients.
Current Therapeutic Approaches and Limitations in Diabetic Wound Management
This aspect presents various management of diabetic wounds based on the goal of reducing infections, tissue healing and complications. Even though some therapeutic options are in place, they fail to work effectively in the presence of diabetes as there are metabolic and molecular abnormalities that compromise their efficacy in diabetic conditions.
Conventional Wound Dressings
Conventional wound dressings, such as gauze, hydrocolloids, foams, alginates, and hydrogel dressings are frequently applied to the wound bed to maintain a moist wound environment and protect the wound site from external contamination. There are also advanced dressings that can be used to permit autolytic debridement and removing of excess exudate. Although such dressings offer the fundamental wound care value, they do not actively mitigate underlying molecular defects of the wound like oxidative stress, persistent inflammation, and dysfunctional angiogenesis typical of diabetic wounds. Consequently, the recovery process is slow and incomplete among most of the patients.13,21
Antibiotics and Growth Factors
In terms of antibiotics and growth factors has been widely used in the treatment of bacterial infection and diabetic wounds. Recombinant platelet-derived growth factor (PDGF), which is growth factor-based therapy, has also been given to promote cell proliferation and the formation of granulation tissue. Although the methods may be productive in specific situations, their general effectiveness in clinical practice is inconsistent Antibiotic therapy does not directly promote tissue reconstruction and prolonged usage can result in antimicrobial resistance. On the same note, the growth factor therapies are usually not very effective because they are quickly degraded, they show limited penetration into ischemic tissue and the diabetic cells do not respond to growth factor signalling in the most effective way.8,22
Limitations of current Therapies
Modern technologies do not eliminate the fact that current therapeutic methods of diabetic wound healing have several major drawbacks. The cost of treatments and the fact that patients require long-term care exert a huge financial strain on the patients and medical systems. The threat of antibiotic resistance and infection is an important issue especially when antibiotics are used long-term or repeatedly. Furthermore, there are adverse effects and limited availability of growth factor therapies and sophisticated wound-care products. Most importantly, many current therapies are not able to address both oxidative stress and chronic inflammation, which are major causes of delay healing in diabetic wounds. These drawbacks point to the fact that alternative or complementary therapies that are safe and can regulate various pathological pathways are needed.11,23
Therapeutic Potential of Medicinal Plants in Diabetic Wound Healing
Medicinal plants have been traditionally used for centuries to treat wounds and skin diseases. In recent years, there has been growing scientific interest in their therapeutic value in the management of diabetic wounds because they are capable of affecting a number of pathological pathways that cause delayed wound healing.
Rationale for Using Medicinal Plants
Multi-Target Action
Diabetic wound healing is Impaired by a complex set of factors such as oxidative stress, chronic inflammation, impaired angiogenesis and decreasing collagen synthesis. In contrast to single-target synthetic drugs, medicinal plants possess a variety of bioactive compounds, which can act simultaneously on multiple molecular pathways. This principle of multi-target is specifically effective in diabetic wounds, where the complex pathophysiological mechanisms are simultaneously present and must be modulated in a coordinated manner.24
Antioxidant and Anti-Inflammatory Properties
Polyphenols, flavonoids, alkaloids, and terpenoids are abundant in a number of medicinal plants and possess strong antioxidant and anti-inflammatory properties. These antioxidants neutralise reactive oxygen species (ROS), augment endogenous antioxidant responses and inhibit the synthesis of pro-inflammatory cytokines including TNF-α, IL-6, and IL-1β. Plant-based therapies help restore a favourable wound microenvironment by minimizing oxidative stress and inflammation and enhancing tissue healing in diabetic patients.7,25
Affordable and Easy to access
