Impact of Seed Pretreatments in Lablab purpureus (L.) Sweet Seedlings for Mitigating In vitro Salinity Stress.
Department of Botany, Fergusson College (Autonomous), Pune, India.
Corresponding Author E-mail: rupaliseth@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3510
ABSTRACT:The present study was conducted to evaluate the impact of different seed pretreatment agents on germination and seedling growth in Lablab purpureus (L.) Sweet (variety Kokan Bhushan), under in vitro saline conditions. Five different pretreatments were provided to the seeds: distilled water, NaCl (100 mM), NaCl (500 mM), KCl (100 mM) and KCl (500 mM). Untreated seeds served as control. The pretreated and untreated seeds were germinated in vitro on half strength Murashige and Skoog’s basal medium supplemented with graded series of salt stress (0, 60, 80 and 100 mM). The germinated seedlings were maintained in saline stress for six weeks. The results demonstrated that pretreatments with distilled water, NaCl (100 mM) and KCl (100 mM) significantly enhanced the germination percentage, shoot and root length, fresh and dry weight of the seedlings both in presence and absence of salinity stress. Pretreated seeds also exhibited improved seedling vigour index and salt tolerance indices. Seed pretreatment with KCl (100 mM) proved most effective followed by NaCl (100 mM) and distilled water. This study substantiates that KCl and NaCl seed pretreatments optimized under in vitro conditions, represent a simple, cost-effective and practical approach in Lablab purpureus (L.), for upscaling to salinity affected fields.
KEYWORDS:In vitro; KCl; Lablab purpureus (L.); NaCl; Salt stress; Seed pretreatment
Introduction
Legumes are indispensable source of plant-based protein for human and animal consumption. Lablab purpureus (L.) Sweet is a minor, underutilized, versatile legume with various synonyms such as Hyacinth bean, Indian Bean, Lablab bean, Field bean, Sem, Wal etc.1,2 Lablab is a store-house of nutrients, minerals, pharmaceuticals supporting health and wellness in mankind as well as livestock.3,4 In order to meet the growing demands for plant proteins, Lablab an underutilized grain legume should be brought into mainstream cultivation. The production of legumes is threatened due to changing climatic conditions resulting in excessive heat and altered rainfall patterns causing drought, salinity, and flooding.5 These climate induced stresses are detrimental for attaining full potential of food crops along with legumes and are expected to decline the global food production up to 2.3% by 2050.6 Amongst these, salinity is one of the major abiotic stresses which affects nearly 20% of total cultivated and 33% of irrigated agriculture land worldwide.7 It is expected that nearly 50% of cultivable land will become saline and unfit for agriculture by 2050.8 Lablab is moderately sensitive to salt stress.9,10 Seed germination and early seedling phase are the most vulnerable stages to salinity stress in majority of legumes.11 To overcome the detrimental effects of salt stress, seed pretreatment is an effective, low cost, simple and ecofriendly technique. Seeds are soaked for a specific time span in different agents such as distilled water, osmotic solutions, hormones etc., which induces a mild stress that becomes engrained in their epigenetic memory and improves tolerance potential towards future stresses.12,13 Seed pretreatment improves the expression of germination related and antioxidant genes, which scavenge reactive oxygen species (ROS) and accelerate seed germination in saline ecosystems.14 Pretreating the seeds synchronizes germination, ensures uniform seedling emergence and stand establishment thus improving productivity in salt affected environment.15,16 Soaking seeds in water prior to sowing is a traditional method for improving germination and seedling growth by lowering the reactive oxygen species produced in presence of salt stress.13 Seed pretreatment with inorganic salt solutions like KCl or NaCl lowers osmotic stress by reducing Na+ uptake and increasing K+ intake, maintaining ion homeostasis and chlorophyll content in saline conditions consequently improving seedling growth.17 Earlier studies showed that pretreatment of Lablab seedlings with spermidine improved their tolerance under saline stress.18 The present study was conducted to evaluate the impact of different seed pretreatment agents (distilled water, NaCl and KCl) at variable concentration on germination and seedling growth in Lablab in presence of graded series of salt stress under in vitro conditions. By analyzing the effects of different seed pretreatment agents, the study aims to identify the most effective agent and optimal dose for enhancing salinity stress tolerance in Lablab. This investigation provides valuable insights for mitigating salt stress in Lablab using seed pretreatment, a novel approach not previously explored in this underutilized and multipurpose legume.
