In Vitro Anticancer Potential of Kalanchoe pinnata (Lam.) Pers.: Against PANC-1 Cells Assessed by Flow Cytometry
Department of Biochemistry, Sree Narayana Guru College, KG Chavadi, Tamil Nadu, India.
Corresponding Author E-mail:parvathypayimenon@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3507
ABSTRACT:Kalanchoe pinnata (Lam.) Pers.: a medicinal plant traditionally used for various ailments, has gained attention for its curing various infectious diseases. This plant being a succulent herb with various medicinal applications for several diseases especially cancer have shown its mechanism by several approaches. This herb contains gallic acid, caffeic acid, coumaric acid, stigmasterol, campesterol, and other elements; it is these phytochemicals that participate in the regulation of cell proliferation, regulation, oxidative stress, and apoptosis. They also have the potentiality to act as a drug agents for tumours resistance therapy. Cancer is a global health problem that is associated with the life style changes. Out of which pancreatic cancer represents the most lethal among all; despite being all the advances made in these years. This present work assessed the effect of Kalanchoe pinnata (Lam.) Pers.: extract against the human pancreatic cancer cell line PANC-1 using flow cytometry based analyses. PANC-1 cells were treated with the increasing concentrations of Kalanchoe pinnata (Lam.) Pers. :( 150-450µg/ml). Cell viability were examined using the MTT assay while the cell cycle and apoptosis were determined using the flow cytometry. The results indicated the significance in the increase of cytotoxicity; decrease in the cell viability with the change of concentration. Flow cytometric analysis were carried out for the induction of the apoptosis which demonstrated that the extracts were capable for the induction of cell cycle arrest. The Study outcomes suggests that Kalanchoe pinnata (Lam.) Pers.: exhibits potent in vitro anticancer activity against PANC-1 cells through dose-dependent induction of apoptosis.
KEYWORDS:Apoptosis Cytotoxicity; Flow cytometry; Kalanchoe pinnata (Lam.) Pers.; Pancreatic cancer; PANC-1 cells
Introduction
Long ago, medicinal plants have been regarded as an important sources of bioactive compounds with promising anticancer properties. Phytochemicals such as flavonoids, alkaloids, terpenoids, and phenolic compounds have been shown to inhibit cancer cell proliferation, trigger apoptosis, and modulate key signalling pathways involved in tumors development. Approximately 125 perennial succulent species belong to the genus Kalanchoe most of which originate from Madagascar. 1 The genus is taxonomically intricate, with an inconsistent number of recognized species and multiple synonyms. The diversity within the genus, along with its widespread distribution and adaptability, has contributed to its ecological and pharmacological significance, making it an important focus for research into bioactive compounds and medicinal properties.2 Various Kalanchoe species have also been widely introduced by humans, as their leaves are traditionally used to treat cancer .3, 4 Research studies shows that some of them can exert cytotoxicity against wide range of tumours cell lines, such as those originating from the cervix, breast, lung, liver, colon, and blood. 5,6,7Kalanchoe pinnata (Lam.) Pers.: Commonly called as “Mother of Thousands”, this plant produces new plants along the edges of the leaves that can be removed and propagated separately in pots or bare soil8.Growing 1-1.5 tall this perennial stores water in its thick, scalloped green leaves and produces pendulous, bell shaped flowers on tall hollow stems.9 This plant is commonly found in the plains and tropical and temperate regions of Africa, Australia, and the Americas. It is also widely used in traditional Asian medicine for its ethnomedicinal properties.10 The leaves and stems have a bitter taste and, owing to their astringent properties, are effective in treating diarrhoea, flatulence, and vomiting.11These leaves are used in different countries for several diseases such as hypertension, kidney and urinary diseases, cough and asthma and for the treatment of the burns, ulcers, diarrhoea and insect bites.