Medicinal plants are typically inexpensive and widely accessible, particularly in developing and low-resource countries where the prevalence of diabetes is increasing rapidly. Plant-based treatments are a cheaper alternative to more complex wound-care products and growth factor therapies, with minimal reported side effects. This ease of access makes medicinal plants a compelling option for long-term management of chronic diabetic wounds.26
Mechanisms of Action
Free Radical Scavenging: Antioxidants present in medicinal plants have the ability to counter excess ROS produced in excess under hyperglycaemic conditions. The medicinal plants prevent oxidative damage of cellular proteins and DNA and which helps maintain fibroblasts and keratinocytes intact that is necessary to maintain proper wound healing.27
Cytokine Suppression
Experimental studies in diabetic animal models (in vivo) have demonstrated that certain phytochemicals, such as curcumin and flavonoids, can modulate NF-κB signalling pathways. In vitro studies further support their anti-inflammatory effects, while in silico docking studies suggest potential molecular interactions with inflammatory targets.7,43
Enhancement of Collagen Synthesis
A number of medicinal plants can induce growth of fibroblasts and increase the production of collagen, which helps in improved granulation tissue formation and high tensile strength of the healed wound. The improved collagen deposition restores the integrity and speed of wound contraction in diabetic models.29
Angiogenesis Promotion
Angiogenesis plays a vital role in the provision of oxygen and nutrients to the healing tissue. Phytochemicals, including flavonoids and alkaloids, stimulate endothelial cell migration and increase the expression of pro-angiogenic factors, including vascular endothelial growth factor (VEGF). Better angiogenesis facilitates the development of granulation tissue and accelerated wound healing in diabetic wounds.30,42
Experimental Evidence from Medicinal Plants in Diabetic Wound Healing
In vivo studies using streptozotocin (STZ)-induced diabetic rat and mouse models have demonstrated improved wound contraction, epithelialisation, and collagen deposition following treatment with medicinal plant extracts.
Commonly Studied Medicinal Plants
Argemone mexicana
Argemone mexicana has traditionally been used for the treatment of skin disorders and wounds. In experimental studies using diabetic rodents, topical or oral application of A. mexicana extracts has shown significantly faster wound contraction and epithelialisation compared with controls. These effects are attributed to the high content of antioxidant and anti-inflammatory alkaloids and flavonoids. The diabetic wounds treated have consistently recorded a reduction in lipid peroxidation, improved collagen deposition, and enhancement of the formation of granulation tissue.16,31
Aloe vera
Aloe vera is among the widely researched medicinal plants in treating wounds. Aloe vera gel has been reported to stimulate fibroblast growth, collagen production, and angiogenesis in STZ induced diabetic models. It has bioactive components such as polysaccharides and phenolic compounds that inhibit oxidative stress, inhibit inflammatory cytokine production leading to accelerated wound healing and improve tissue architecture.32
Moringa oleifera
Moringa oleifera has a strong antioxidant and anti-inflammatory property as it contains high level of polyphenols, vitamins, and flavonoids. There is evidence that M. oleifera extracts in animal models of diabetic wounds enhance wound contractions, antioxidant enzymes (SOD, CAT, GSH), and reduced inflammatory cytokines (TNF-α, IL-6). Histology shows increased neovascularisation and collagen deposition in groups where treatment is done.33
Curcuma longa
Based on its remarkable wound-healing properties, Curcuma longa (turmeric) and curcumin as its active constituent were tested in diabetic animal models. Curcumin has a strong antioxidant and anti-inflammatory effect, which suppresses NF-κB signalling and leads to a reduction in oxidative stress. Curcumin therapy proves to be more effective in wound contraction, tensile strength and collagen organization and reduces inflammatory cells infiltration in excision and incision wound model.34
To improve translational clarity, studies on medicinal plants in diabetic wound healing can be categorized based on route of administration (topical vs systemic), as summarized in Table 1.
