Impact of Various Receptors: GPCR, PACAP, CGRP, Purinergic Receptors and Phosphodiesterase in Migraine.
Department of Pharmacology, SRM College of Pharmacy, Faculty of Medicine and Health sciences, SRM Institute Of Science and Technology, Kattankulathur, India.
Corresponding Author E-mail: gowrik@srmist.edu.in
DOI : http://dx.doi.org/10.13005/bbra/3452
ABSTRACT:The study of migraine is a neurological condition that may cause headaches, followed by vomiting and sensitivity reactions of light and sound. In this review, we present a comprehensive and systematic analysis of current pathophysiology development strategies to treat migraine, highlighting their mechanisms of action, such as triptans, CGRP antagonists, anti-inflammatory drugs, and antidepressants. In addition, this review explores the pathophysiological mechanisms responsible for monogenic and polygenic migraine disorders, showing the involvement of the trigeminovascular pathway. Migraine is generally categorised into three important types: Migraine with Aura, Migraine without Aura and Hemiplegic migraine. A Migraine is divided into four phases: premonitory phase, aura phase, headache phase, and postdromal phase. Further, we have shown that GPCR, cAMP, CGRP, and PACAP signalling pathways play crucial roles in both physiological and pathophysiological processes. In migraine, cAMP causes inflammation and vasodilation. Increased cAMP levels activate receptors like CGRP and PACAP that trigger migraines. These receptors interact with G proteins to increase cAMP inside the cells. To treat migraine, we used purinergic receptors, which help in lowering the cAMP levels to reduce migraine symptoms. Additionally ,this review highlights, the phosphodiesterases (PDE) activators for migraine treatment, which help in reducing the accumulation of intracellular cAMP levels. Activation of cAMP-selective phosphodiesterases (PDE3 and PDE5) is used in the treatment of migraine. PDE4 acts as a modulator that, on inhibition, leads to elevated cAMP levels and releases the inflammatory neuropeptides like CGRP, which play a major role in migraine by promoting sensitisation of pain pathways. On the other hand, PDE5 is an enzyme that breaks down cGMP levels, which influence the vasodilation of cerebral blood vessels. Excessive vasodilation contributes to migraine headaches by activating pain-sensitive structures in the brain (like the trigeminovascular system). This review highlights the basic pathophysiology of migraine, the effects of inflammatory mediators and their mechanisms.1,2
KEYWORDS:cAMP; CGRP; Gepants; GPCR; Monoclonal antibodies; PACAP; PDE4; PDE5
Introduction
Migraine is a multicausal disease caused by the interplay of genetic predisposition with various environmental and physiological provocative factors. Common risk factors/triggers include female sex and hormonal changes (puberty, menstrual cycle), family history, stress, sleep disturbance, certain foods and alcohol, sensory stimuli-lights/smells/noise-and co-morbid medical conditions-anxiety, depression, obesity. These all decrease the threshold for an attack but do not explain its biology in full. Historically debated theories converged toward a neurovascular disorder with a main neuronal origin: abnormal cortical or brainstem excitability (genetically influenced) → CSD in some patients → activation of meningeal nociceptors and trigeminal pathways → peripheral and central neurogenic inflammation and sensitisation. Recent work emphasises the role of specific molecular mediators-especially CGRP immune/inflammatory signalling, as modulators rather than simple “blood-vessel” problems. It is estimated that ~14–15% of the global population has migraine, or approximately 1.1–1.2 billion people affected in recent GBD analyses. Migraine is a leading cause of years lived with disability, mainly among women. (GBD / epidemiology reviews). In India, recent national and regional reviews/scoping studies are reporting higher prevalence observed in India, with some syntheses reporting figures around ~20–25% in selected populations/studies, though estimates are varied by study methodology and region. One scoping review from 2025 reported a pooled prevalence of 25% in India. Interpret with caution – heterogeneity, rural/urban differences, and case-finding methods. This means that the pattern repeated at the C-terminal region can serve as an epitope for several antibodies.2
Migraine result from abnormal activation of the trigeminovascular system, whereby hyperexcitable areas of the brain initiate cortical spreading depression (CSD), especially during aura. This wave of neuronal disturbance activates trigeminal nerve fibres supplying the meninges, promoting the release of neuropeptides such as CGRP, substance P, and neurokinin A, which produce vasodilation and neurogenic inflammation in cranial blood vessels. These processes powerfully activate pain signals to the brainstem and thalamus, giving rise to central sensitisation, amplifying sensitivity to light, sound, and touch. Thus, migraine headache reflects combined neuronal dysfunction and vascular–inflammatory responses in the head. Recent advances in migraine biology highlight that its pathogenesis extends beyond CGRP, with mediators such as PACAP and diverse cytokine networks emerging as key players. These molecules modulate neuronal excitability, vascular tone, and immune signalling, reinforcing the concept that migraine is not merely a vascular disorder but an immune–neurovascular interface. By linking hyperexcitable cortical and trigeminal pathways with inflammatory cascades, this perspective opens new avenues for targeted therapies that address both neuronal dysfunction and immune driven sensitisation.3
The novelty of my article is the effects of the G protein-coupled receptors, PACAP and CGRP, in the context of migraine, is the independent intracellular signalling pathway and the potential for therapeutic targeting. Preclinical studies have shown that CGRP and PACAP induce similar migraine like symptoms but they do so by various other and different mechanisms.