Materials and Methods
Pretreatment and in vitro germination
The seeds of Lablab purpureus (L.) Sweet variety Kokan Bhushan, were procured from certified seed agency, Pune, Maharashtra. Five different pretreatments were provided to the seeds: a) distilled water, b) NaCl (100 mM), c) NaCl (500 mM), d) KCl (100 mM), e) KCl (500 mM). Untreated seeds served as control. Seeds were soaked in the pretreatment solution for 2 hours and maintained on a rotary shaker (DBK instruments, India) at room temperature to ensure continuous aeration and uniform exposure to the pretreatment agent. After pretreatment, the seeds were rinsed with sterile distilled water and blotted on filter paper. For in vitro maintenance of Lablab seeds, half strength Murashige and Skoog’s (1962)19 (HiMedia, India) basal medium supplemented with sucrose (3%), agar (0.8%) and NaCl (0, 60, 80 and 100 mM) was prepared in culture tubes (25 x150 mm). The pH of medium was adjusted at 5.8 ± 0.2 and it was autoclaved (Equitron®, Media instruments, India) at 121°C and 15 lbs. pressure for 20 minutes. Analytical grade chemicals (SRL, India) were used throughout the present study.
In order to establish seeds under in vitro conditions (Figure 1), both pretreated and control seeds were surface sterilized with 70% (v/v) ethanol for 30 seconds, followed by 0.1% (w/v) mercuric chloride for 8 minutes, and rinsed three times with sterile distilled water. The surface sterilized seeds were briefly blotted and inoculated in the culture medium. The seeds were kept in dark conditions for germination in the culture room. Germinated seedlings were maintained under 16 h light and 8 h dark photoperiod provided by cool white fluorescent tube light of 32 µmol m-2 s-1 intensity and at 25 ± 2°C temperature. Seeds were considered germinated when the healthy white radicle appeared through the integuments. The number of germinated seeds were recorded daily for two weeks. The germination percentage was calculated using the following formula: 20
Germination % = Number of germinated seeds/ Total inoculated seeds *100.
Growth parameters
The in vitro grown seedlings were harvested after six weeks of culture to study the growth parameters. The shoot and root length of seedlings was measured using graduated ruler (cm). Fresh weight was recorded immediately after the harvest using a digital balance (g) (CAH-223, CONTECH, India). Six weeks old seedlings were oven (Lab Hosp™ Oven digital, India) dried at 75°C for 48 to 72 hours till constant dry weight was achieved. To evaluate the performance of the pretreated and untreated Lablab seedlings in presence of stress the seedling vigor index (SVI) and salt tolerance index (STI) were calculated using the following formula respectively:
SVI = Germination percentage (G %) x Seedling length (SL). 21
STI = Value of trait under salt stress after pretreatment (Vin)/ Value of trait under control after pretreatment (Vic).22
Data Analysis
The experimental layout was completely randomized design. Each experiment comprised of six treatments containing five replicates and the experiments were repeated twice. The data is presented as means of five replicates per treatment and standard error. Statistical analysis was performed using Two-Way analysis of variance (ANNOVA) with replicates (p<0.05).
 |
Figure 1: An overview of the experimental design for seed pretreatment and in vitro establishment of Lablab pupureus (L.) Sweet.
|
Results
Germination percentage
The impact of seed pretreatment in Lablab on germination percentage in presence of in vitro salinity stress is depicted in Graph 1. The germination percentage increased on pretreatment with 100 mM KCl (100%), 100 mM NaCl (100%) and distilled water (91.7%) as compared to untreated seeds (83.34%). There was no improvement in germination percentage for pretreatment with 500 mM KCl, and pretreatment with 500 mM NaCl proved inhibitory. In presence of 80 mM salt stress, the germination percentage enhanced by 48.84% for 100 mM KCl, 41.86% for 100 mM NaCl and 23.26% for distilled water pretreatments as compared to untreated control. Seeds pretreated with 100 mM NaCl, 100 mM KCl and distilled water performed well and were able to mitigate the effect of high salinity stress (100 mM), whereas germination percentage declined drastically for untreated seeds. The Two-Way ANOVA for germination percentage (Table 1) showed statistically significant difference for seed pretreatments (F=108.90, p=5.6E-38) and NaCl concentration (F=1165.76, p= 2.4E-75) with non-significant interaction of seed pretreatments × NaCl concentrations (F= 3.96, p =1.6E-05).
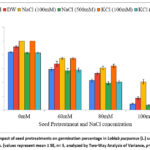 |
Graph 1: Impact of seed pretreatments on germination percentage in Lablab purpureus (L.) under saline stress. (values represent mean ± SE, n= 5, analyzed by Two-Way Analysis of Variance, p<0.05).
|
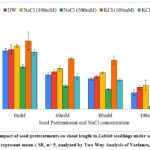
Root length
There was increment in root length of seedlings pretreated with 100 mM KCl (6.36 cm), 100 mM NaCl (5.22 cm) and distilled water (5.00 cm) in comparison with untreated seedlings (4.78 cm) under non-saline conditions (Graph 2). Root length exhibited increment of 33.05 %, 9.20 % and 4.60 % on pretreatment with 100 mM KCl, 100 mM NaCl and distilled water respectively in absence of salt stress. Under the influence of 80 mM salt stress, the root length increased by 56.75 % for 100mM KCl, 19.59 % for 100 mM NaCl and 10.13 % for distilled water pretreatments. The root length was 2.92 cm for 100 mM KCl followed by 2.24 cm for 100 mM NaCl and 2.02 cm for distilled water pretreatments over 1.68 cm under highest saline stress of 100 mM. The Two-Way ANOVA for root length (Table 1) showed statistically significant difference for seed pretreatments (F=85.27, p= 9.37E-24) and NaCl concentration (F= 149.62, p= 4.59E-36) with non-significant interaction of seed pretreatments × NaCl concentrations (F= 0.52, p= 0.920).