Table 1: Summary of medicinal plants used in diabetic wound healing (topical vs systemic evidence)
| Plant Name | Active Constituents | Experimental Model | Route of Administration | Dose/Formulation | Key Outcomes |
| Argemone mexicana | Alkaloids, flavonoids | STZ-induced diabetic rats | Topical | Ointment/extract | Enhanced wound contraction and reduced inflammation |
| Aloe vera | Polysaccharides, phenolics | STZ-induced diabetic rats | Topical | Gel | Increased collagen synthesis and epithelialization |
| Moringa oleifera | Polyphenols, flavonoids | STZ-induced diabetic rats | Oral (systemic) | Extract | Improved antioxidant enzyme levels (SOD, CAT, GSH) |
| Curcuma longa | Curcumin | STZ-induced diabetic mice | Oral and topical | Extract/gel | Reduced NF-κB activity, improved wound healing and collagen deposition |
Evidence from Animal Models
STZ-Induced Diabetic Wound Models
The STZ-induced diabetic rodent model is commonly used to replicate chronic hyperglycaemia and compromised wound healing in human diabetes. This model is characterized by a sustained increase in blood glucose levels along with a reduction in oxidative stress markers following treatment with medicinal plant therapies which promote improved wound healing responses. Improved antioxidant defence levels and reduced malondialdehyde levels are also commonly observed outcomes.4,35
Excision and Incision Wound Studies
Excision wound models are typically used to determine wound contraction, epithelialisation time, and granulation tissue formation whereas incision models determine tensile strength and collagen maturation. Medicinal plant studies indicate enhancement of wound healing, a reduction in epithelialisation time as well as increased tensile strength in comparison to non-treated diabetic controls implying that healed wound tissue has a greater structural integrity.5
Biomarkers and histopathological findings
The typical biochemical findings in plant-treated diabetic wounds include reduced levels of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) and oxidative stress markers, along with increased antioxidant enzyme activity. The further confirmation of the enhanced healing is obtained through the histopathological analysis which demonstrates the existence of organized collagen Fibers, improved angiogenesis, decrease in inflammatory infiltration, and normal tissue architecture.8
Molecular and in Silico Approaches Supporting Herbal Therapy in Diabetic Wound Healing
Recent advances in computational biology have enabled the application of in silico methods especially the molecular docking method to help justify the use of medicinal plants in the treatment of diabetic wounds and their therapeutic potential. These methods complement of in vivo and in vitro experiments that can give molecular details on the interaction between phytochemicals and the important biological targets associated with oxidative stress and inflammation.
Role of Molecular Docking
Molecular docking is a computational technique applied to estimate binding orientation, affinity and interaction patterns of small molecules (ligands) and target proteins (receptors). In the context of diabetic wound healing docking research assists in determining the interaction of the plant-derived bioactive compounds with enzymes, transcription factors and cytokines involved in the oxidative stress pathways associated with inflammatory mechanisms. This method enables the rapid screening of phytochemicals, reduces experimental costs and offers mechanistic data to support the traditional use of herbs.36
Interaction of Phytochemicals with Inflammatory and Oxidative Targets
In silico molecular docking studies suggest potential binding interactions between phytochemicals and inflammatory or oxidative stress–related targets; however, these findings remain predictive and require validation through in vitro and in vivo studies. Notably, docking scores against NF-κB and COX-2 are high for curcumin from Curcuma longa and quercetin-like flavonoids in agreement with their anti-inflammatory actions. antioxidant and inflammatory targets have shown early signs of binding by alkaloids and phenolics of Argemone mexicana, which supports its reported wound-healing activity.15,37
Supporting Experimental Findings
The scientific inferences derived from molecular docking are generally consistent with experimental data. Phytochemicals with high binding affinity to the inflammatory or oxidative stress related target in silico are often correlated with lower levels of cytokines, reduced of lipid peroxidation and increased levels of antioxidant enzyme activity in animal wound models. The results of these studies show again the biological applicability of docking outcomes and confirm the use of phytochemicals as multi-target agents in diabetic wound healing.
Combination of in silico docking with in vivo data therefore offers a comprehensive perspective in the entirety of the molecular processes of herbal therapy. Such a combination not only helps to validate traditional medicinal claims but also helps to identify lead compounds upon which future drug development may be made.38
Challenges and Limitations in the Use of Medicinal Plants for Diabetic Wound Healing
The use of medicinal plants is promising in experimental research but has various limitations but faces several limitations that impede its application in global clinical practice. These drawbacks are mainly attributed to extract variability, non-standardization, and inadequate clinical validation.
