Protective Effects of Phenolic-Rich Moringa oleifera Extract on Streptozotocin–Nicotinamide-Induced Endothelial Dysfunction: Involvement of Arginase-1 and Nrf2-Mediated Antioxidant Pathways
1Department of Bio-technology, K L E F Deemed to be University, Vijayawada, Andhra Pradesh, India.
2Department of Bio Technology, Centre for Innovative Research, JNTUH University College of Engineering, Science & Technology Hyderabad (UCESTH), Telangana, India.
Corresponding Author Email: koteswarareddy@kluniversity.in
DOI : http://dx.doi.org/10.13005/bbra/3509
ABSTRACT:Endothelial dysfunction is a critical precursor to vascular complications in type 2 diabetes, largely driven by oxidative stress, impaired nitric oxide (NO) bioavailability, and inflammation. This study evaluated the protective effects of phenolic-rich Moringa oleifera extract (PMOE) against streptozotocin-nicotinamide-induced endothelial dysfunction in rats, focusing on the arginase 1/eNOS axis and the Nrf-2 pathway. Male Wistar rats were divided into four groups (n = 6): normal control, diabetic control, and diabetic rats treated with PMOE (250 mg/kg or 500 mg/kg) orally for 45 days. Diabetic controls exhibited significant hyperglycemia, elevated HbA1c, reduced serum insulin, increased blood pressure, and higher serum arginase activity compared to normal rats. These changes were accompanied by reduced NO levels, decreased NO synthase (NOS) activity, and enhanced reactive oxygen species (ROS) generation with concomitant depletion of endogenous antioxidants (reduced glutathione, superoxide dismutase, catalase, glutathione peroxidase). PMOE administration significantly lowered fasting glucose, HbA1c, blood pressure, and arginase activity while restoring insulin, NO concentrations, NOS activity, and antioxidant status in a dose-dependent manner. RT-qPCR analysis revealed that PMOE suppressed ARG1, VCAM-1, and ICAM-1 expression, while upregulating eNOS, Nrf-2, HO-1, and NQO1 transcripts. Histopathological examination confirmed severe vascular disruption in diabetic controls, whereas PMOE treatment preserved endothelial integrity and medial structure, particularly at the higher dose, which closely resembled normal controls. These findings indicate that PMOE mitigates diabetes-induced endothelial dysfunction by reducing arginase activity, restoring NO bioavailability, and activating the Nrf-2 antioxidant pathway.
KEYWORDS:Arginase 1; Endothelial dysfunction; Moringa oleifera; Nrf-2 pathway; Oxidative stress
Introduction
Diabetes mellitus (DM) is a chronic metabolic disorder characterized by persistent hyperglycemia resulting from impaired insulin secretion, action, or both. 1,2 A central and devastating complication of DM is endothelial dysfunction, a pathological state marked by reduced bioavailability of nitric oxide (NO), increased oxidative stress, and inflammation, which together promote atherogenesis and microvascular damage. 3,4 Endothelial dysfunction is not only an early biomarker but also a critical driver of diabetic vascular complications such as retinopathy, nephropathy, neuropathy, and cardiovascular disease. 3 Despite advances in pharmacotherapy, the burden of diabetic vascular disease remains high, underscoring the need for novel and effective therapeutic strategies that specifically target endothelial health.5
At the molecular level, endothelial dysfunction in DM is closely associated with dysregulated arginine metabolism and redox imbalance. Arginase 1 (ARG1) competes with endothelial nitric oxide synthase (eNOS) for the common substrate L-arginine, diverting it away from NO production toward ornithine and polyamines. 6 Upregulation of ARG1 in diabetes leads to reduced NO bioavailability and exacerbated vascular tone dysregulation, inflammation, and oxidative stress. Meanwhile, the nuclear factor erythroid 2-related factor 2 (Nrf-2) pathway plays a pivotal role in cellular antioxidant defense by regulating the transcription of genes encoding antioxidant enzymes such as heme oxygenase-1 (HO-1), NAD(P)H quinone oxidoreductase-1 (NQO1), and glutathione-related enzymes. 7,8 However, hyperglycemia and oxidative stress in diabetes impair Nrf-2 signaling, diminishing the endothelium’s capacity to counteract reactive oxygen species (ROS) mediated injury. 9,10 The coordinated dysregulation of ARG1 and Nrf-2 pathways thus represents a mechanistic nexus underlying diabetic endothelial dysfunction.3,8
Given this mechanistic insight, there is growing interest in the use of plant-derived, phenolic-rich extracts as adjunctive or alternative therapies for vascular complications in DM. 11 Polyphenols are well-known for their potent antioxidant, anti-inflammatory, and vasoprotective properties. 12 They have been shown to restore NO bioavailability, inhibit ARG1 activity, and activate Nrf-2-dependent antioxidant pathways, thereby counteracting the multifaceted insults of hyperglycemia-induced endothelial dysfunction. 13,14 Among medicinal plants, Moringa oleifera stands out as a rich source of phenolic compounds such as flavonoids, phenolic acids, and tannins. These phytochemicals have demonstrated diverse bioactivities, including modulation of oxidative stress, inflammation, and glucose metabolism, making Moringa a promising candidate for mitigating diabetic complications.15
Despite extensive ethnomedicinal use and emerging evidence of Moringa’s anti-diabetic and cardioprotective effects, the specific role of phenolic-rich Moringa oleifera (PMOE) extract in ameliorating endothelial dysfunction via the ARG1 and Nrf-2 pathways remains inadequately elucidated. Addressing this knowledge gap is crucial for validating the therapeutic potential of Moringa in diabetic vasculopathy and informing the development of phytopharmaceutical interventions targeting molecular drivers of endothelial dysfunction. The present study is therefore designed with the hypothesis that PMOE can attenuate streptozotocin (STZ)-nicotinamide (NAD)-induced endothelial dysfunction in a diabetic model by downregulating ARG1 expression and activity while upregulating Nrf-2-mediated antioxidant defenses. By systematically evaluating the extract’s effects on vascular function, oxidative stress markers, ARG1/Nrf-2 signaling pathways, and related molecular endpoints, this work aims to provide mechanistic evidence supporting the use of PMOE as a promising plant-based therapeutic strategy for diabetic endothelial dysfunction.