 |
Graph 2: Impact of seed pretreatments on root length in Lablab seedlings under saline stress. (values represent mean ± SE, n= 5, analyzed by Two-Way Analysis of Variance, p<0.05).
|
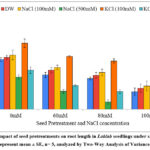
Shoot length
The shoot length increased on pretreatments with 100 mM KCl (16.42 cm), 100 mM NaCl (15.22 cm) and distilled water (14.32 cm) in comparison with untreated seeds (14.02 cm) under non-saline conditions (Graph 3). The shoot length improved by 17.11 %, 8.55 % and 2.13 % on pretreatment with 100mM KCl, 100 mM NaCl and distilled water in absence of stress. The shoot length declined upon pretreatment with 500 mM KCl and 500 mM NaCl demonstrating repressive nature of these agents at higher doses. Under 80 mM saline stress, the shoot length reached 11.78 cm for 100 mM KCl, 8.76 cm for 100 mM NaCl, 7.82 cm for distilled water pretreatments as compared to 7.30 cm for untreated seeds. The shoot length improved by 61.37 % for 100mM KCl, 20 % for 100 mM NaCl and 7.12 % for distilled water pretreatments. Maximum improvement in shoot length was obtained for pretreatment with 100 mM KCl, followed by 100 mM NaCl and distilled water in presence and absence of stress. The Two-Way ANOVA for shoot length (Table 1) showed statistically significant difference for seed pretreatments (F=32.27, p= 3.6E-19) and NaCl concentration (F= 179.84, p= 2.9E-39) with non-significant interaction of seed pretreatments × NaCl concentrations (F= 0.85, p= 0.615).
 |
Graph 3: Impact of seed pretreatments on shoot length in Lablab seedlings under saline stress. (values represent mean ± SE, n= 5, analyzed by Two-Way Analysis of Variance, p<0.05).
|
Seedling length
The pretreated seedlings displayed improved growth maintaining their superiority over untreated seedlings (Figure 2). The seedling length increased on pretreatment with 100 mM KCl (22.78 cm), 100 mM NaCl (20.44 cm) and distilled water (19.32) in absence of salt stress (Graph 4). In non-saline conditions seedling length enhanced by 21.17 % on pretreatment with 100 mM KCl. In presence of 80 mM saline stress seedling length was 16.42 cm for KCl, 12.30 cm for 100 mM NaCl and 11.08 cm for distilled water pretreated seedlings as compared to 10.26 cm for untreated seedlings. Maximum increment in seedling length was exhibited on pretreatment with 100 mM KCl (60.04%), followed by 100 mM NaCl (19.88%) and distilled water (7.99%) under the influence of salt stress (80 mM). The Two-Way ANOVA for seedling length (Table 1) showed statistically significant difference for seed pretreatments (F=72.45, p= 4.8E-31) and NaCl concentration (F= 299.60, p=1.4E-48) with nonsignificant interaction of seed pretreatments × NaCl concentrations (F= 0.90, p= 0.56).
 |
Figure 2: Effect of KCl (100 mM) pretreatment on seedling length in untreated (i) and pretreated (ii) Lablab seedlings under graded series of salt stress (a: control; b: 60 mM; c: 80 mM; d: 100 mM)
|
 |
Graph 4: Impact of seed pretreatments on seedling length in Lablab under saline stress. (values represent mean ± SE, n= 5, analyzed by Two-Way Analysis of Variance, p<0.05).
|
Seedling vigor index
The seedling vigour index shows the potential for germination and growth in the seedlings 23. Seed pretreatment significantly enhanced seedling vigour index both in presence and absence of salt stress (Graph 5). The seedling vigor index increased on pretreatment with 100 mM KCl (2278), 100 mM NaCl (2044) and distilled water (1773) over untreated seedlings (1579) in absence of salinity stress. At 80 mM saline stress, the seedling vigor index improved on pretreatment with 100 mM KCl (1046), 100 mM NaCl (753) and distilled water (588) as compared to untreated control (441). The Two-Way ANOVA for seedling vigor index (Table 1) showed statistically significant difference for seed pretreatments (F= 87.51, p= 3.4E-34) and NaCl concentration (F= 631.11, p= 5E-63), with the nonsignificant interaction of seed pretreatments × NaCl concentrations (F= 3.16, p= 0.00).