Variability in Plant Extracts
The considerable variability in herbal extracts is one of the biggest issues of herbal therapy. The phytochemical content of medicinal plants may differ widely depending on the plant species, geographical and climatic conditions, soil condition, time of harvest, and mode of extraction. This inconsistency arises from the variability of concentrations of bioactive compounds which leads to unpredictable therapeutic effects and reproducibility problems across various studies. This inconsistency in diabetic wound healing makes the comparison of experimental results more complicated and limits the validity of the plant-based interventions.38
Lack of Standardization
Standardization of herbal formulations remains a major limitation. In comparison to synthetic drugs, the majority of medicinal plant extracts have no standard protocols for dosage, formulation, and quality control. Differences in extraction solvents, handling methods and storage conditions also influence stability and bioactivity. It is challenging to achieve batch-to-batch consistency, safety, and optimum dosing when treating diabetic wound because of the lack of validated biomarkers that can be used for standardization. There are also regulatory issues that surround herbal products that hinder their translation from laboratory research to clinical application.39
limited Clinical Trials
There is no doubt that there are several in vivo studies that have used medicinal plants to establish a wound-healing property in diabetic animal models; however, there is a dearth of well-designed clinical trials in humans. The Majority of existing data are preclinical and it is difficult to extrapolate such studies to diabetic wounds in humans due to physiological and metabolic differences. Also, the clinical trials are usually plagued by small sample sizes, absence of adequate controls and sufficient duration of follow-up. The limited availability of large, randomized, controlled clinical trials evaluating herbal treatments restricts the adoption of herbal therapeutics in traditional diabetic wound treatment.36
Future Perspectives in Medicinal Plant–Based Therapy for Diabetic Wound Healing
Medicinal plants have demonstrated a considerable of promise in enhancing the healing of diabetic wounds; however, to accomplish their clinical implementation in the future, future research should address several critical gaps. The therapeutic potential of plant-based interventions needs to be fully maximised, and this can only be effectively done through advancements made in the areas of standardization, integrative research methods and well-designed clinical studies.
Need for Standardized Formulations
Standardisation of herbal preparations is among the primary concerns that future studies need to focus on. Standardization refers to the definition of the phytochemical profile, proper identification of the bioactive markers, batch-to-batch consistency and optimised dosing regimens. Quality control and reproducibility may be supported by advanced analytical methods, including high-performance liquid chromatography (HPLC), liquid chromatography-mass spectrometry (LC-MS), nuclear magnetic resonance (NMR) spectroscopy. This will increased safety, efficacy, and regulation acceptability of the medicinal plant products in the treatment of diabetic wounds because of the use of standardized formulations.40
Integration of Molecular and Experimental Studies
Future research should focus on integrative approaches that incorporating in silico, in vitro and in vivo techniques. Network pharmacology and molecular docking can be used to identify essential phytochemicals and forecast their behaviour with harmful consequences of oxidative stress and inflammatory age predict their interactions with key targets involved in oxidative stress and inflammatory processes. Such findings must then be confirmed by conducting experimental studies on diabetic wound models supported by biomarker and histopathological analyses. This kind of integration will result in a deeper, mechanistically grounded scientific understanding and faster identifying of lead compounds that can be used to develop some therapeutic benefits.38,40
Translation from Animal Models to Clinical Use
Animal studies offer valuable information on the mechanisms of wound-healing but significant challenges remain in translating this information to clinical practice. Any future study should aim at implementing well designed clinical trials to determine the safety, efficacy and long-term effects of the medicinal plant-based interventions in diabetic patients. Further translational success in deliberation of proper delivery systems, interpatient variability and incorporation of combination therapy with conventional therapies must also be addressed. Bridging studies between preclinical and clinical research are required to fill this gap to incorporate herbal remedies in the evidence-based approach to diabetic wound care.8,40
Discussion
Diabetic wound healing remains a significant clinical challenge due to persistent hyperglycaemia, oxidative stress, chronic inflammation, and impaired angiogenesis, all of which collectively delay tissue repair. Increasing evidence indicates that oxidative stress and inflammation act synergistically to maintain diabetic wounds in a prolonged inflammatory phase, preventing progression to proliferation and remodelling. Excessive production of reactive oxygen species (ROS) activates redox-sensitive signalling pathways such as NF-κB, leading to sustained release of pro-inflammatory cytokines including TNF-α, IL-6, and IL-1β, which impair fibroblast function, collagen synthesis, keratinocyte migration, and neovascularisation.
Medicinal plants have emerged as promising multi-target therapeutic agents capable of addressing these interconnected pathological mechanisms. Bioactive phytochemicals such as flavonoids, alkaloids, phenolics, and terpenoids have been reported to exhibit antioxidant and anti-inflammatory activities. Evidence from in vitro and in vivo studies suggests that medicinal plants may modulate oxidative stress and inflammatory pathways; however, most findings remain preclinical and require clinical validation. These compounds are reported to scavenge free radicals, enhance endogenous antioxidant defences (SOD, CAT, GSH), suppress inflammatory cytokine production, and promote collagen deposition and angiogenesis. Experimental findings from streptozotocin-induced diabetic wound models consistently report accelerated wound contraction, improved epithelialisation, enhanced tensile strength, and better histopathological organization following plant-based treatment.