Materials and Methods
Preparation of PMOE
Fresh leaves of M. oleifera were collected from a cultivated garden in Hyderabad, Telangana, India (17.418974° N, 78.526596° E) and authenticated by a taxonomist at Osmania University (Voucher specimen: OUH/BDH/11231). The leaves were thoroughly washed, shade-dried at ambient temperature to prevent thermal degradation of phytoconstituents, and milled to obtain a fine powder. The powdered material was extracted using 70% ethanol through cold maceration for 72 h with intermittent stirring to enhance solvent penetration. The resulting extract was filtered, concentrated under reduced pressure at 40 °C using a rotary evaporator, and lyophilized to yield a dry powder.
Preliminary phytochemical analysis
Preliminary phytochemical screening of the PMOE was conducted using standard qualitative tests to identify major secondary metabolite classes. Small aliquots of the extract were subjected to established chemical assays to detect alkaloids (Mayer’s and Wagner’s reagents), flavonoids (alkaline reagent and Shinoda tests), tannins and phenolics (Ferric chloride reaction).
Quantitative analysis of phytochemicals
Quantitative analysis of major phytochemical groups in the PMOE was performed using standard spectrophotometric methods. Total phenolic content was determined by the Folin-Ciocalteu assay and expressed as milligrams of gallic acid equivalents per gram of extract (mg GAE/g). Total flavonoid content was measured using the aluminum chloride colorimetric method, calculated as milligrams of quercetin equivalents per gram of extract (mg QE/g). Condensed tannins were estimated by the vanillin-HCl assay, expressed as catechin equivalents (mg CE/g). Each analysis was conducted in triplicate to ensure reproducibility.
Animal experimental design
Male Wistar rats (8 weeks old, 180-220 g) were maintained under controlled conditions (22 ± 2 °C, 50-60% humidity, 12 h light/dark cycle) with ad libitum access to standard diet and water. Experimental procedures were approved by the Institutional Animal Ethics Committee, Jeeva Life Sciences, Hyderabad, Telangana, India (Approval No: CCSEA/IAEC/JLS/21/04/24/043). DM was induced by intraperitoneal administration of NAD (110 mg/kg), followed after 15 min by STZ (55 mg/kg) in citrate buffer (pH 4.5). Fasting blood glucose was measured 72 h later, and animals with levels > 250 mg/dL were included as diabetic. Rats were randomly assigned to four groups (n = 6): normal control (saline, 2 mL/kg), diabetic control (saline, 2 mL/kg), diabetic + PMOE (250 mg/kg), and diabetic + PMOE (500 mg/kg) and respective doses were orally administered for 45 days.
Determination of blood glucose, insulin and glycated hemoglobin
Fasting blood glucose was measured using the glucose oxidase-peroxidase method with a standard diagnostic kit (Cat. No: G3660-1CAP, Sigma-Aldrich®, India). Serum insulin concentrations were quantified by a rat-specific ELISA kit (Cat. No: RAB0904, Sigma-Aldrich®, India) according to the manufacturer’s instructions. Glycated hemoglobin (HbA1c) levels were determined in whole blood samples using an ion-exchange resin method and expressed as a percentage of total hemoglobin.
Measurement of blood pressure
Systolic blood pressure was assessed non-invasively using the tail-cuff method in conscious, minimally restrained rats. Animals were acclimatized to the warming chamber (35 °C), restrainer, and tail cuff for 20 min per day over three consecutive days prior to recording. On the day of measurement, rats were placed in the warming chamber and restrainer for a 5-10 min equilibration period. Blood pressure was then recorded using an automated tail-cuff system with 10 inflation-deflation cycles. The mean of six consecutive readings falling within a 5-10 mmHg variation range was considered the final blood pressure value.
Determination of serum arginase activity and advanced glycation end products (AGEs)
Serum arginase activity was quantified by measuring urea formation, the enzymatic product of arginase-mediated hydrolysis of L-arginine. Briefly, serum samples were incubated for 1 h in carbonate buffer (pH 9.5) containing manganese sulfate as an activator and L-arginine as the substrate. The urea generated was reacted with diacetyl monoxime under acidic conditions in the presence of ferric chloride (oxidizing agent) and carbazide (color development enhancer). The resulting chromophore was measured spectrophotometrically at 525 nm. Serum AGEs were analyzed following the method described by Sampathkumar et al. 16 Serum samples were diluted (1:15) in saline, and fluorescence intensity was recorded using a LS45 fluorescence spectrophotometer (PerkinElmer®, UK) at an excitation wavelength of 370 nm and emission at 440 nm.
Assessment of intracellular ROS levels
Intracellular ROS generation in aortic tissues was evaluated using the fluorescent probe 2′,7′-dichlorofluorescein diacetate (DCF-DA) with slight modifications to the method of Ugochukwu and Cobourne. 17 Frozen aortic samples were homogenized in 0.32 mol/L sucrose solution (1:20, w/v) on ice and centrifuged at 2000 rpm for 15 min at 4 °C. An aliquot of the resulting supernatant (100 µL) was mixed with 1.9 mL of Tris-HEPES buffer (40 mmol/L Tris, pH 7.4; containing 10 mmol/L HEPES, 120 mmol/L NaCl, 2.5 mmol/L KCl, 1.2 mmol/L NaH2PO4, 0.1 mmol/L MgCl2, 5 mmol/L NaHCO3, 6 mmol/L glucose, and 1 mmol/L CaCl2) supplemented with 5 µM DCF-DA and 0.1% pluronic F-127. Fluorescence intensity was recorded at an excitation wavelength of 485 nm and emission wavelength of 515 nm both prior to and following 2 h incubation at 37 °C. The hourly change in fluorescence was used as an index of ROS production.