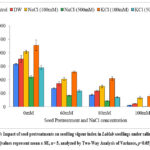 |
Graph 5: Impact of seed pretreatments on seedling vigour index in Lablab seedlings under saline stress. (values represent mean ± SE, n= 5, analyzed by Two-Way Analysis of Variance, p<0.05).
|
Fresh weight
There was significant increment in fresh weight of seedlings on pretreatment in presence and absence of saline stress (Graph 6). The fresh weight increased upon pretreatments with 100 mM KCl (58.97%), 100 mM NaCl (43.63%) and distilled water (21.15%) as compared to untreated seedlings in absence of saline stress. At 80 mM stress, the fresh weight was 1.01 g, 0.89 g and 0.72 g for pretreatments with 100 mM KCl, 100 mM NaCl and distilled water as compared with untreated control (0.52 g). The fresh weight of seedlings doubled upon pretreatment with 100 mM KCl exhibiting positive influence of pretreatment agent in presence of salinity stress (80 mM). The Two-Way ANOVA for fresh weight (Table 1) showed statistically significant difference for seed pretreatments (F= 28.88, p= 9E-18) and NaCl concentration (F= 149.53, p= 4.7E-36) with the non-significant interaction of seed pretreatments × NaCl concentrations (F= 1.50, p= 0.11).
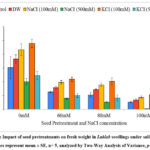 |
Graph 6: Impact of seed pretreatments on fresh weight in Lablab seedlings under saline stress. (values represent mean ± SE, n= 5, analyzed by Two-Way Analysis of Variance, p<0.05).
|
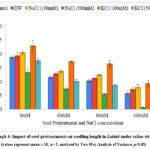
Dry Weight
There was increment in the dry weight of seedlings on pretreatment in presence and absence of salt stress. Under non-saline conditions, maximum increment in dry weight was observed for 100 mM KCl pretreatment registering an increase of 42.29 % as compared to untreated seedlings. Under the influence of salt stress (80 mM) the dry weight was 0.051 g, 0.040 g and 0.035 g for 100 mM KCl, 100 mM NaCl and distilled water pretreatments as compared to untreated control (0.028 g; Graph 7). The dry weight increased by 135.33 %, 40.69 % and 22.27 % for pretreatment with 100 mM KCl, 100 mM NaCl and distilled water respectively while it declined by 10.92 % and 37.69 % for pretreatments with 500 mM NaCl and 500 mM KCl. The Two-Way ANOVA for dry weight (Table 1) showed statistically significant difference for seed pretreatments (F=21.33, p= 2.7E-14) and NaCl concentration (F= 106.43, p= 2E-30) with non-significant interaction of seed pretreatments × NaCl concentrations (F= 1.19, p= 0.28).
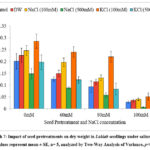 |
Graph 7: Impact of seed pretreatments on dry weight in Lablab seedlings under saline stress. (values represent mean ± SE, n= 5, analyzed by Two-Way Analysis of Variance, p<0.05).
|
Table 1: Two -Way analysis of variance (mean of squares) for growth parameters under saline stress in pretreated seedlings of Lablab purpureus (L.)
| Source of Variation | DF | GP | RL | SL | SDL | SVI | FW | DW |
| Seed pretreatment | 5 | 2372.33 | 30.39 | 93.82 | 226.85 | 2289370.80 | 2.14 | 0.03 |
| NaCl concentration | 3 | 25394.20 | 53.34 | 522.78 | 938.06 | 9078209.7 | 11.10 | 0.18 |
| Interaction | 15 | 86.27 | 0.18 | 2.48 | 2.83 | 33917.05 | 0.11 | 0.00 |
DF: degree of freedom; GP: germination percentage; RL: root length; SL: shoot length; SDL: seedling length; SVI: seedling vigour index; FW: fresh weight, DW: dry weight (p < 0.05).
Stress Tolerance Index
The stress tolerance index (Graph 8) for germination percentage at 80 mM salt stress showed maximum improvement upon pretreatment with 100 mM KCl (0.64), followed by 100 mM NaCl (0.61) and distilled water (0.58) over untreated (control) seeds (0.51). In presence of saline conditions (80 mM) stress tolerance index for root length improved by 17.81 %, 9.51 % and 5.29 % upon pretreatments with 100 mM KCl, 100mM NaCl and distilled water as compared to control (untreated). Seed pretreatment had positive impact on the shoot length stress tolerance index in presence of salt stress (80 mM) with 100 mM KCl being superior (0.72) as compared to 100 mM NaCl (0.58) and distilled water (0.55). The fresh weight stress tolerance index enhanced by 21.09 %, 18.06 % and 13.85 % on pretreatments with 100 mM KCl, 100 mM NaCl and distilled water over untreated seeds under the influence salt stress (80 mM). The dry weight stress tolerance index at 80 mM salt stress was maximum for pretreatment with 100 mM KCl (65.39 %) followed by 100 mM NaCl (15.14 %) and distilled water (8.74 %).