Furthermore, molecular docking studies provide mechanistic support by demonstrating strong binding interactions between phytochemicals and key inflammatory and oxidative stress–related targets such as NF-κB, COX-2, TNF-α, and IL-6. These in silico findings complement in vivo data and reinforce the concept that medicinal plants exert therapeutic effects through modulation of multiple molecular pathways rather than single-target mechanisms.
Although the majority of current evidence remains preclinical, the accumulated data highlight the therapeutic potential of medicinal plant–derived compounds as complementary or alternative strategies for diabetic wound management. However, translation into clinical practice requires standardized formulations, comprehensive toxicity profiling, well-designed randomized controlled trials, and optimized delivery systems. Future integrative approaches combining molecular, experimental, and clinical investigations will be essential to establish safety, efficacy, and reproducibility in human diabetic wound care.
Although the present review considers multiple mechanistic interpretations linking oxidative stress and inflammation to impaired diabetic wound healing, alternative explanations may also contribute to the observed outcomes. For instance, factors such as microbial infection, neuropathy, and vascular insufficiency may independently or synergistically influence wound healing processes. Furthermore, most of the available evidence is derived from preclinical animal models, which may not fully replicate human pathophysiology. Therefore, additional studies, particularly well-designed clinical trials, are required to validate these findings and explore other potential mechanisms involved in diabetic wound repair.
While antioxidant and anti-inflammatory strategies show considerable promise, several important limitations must be acknowledged. Excessive suppression of reactive oxygen species (ROS) may impair essential host defense mechanisms, including microbial clearance and immune cell function. In addition, certain phytochemicals may exhibit dual biological effects; for example, while promoting angiogenesis, they may also contribute to excessive fibrosis depending on dose, duration, and wound environment. Another key limitation is the poor bioavailability of some compounds, such as curcumin, which may restrict their clinical applicability despite strong preclinical efficacy.
Safety considerations are also critical, as plant-derived therapies may cause local irritation, allergic reactions, or systemic toxicity, particularly in the case of alkaloid-containing extracts. Furthermore, potential herb–drug interactions must be considered in diabetic patients receiving multiple medications, as such interactions may alter therapeutic efficacy or increase adverse effects.
Additionally, diabetic wounds are heterogeneous, including neuropathic, ischemic, and neuro-ischemic types, each characterized by distinct pathophysiological mechanisms. Therefore, therapeutic responses to medicinal plant–based interventions may vary depending on wound type, highlighting the need for more personalized and condition-specific treatment strategies.
As discussed earlier, oxidative stress and inflammation interact through pathways such as NF-κB to impair wound healing.
Safety and Translational Challenges
Despite the promising therapeutic potential of medicinal plants in diabetic wound healing, several safety and translational challenges must be carefully considered. Certain phytochemicals may produce adverse effects, including local skin irritation, allergic reactions, and, in some cases, systemic toxicity, particularly with prolonged use or high doses. Alkaloid-containing plants, for example, may exhibit cytotoxic or hepatotoxic effects if not properly standardized.
In addition, herb–drug interactions represent a significant concern in diabetic patients who are often on multiple medications. Some plant-derived compounds can modulate cytochrome P450 enzymes, potentially altering the metabolism of commonly prescribed drugs such as antidiabetic agents, anticoagulants, and antiplatelet medications. This may lead to either reduced therapeutic efficacy or increased risk of adverse effects. For instance, certain phytochemicals may enhance bleeding risk when used concomitantly with anticoagulant therapy.
Furthermore, regulatory and quality control issues pose major barriers to clinical translation. Variability in plant sources, extraction methods, and formulation processes can lead to inconsistent concentrations of active constituents. The presence of contaminants such as heavy metals, pesticides, or microbial toxins further complicates safety assessment. Therefore, the development of standardized formulations with well-characterized bioactive markers, along with stringent quality control and regulatory oversight, is essential for ensuring reproducibility, safety, and clinical applicability of plant-based therapies.
Quantitative outcomes reported in preclinical studies further support the therapeutic potential of medicinal plants; however, variability across models and study designs should be considered.