Cancer is a multifactorial pathological condition involving dysregulated cell cycle control, apoptosis resistance, sustained neovascularization, and invasive and metastatic behaviour. These abnormalities arise from cumulative genetic mutations and epigenetic dysregulation that disrupt critical regulatory networks governing cell cycle progression, DNA repair, apoptosis, and signal transduction that can be quantitatively assessed by flow cytometric analysis. The development of cancer can be by: exogenous factors like usage of tobacco, chemical carcinogens, radiations and oncogenic pathogens Endogenous factors that include the hormonal imbalances, mutations that are inherited, dysregulation in immune system and the mutations arising during the normal metabolic process. These factors jointly contribute for the initiation or the promotion of the cancer. There are types of cancers that can be eliminated by avoiding the tobacco consumption and the other factors that regulate these processes. Other type of malignancies have been to detected before the cells become cancerous which is suitable time for the treatment; that includes chemotherapy, radiation or immunotherapy.12
Pancreatic Cancer is an aggressive malignancy that’s predominantly composed of pancreatic duct adenocarcinoma which constitutes the major of the mortality rate in most of the cases. An important aspect of the pancreatic cancer is the poor prognosis to the therapies. Many plants and herbal medicines have been used for the treatment of the pancreatic cancer in the centuries ago. Traditional Chinese medicine and other plant medicines from all over the world have gained the importance for their role in cancer therapy. Over the decades the experimental investigation with a lesser extent to the clinical trials have expanded in higher rate, highlighting the importance of the plant-derived compounds.13 Kalanchoe pinnata (Lam.) Pers., a member of the Crassulaceae family, have been traditionally used for the treatment of the inflammation, infectious wounds, and tumours .Phytochemical analysis of the Kalanchoe pinnata (Lam.) Pers revealed that has the presence of bioactive compounds such as flavonoids, bufadienolides, alkaloids, and phenolic compounds, all contributing in a way to anti-oxidant, anti-inflammatory,anti-cancer which makes this plant suitable for the investigation of pancreatic cancer. In this study, we have utilized the plant with PANC-1 cells, a human pancreatic ductal carcinoma cell line that is extensively used for the in-vitro model of pancreatic research to show their cell viability and cytotoxicity and the incorporation of the flow cytometry technique for the assessment arrest of cell cycle, apoptosis and changes in the cell viability.
Materials and Methods
Collection of Plant and Identification
Kalanchoe pinnata (Lam.) Pers was collected from the botanical garden of Centre for Bioscience and Nano science Research (CBNR) Echanari, Coimbatore and were authenticated by Botanical Survey of India at Coimbatore.(BSI/SRC/5/23/2023/Tech-475)
Preparation of the Plant Extract
The leaves of Kalanchoe pinnata (Lam.) Pers were cleaned with distilled water, made into fine powder the washed leaves were shade dried at room temperature. The leaves were suspended in the ethanol for several days, filter and evaporate the solvent to get a crude extract. Dissolved a known amount of the crude extract in a suitable volume of DMSO to create stock solution.
In Vitro Assesment of Anti-Cancer Activity Using MTT Assay
The anticancer activity of sample is determined by the Pancreas cancer cell line (PAN-C1 cell lines) . Cytotoxicity activity of the sample on different concentrations (150, 250, 350 and 450 μg/mL) of cancer cell line was evaluated in-vitro. It is based on the formation of the tetrazolium salt 3-[4, 5-dimethylthiazolyl-2]-2, 5-diphenyl tetrazolium bromide (MTT), that’s converted to blue formazan an insoluble product by mitochondrial enzymes in viable cells.
The cell lines were cultured in 12-well microtitre plates until a confluent monolayer was obtained. The test samples were then applied directly to the established cell monolayer. Prior to cell seeding, the specimens were pre-wetted in 70% aqueous ethanol for 48 h, rinsed twice with ultrapure water, and incubated in 1 mL of DMEM in 24-well plates for 2 h at 37 °C. Subsequently, the specimens were seeded with cells at a density of 10,000 cells per well following standard cell-culture protocols. The plates were maintained at 37 °C in a humidified atmosphere containing 5% CO₂ for 15 days.
At each designated time point, samples were removed from the 24-well plates and transferred to fresh plates for the MTT assay. The MTT solution was prepared by dissolving the reagent in phosphate-buffered saline to a final concentration of 1 mg/mL. Following 1 h of incubation, the resulting purple formazan crystals were solubilized using sodium dodecyl sulfate (SDS) prepared at 20% (w/v) in a 1:1 mixture of water and dimethylformamide (DMF). Subsequently, 1 mL of MTT medium (0.0005 mg/mL) was added to each well and the plates were incubated for 3 h, rinsed, and the dye was desorbed using 100 µL of 70% isopropanol. After agitation at 400 rpm for 40 min, the colored solution was transferred to a 96-well plate and the absorbance was measured at 550 nm. Cell viability and proliferation were calculated, and morphological changes were examined using inverted microscopy. Images of control cells (doxorubicin, 100 µg/mL) and treated samples were recorded and presented separately.