The two-way analysis of variance (Table 2) indicated that seed pretreatments and NaCl concentration significantly influenced the stress tolerance indices (GSTI, RLSTI, SLSTI, FWSTI and DWSTI) in Lablab seedlings however, the interaction between seed pretreatments and NaCl concentration was non-significant.
Table 2: Two -Way analysis of variance (mean of squares) for stress tolerance index under saline stress in pretreated seedlings of Lablab purpureus (L.)
| Source of Variation | DF | GPSTI | RLSTI | SLSTI | FWSTI | DWSTI |
| Seed pretreatment | 5 | 0.10 | 0.41 | 0.29 | 0.08 | 0.15 |
| NaCl concentration | 2 | 1.85 | 1.37 | 0.91 | 0.79 | 3.02 |
| Interaction | 10 | 0.02 | 0.03 | 0.02 | 0.00 | 0.01 |
DF: degree of freedom; GPSTI: germination percentage stress tolerance index; RLSTI: root length stress tolerance index; SLSTI: shoot length stress; FWSTI: fresh weight stress tolerance index, DW: dry weight stress tolerance index (p < 0.05).
Discussion
The present study established that pretreatments with distilled water, NaCl (100 mM) and KCl (100 mM) significantly improved the germination percentage and growth parameters both in presence and absence of salinity stress in Lablab. Seed germination is the most crucial stage for successful establishment of a plant during its life cycle.24 Salinity is detrimental for germination especially in legumes.11 Seed pretreatment prepares seeds to withstand stressful conditions and improve growth parameters.25 Treating seeds before sowing has proved beneficial in several plants such as wheat,26 legumes 27 and maize.28 Seed pretreatment brings about several biochemical changes within the seed by stimulating the metabolic processes essential for germination. Pretreatment improves the activity of enzymes α amylases and proteases leading to efficient mobilization of reserved food material to the germinating embryo thus facilitating increased germination under stressful conditions.29 Pretreated seeds exhibit enhanced activity of antioxidant enzymes like superoxide dismutase, catalase, and ascorbate peroxidases. These enzymes effectively scavenge reactive oxygen species (ROS) and strengthen the defense mechanism in salt stress.30 There is accumulation of osmoprotectants such as proline, soluble sugars and inorganic ions in pretreated seeds which help in maintaining the osmotic balance and cellular stability upon exposure to saline stress.31
Lablab is moderately sensitive to salt stress,32 therefore raising salinity declined the germination percentage, however, pretreatment with KCl (100 mM), NaCl (100 mM) and distilled water showed significant improvement in germination over untreated seeds. Similar results were seen in wheat genotypes Kharchia 65 (salt tolerant) and PI 94341 (salt sensitive) where distilled water, NaCl (50 mM) and KCl (50 mM) pretreated seeds exhibited better germination response under saline conditions.26 Earlier studies in beans and chickpea also reported improvement in germination percentage for seeds primed with KCl (15g/L).27 Pretreatment with KCl (100 mM) proved most effective as potassium is an important vital nutrient that shows attenuating effect on various abiotic stresses such as salinity, water and temperature stress. Potassium improves growth and development in plants as it participates in various metabolic activities such as protein synthesis, photosynthesis, stomatal regulation and enzyme activation.27,33 Growth attributes declined drastically upon pretreatment with 500 mM NaCl compared to 500 mM KCl demonstrating repressive nature of these agents at higher doses. Possibly because the osmotic stress induced by NaCl exerts stronger detrimental effect compared to KCl owing to higher toxicity of sodium ions. At higher concentrations (500 mM), both NaCl and KCl disrupt the antioxidant defence mechanism causing oxidative imbalance and damaging cellular metabolism. Therefore, the embryonal cells are unable to maintain sufficient energy required for germination. Additionally, excessive accumulation of Na+ and K+ damages cell membranes and embryonal tissues impairing seed germination.34,35 This is supported by earlier studies in Capsicum annuum L. where seed germination and seedling development was adversely affected at higher levels (2% and 3%) of KCl.36
High salinity reduces the root and shoot length in growing seedlings.37 Pretreatment with 100 mM KCl improved the root and shoot length in Lablab seedlings under high saline conditions (100mM). This agrees with earlier reports for increment in root length in Sorghum genotype CSV15 upon pretreatment with KCl (150mM).38 Shoot length improvement is supported by previous studies on KCl seed priming (250 and 500 ppm) in Pisum sativum, where seedling shoot length enhanced in primed seeds reducing the impact of salinity stress.39