Treatment, Medication and Mechanism of Drugs in Migraine
Triptans are the first-line migraine-specific drugs and are used in migraine treatment. These drugs serve as agonists for serotonin 5-HT1B and 5-HT1D receptors on blood vessels. Triptans activate the three types of serotonin receptors. The first one is 5HT1B controls the blood vessels in your brain, and 5HT1D receptors release neurotransmitters and affect the parts of the brain affected by the trigeminal system, which plays a role in how you feel pain. Lastly, 5HT1F also releases neurotransmitters, which limit pain signalling. Triptan binds with the 5HT1D receptor in the trigeminal nerve ending, which releases neuropeptide, and further triptan binds with the receptor 5HT1B, which causes vasoconstriction in dural blood vessels.3,4
Topiramate is an antiepileptic drug that is used as a preventive treatment for migraine. The mechanism starts with topiramate blocking the voltage-gated sodium channels on neurons, which decreases neuronal excitability. This enhances the activity of (GABA-A) gamma-aminobutyric acid receptors, causing an increase in inhibitory tone. Further inhibition of AMPA/kainate glutamate receptors decreases excitation, and carbonic anhydrase inhibition causes cortical spreading depression (CSD), which prevents migraine initiation.5
Pathophysiology
Normal pathophysiology
Studies have shown that Single Gene Polymorphism can result in monogenic migraine, of which about 30% to 60% of the cases reported globally are due to genetic reasons. Currently, studies are being done to understand the relationship between migraine and the monogenic or polygenic reasons that are responsible for the condition.6
Trigeminovascular Pathway
The impulses flow through the nociceptors from the cranial blood vessels to the thalamic and cortical regions of the brain using the trigeminal neural pathway. This impulse transduction is supported by the endogenous neuropeptides like CGRP, Substance-P, PACAP, and also by inflammatory mediators like NO (Nitric Oxide). All these are responsible for the meningeal inflammation.7
Migraine can be broadly divided into the following types
Migraine with Aura
The mechanism of migraine can be caused due CSD, this depression is termed as aura, and it propagates across the brain. This induces the state of hyperpolarisation and cell excitation, which induces the partial release of inflammatory mediators and CGRP in the meninges.8
Migraine without Aura
This is the most widespread type of migraine observed in 60% to upto 70% of the reported cases of migraine. The typical symptoms of this type of migraine include the pulsating headache experienced across only a single side of the head, caused due to factors such as physical exertion, photophobia and phonophobia.8
Hemiplegic Migraine
This is the migraine with the highest intensity but the lowest number of reported cases. This migraine subtype causes partial paralysis, which can last up to several days and starts during or before the migraine attacks. The symptoms of hemiplegic migraine include prickling sensation, vertigo, and problems in phonation and swallowing.9
 |
Figure 1: The different possible causes or triggers for migraine (this has very low evidence). |
Phases of migraine
Premonitory Phase
The onset of this phase is 24-72 hours prior to the migraine attack, the symptoms include irritability, neck stiffness and heightened sensitivity to sound. The symptoms are present across all the phases of migraine, and an increased blood flow is observed in the hypothalamus.10
Aura phase
This is only seen in less than half of the patients and is characterised by the presence of the CSD. It has been both rejected and accepted as a significant factor that leads to the initiation of the aura phase of migraine.10
Headache phase
The headache phase of migraine can be identified by the one-sided throbbing pain of variable intensity, due to the stimulation of the trigeminovascular system through the initial activation of the thalamus and hypothalamus. The nerve fibres from the neck region and the nerve fibres of the trigeminal neuron synapses(trigeminal cervical complex) at the same ganglion are considered responsible for the upper neck pain.10,11
Postdormal phase
Patients in this phase experience symptoms similar to the aura phase of migraine, which can be explained by sustained brainstem and diencephalon activity eliciting pain stimuli. However, this is the least studied phase of migraine.10,11
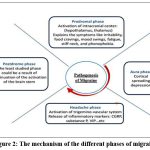 |
Figure 2: The mechanism of the different phases of migraine. |
GPCR