Cell Cycle Analysis by Flow Cytometry
Materials and Methods
Sample Extract, PAN-C1 cell line, Fetal Bovine Serum (FBS), DMEM medium and antibiotic solution (penicillin and streptomycin). Ribonuclease-I (stock 100mg/ml), FITC-annexin V and Propidium iodide (100µg/ml) and 1X PBS. 6 well tissue culture plate and wash beaker (Tarson) and 70% ethanol.
Cell Culture
PAN-C1 cell line were cultured in liquid medium (DMEM) supplemented 10% Fetal Bovine Serum (FBS), 100ug/ml penicillin and 100µg/ml streptomycin, and maintained under an atmosphere of 5% CO2 at 37oC
Cell Cycle Analysis by Flow Cytometry
The sample extract was evaluated for cell cycle analysis in PAN-C1 cells using the propidium iodide (PI) staining method. Briefly, cultured PAN-C1 cells were harvested by trypsinization, pooled into a 15 mL centrifuge tube, and seeded at a density of 2 × 10⁶ cells/mL in 6-well tissue culture plates containing DMEM supplemented with 10% FBS and 1% antibiotic solution. The cells were incubated for 24–48 h at 37 °C in a humidified atmosphere with 5% CO₂.
After incubation, the cells were washed with sterile PBS and treated with the extract at concentrations of 250 µg/mL and 450 µg/mL in serum-free DMEM, followed by further incubation for 24 h under the same conditions. The cells were then harvested by trypsinization and washed with PBS by centrifugation at 1500 rpm for 5 min. The resulting cell pellet was fixed by the dropwise addition of cold 70% ethanol and incubated for 30 min at 4 °C.
Following fixation, the cells were washed twice with sterile PBS by centrifugation at 1500 rpm for 5 min, and the supernatant was discarded. The final cell pellet was resuspended in binding buffer containing RNase (100 µg/mL), FITC-annexin V, and propidium iodide (PI, 100 µg/mL) and incubated for 15 min in the dark. The stained cells were immediately analyzed by flow cytometry using a FACS Canto II flow cytometer (BD Biosciences).
Results
In Vitro Assesment of Anti-Cancer Activity Using MTT Assay
PAN-C1 cancer cell lines were exposed to increasing concentrations (150–450 µg/ml) of sample extract to evaluate their response for the cytotoxicity in untreated and positive control groups. Analysis of the cytotoxicity assay indicated a significant, concentration-dependent increase in cytotoxic effects on PAN-C1 cell lines which is evident in the Table 1 and Fig 1. As the concentration of the sample was increased from 150 µg/ml to 400 µg/ml, there is a significant increase in the cytotoxicity while the percent of cell viability shows a decline from dropping from 72.6 ±0.75% to a minimum of 35.6± 0.75.The untreated group of cells were found to be in normal fusiform shape (Fig. 1A); whereas, the cells treated with Sample produced many white circular dead cells under microscope (Fig. 1B, 1C, 1D to 1E) for their respective concentrations (150µg/ml, 250µg/ml, 350µg/ml and 450µg/ml). Substantially the positive control (Doxorubicin) when compared was observed to almost similar cytotoxicity percentage (66.9 ± 0.57 % – Fig. 1F) was found evident for the higher concentration (450µg/ml) of Sample Extract (64.6 ± 0.75 % Fig. 1E).
Table 1: Cytotoxicity and Cell viability assay of Sample Extract in PAN-C1 cell lines
| Type of cells | PAN-C1 cell lines – Cytotoxicity Assay | Inference
|
||
| Concentration (µg/ml) | Cytotoxicity (%) | Cell viability (%) | ||
| Cancer
(A) |
Untreated | 0 | > 99 | No Cytotoxicity |
| Cancer
(B) |
150 | 27.6 ± 0.75 | 72.6 ± 0.75 | Cytotoxicity |
| Cancer
(C) |
250 | 32.3 ± 1.05 | 67.9 ± 0.57 | |
| Cancer
(D) |
350 | 46.9 ± 0.57 | 53.3 ± 1.05 | |
| Cancer
(E) |
450 | 64.6 ± 0.75 | 35.6 ± 0.75 | |
| Control
(F) |
100 | 66.9 ± 0.57 | 33.3 ± 1.05 | |
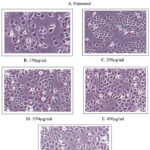 |
Figure 1: Microscopic images of PAN-C1 cell lines
|
Cell Cycle Analysis by Flow Cytometry
Flow cytometric analysis (Fig 2,3, 4 and 5) revealed that treatment of PAN-C1 cells with the extracts induced a concentration-dependent apoptotic response. The differences in viable, early apoptotic, late apoptotic, and necrotic cell populations between untreated controls and extract-treated cells are summarized in Table 2 which is determined by Annexin V and Propidium Iodide (PI) staining.