Maximum increment in seedling length was evident in Lablab seedlings upon pretreatment with 100 mM KCl. Similar findings were reported in rice cultivars which demonstrated improved tolerance towards salt stress upon exposure to 2.2 % KCl for 36 hrs by increasing germination capacity, germination speed, seedling length and dry weight in saline medium.40 Earlier reports on Cucumis sativus support the present findings where priming with 20 mM KCl exhibited increment in seedling length. 41
The overall expression of seed germination and seedling growth can be ascertained by studying the seedling vigor index. 42 Lablab seedlings performed better with 100 mM KCl pretreatment both in presence (80mM) and absence of saline stress. These results are in accordance to previous reports on Wheat where seedling vigor index enhanced on pretreatment with 2 % KCl.43 Seed germination and seedling vigor improved in seeds pretreated with KCl (10,000 and 20,000 ppm) in Wheat.44
Seeds pretreated with 100 mM KCl reflected enhancement in seedling fresh weight under saline and non-saline situations. Fresh weight of pretreated seedlings displayed the following trend 100 mM KCl >100 mM NaCl > distilled water in presence and absence of salt stress. These results are in agreement with earlier reports where KCl (15 mM) priming increased seedling fresh and dry weight in Cicer arietinum, Pisum sativum and Vicia faba under salinity stress.27 Effect of NaCl priming (5 g/L) increased the fresh and dry weights in Zea mays, diminishing the impact of salt stress during seedling stage.45
The dry weight parameters showed maximum values under saline (80mM) and non-saline conditions in seedlings pretreated with KCl (100 mM) and NaCl (100 mM). Studies done in Safflower showed better growth parameters such as fresh and dry weight, seedling length, seedling vigor index on priming with NaCl and KCl (5g/L).46 Seed pretreatment with KCl (5 g/L) and NaCl (5 g/L) raised the dry weight in turnip under high saline stress.47 The stress tolerance index (STI) for germination and growth attributes improved after seed pretreatment in Lablab exhibiting that pretreatments were effective in alleviating the adverse impact of salinity. Our results find similarity with earlier studies in mung bean (Vigna radiata L.) where significant improvement in stress tolerance indices was obtained followed by seed pretreatment. 48
Conclusion
Lablab purpureus (L.) Sweet is moderately sensitive to salt stress. Hence, the present study was conducted to determine the impact of different seed pretreatment agents such as distilled water, KCl and NaCl at variable doses for determining the most effective agent and optimal dose for better adaptability of Lablab seedlings in presence of in vitro salinity stress. The results suggested that seed pretreatment with KCl (100 mM), NaCl (100 mM) and distilled water considerably improved the germination percentage and morphological parameters like shoot length, root length and seedling length as compared to untreated (control) seeds in presence of in vitro graded series of salt stress. The seedling vigor index and stress tolerance index also improved upon seed pretreatment with 100 mM KCl, 100 mM NaCl and distilled water. However, seed pretreatment with high salt concentrations (500 mM KCl and NaCl) probably damaged the embryo and inhibited germination and seedling growth. This investigation provides valuable insights for optimizing the dose and duration of KCl and NaCl pretreatments in Lablab through in vitro approach, enabling rapid, ecofriendly and cost-effective upscaling in salt affected ecosystems.
Acknowledgement
The authors are thankful to The Principal, Fergusson College (Autonomous), Pune and Head, Department of Botany, Fergusson College (Autonomous), Pune for providing the necessary infrastructure and laboratory facilities during the research work. KDS is thankful to Dr. Babasaheb Ambedkar Research and Training Institute (BARTI), Pune for providing Junior research fellowship.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources –
Not Applicable
Author Contributions
Kalyani Deepak Salunke: Conceptualization, Methodology, Data Collection, Analysis, Writing-original draft.
Rupali Seth: Writing-Review and Editing, Visualization, Resources, Supervision.
References
- Maass B L, Robotham O, Chapman M A. Evidence for two domestication events of hyacinth bean (Lablab purpureus (L.) Sweet): A comparative analysis of population genetic data. Resour. Crop Evol. 2016;64(4):1221-1230.
CrossRef - Ramesh S, Byregowda M. Dolichos bean (Lablab purpureus Sweet var. lignosus) genetics and breeding – present status and future prospects. Mysore J. Agric. Sci. 2016; 50(3):481-500.
- Das S, Bose A, Reang M, Datta B K. Underutilized legumes of India: a potential food resource for the future. Plants. 2024;1(1):168-170.
CrossRef - Bhat S S, Shivamallu C, Prasad K S, Prasad S K. Biomedical importance of Lablab purpureus: A review. Med. Plants. 2022;14(1):20-29.
CrossRef - Eswar D, Karuppusamy R, Chellamuthu S. Drivers of soil salinity and their correlation with climate change. Opin. Environ. Sustain. 2021;50:310-318.