Determination of NO, NOS activity, and argininosuccinate enzyme activities
Serum NO levels were determined using the Griess reaction. Nitric oxide synthase (NOS) activity was measured with a commercial assay kit (Cat. No: MAK532, Sigma-Aldrich®, India). Argininosuccinate synthetase (AS) and argininosuccinate lyase (ASL) activities were assessed according to the methods as described by Swamy et al. 18
Assessment of oxidative stress markers in aortic tissue
Aortic tissues were rapidly excised, rinsed in ice-cold saline to remove residual blood, blotted dry, and weighed. Samples were homogenized in ice-cold buffer containing 0.25 M sucrose and 0.02 M triethanolamine hydrochloride (pH 7.4), followed by centrifugation at 1000 g for 10 min at 4 °C. The resulting supernatant was collected and used for the determination of thiobarbituric acid-reactive substances (TBARS), lipid hydroperoxides, reduced glutathione (GSH), superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) levels. All biochemical parameters were measured using standard assay kits (Sigma-Aldrich®, India) according to the manufacturer’s instructions.
Histopathological examination of aortic tissue
Aortic tissues were collected, fixed in 10% neutral-buffered formalin, dehydrated through graded ethanol, and embedded in paraffin. Sections of 5 µm thickness were cut using a rotary microtome and stained with hematoxylin and eosin (H&E) for microscopic evaluation.
Quantitative real-time PCR (qRT-PCR) analysis
Total RNA was isolated from frozen aortic tissues using TRIzol reagent (Invitrogen, USA) according to the manufacturer’s instructions. The purity and concentration of RNA were determined spectrophotometrically by measuring absorbance at 260/280 nm. One microgram of total RNA was reverse-transcribed into cDNA using a High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, USA). qRT-PCR was performed using SYBR Green Master Mix (Applied Biosystems) in a StepOnePlus Real-Time PCR System (Applied Biosystems) under standard cycling conditions. Gene-specific primers were designed to target endothelial dysfunction-related and oxidative stress-associated transcripts, including arginase 1 (ARG1), endothelial nitric oxide synthase (eNOS), Nrf-2, heme oxygenase-1 (HO-1), NAD(P)H quinone oxidoreductase 1 (NQO1), vascular cell adhesion molecule-1 (VCAM-1), and intercellular adhesion molecule-1 (ICAM-1). β-actin was used as the internal control. Relative mRNA expression levels were calculated using the 2^−ΔΔCt method.
Table 1: Primer sequence used for the qRT-PCR analysis
| Gene | Primer sequence |
| ARG1 | F: 5′-TGGACAGACTAGGAATTGGCA-3′
R: 5′-CCAGTCCGTCAACATCAAAACT-3′ |
| eNOS | F: 5′-CTGCAGGTCTTTGACGCTCGG-3′
R: 5′-GTGGAACACAGGGGTGATGCT-3′ |
| Nrf-2 | F: 5′-GAGAGCCCAGTCTTCATTGC-3′
R: 5′-TGCTCAATGTCCTGTTGCAT-3′ |
| HO-1 | F: 5′-TTCTACCTGTTCGAGCATGTGG-3′
R: 5′-TGTTAGCATGGAGCCAGCCT-3′ |
| NQO1 | F: 5′-ATCCTGGAAGGATGGAAG-3′
R: 5′-CCTGCCTGGAAGTTTAGG-3′ |
| VCAM-1 | F: 5′-GAATTCTCCCAAATCGACATATTCCC-3′
R: 5′-CTCGAGTTATTTCTCTTGAACAGTTAATT-3′ |
| ICAM-1 | F: 5′-TCTCAGCAGACTCTTACATC-3′
R: 5′-ATATCCTGATCTTCCTCTGG-3′ |
| β-actin | F: 5′-GGCACCACACTTTCTACAAT-3′
R: 5′-AGGTCTCAAACATGATCTGG-3′ |
Statistical analysis
All data were expressed as mean ± SD. Statistical significance was determined by one-way ANOVA followed by Tukey’s post hoc test. A p-value < 0.05 was considered significant. Analyses were performed using GraphPad Prism (version 8).
Results
PMOE exhibits high polyphenol content and diverse phytochemical composition
The prepared PMOE demonstrated a substantial yield and contained a rich profile of secondary metabolites. Preliminary phytochemical screening confirmed the presence of alkaloids, flavonoids, tannins and phenolics, with intense coloration reactions indicative of abundant flavonoid and phenolic constituents. Quantitative spectrophotometric analyses revealed a high total phenolic content (195.73 ± 1.25 mg GAE/g extract), prominent flavonoid levels (24.82 ± 0.48 mg QE/g), and considerable tannin (37.89± 0.94 mg GAE/100 g DM), validating the phenolic-enriched nature of the extract.
PMOE ameliorates hyperglycemia, improves insulin levels, and reduces HbA1c in diabetic rats
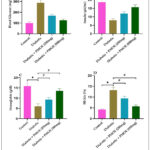
STZ and NAD administration significantly (P< 0.05) increased fasting blood glucose in diabetic control rats compared to normal controls. Oral administration of PMOE at both 250 mg/kg and 500 mg/kg markedly (P< 0.05) reduced fasting glucose levels throughout the 45-day treatment period, with the higher dose producing a more pronounced effect (Fig. 1A). Consistent with glucose regulation, diabetic rats exhibited reduced serum insulin, hemoglobin and elevated HbA1c levels, all of which were significantly corrected by PMOE treatment (Fig. 1B-D).