GPCR is among the extensively studied classes of membrane-bound receptors, serving vital functions in various physiological and pathological processes. These receptors, also known as 7-transmembrane (7TM) receptors, contain seven transmembrane helices and are activated by a vast variety of extracellular ligands. There are several varieties of GPCR proteins, such as Go, Gi, and Gq, but Gq are predominantly used in migraine pathways. The standard GPCR signalling pathway involves the interaction of ligand-activated GPCRs with intracellular G proteins, which transduce and amplify GPCR signals via secondary messengers, leading to different cellular effects. Adenylyl cyclase is an enzyme that facilitates the transformation of ATP to cAMP.12
cAMP is a secondary messenger molecule that activates protein kinase A. It is a serine/threonine kinase that serves an essential function in various cellular processes. PKA is activated when cAMP binds to its regulatory subunits. The active catalytic subunits of PKA then add phosphate groups and activate various downstream targets, which include transcription factors, ion channels, and other enzymes.13
The mechanism results in diverse cellular responses that include changes in gene expression, cellular metabolism, and electrical activity. The activation of PKA by cAMP has been implicated in the transmission of pain signals and the development of inflammation. Therefore, modulating the GPCR-PKA pathway may be a potential strategy for the development of new migraine therapies.14
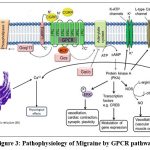 |
Figure 3: Pathophysiology of Migraine by GPCR pathway. |
Activation of GPCR Receptors
GPCRs can also be activated through novel modes, which deviate from the traditional ‘GPCR-G protein’ activation mechanism. One such mechanism is ‘biased activation’. This mode of activation is mediated by the multifunctional adaptor β-arrestins, which can trigger concurrent G protein-independent signalling pathways. Another novel mode of GPCR activation is ‘intracellular activation’. This mode of activation was discovered when it was found that some GPCRs can be activated inside cells, rather than solely on the cell surface. This mode of activation highlights the complexity and diversity of GPCR activity.15
In addition to biased activation and intracellular activation, GPCRs can also be activated through ‘dimerization activation.’ This mode of activation refers to the ability of GPCRs to form dimers, which can assume different states and produce different signals and functions. GPCRs can also be activated through ‘transactivation’ of other receptors. This mode of activation refers to the ability of ligands that bind to GPCRs to activate other classes of receptors, such as tyrosine kinase receptors (RTKs). This form of activation broadens the range of GPCR signalling and functions and highlights the complexity and diversity of GPCR signalling pathways.16
The CGRP receptor is the most studied GPCR in relation to migraine. CGRP vasodilator released from trigeminal nerve terminals during migraine attacks, leading to activation of the CGRP receptor and the transmission of pain signals and dilation of blood vessels. The effectiveness of CGRP receptor antagonists in alleviating migraine pain underscores the significance of this receptor in migraine pathophysiology. Alongside the CGRP receptor, the serotonin receptor has also been implicated in migraine.17
Serotonin is a chemical messenger that has an essential role in pain modulation and mood control, and its receptor is a GPCR that is activated by serotonin, leading to the transmission of pain signals and the regulation of mood. The efficacy of serotonin receptor agonists, such as triptans, in reducing migraine pain highlights the importance of this receptor in migraine treatment. Furthermore, other GPCRs, including the adrenergic receptor and the dopamine receptor, have also been implicated in migraine. The adrenergic receptor, which is activated by adrenaline, has been found to contribute to migraine pain modulation, while the dopamine receptor, which is activated by dopamine, has been implicated in mood regulation and motivation in migraine. GPCRs are appealing targets for drug design owing to their participation in diverse physiological processes, including pain processing, inflammation, and vasodilation.18
cAMP
Cyclic Adenosine Monophosphate is a secondary messenger molecule that regulates different cellular processes, signal transduction, and cellular metabolism. In the context of migraine, cAMP causes the release of various pain signals, inflammation, and vasodilation. The increased cAMP levels can also stimulate migraine attacks in individuals. Phosphodiesterase inhibitors are used, which increase cAMP levels by inhibiting its breakdown, and can initiate migraine-like symptoms in patients. Furthermore, the efficacy of triptans, a class of migraine medications, is thought to be mediated, at least in part, by their ability to decrease cAMP.19
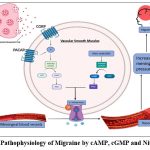 |
Figure 4: Pathophysiology of Migraine by cAMP, cGMP and Nitric oxide. |