CrossRef - Kiyani A, Lei M, Rabbani G, Sardar M M, Ilyas M K, Iqbal U, Tahir A T. Legume production under the changing climate. In: Climate Smart Agric. Future Food Secur. (Faiz S, Ashraf U, Attia KA, Amir RM, ed) Singapore: Springer. 2025;479–505.
CrossRef - Mukhopadhyay R, Sarkar B, Jat H S, Sharma P.C., Bolan N.S. Soil salinity under climate change: Challenges for sustainable agriculture and food security. Environ. Manage. 2021;280(6):111736.
CrossRef - Kumar P, Sharma P K. Soil salinity and food security in India. Sustain. Food Syst. 2020; 4:533781.
CrossRef - Mass E V. Testing crops for salinity tolerance. In: Proc. Workshop on Adaptation of Plants to Soil Stresses (Maranville JW, Baligar BV, Duncan RR, Yohe JM, ed). INTSORMIL. Pub. No. 94-2, University of Nebraska, Lincoln. 1993;234-247.
- D’Souza M R, Devaraj V R. Biochemical responses of Hyacinth bean (Lablab purpureus) to salinity stress. Acta Physiol. Plant. 2010; 32:341–353.
CrossRef - Bouzroud S, Henkrar F, Fahr M, Smouni A. Salt stress responses and alleviation strategies in legumes: a review of the current knowledge. 3 Biotech. 2023;13(8):287.
CrossRef - Paparella S, Araújo S S, Rossi G. Seed priming: state of the art and new perspectives. Plant Cell Rep. 2015; 34:1281-1293.
CrossRef - Biswas S, Seal P, Majumder B, Biswas A K. Efficacy of seed priming strategies for enhancing salinity tolerance in plants: An overview of the progress and achievements. Plant Stress. 2023; 9:100186.
CrossRef - Banerjee A, Roychoudhury A. Seed priming technology in the amelioration of salinity stress. In: Advances in Seed Priming (Rakshit A, Singh HB, ed). Springer Nature Singapore Pte Ltd.2018;81-93.
CrossRef - Seth R. Seed priming to improve tomato productivity in salinity stressed environments: A Review. Biotech. Res. Asia. 2023;20(3):817-826.
CrossRef - Adhikari B, Olorunwa O J, Barickman T C. Seed priming enhances seed germination and morphological traits of Lactuca sativa under salt stress. Seeds. 2022;1(2):74-86.
CrossRef - Purwestri Y A, Nurbaiti S, Putri S P M, Wahyuni I M, Yulyani S R, Sebastian A, Nuringtyas T. R., Yamaguchi N. Seed halopriming: A promising strategy to induce salt tolerance in Indonesian pigmented Rice. Plants. 2023;12(15):2879.
CrossRef - D’souza M, Devaraj V R. Pre-treatment with spermidine reverses inhibitory effects of salt stress in Hyacinth bean (Lablab purpureus). Chem. Pharm. Res. 2015;7(1):504-509.
- Murashige T, Skoog F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Plant. 1962;15(3):473-497.
CrossRef - Kandil A A, Sharief, A E, Abido W A E, Ibrahim M M. Effect of salinity on seed germination and seedling characters of some forage sorghum cultivars. J. Agric. Sci. 2012;4(7):306-311.
CrossRef - Abdul-Baki A, Anderson J D. Vigor determination in soybean seed by multiple criteria. Crop Sci.1973; 13:630-633.
CrossRef - Li W, Zhang H, Zeng Y. A salt tolerance evaluation method for Sunflower (Helianthus annuus) at the seed germination stage. Sci. Rep. 2020; 10:10626.
CrossRef - Ghanad M, Nulit R, Go R, Yong C. The effects of NaCl, KCl and MgCl2 on the germination of Brassica rapa parachinensis seed. Int. J. Biol. Res. 2016; 4(1)52-55.
CrossRef - Ghosh T, Atta K, Mondal S, et al. Hormonal signaling at seed germination and seedling stage of plants under salinity stress. Plant Growth Regul.2025;105:583-600. doi:10.1007/s10725-025-01305-7
CrossRef - Paulikienė S, Benesevičius D, Benesevičienė K, Ūksas T. Review- Seed treatment: Importance, application, impact, and opportunities for increasing sustainability. 2025;15(7):1689. doi:10.3390/agronomy15071689
CrossRef - Saddiq M S, Iqbal S, Afzal I, Ibrahim A H, Bakhtavar M A, Hafeez M B, Jahanzaib, Maqbool M M. Mitigation of salinity stress in Wheat (Triticum aestivum) seedlings through physiological seed enhancements. J. Plant Nutr. 2019;42(10):1192-1204.
CrossRef - Buhwish R. Seed priming with potassium chloride to alleviate salinity stress of some legume plants. J.Basic Sci. 2023;20:1-12.
- Mahara G, Bam R, Kandel M, Timilsina S. Seed priming with NaCl improves germination in Maize under saline soil conditions. Eurasian J. Soil Sci. 2022;11(2):151-156.