PMOE ameliorates hypertension
Tail-cuff plethysmography revealed a significant (P<0.05) elevation in both systolic and diastolic blood pressure in diabetic control animals compared to normal controls, indicating compromised vascular tone and endothelial dysfunction (Fig. 2A&B). At the same time, treatment with PMOE dose-dependently attenuated these elevations. In the 250 mg/kg group, both systolic and diastolic pressures were moderately reduced, while the 500 mg/kg PMOE-treated group demonstrated near-normalization of both parameters (P<0.05 vs. diabetic control), suggesting restoration of vascular homeostasis and improved endothelial function.
PMOE suppresses arginase activity and lowers circulating AGEs
Serum arginase activity was markedly (P< 0.05) elevated in diabetic controls (Fig. 2C), indicating enhanced L-arginine diversion from NO synthesis. PMOE treatment significantly reduced arginase activity in a dose-dependent manner (P< 0.05), thereby preserving substrate availability for eNOS. Similarly, AGEs, which were significantly (P< 0.05) increased in diabetic rats (Fig. 2D), were reduced following PMOE administration, suggesting attenuation of non-enzymatic protein glycation and reduced glycoxidative stress.
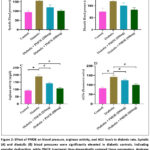
PMOE diminishes intracellular ROS levels and enhances nitric oxide bioavailability
Aortic homogenates from diabetic rats displayed elevated ROS generation (Fig. 3A), as evidenced by increased DCF fluorescence. PMOE treatment significantly (P< 0.05) reduced intracellular ROS levels, indicating potent antioxidant activity. Concurrently, serum NO concentrations were restored, and NOS enzymatic activity was significantly (P< 0.05) enhanced compared to diabetic controls (Fig. 3B&C), suggesting that PMOE improves endothelial NO bioavailability by reducing oxidative degradation and arginase competition. On the other hand, diabetic rats exhibited markedly reduced activities of AS and ASL, but PMOE treatment dose-dependently restored these enzymes, with the 500 mg/kg group approaching normal values (Fig. 3D&E).
PMOE restores antioxidant enzyme activities and lowers lipid peroxidation in aortic tissue
Diabetic rats exhibited marked oxidative stress characterized by increased TBARS and lipid hydroperoxide levels, along with significant depletion of GSH, SOD, CAT, and GPx activities (Fig. 4A-E). Treatment with PMOE resulted in significant (P< 0.05) reductions in lipid peroxidation products and a dose-dependent restoration of endogenous antioxidant defenses.
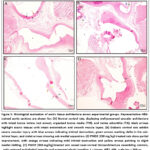
PMOE preserves aortic histological architecture and reduces structural damage
Histopathological examination of aortic tissues from the normal control group revealed well-preserved vascular architecture characterized by an intact endothelial lining, regular elastic lamellae, and uniform medial thickness without any evidence of edema, inflammatory infiltration, or structural disruption (Fig. 5A). In contrast, sections from the diabetic control group exhibited marked pathological alterations, including endothelial cell detachment, fragmentation of elastic fibers, subendothelial edema, and pronounced medial thickening (Fig. 5B). Treatment with PMOE produced a clear dose-dependent improvement in vascular morphology. Aortic tissues from rats receiving 250 mg/kg PMOE showed partial restoration of endothelial continuity, reduced medial hypertrophy, and improved elastic fiber organization compared to diabetic controls (Fig. 5C). The 500 mg/kg PMOE group demonstrated near-normal histological features with a continuous endothelial layer, preserved elastic lamellae, and minimal structural abnormalities, closely resembling those observed in the normal control group (Fig. 5D).
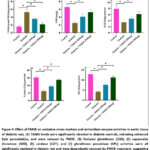
PMOE downregulates arginase 1 expression and upregulates Nrf-2-mediated antioxidant genes
qRT-PCR showed that diabetic rats had markedly (P< 0.05) elevated ARG1 and reduced eNOS expression compared to normal control group (Fig. 6A&B). PMOE dose-dependently suppressed ARG1 and restored eNOS. Antioxidant genes Nrf-2, HO-1, and NQO1 were downregulated in diabetic rats but increased (P< 0.05) with PMOE treatment (Fig. 6C-E). In addition, inflammatory markers VCAM-1 and ICAM-1 were upregulated in diabetic rats but significantly (P< 0.05) downregulated by PMOE (Fig. 6F&G), indicating improved endothelial function.
Discussion
The findings from the present investigation clearly demonstrate that chronic hyperglycemia induced by STZ and NAD administration profoundly disrupts systemic metabolic homeostasis and vascular function, consistent with the well-established role of glucotoxicity in the pathogenesis of diabetic complications. In the diabetic control group, fasting blood glucose levels remained markedly elevated throughout the study period, accompanied by a significant rise in HbA1c and a notable decline in circulating insulin concentrations. These alterations indicate both pancreatic β-cell impairment and peripheral insulin resistance, leading to persistent glucose overload and non-enzymatic protein glycation. 19 Elevated HbA1c reflects cumulative exposure of hemoglobin to glucose and is a surrogate marker for chronic hyperglycemia-driven biochemical stress. This persistent hyperglycemia sets in motion a cascade of pathogenic events including enhanced mitochondrial superoxide generation, activation of polyol and hexosamine. pathways, and the accumulation of AGEs, all of which collectively impair endothelial integrity 4,20,21 Treatment with PMOE produced a clear improvement in these indices. Animals receiving PMOE displayed a dose-dependent reduction in fasting blood glucose and HbA1c, along with a partial restoration of serum insulin levels. Such effects strongly suggest that the extract exerts dual antidiabetic actions first, by preserving pancreatic β-cell viability against oxidative insult, and second, by enhancing peripheral glucose utilization. Phenolic compounds such as quercetin, kaempferol, and chlorogenic acid known constituents of M. oleifera have been reported to improve insulin signalling through modulation of the PI3K/Akt pathway, increase GLUT4 translocation in muscle and adipose tissues, and suppress hepatic gluconeogenesis. 22 By lowering systemic glucose levels, PMOE indirectly reduces glucose auto-oxidation and the formation of reactive carbonyl intermediates, which are primary contributors to glycoxidative vascular injury. 23
The elevated blood pressure observed in diabetic control animals is consistent with endothelial dysfunction and impaired vasodilatory capacity, which are hallmark features of vascular complications in type 2 DM. 24 Chronic hyperglycemia promotes structural and functional changes in large arteries, including increased vascular stiffness, reduced elastin content, and enhanced smooth muscle tone, all contributing to systolic hypertension. 25,26 In the present study, PMOE administration significantly reduced systolic blood pressure toward near-normal levels, indicating an improvement in vascular compliance. This antihypertensive effect can be mechanistically attributed to the restoration of eNOS activity and enhanced NO bioavailability, both of which are critical for vasorelaxation. 27,28 Additionally, the suppression of oxidative stress by PMOE may prevent the rapid inactivation of NO by superoxide radicals, thereby prolonging its biological half-life and improving vascular tone.