Conclusion
Prolonged hyperglycaemia, oxidative stress, chronic inflammation, poor angiogenesis, and imbalanced collagen remodelling are among the key factors that adversely effect on diabetic wound healing, a multifactorial process. The wound healing and repair process involves a finely tuned balance of haemostasis, inflammation, proliferation, and remodelling, but in diabetes, chronic non-healing ulcers are caused by a lengthening of an inflammatory response time, high production of reactive oxygen species (ROS), and immune dysfunction.
Oxidative stress and inflammation develop as key, interconnected contributors of poor wound healing in diabetes. ROS generated by hyperglycaemia trigger redox-sensitive inflammatory mechanisms including NF-κB, leading to chronic expression of pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1β. These mediators also augment oxidative injury, inhibit fibroblast and keratinocyte activities, hamper angiogenesis and accelerated degeneration of the extra cellular matrix. The subsequent vicious cycle of oxidative stress and inflammation inhibits wound healing and increases morbidity and risk of limb amputation in diabetic individuals.
Concurrent targeting of oxidative stress and inflammation is therefore a sound and promising method of therapy. Multi-target actions provide medicinal plants with a clear advantage in this respect. Medicinal plants show significant therapeutic potential as complementary approaches in diabetic wound management; however, their routine clinical use requires further validation through well-designed clinical studies. Although there are current problems associated with standardization and limited availabilities of clinical trials, a vast pool of preclinical and mechanistic data shows the great potential of medicinal plants to be used as an addition or alternative treatment to diabetic wound care. These promising discoveries will require the future incorporation of standardized formulations, molecular docking, experimental validation, and well-designed clinical studies that will translate the promising findings into effective, affordable and accessible therapies in diabetic wound healing.
The interplay between oxidative stress and inflammation remains central to diabetic wound pathology.
Future Perspectives
 |
Figure 4: Proposed research pipeline for the development of medicinal plant–based therapies for diabetic wound healing.
|
Acknowledgement
The authors would like to acknowledge the Department of Pharmacology, Navsahyadri Institute of Pharmacy, for providing the necessary facilities to conduct this review.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement-
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Suraj Jadhao: Conceptualization, literature review, and manuscript preparation including Abstract, Introduction, Pathophysiology of Diabetic Wound Healing, Role of Oxidative Stress and Inflammation, Therapeutic Potential of Medicinal Plants, Experimental Evidence, Molecular and In Silico Approaches, and preparation of figures and tables.
Ajay Kale: Supervision, critical revision of the manuscript for important intellectual content, and overall guidance throughout the study.
Kishor Otari: Literature collection, data organization, reference management, and assistance in manuscript formatting.
All authors contributed to the conception, design, literature search, and writing of the manuscript. All authors have read and approved the final version of the manuscript.
References
- Falanga V. Wound healing and its impairment in the diabetic foot. Lancet. 2005;366(9498):1736-1743. doi:10.1016/S0140-6736(05)67700-8
CrossRef - Brem H, Tomic-Canic M. Cellular and molecular basis of wound healing in diabetes. J Clin Invest. 2007;117(5):1219-1222. doi:10.1172/JCI32169
CrossRef - Guo S, DiPietro LA. Factors affecting wound healing. J Dent Res. 2010;89(3):219-229. doi:10.1177/0022034509359125
CrossRef - Sen CK. Wound healing essentials: Let there be oxygen. Wound Repair Regen. 2009;17(1):1-18. doi:10.1111/j.1524-475X.2008.00436.x
CrossRef - Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature. 2001;414(6865):813-820. doi:10.1038/414813a
CrossRef - Nishikawa T, Edelstein D, Du XL, et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature. 2000;404(6779):787-790. doi:10.1038/35008121
CrossRef - Vincent AM, Russell JW, Low P, Feldman EL. Oxidative stress in the pathogenesis of diabetic complications. Endocr Rev. 2004;25(4):612-628. doi:10.1210/er.2003-0019
CrossRef - Calixto JB. Efficacy, safety, quality control, marketing and regulatory guidelines for herbal medicines. Braz J Med Biol Res. 2000;33(2):179-189. doi:10.1590/S0100-879X2000000200004
CrossRef - Ekor M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol. 2014;4:177. doi:10.3389/fphar.2013.00177
CrossRef - World Health Organization. Quality Control Methods for Herbal Materials. Geneva, Switzerland: WHO Press; 2011.