CrossRef - Rhaman M S. Seed priming before the sprout: revisiting an established technique for stress resilient germination. Seeds. 2025; 4(3): 29. https://doi.org/10.3390/seeds4030029
CrossRef - Jatana B S, Grover S, Ram H, Baath GS. Seed priming: molecular and physiological mechanisms underlying biotic and abiotic stress tolerance. Agronomy. 2024;14(12):2901. https://doi.org/10.3390/agronomy14122901
CrossRef - Anghla LC, Rehan, Das S, Sharma A, Thakur S, Rana N, Sharma S. Assessment of seed priming for mitigating abiotic stress and improving growth of horticultural crops: a review. Appl. Sci. 2025; 7:1423. https://doi.org/10.1007/s42452-025-08054-2
CrossRef - D’souza M, Devaraj V R. Role of calcium in increasing tolerance of hyacinth bean to salinity. Appl. Biol. Biotechnol. 2013; 1:11-20. https://dx.doi.org/10.7324/JABB.2013.1301
- Wang M, Zheng Q, Shen Q, Guo S. The critical role of potassium in plant stress response. Int. J. Mol. Sci. 2013;14(4):7370-90.
CrossRef - Ibrahim EA. Seed priming to alleviate salinity stress in germinating seeds. Plant Physiol. 2016;15;192: 38-46. https://doi.org/10.1016/j.jplph.2015.12.011
CrossRef - Sghayar S, Debez A, Lucchini G, Abruzzese A, Zorrig W, Negrini N, et.al. Seed priming mitigates high salinity impact on germination of bread wheat (Triticum aestivum) by improving carbohydrate and protein mobilization. Plant Direct. 2023;4;7(6): e497.doi: 10.1002/pld3.497
CrossRef - Joseph E, Pius C, Mendez A. Effect of potassium chloride stress on seed germination and growth of Capsicum annuum Int. J. Res. Eng. Sci. 2021;9(7):30-34.
- Gaur K, Saxena K. Exploring salinity effects on moth beans in response to physiological parameters. Ecol. Eng. 2025; 26:261-273. doi:10.12911/22998993/203400
CrossRef - Chauhan P, Pandey G, Pandey P K. Priming with potassium solutions improves seedling growth and vigor in forage Sorghum (Sorghum bicolor) J.Appl.Nat.Sci.2016; 8(4):1937-1940.
CrossRef - Naz F, Gul H, Hamayun M, Sayyed A, Khan H, Khan S S. Effect of NaCl stress on Pisum sativum germination and seedling growth with the influence of seed priming with potassium (KCl and KOH). Eurasian J. Agric. Environ. Sci. 2014;(11):1304-1311.
- Afzal I, Butt A, Rehman A.U, Basra S M A, Afzal A. Alleviation of salt stress in fine aromatic rice by seed priming. J. Crop Sci. 2012;6(10):1401-1407.
- Mohammed S J, Nulit R, Fayed M I A. Effects of using salicylic acid, potassium chloride, and a mixture of salicylic acid and potassium chloride treatments on salt-stressed Cucumis sativus Malaysia timun 2 germination. Plant Biotechnol. Persa. 2023; 5:56-67.
CrossRef - Barpete S, Oguz M, Ozcan S, Anayol E, Ahmed HA, Khawar KM, Ozcan S. Effect of temperature on germination, seed vigor index and seedling growth of five Turkish cotton (Gossypium hirsutum L) cultivars. Fresenius Environ Bull. 2015; 24:2561-2566.
- Haque Md A. Effect of seed priming on germination behavior and emergence of Wheat (Triticum aestivum). J. Agric. Ecol. Res. Int. 2024;25(2):53-61.
CrossRef - Anwar M P, Akhter M, Aktar S, et.al. Relationship between seed priming mediated seedling vigor and yield performance of spring Wheat. J. Exp.Bot. 2024;93(6):1159-1177.
CrossRef - Abraha B, Yohannes G. The role of seed priming in improving seedling growth of maize (Zea mays) under salt stress at field conditions. Agric. Sci. 2013;4(12):666-672.
CrossRef - Elouaer M A, Hannachi C. Seed priming to improve germination and seedling growth of safflower (Carthamus tinctorius) under salt stress. J. BioSci. 2012;6:76-84.
CrossRef - Mohammed S, Nulit R. Seed priming improves the germination and early growth of turnip seedlings under salinity stress. Periodico Tche Quim. 2020;17(35):73-82.
CrossRef - Sivakumar R, Nandhitha G K. Impact of PGRS and nutrients pre-soaking on seed germination and seedling characters of mung bean under salt stress. Legume Res. 2017;40(1):125-131.
Accepted on: 25-03-2026
Second Review by: Dr. Shashikala Metri
Final Approval by: Dr. Wagih Ghannam