The cAMP pathway is activated by various receptors, like CGRP and PACAP. These types of receptors are coupled with G-proteins that stimulate adenylate cyclase, leading to an elevation in cAMP production. CGRP and the PACAP are both known inducers of migraine, and their ability to increase cAMP levels may contribute to their migraine-inducing effects. cAMP roles in pain release have also been implicated in the formation of inflammation and vasodilation. These mediators can cause inflammation and pain in migraine. Inhibitors of adenylate cyclase that decrease cAMP production, and activators of phosphodiesterases, which increase cAMP breakdown, may be effective in reducing migraine frequency and severity.20
Three ways to target the cAMP pathways:
Administration of agonists that couple to Gi receptors;
Inhibiting the adenylate cyclase causes a reduced level in cAMP; and
Breakdown of cAMP by PDE stimulation.20,21
Purinergic Receptor
Purinergic receptors, particularly P2Y12, P2Y13, and P2Y14 receptors, are potential targets for migraine treatment due to their ability to decrease intracellular cAMP levels. The receptors couple through GPCR(Gi) proteins, which induce adenylate cyclase and reduce cAMP production. Agonists of these receptors may be effective anti-migraine agents. Preliminary studies have shown promising results for P2Y13 receptor agonists. However, the potential anti-migraine effects of P2Y13 receptor agonists. In contrast, P2Y12 receptors are unlikely to be suitable candidates due to their role in sensitising platelet aggregation. Activation of P2Y12 receptors can increase the risk of thrombotic events, making them an unsuitable target for treatment.21
CGRP
Calcitonin Gene Related Petide is an effective vasodilator, activation role in the trigeminovascular system, and migraine. It is plentiful in the CNS and has a wide distribution throughout all. It is secreted through trigeminal nerves for responding to the local cerebral vasoconstriction, causing dilation and maintaining blood circulation in cerebral region. CGRP contains 37 Amino Acid of related molecules. including adrenomedullin and amylin. These peptides share structural homology and similarities in biological activities. CGRP is primarily present in nerves, while calcitonin is expressed in the thyroid. Studies have demonstrated that CGRP activates Adenylyl Cyclase of smooth muscle cells.22 CGRP is produced in neurons through specific mRNA splicing from the calcitonin gene, forming two types: α-CGRP (linked to brain and migraine) and β-CGRP (mainly in the gut). It helps regulate cerebral blood flow by triggering vasodilation during local constriction via the trigeminovascular reflex. This protective mechanism makes CGRP a key player in migraine development and treatment strategies 22.CGRP contributes to migraines by dilating cerebral and dural vessels, triggers inflammatory mediators released by mast cells, and transmitting pain signals from brain vessels to the nervous system. These combined actions make CGRP a central player in migraine pathophysiology and a key target for treatment.During migraine attacks, CGRP levels increase in the saliva, plasma, and tear fluid. CGRP can cause dilations of blood vessels present in the brain and dura mater, release of inflammatory mediators by the mast cells, transmission of nociceptive information by the blood vessels to our nervous system, and sensitisation of the trigeminal nerves. CGRP receptor antagonists are medications that can treat and prevent migraines. These antagonists block the effects of CGRP at its receptor sites, thereby preventing the vasodilation and pain transmission associated with migraine.CGRP doesn’t seem to cross the Blood Brain Barrier, and not all vasodilators—like VIP-can trigger migraines. This suggests that CGRP-induced vasodilation alone isn’t the direct cause of migraine attacks. Anti-migraine therapies targeting CGRP fall into two main types: gepants (small molecule blockers) and monoclonal antibodies.23
Gepants
Gepants are an important class of drugs that target CGRP receptors, which plays important role in migraine pathophysiology. CGRPs are potent vasodilators that are released from trigeminal nerves during migraine episodes, leading to inflammation, pain, and sensitisation of trigeminal nerves. Gepants work by blocking the CGRP receptor, thereby preventing the transmission of pain signals and reducing inflammation. This mechanism of action is distinct from the triptans, which constrict blood vessels and block pain pathways. Gepants have shown efficacy in treating acute migraine attacks, with a rapid onset of action and sustained pain relief. Several gepants, including ubrogepant, rimegepant, and atogepant, are currently in various stages of clinical development. The drugs used shown better results in clinical trials, with a safety and tolerability profile. Gepants may offer a new treatment option for patients who don’t respond to or cannot handle existing migraine therapies.24