The marked elevation of arginase activity observed in the diabetic control group provides mechanistic insight into the impaired NO signaling that characterizes diabetic endothelial dysfunction. Arginase competes with eNOS for their common substrate, L-arginine, diverting it toward urea and ornithine production rather than NO synthesis. 29,30 This substrate depletion limits NO availability, while excessive arginase activity further promotes “eNOS uncoupling,” a pathological state in which eNOS, instead of generating NO, produces superoxide radicals. The combined effects of substrate competition and eNOS uncoupling contribute to a profound reduction in vascular NO levels, which compromises vasodilatory capacity, increases vascular resistance, and exacerbates oxidative stress. 31 In this study, diabetic rats showed significantly elevated serum arginase activity along with decreased NOS activity and lower circulating NO concentrations, clearly reflecting this dysfunctional pathway. PMOE treatment significantly suppressed arginase activity in a dose-dependent manner while enhancing NOS activity and restoring NO levels. The transcriptional data reinforce these biochemical findings: ARG1 gene expression was markedly upregulated in diabetic rats but reduced toward normal levels following PMOE administration. By lowering ARG1 expression and enzymatic activity, PMOE likely increased intracellular L-arginine availability, allowing eNOS to remain in a coupled, NO-producing state and these results are in line with previous study. 32 The resulting improvement in NO bioavailability explains, at least in part, the observed blood pressure reduction and better endothelial function in PMOE-treated animals. Furthermore, phenolic compounds are known to modulate arginase activity directly through competitive inhibition and indirectly by reducing oxidative stimuli that otherwise enhance arginase expression via inflammatory transcription factors such as NF-κB. 14,33
Parallel to changes in arginase activity, diabetic rats exhibited pronounced oxidative stress, as evidenced by significantly elevated ROS generation in aortic tissues, increased lipid peroxidation products (TBARS and lipid hydroperoxides), and depletion of critical endogenous antioxidants including GSH, SOD, CAT, and GPx. Persistent hyperglycemia is known to increase ROS production through mitochondrial electron transport chain overload, activation of NADPH oxidase, and auto-oxidation of glucose. These excessive free radicals damage lipids, proteins, and DNA, 34,35 and further impair eNOS function by oxidizing its essential cofactor tetrahydrobiopterin (BH4), reinforcing the cycle of endothelial dysfunction. 4,36 Administration of PMOE markedly reduced ROS levels and lipid peroxidation while restoring GSH and the activities of SOD, CAT, and GPx. This strong antioxidant effect is likely mediated by both direct and indirect mechanisms. The direct mechanism involves the radical scavenging ability of phenolic hydroxyl groups, which neutralize reactive species before they can initiate lipid peroxidation. 37,38 The indirect mechanism is supported by the observed upregulation of Nrf-2 expression and its downstream antioxidant genes HO-1 and NQO1. Activation of the Nrf-2 pathway enhances cellular defense by promoting the transcription of phase II detoxifying enzymes and antioxidant proteins. 8,10,39 These molecular findings suggest that PMOE not only provides immediate antioxidant activity but also triggers sustained endogenous protection, which is particularly important for chronic diseases like DM where ongoing oxidative stress is a key driver of vascular damage and these findings were in line with previous study. 40
A critical observation from this study is the substantial increase in AGEs in the diabetic control group. Chronic hyperglycemia promotes nonenzymatic glycation of proteins, leading to irreversible AGE formation. These AGEs contribute to vascular pathology through two principal mechanisms: structural crosslinking of extracellular matrix proteins, which reduces arterial elasticity and increases stiffness, and activation of the receptor for AGEs (RAGE), which triggers intracellular signaling cascades that enhance oxidative stress and inflammatory gene expression. 41 The elevated AGE levels observed in diabetic rats are therefore a major factor underlying their increased vascular rigidity, heightened oxidative burden, and endothelial activation. Treatment with PMOE significantly lowered AGE accumulation in a dose-dependent manner, an effect that likely stems from its dual ability to improve glycemic control and directly scavenge reactive carbonyl intermediates that participate in glycation reactions. Moreover, phenolic compounds have been shown to inhibit the binding of AGEs to RAGE, thereby attenuating downstream signaling events that would otherwise amplify ROS production and NF-κB-dependent transcription of proinflammatory mediators. 42
This suppression of AGE-RAGE-mediated inflammatory signaling is reflected in the expression patterns of vascular adhesion molecules. 43 Diabetic rats exhibited a marked upregulation of VCAM-1 and ICAM-1 mRNA in aortic tissues. These molecules are central to the early stages of atherogenesis, as they facilitate leukocyte rolling, adhesion, and transmigration into the vascular wall, leading to chronic inflammation and endothelial injury. 44 Their increased expression in diabetic controls is consistent with sustained oxidative stress, NF-κB activation, and AGE-RAGE engagement. PMOE administration resulted in a significant reduction in VCAM-1 and ICAM-1 expression, suggesting that the extract interrupts these inflammatory pathways. By restoring Nrf-2 signaling, reducing ROS, and enhancing NO bioavailability, PMOE may prevent the activation of NF-κB and other redox-sensitive transcription factors that drive adhesion molecule gene transcription. The simultaneous upregulation of antioxidant genes such as HO-1 and NQO1 further supports the notion that PMOE shifts the vascular redox environment from a pro-oxidant, pro-inflammatory state to one favoring endothelial quiescence and structural stability. 45,46 Histopathological analysis of aortic tissue corroborates these biochemical and molecular findings. At the molecular level, the coordinated gene expression changes induced by PMOE provide a mechanistic framework for its vascular protective effects.