- Rahman MM, Hossain MS, Islam MR, et al. Pharmacological activities and phytochemical constituents of Argemone mexicana Linn.: A review. J Ethnopharmacol. 2019;236:237-252. doi:10.1016/j.jep.2019.02.034
CrossRef - Ayele TM, Mekonnen A, Desta Z. Evaluation of in vivo wound-healing and anti-inflammatory activities of solvent fractions of Argemone mexicana L. J Inflamm Res. 2022;15:5417-5432. doi:10.2147/JIR.S383217
CrossRef - Heggers JP, et al. Beneficial effect of Aloe vera on wound healing in an excisional wound model. J Altern Complement Med. 1996;2(2):271-277.
CrossRef - Chithra P, Sajithlal GB, Chandrakasan G. Influence of Aloe vera on collagen characteristics in healing dermal wounds in rats. Mol Cell Biochem. 1998;181:71-76.
CrossRef - Jagetia GC, Rajanikant GK. Curcumin treatment enhances the repair and regeneration of wounds in diabetic rats. Wound Repair Regen. 2012;20(4):481-494. doi:10.1111/j.1524-475X.2012.00797.x
CrossRef - Sidhu GS, et al. Enhancement of wound healing by curcumin in animals. Wound Repair Regen. 1999;7(6):362-374.
CrossRef - Gupta A, Upadhyay NK, Kumar R. Antioxidant and wound-healing potential of medicinal plants in diabetic conditions. Biomed Pharmacother. 2019;109:232-245. doi:10.1016/j.biopha.2018.10.090
CrossRef - Kitchen DB, Decornez H, Furr JR, Bajorath J. Docking and scoring in virtual screening for drug discovery. Nat Rev Drug Discov. 2004;3(11):935-949. doi:10.1038/nrd1549
CrossRef - Meng XY, Zhang HX, Mezei M, Cui M. Molecular docking: A powerful approach for structure-based drug discovery. Curr Comput Aided Drug Des. 2011;7(2):146-157. doi:10.2174/157340911795677602
CrossRef - Lionta E, Spyrou G, Vassilatis DK, Cournia Z. Structure-based virtual screening for drug discovery. Curr Top Med Chem. 2014;14(16):1923-1938. doi:10.2174/1568026614666140929124445
CrossRef - Armstrong DG, Boulton AJM, Bus SA. Diabetic foot ulcers and their recurrence. N Engl J Med. 2017;376(24):2367-2375. doi:10.1056/NEJMra1615439
CrossRef - Eming SA, Martin P, Tomic-Canic M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci Transl Med. 2014;6(265):265sr6. doi:10.1126/scitranslmed.3009337
CrossRef - Loots MAM, et al. Differences in cellular infiltrate and extracellular matrix of chronic diabetic and venous ulcers versus acute wounds. J Invest Dermatol. 1998;111(5):850-857. doi:10.1046/j.1523-1747.1998.00381.x
CrossRef - Berlanga-Acosta J, et al. Cellular and molecular mechanisms of diabetic foot ulcer healing: Implications for advanced wound therapy. Diabetes Metab Res Rev. 2013;29(6):491-507. doi:10.1002/dmrr.2443
CrossRef - Schäfer M, Werner S. Oxidative stress in normal and impaired wound repair. Pharmacol Res. 2008;58(2):165-171. doi:10.1016/j.phrs.2008.06.004
CrossRef - Lan CCE, et al. High-glucose environment reduces human keratinocyte mobility. J Invest Dermatol. 2008;128(1):103-111. doi:10.1038/sj.jid.5700994
CrossRef - Zhao R, Liang H, Clarke E, Jackson C, Xue M. Inflammation in chronic wounds. Int J Mol Sci. 2016;17(12):2085. doi:10.3390/ijms17122085
CrossRef - Wilkinson HN, Hardman MJ. Wound healing: Cellular mechanisms and pathological outcomes. Open Biol. 2020;10(9):200223. doi:10.1098/rsob.200223
CrossRef - Ridiandries A, Tan JTM, Bursill CA. The role of macrophages in wound healing. Int J Mol Sci. 2018;19(9):2596. doi:10.3390/ijms19092596
CrossRef - Frykberg RG, Banks J. Challenges in the treatment of chronic wounds. Adv Wound Care (New Rochelle). 2015;4(9):560-582. doi:10.1089/wound.2015.0635
CrossRef - Shukla A, Rasik AM, Dhawan BN. Asiaticoside-induced elevation of antioxidant levels in healing wounds. Phytother Res. 1999;13(1):50-54. doi:10.1002/(SICI)1099-1573(199902)13:1<50::AID-PTR384>3.0.CO;2-F
CrossRef - Upadhyay NK, et al. Antioxidant, antimicrobial and wound-healing activity of Ficus religiosa. J Ethnopharmacol. 2011;135(1):183-190. doi:10.1016/j.jep.2011.03.021
CrossRef - Kumar B, et al. Wound-healing activity of Tridax procumbens in diabetic rats. Indian J Exp Biol. 2007;45:1070-1076.