Monoclonal antibodies
Monoclonal antibody (mAb) treatments that target CGRP or its receptors has successful for treating migraine. These treatments, including galcanezumab, fremanezumab, eptinezumab, and erenumab, have proven activity and safety in clinical trials. The lack of adverse effects is partially because of the limited permeability of antibodies through the Blood-Brain Barrier. The anti-migraine effects of CGRP targeting are predominant evidence for its involvement in migraine pathology. CGRP antibodies are safer than triptans for blood pressure and vasoconstriction, but blocking CGRP during an ischemic event may carry uncertain risks 25.CGRP antagonists, including mAbs and gepants, may target different mechanisms than triptans. Targeting the CGRP signalling can lead to fewer adverse events and improved migraine outcomes. Clinical trials have shown that CGRP/CGRP receptor mAbs can reduce migraine days, even in patients using other preventive medications. However, it is unclear whether combining triptans and mAbs could be beneficial for some patients. Further research is needed for the understanding of the differences between treatment approaches and to identify patients who may benefit from combination therapy.26
PACAP
Pituitary Adenylate Cyclase Activating Peptides are neuropeptides that are crucial in the pathophysiology of migraine. PACAP is a vasodilator secreted from trigeminal neurons during migraine, leading to vasodilation and the generation of pain signals. Studies have shown that PACAP is elevated in cerebrospinal fluid and blood in patients who have migraine, and that it is released from trigeminal nerve endings in response, migraine triggers such as stress and inflammation.27
Furthermore, a small clinical trial reveals that a PACAP receptor antagonist reduced migraine frequency and severity for patients having chronic migraine. PACAP has also been implicated in other neurological disorders, including cluster headache, trigeminal neuralgia, and fibromyalgia. PACAP has a vital role in various physiological functions, also pain processing, inflammation, and vasodilation.28
PACAP exerts its effects by the activation of specific receptors, which include the PAC-1 receptor and the VPAC-1 receptor. The PAC-1 receptor is primarily responsible for the vasodilatory and pain-producing effects of PACAP, and is shown in trigeminal nerve terminals and blood vessels. PACAP activates PAC-1, leading to the activation of AC, which increases the formation of cyclic adenosine monophosphate. Then initiates protein kinase A, phosphorylates and also activates various downstream targets and processes, including the transcription factor CREB.28
Studies have shown that PACAP38 is an efficient vasodilator present in cerebral and dural arteries, and it has a crucial role in the initiation of the trigemino-vascular system, an important player in migraine studies. PACAP38 triggers migraine-like symptoms and strong blood vessel changes, unlike its milder cousin VIP. In the results, 73% subjects developed migraine-like attacks after infusion of PACAP 3,8, and for VI, O, it was 18%. PACAP38 and VIP dilate extracranial arteries, but PACAP38 causes longer-lasting effects and elevated plasma levels in migraineurs, unlike VIP’s short-lived response.29
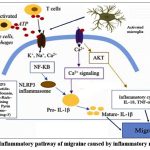 |
Figure 5: Inflammatory pathway of migraine caused by inflammatory mediators. |
Phosphodiesterase (PDE) Activator
Phosphodiesterase (PDE) activator is another potential target for migraine treatment. PDEs are enzymes that break down cAMP and cGMP, which in turn reduces the accumulation of these cAMP and cGMP. Sildenafil (PDE5 inhibitor) and cilostazol (PDE3 inhibitor) have been observed to induce migraines, indicating that cAMP and cGMP accumulation might contribute to migraine pathophysiology. Therefore, activating cAMP-selective PDEs (PDE3 or PDE5) would be an important migraine treatment. However, no such activators have been developed for PDE3 or PDE5, and the risk of serious adverse events is high due to the proper overall distribution of these enzymes in the body.30,31
PDE-4
cAMP has a crucial role in the activity of systemic circulation. In the heart, cAMP affects various processes, like the contractility of myocytes to the death of myocytes. In the case of blood vessels, cAMP can change the state of smooth muscle cells, namely into contracted and relaxed form, and the permeability of endothelium can also be altered.32
cAMP produces vastly varying cellular activity by activating 4 types of proteins