Despite the strengths of the current study, certain limitations should be acknowledged. The research focused primarily on biochemical, molecular, and histological endpoints, and although these parameters provide strong mechanistic evidence, additional functional assays such as vascular reactivity studies or measurements of arterial stiffness would provide further insight into the physiological relevance of the observed molecular changes. Moreover, while the extract was characterized for total phenolic content, the specific contributions of individual phenolic compounds remain to be determined. Detailed phytochemical profiling, along with fractionation and bioactivity-guided studies, could help identify the most potent constituents responsible for the observed effects. Finally, although the STZ-NAD rat model replicates several features of human type 2 DM, differences in metabolic rate, immune responses, and long-term disease progression mean that clinical validation is necessary before therapeutic recommendations can be made.
Conclusion
In conclusion, the results obtained from this study provide strong experimental evidence that PMOE protects against DM-induced endothelial dysfunction by reducing hyperglycemia, suppressing arginase activity, restoring NO bioavailability, activating the Nrf-2 antioxidant pathway, and attenuating vascular inflammation and structural damage. These findings highlight the potential of PMOE as a promising phytotherapeutic candidate for preventing or delaying diabetic vascular complications and underscore the importance of plant-derived phenolic compounds as multi-target modulators of vascular health. Further studies aimed at isolating active constituents, optimizing dosage, and evaluating long-term safety and efficacy in clinical settings are warranted to translate these preclinical findings into practical therapeutic strategies.
Acknowledgement
Authors would like to express their sincere gratitude to the management of Koneru Lakshmaiah Education Foundation (KL University), Vijayawada for providing necessary facilities to carry out this work and for their constant support.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants. The study involved animal experiments and the protocol was approved by the Institutional Animal Ethics Committee, Jeeva Life Sciences, Hyderabad, Telangana, India (Approval No: CCSEA/IAEC/JLS/21/04/24/043).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contribution
Lakshmi Sowjanya: Conceptualization, Methodology, Resources, Data Collection, Analysis, Visualization, Writing – Original Draft.
Koteswara Reddy: Conceptualization, Methodology, Supervision, Project Administration, Writing – Review & Editing.
Chandrasekharnath: Data Collection, Analysis
Uma: Data Collection, Analysis
References
- Uddandrao VVS, Parim B, Ramavat R, et al. Effect of S-allylcysteine against diabetic nephropathy via inhibition of MEK1/2-ERK1/2-RSK2 signalling pathway in streptozotocin-nicotinamide-induced diabetic rats. Arch Physiol Biochem. 2023;129(1):213-221. doi:10.1080/13813455.2020.1811731
CrossRef - Sethi S, Cheong I, Bussey CT, et al. Central regulation of the heart in type 2 diabetes mellitus. J Mol Cell Cardiol. 2026;211:43-52. doi:10.1016/j.yjmcc.2025.11.012
CrossRef - Yang DR, Wang MY, Zhang CL, Wang Y. Endothelial dysfunction in vascular complications of diabetes: a comprehensive review of mechanisms and implications. Front Endocrinol (Lausanne). 2024;15. doi:10.3389/fendo.2024.1359255
CrossRef - Brahmanaidu P, Uddandrao VVS, Sasikumar V, et al. Reversal of endothelial dysfunction in aorta of streptozotocin-nicotinamide-induced type-2 diabetic rats by S-Allylcysteine. Mol Cell Biochem. 2017;432(1-2):25-32. doi:10.1007/s11010-017-2994-0
CrossRef - Siam NH, Snigdha NN, Tabasumma N, Parvin I. Diabetes Mellitus and Cardiovascular Disease: Exploring Epidemiology, Pathophysiology, and Treatment Strategies. Rev Cardiovasc Med. 2024;25(12). doi:10.31083/j.rcm2512436
CrossRef - Heuser SK, LoBue A, Li J, et al. Downregulation of eNOS and preserved endothelial function in endothelial-specific arginase 1-deficient mice. Nitric Oxide. 2022;125-126:69-77. doi:10.1016/j.niox.2022.06.004
CrossRef - Wu J, Sun X, Jiang Z, et al. Protective role of NRF2 in macrovascular complications of diabetes. J Cell Mol Med. 2020;24(16):8903-8917. doi:10.1111/jcmm.15583
CrossRef - Uddandrao VVS, Parim B, Singaravel S, et al. Polyherbal Formulation Ameliorates Diabetic Cardiomyopathy Through Attenuation of Cardiac Inflammation and Oxidative Stress Via NF-κB/Nrf-2/HO-1 Pathway in Diabetic Rats. J Cardiovasc Pharmacol. 2022;79(1):75-86. doi:10.1097/FJC.0000000000001167
CrossRef - Jansy Isabella Rani A, Uddandrao VVS, Sangeethadevi G, et al. Biochanin A attenuates obesity cardiomyopathy in rats by inhibiting oxidative stress and inflammation through the Nrf-2 pathway. Arch Physiol Biochem. 2023;129(3):788-798. doi:10.1080/13813455.2021.1874017