Table 2: Cell cycle analysis by flow cytometry
|
S. No. |
Stages of apoptosis |
Cell cycle analysis by flow cytometry (%) | ||||
| PAN-C1 cells
Untreated (Control) |
PAN-C1 cells
Treated with extracts (150µg/ml) |
PAN-C1 cells
Treated with extracts (250µg/ml) |
PAN-C1 cells
Treated with extracts (350µg/ml) |
PAN-C1 cells
Treated with extracts (450µg/ml) |
||
| 1 | Viable cells | 99 | 81.0 | 70.5 | 51.5 | 19.5 |
| 2 | Necrosis | 0.5 | 1.0 | 1.5 | 1.5 | 1.0 |
| 3 | Early apoptosis | 0.5 | 10.5 | 18.5 | 26.5 | 3.0 |
| 4 | Late apoptosis | 0 | 7.5 | 10.5 | 20.5 | 76.5 |
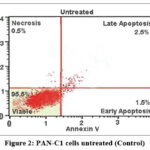 |
Figure 2: PAN-C1 cells untreated (Control)
|
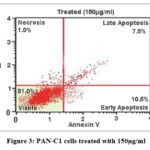 |
Figure 3: PAN-C1 cells treated with 150µg/ml
|
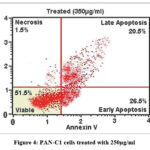 |
Figure 4: PAN-C1 cells treated with 250µg/ml
|
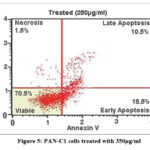 |
Figure 5: PAN-C1 cells treated with 350µg/ml
|
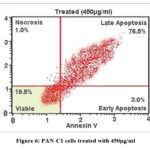 |
Figure 6: PAN-C1 cells treated with 450µg/ml
|
Discussion
The results demonstrate a clear dose-dependent cytotoxic response in PAN-C1 cancer cells, with increasing concentrations enhancing growth inhibition and promoting cell death. Lower concentration (150–250 µg/ml) of the extracts showed a partial growth inhibition with moderate cytotoxicity. While the high concentration (350–450 µg/ml)) was observed to a greater reduction in cell viability suggesting for the mechanism of apoptosis. While comparing the concentration of 450 µg/ml compared with positive control Doxorubicin highlights the efficiency of the extract for the potential anti-cancer activity. From these data the IC50 is estimated approximately to be 368 µg/ml, indicating a moderate cytotoxicity. This is further studied by the assay of flow cytometry.
In Flow cytometry The untreated control showed 99% viability with negligible levels of necrosis (0.5%) and apoptosis (0.5%), confirming the baseline health and structural integrity of the PAN-C1 cell population. At lower concentration of 150 µg/ml-350 µg/ml there is decrease of the cell viability with increase in the early apoptosis (10.5%, 18.5%, and 26.5%) and late apoptosis (7.5%, 10.5%, 20.5%) indicating the initiation and stepwise progression of apoptosis. At higher dose of 450 µg/ml (Fig 6) at the stage of the Early Apoptosis drops to 3.0% while late apoptosis surged to 76.5%, meaning that the extract pushing for the apoptotic pathway very rapidly. At all the concentrations the necrotic population remain low <1.5 % that is good result suggesting that the cells are dead by a controlled mechanism of apoptosis. The LC₅₀ of the extract against PAN-C1 cells was calculated to be approximately 368 µg/ml, indicating moderate cytotoxic activity. With the cytotoxicity and apoptosis data suggest that the extract inhibits pancreatic cancer cell growth through a dose-dependent induction of apoptosis and potential cell cycle arrest.