Protein kinase A – phosphorylated substrate protein, Exchange protein directly activated by cAMP – activates the RAS superfamily of enzymes,Cyclic nucleotide-activated ion channels, Popeye domain containing proteins, cAMP is synthesised by mAC and sAC in the presence of a stimulus. It quickly diffuses throughout the cell cytoplasm, while the diffusion of cAMP activates all effector proteins instantly. It can also show multiple discrete receptor-specific responses due to the action of cAMP.32
cAMP-degrading proteins have subcellular localisation in the cells; this type of enzyme falls under the superfamily phosphodiesterase (PDE). PDE is the only known protein that can hydrolyse cAMP. Thus reduction in cAMP levels in the specific area leads to inadequate phosphorylation of PKA in normal conditions 32. PDE4 is an example of a cAMP-specific PDE present in inflammatory cells. Reduced activity of PDE4 can result in elevated cAMP levels in the cell, which leads to decreased inflammatory response.33
PDE is a large superfamily of enzymes composed of products of 11 different genes on the basis of structure, function and affinity of cGMP and cAMP. PDEs have common carboxy-terminal catalytic cores, while the amino-terminal region differs between the various families, sub-families and specific isoforms. The N-terminal or the amino-terminal acts as the targeting moiety.33 cAMP-specific PDE4s constitute the biggest family with more than 20 isoforms encoded by 4 genes, which are A, B, C, and D. Each type of isoform consists of a unique N-terminal region, composed of an N-terminal targeting domain (TD).34
N-terminal consists of Upstream-Conserved Region 1 and Upstream-Conserved Region 2, which are bound together with catalytic domains via Linker Region 1 and Linker Region 2, respectively. Based on this, PDE4s can be divided into 4 types.34,35
Long isoform – has UCR1 +UCR2.
Short isoform – only UCR2.
Super-short isoform – shortened UCR2.
Dead-short isoform – lacks the UCR domain and the shortened catalytic domain.
UCR1 possesses a specific site for phosphorylation of cAMP-dependent PKA, whereas UCR2 is considered to possess an inhibitory PDE4-catalytic unit and engage in interactions with UCR1, thereby regulating the functional activity of UCR1. Therefore, the various UCR1/2 regions have regulated pathways for PDE4 subfamilies. Only PDE4B, PDE4C and PDE4D possess signal-regulated kinase (ERK)-phosphorylation subunits outside the cell, which are present in the catalytic PDE4 unit. ERK unit phosphorylation regulates the hydrolysable activity of PDE4 isoforms, specifically inhibiting the long isoform, enhancing the activity of the short isoform and having a negligible impact on the super-short isoform.36
Rolipram, which is an early PDE4 inhibitor, had restricted therapeutic usage due to the presence of side effects like nausea and emesis q which can be prevented using the dose-limiting approach. Second-generation medications like Cilomilast have been reported to possess reduced side effects. Indeed, Cilomilast shows effective therapeutic activity in clinical trials for the treatment of asthma and COPD and is an example of most advanced selective PDE4 inhibitors.37,38
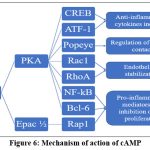 |
Figure 6: Mechanism of action of cAMP. |
cAMP pathophysiology through inflammation may result in different outcomes. cGMP activity can be prevented by PDE5 and PDE5 inhibitors, such as Zaprinast and Sildenafil. The synthesis of sildenafil and its effect on penile erection is utilised for the treatment of Erectile Dysfunction. Sildenafil (PDE5 inhibitor) and Cilostazol (PDE3 inhibitor) exhibit side effects like inducing migraines, further reinforcing the idea of cAMP (and cGMP) accumulation as a major pathway that can trigger migraine. Hence, cAMP-selective PDE (PDE3 or PDE5) activation can be used for migraine treatment. Till now, no such compound has been synthesised, identified or isolated for PDE3 or PDE5. But this is not the case for PDE4, exhibiting as a proof of concept. In spite of the potential therapeutic effect, there is a risk of prominent adverse reactions due to the wide distribution of this system all over the body.39,40
PDE5
Phosphodiesterase type 5 is a dimeric protein consisting of a catalytic domain that catalyses the breakdown of cGMP to 5-GMP, and a regulatory part. PDE5 enzymes that break down cGMP. It increases cGMP levels, causes vasodilation of cerebral blood vessels, and causes migraine attacks by stimulating the trigeminovascular system.PDE5 inhibitors are not used for migraine treatment as sildenafil and tadalafil can trigger migraine, whereas activating PDE5 reduces cGMP levels, limiting vasodilation and reducing migraine.PDE5 inhibitors are used for headaches, including migraine-like headaches, as a side effect.41,42
Discussion