CrossRef - Sathibabu Uddandrao V V., Brahmanaidu P, Nivedha PR, Vadivukkarasi S, Saravanan G. Beneficial Role of Some Natural Products to Attenuate the Diabetic Cardiomyopathy Through Nrf2 Pathway in Cell Culture and Animal Models. Cardiovasc Toxicol.Humana Press Inc. 2018;18(3):199-205. doi:10.1007/s12012-017-9430-2
CrossRef - Uddandrao VVS, Brahmanaidu P, Saravanan G. Therapeutical Perspectives of S-Allylcysteine: Effect on diabetes and other disorders in Animal Models. Cardiovasc Hematol Agents Med Chem. 2016;15(2):71-77. doi:10.2174/1871525714666160418114120
CrossRef - Kour N, Bhagat G, Singh S, et al. Polyphenols mediated attenuation of diabetes associated cardiovascular complications: A comprehensive review. J Diabetes Metab Disord. 2023;23(1):73-99. doi:10.1007/s40200-023-01326-x
CrossRef - Serreli G, Deiana M. Role of Dietary Polyphenols in the Activity and Expression of Nitric Oxide Synthases: A Review. Antioxidants. 2023;12(1):147. doi:10.3390/antiox12010147
CrossRef - Minozzo B, Fernandes D, Beltrame F. Phenolic Compounds as Arginase Inhibitors: New Insights Regarding Endothelial Dysfunction Treatment. Planta Med. 2018;84(05):277-295. doi:10.1055/s-0044-100398
CrossRef - Villegas-Vazquez EY, Gómez-Cansino R, Marcelino-Pérez G, Jiménez-López D, Quintas-Granados LI. Unveiling the Miracle Tree: Therapeutic Potential of Moringa oleifera in Chronic Disease Management and Beyond. Biomedicines. 2025;13(3):634. doi:10.3390/biomedicines13030634
CrossRef - Sampathkumar R, Balasubramanyam M, Rema M, Premanand C, Mohan V. A novel advanced glycation index and its association with diabetes and microangiopathy. Metabolism. 2005;54(8):1002-1007. doi:10.1016/j.metabol.2005.02.017
CrossRef - Ugochukwu NH, Cobourne MK. Modification of renal oxidative stress and lipid peroxidation in streptozotocin-induced diabetic rats treated with extracts from Gongronema latifolium leaves. Clin Chim Acta. 2003;336(1-2):73-81. doi:10.1016/S0009-8981(03)00325-5
CrossRef - Swamy M, Zakaria AZ, Govindasamy C, Sirajudeen KNS, Nadiger HA. Effects of acute ammonia toxicity on nitric oxide (NO), citrulline–NO cycle enzymes, arginase and related metabolites in different regions of rat brain. Neurosci Res. 2005;53(2):116-122. doi:10.1016/j.neures.2005.06.005
CrossRef - Sathibabu Uddandrao V V., Brahmanaidu P, Ravindarnaik R, Suresh P, Vadivukkarasi S, Saravanan G. Restorative potentiality of S-allylcysteine against diabetic nephropathy through attenuation of oxidative stress and inflammation in streptozotocin–nicotinamide-induced diabetic rats. Eur J Nutr. 2019;58(6):2425-2437. doi:10.1007/s00394-018-1795-x
CrossRef - Naidu PB, Sathibabu Uddandrao V V., Naik RR, et al. Effects of S-Allylcysteine on Biomarkers of the Polyol Pathway in Rats with Type 2 Diabetes. Can J Diabetes. 2016;40(5):442-448. doi:10.1016/j.jcjd.2016.03.006
CrossRef - Qambari H, Yu PK, Balaratnasingam C, Dickson J, Yu DY. Endothelial function can be modulated by acute hyperglycemia. Sci Rep. 2025;15(1):30559. doi:10.1038/s41598-025-12612-4
CrossRef - Abdelazim AM, Afifi M, Abu-Alghayth MH, Alkadri DH. Moringa oleifera: Recent Insights for Its Biochemical and Medicinal Applications. Dhull SB, ed. J Food Biochem. 2024;2024:1-21. doi:10.1155/2024/1270903
CrossRef - Chen Y, Meng Z, Li Y, Liu S, Hu P, Luo E. Advanced glycation end products and reactive oxygen species: uncovering the potential role of ferroptosis in diabetic complications. Mol Med. 2024;30(1):141. doi:10.1186/s10020-024-00905-9
CrossRef - Chen YF, Qiu Q, Wang L, et al. Quercetin Ameliorates Myocardial Injury in Diabetic Rats by Regulating Autophagy and Apoptosis through AMPK/mTOR Signaling Pathway. Am J Chin Med. 2024;52(03):841-864. doi:10.1142/S0192415X24500344
CrossRef - De la Maza-Bustindui NS, León-Álvarez M, Ponce-Acosta C, et al. Impact of cardiometabolic risk factors and its management on the reversion and progression of arterial stiffness. npj Cardiovasc Heal. 2025;2(1):36. doi:10.1038/s44325-025-00074-6
CrossRef - Hussain A. Chronic hyperglycemia and cardiovascular dysfunction: an in-depth exploration of metabolic and cellular pathways in type 2 diabetes mellitus. Cardiovasc Diabetol – Endocrinol Reports. 2025;11(1):39. doi:10.1186/s40842-025-00247-3
CrossRef - El-Bassossy HM, El-Fawal R, Fahmy A. Arginase inhibition alleviates hypertension associated with diabetes: Effect on endothelial dependent relaxation and NO production. Vascul Pharmacol. 2012;57(5-6):194-200. doi:10.1016/j.vph.2012.01.001
CrossRef - Chatturong U, Nuengchamnong N, Inchan A, et al. Vasorelaxant and Hypotensive Mechanisms of Nelumbo nucifera Seed Extract: Roles of Nitric Oxide, Calcium Channel Blockade and eNOS Interaction with Active Compounds. Pharmaceuticals. 2025;18(10):1500. doi:10.3390/ph18101500