- Dwivedi D, et al. Medicinal plants in wound healing. Int J Pharm Sci Res. 2013;4(11):4037-4048.
- Bairy KL, et al. Evaluation of wound-healing activity of herbal formulations in diabetic rats. J Ayurveda Integr Med. 2017;8(4):256-263. doi:10.1016/j.jaim.2017.03.001
- Süntar I. Importance of ethnopharmacological studies in wound healing. Front Pharmacol. 2020;11:1-14. doi:10.3389/fphar.2020.00152
CrossRef - Pazyar N, et al. Skin wound healing and phytomedicine. Int J Prev Med. 2014;5(7):853-864.
- Thakur R, Jain N, Pathak R, Sandhu SS. Practices in wound healing studies of plants. Evid Based Complement Alternat Med. 2011;2011:438056. doi:10.1093/ecam/nep023
CrossRef - Süntar I, et al. Wound healing potential of plants used in Turkish folk medicine. J Ethnopharmacol. 2012;134(2):486-496. doi: 10.1016/j.jep.2010.12.027
CrossRef - Ghosh PK, et al. Herbal therapeutics in wound healing: A review. Phytomedicine Plus. 2021;1(4):100036. doi: 10.1016/j.phyplu.2021.100036.
CrossRef - Li M, Yu H, Pan H, et al. Nrf2 suppression delays diabetic wound healing through sustained oxidative stress and inflammation. Front Pharmacol. 2019;10:1099. doi:10.3389/fphar.2019.01099
CrossRef - Guo YL, Niu WJ, Jiao HR, Li YP, Xu C, Zhou X, Wang J. Crosstalk between oxidative stress and inflammatory pathways: Natural therapeutic approaches for diabetic wound healing. World J Diabetes. 2025;16(11):111400. doi:10.4239/wjd.v16.i11.111400
CrossRef - Amer MS, El-Nesr KA, Abo El-Ela FI, Zanaty MI. Topical treatment of diabetic foot ulcers using a novel quercetin-loaded hyaluosome gel nanoformulation. Sci Rep. 2026;16(1):7180. doi:10.1038/s41598-026-37537-4
CrossRef
Abbreviation
AGEs – Advanced Glycation End Products AP-1 – Activator Protein-1 CAT – Catalase COX-2 – Cyclooxygenase-2 ECM – Extracellular Matrix FGF – Fibroblast Growth Factor GSH – Reduced Glutathione HPLC – High-Performance Liquid Chromatography IL-1β – Interleukin-1 Beta IL-6 – Interleukin-6 iNOS – Inducible Nitric Oxide Synthase Keap1 – Kelch-like ECH-associated Protein 1 LC-MS – Liquid Chromatography–Mass Spectrometry MDA – Malondialdehyde MMPs – Matrix Metalloproteinases NADPH – Nicotinamide Adenine Dinucleotide Phosphate NF-κB – Nuclear Factor Kappa B NMR – Nuclear Magnetic Resonance Nrf2 – Nuclear Factor Erythroid 2–Related Factor 2 PDGF – Platelet-Derived Growth Factor ROS – Reactive Oxygen Species SOD – Superoxide Dismutase STZ – Streptozotocin TGF-β – Transforming Growth Factor Beta TNF-α – Tumour Necrosis Factor Alpha VEGF – Vascular Endothelial Growth Factor
Accepted on: 25-03-2026
Second Review by: Dr. Ugwu Valentine
Final Approval by: Dr. Wagih Ghannam