Migraine is an intricate and widespread neurological disorder, highlighting the pathophysiology, genetics, and treatment options. Affecting over a billion people worldwide, migraine poses significant personal, economic, and social challenges. The exact mechanisms underlying migraine remain poorly understood, making it challenging to treat and diagnose. Studies have shown several genetic variants contribute to the symptoms of migraine, particularly the neurotransmitters and blood vessels. For instance, the GPCR pathway plays a pivotal role in migraine initiation. The trigeminovascular pathway, which involves activation of trigeminal nerve fibres and releases the vasoactive neuropeptides, is also thought to play a key role in migraine initiation. Cortical spreading depression is are waves of abnormal brain activity that propagate across the cortex and is associated with aura phase of migraine. The activation of GPCR will stimulate the trigeminal nerve, which starts CSD and also the formation and accumulation of cAMP in the intracellular region. cAMP promotes the formation of inositol triphosphate (IP3), which causes calcium ions to influx into the cytosolic region. The endoplasmic reticulum releases the calcium ions, and the influx of extracellular calcium leads to contraction of cells of the endothelium, which in turn causes an increase in blood pressure of meningeal blood vessels. This leads to an increase in pressure on the brain and meninges that can initiate migraine-like symptoms. CGRP it’s a neurotransmitter that helps in the vasodilation of blood vessels. The trigeminal nerve releases the CGRP when the brain processes the input the action potential is passed through the trigeminal nerve. The CGRP are released from both peripheral and central parts of the brain. These receptors initiate the pain, artery dilation and inflammatory symptoms. The CGRP bind with the meningeal mast cell, which triggers the inflammatory cascade, making the neuron hyperexcitable. The brain stem receptor is activated by CGRP, which increases the firing of postsynaptic neurons. This causes the release of pain signals, causing migraine episodes. The CGRP increases the cAMP level, causing the secretion of NO, which may lead to inflammatory symptoms. PDE4 inhibitors cause a fall in the level of PDE4, contributing to a rise in the level of cAMP in the intracellular region, which leads to increased PKA levels. The increased pKA levels will increase cAMP Response Element Binding protein, which causes a decrease in inflammation. This increase in the pKA level inhibits NF-kb and CGRP level in the cells, which decreases the pro-inflammatory mediators and inflammatory gene expression this leading to a decrease in migraine-like symptoms. PDE5 inhibitors are migraine inducers that regulate cGMP levels, causing vasodilation and inflammation in blood vessels, which results in migraine.
Conclusion
Migraine is a complex neurological trick that makes you have headaches over and over again, causes you to be super sensitive to light and sound and screws with your blood vessels. Here I’m (looking) at the main molecules and meds determining how it works and how we treat it. Some important signals are GPCRs, cAMP, CGRP and PACAP. When cAMP goes up it causes the vessels to widen and the inflammation to release pain chemicals – this is why migraine feels so intense. When it hits the trigeminovascular system kicks in and releases CGRP. CGRP binds with cells in the outer layer of the brain and the walls of blood vessels, in effect making the nerves even more excited and inflamed. PACAP does much of the same job and adds in more of the chemical cAMP. Because of that, cAMP and CGRP are attempted to be subdued with treatments that try to block these. Purinergic receptors can reduce cAMP and phosphodiesterases (PDEs) break down cAMP. PDE3 and PDE5 are promising – blocking PDE4 actually causes cAMP to go up and symptoms to get worse. PDE5 also works with cGMP, including on how blood vessels in the brain relax, and we feel pain. New treatments focus on CGRP. Small pills (gepants) block the receptors of CGRP for fast relief. Big antibodies that target CGRP or CGRP receptor are long term preventatives. Old-school drugs such as triptans remain strong – these drugs work by hitting the serotonin receptors to make the blood vessels tighter and preventing pain chemicals from leaking out of the body. Painkillers that have anti inflammatory effects and anti depressants help as well. Migraine isn’t a one size fits all Some get the aura, some don’t and some end up having the rare hemiplegic type. It comes in four normal stages: premonitory (early warning signs), aura or visual stuff, headache, and postdromal (fatigue and grogginess). It’s important to know what type and stage a person is in in order to be able to treat them accordingly. To sum it up, migraines are caused by precisely-controlled signalling that results in inflammation and pain. The current best treatments are directed towards CGRP blockers and PDE drugs showing promising results for each patient.