CrossRef - Collado A, Humoud R, Kontidou E, et al. Erythrocyte-derived extracellular vesicles induce endothelial dysfunction through arginase-1 and oxidative stress in type 2 diabetes. J Clin Invest. 2025;135(10). doi:10.1172/JCI180900
CrossRef - Mazrouei S, Sharifpanah F, Caldwell RW, et al. Regulation of MAP kinase-mediated endothelial dysfunction in hyperglycemia via arginase I and eNOS dysregulation. Biochim Biophys Acta – Mol Cell Res. 2019;1866(9):1398-1411. doi:10.1016/j.bbamcr.2019.05.004
CrossRef - Costantino S, Mohammed SA, Paneni F. Endothelial dysfunction in patients with type 2 diabetes: the truth is in the blood. J Clin Invest. 2025;135(10). doi:10.1172/JCI193128
CrossRef - Cheng H, Lu T, Wang J, et al. HuangqiGuizhiWuwu Decoction Prevents Vascular Dysfunction in Diabetes via Inhibition of Endothelial Arginase 1. Front Physiol. 2020;11. doi:10.3389/fphys.2020.00201
CrossRef - Minozzo BR, de Andrade EA, Vellosa JCR, et al. Polyphenolic compounds of Euphorbia umbellata (Pax) Bruyns (Euphorbiaceae) improved endothelial dysfunction through arginase inhibition. Phyther Res. 2021;35(5):2557-2567. doi:10.1002/ptr.6986
CrossRef - Uddandrao VVS, Eraniappan S, Balakrishnan Ramajayam A, et al. Hydroxycitric acid and capsaicin combination alleviates obesity-induced testicular apoptosis, oxidative stress and inflammation. Syst Biol Reprod Med. 2024;70(1):20-37. doi:10.1080/19396368.2024.2306403
CrossRef - Tamilmani P, Sathibabu Uddandrao V V., Chandrasekaran P, et al. Linalool attenuates lipid accumulation and oxidative stress in metabolic dysfunction-associated steatotic liver disease via Sirt1/Akt/PPRA-α/AMPK and Nrf-2/HO-1 signaling pathways. Clin Res Hepatol Gastroenterol. 2023;47(10). doi:10.1016/j.clinre.2023.102231
CrossRef - Roy B. Pathophysiological Mechanisms of Diabetes-Induced Macrovascular and Microvascular Complications: The Role of Oxidative Stress. Med Sci. 2025;13(3):87. doi:10.3390/medsci13030087
CrossRef - Lin X jing, Lai Z shi yu, Luo Q, et al. Correlation between Polyphenol Contents and Antioxidant Activities in Different Echinacea Purpurea Varieties. Curr Med Sci. 2023;43(4):831-837. doi:10.1007/s11596-022-2647-8
CrossRef - Pavithra K, Sathibabu Uddandrao V V., Chandrasekaran P, et al. Phenolic fraction extracted from Kedrostis foetidissima leaves ameliorated isoproterenol-induced cardiotoxicity in rats through restoration of cardiac antioxidant status. J Food Biochem. 2020;44(11). doi:10.1111/jfbc.13450
CrossRef - Uddandrao, V. V. S. Phytoformulation with hydroxycitric acid and capsaicin protects against high-fat-diet-induced obesity cardiomyopathy by reducing cardiac lipid deposition and ameliorating inflammation and apoptosis in the heart. J Tradit Complement Med. 2024;14(2):162-172. doi:10.1016/j.jtcme.2023.08.004
CrossRef - Umar U, Ahmed S, Iftikhar A, et al. Phenolics Extracted from Jasminum sambac Mitigates Diabetic Cardiomyopathy by Modulating Oxidative Stress, Apoptotic Mediators and the Nfr-2/HO-1 Pathway in Alloxan-Induced Diabetic Rats. Molecules. 2023;28(14):5453. doi:10.3390/molecules28145453
CrossRef - Apte MM, Khattar E, Tupe RS. Mechanistic role of Syzygium cumini (L.) Skeels in glycation induced diabetic nephropathy via RAGE–NF–κB pathway and extracellular proteins modifications: A molecular approach. J Ethnopharmacol. 2024;322:117573. doi:10.1016/j.jep.2023.117573
CrossRef - Maisto M, Tenore GC. Polyphenols as a Useful Tool to Ameliorate Advanced Glycation End-product Formation: A Focus on Molecular Mechanisms of Action. Front Biosci. 2024;29(12). doi:10.31083/j.fbl2912424
CrossRef - Shen CY, Lu CH, Wu CH, et al. The Development of Maillard Reaction, and Advanced Glycation End Product (AGE)-Receptor for AGE (RAGE) Signaling Inhibitors as Novel Therapeutic Strategies for Patients with AGE-Related Diseases. Molecules. 2020;25(23):5591. doi:10.3390/molecules25235591
CrossRef - Vogel ME, Idelman G, Konaniah ES, Zucker SD. Bilirubin Prevents Atherosclerotic Lesion Formation in Low‐Density Lipoprotein Receptor‐Deficient Mice by Inhibiting Endothelial VCAM‐1 and ICAM‐1 Signaling. J Am Heart Assoc. 2017;6(4). doi:10.1161/JAHA.116.004820
CrossRef - Kotturu SK, Uddandrao VVS, Ghosh S, Parim B. Bioactive Compounds in Diabetic Cardiomyopathy: Current Approaches and Potential Diagnostic and Therapeutic Targets. Cardiovasc Hematol Agents Med Chem. 2020;19(2):118-130. doi:10.2174/1871525718666200 421114801
CrossRef - Parim B, Sathibabu Uddandrao V V., Saravanan G. Diabetic cardiomyopathy: molecular mechanisms, detrimental effects of conventional treatment, and beneficial effects of natural therapy. Heart Fail Rev.Springer New York LLC. 2019;24(2):279-299. doi:10.1007/s10741-018-9749-1
CrossRef
Accepted on: 12-03-2026
Second Review by: Dr. Ugwu Valentine
Final Approval by: Dr. Wagih Ghannam