Conclusion
The present study demonstrates that Kalanchoe pinnata (Lam.) Pers. exhibits significant in vitro anticancer potential against PANC-1 pancreatic cancer cells. Untreated cells maintained over 99% viability, indicating the absence of inherent cytotoxic effects under control conditions. In contrast, the treated cells showed a progressive reduction in cell viability with the increasing concentrations. On treatment, it was seen a gradual increase of the cytotoxicity with the increase in the concentration rising from 27.6 ± 0.75% at 150 µg/ml to 64.6 ± 0.75% at 450 µg/ml, accompanied by a corresponding decrease in cell viability. Similarly increase for the apoptotic population was observed Flow cytometric analysis employing Annexin V/PI staining confirmed that the extract significantly inhibits cancer cell proliferation by inducing apoptosis in a potent and concentration-dependent manner. It was mainly observed in the lower concentrations that triggers a transition to early apoptosis while of the higher concentrations drive a majority of the cell population to the later stage of the programmed cell death. Also the Necrotic cell populations remained negligible across all treatment concentrations, indicating that cell death was predominantly driven by apoptotic mechanisms rather than nonspecific necrosis.
Overall these findings likely attributes to the rich phytochemical profile such as flavonoids, bufadienolides that contributes to the cell cycle arrest and oxidative stress. Accordingly, Kalanchoe pinnata (Lam.) Pers. emerges as a promising candidate for further pharmacological development, with in vivo studies required to substantiate its therapeutic efficacy and safety as a targeted treatment for pancreatic cancer.
Acknowledgement
The author would like express gratitude to Mr.Illayaraja and his laboratory staff for providing the cell lines and providing the guidance during the experimental research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Parvathy Menon PR: Conceptualization, Methodology, Writing;
Nirmala Devi N: Supervision, Project Administration.
References
- Descoings B. Le genre Kalanchoe (Crassulaceae): structure et définition. Journal de Botanique de la Société Botanique de France. 2006; 33:3–28.
CrossRef - Herrando-Moraira S, Vitales D, Nualart N, Gómez-Bellver C, Ibáñez N, Massó S, et al. Global distribution patterns and niche modelling of the invasive Kalanchoe × houghtonii (Crassulaceae). Scientific Reports. 2020; 10(1):3143.
CrossRef - Assis de Andrade E, Machinski I, Terso Ventura AC, Barr SA, Pereira AV, Beltrame FL, et al. A review of the popular uses, anatomical, chemical, and biological aspects of Kalanchoe (Crassulaceae): A genus of plants known as Miracle leaf. Molecules. 2023; 28(14):5574.
CrossRef - Hsieh YJ, Huang HS, Leu YL, Peng KC, Chang CJ, Chang MY. Anticancer activity of Kalanchoe tubiflora extract against human lung cancer cells in vitro and in vivo. Environmental Toxicology. 2016; 31(11):1663–1673.
CrossRef - Kuete V, Fokou FW, Karaosmanoğlu O, Beng VP, Sivas H. Cytotoxicity of the methanol extracts of Elephantopus mollis, Kalanchoe crenata and four other Cameroonian medicinal plants towards human carcinoma cells. BMC Complementary and Alternative Medicine. 2017; 17(1):280.
CrossRef - Lai ZR, Ho YL, Huang SC, Huang TH, Lai SC, Tsai JC, et al. Antioxidant, anti-inflammatory and antiproliferative activities of Kalanchoe gracilis (L.) DC stem. The American Journal of Chinese Medicine. 2011; 39(6):1275–1290.
CrossRef - Mahata S, Maru S, Shukla S, Pandey A, Mugesh G, Das BC, et al. Anticancer property of Bryophyllum pinnata (Lam.) Oken leaf on human cervical cancer cells. BMC Complementary and Alternative Medicine. 2012; 12:15.
CrossRef - Kaur N, Bains R, Niazi JA. Review on Bryophyllum pinnatum: A medicinal herb. Journal of Medical and Pharmaceutical Innovation. 2014; 1:13–19.
- Okwu DE, Nnamdi FU. Two novel flavonoids from Bryophyllum pinnatum and their antimicrobial activity. Journal of Chemical and Pharmaceutical Research. 2011; 3:1–10.
- Afzal M, Kazmi I, Khan R, Singh R, Chauhan M, Bisht T, Anwar F. Bryophyllum pinnatum: A review. International Journal of Research in Biological Sciences. 2012; 2:143–149.
- Kamboj A, Saluja AK. Bryophyllum pinnatum (Lam.) Kurz: Phytochemical and pharmacological profile: A review. Pharmacologyonline. 2009; 3:364–374.
- American Cancer Society. Global cancer facts and figures. American Cancer Society. 2007.
- Saif MW. Is there a role for herbal medicine in the treatment of pancreatic cancer Journal of the Pancreas.2008; 9(4):403–407.
Accepted on: 06-03-2026
Second Review by: Dr. Nafees Ahamad
Final Approval by: Dr. Wagih Ghannam