Acknowledgment
The authors would like to thank SRM College of Pharmacy, SRM Institute of Science and Technology for the necessary facilities provided.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contribution
Parekh Nirmit Jigneshkumar– Mechanism of GPCR, cAMP, CGRP, PACAP and citations.
Patel Mahek Devangbhai – Introduction of migraine, Discussion of Gepants & Purinergic receptors
Ayyan Dutta – PDE activators mechanism, Discussion of migraine pathway, Diagrams
Arnab Dutta – Pathophysiology of Migraine, Phases of Migraine and Diagrams
Debadrita Ghosh – Treatment and mechanism of drugs, Brief introduction of PDE5, Conclusion and Editing of article.
Reference
- The Global Burden of Migraine: A 30-Year Trend Review and Future Projections by Age, Sex, Country, and Region | Pain and Therapy. Accessed December 4, 2025. https://link.springer.com/article/ 10.1007/s40122-024-00690-7
- Migraine and the trigeminovascular system—40 years and counting – The Lancet Neurology. Accessed December 6, 2025. https://www.thelancet.com/journals/laneur/article/PIIS1474-4422(19)30185-1/abstract
- Advances in genetics of migraine | The Journal of Headache and Pain. Accessed December 5, 2025. https://link.springer.com/article/10.1186/s10194-019-1017-9
- Targeting the 5-HT1B/1D and 5-HT1F receptors for acute migraine treatment. In: Progress in Brain Research. Vol 255. Elsevier; 2020:99-121. doi:10.1016/bs.pbr.2020.05.010
CrossRef - Fariba KA, Saadabadi A. Topiramate. In: StatPearls. StatPearls Publishing; 2025. Accessed December 6, 2025. http://www.ncbi.nlm.nih.gov/books/NBK554530/
- Khan J, Asoom LIA, Sunni AA, et al. Genetics, pathophysiology, diagnosis, treatment, management, and prevention of migraine. Biomed Pharmacother. 2021;139:111557. doi:10.1016/j.biopha.2021.111557
CrossRef - The vessel-to-neuron trigeminovascular hypothesis of migraine pathogenesis – the ‘pro’ argument | The Journal of Headache and Pain. Accessed December 6, 2025. https://link.springer.com/article/ 10.1186/s10194-025-02130-z
- Close LN, Eftekhari S, Wang M, Charles AC, Russo AF. Cortical spreading depression as a site of origin for migraine: Role of CGRP. Cephalalgia. 2019;39(3):428-434. doi:10.1177/0333102418774299
CrossRef - Kumar A, Samanta D, Emmady PD, Arora R. Hemiplegic Migraine. In: StatPearls. StatPearls Publishing; 2025. Accessed December 6, 2025. http://www.ncbi.nlm.nih.gov/books/NBK513302/
- Pescador Ruschel MA, De Jesus O. Migraine Headache. In: StatPearls. StatPearls Publishing; 2025. Accessed December 6, 2025. http://www.ncbi.nlm.nih.gov/books/NBK560787/
- Hougaard A, Ayata C, Brennan KC, van den Maagdenberg AMJM, Ashina M. The mysterious link between migraine aura and migraine headache. PLOS Biol. 2025;23(6):e3003168. doi:10.1371/journal.pbio.3003168
CrossRef - Structure, function and drug discovery of GPCR signaling | Molecular Biomedicine. Accessed December 6, 2025. https://link.springer.com/article/10.1186/s43556-023-00156-w
- cAMP-dependent Protein Kinase – an overview | ScienceDirect Topics. Accessed December 6, 2025. https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/camp-dependent-protein-kinase
- Gao F, Yang S, Wang J, Zhu G. cAMP-PKA cascade: An outdated topic for depression? Biomed Pharmacother. 2022;150:113030. doi:10.1016/j.biopha.2022.113030
CrossRef - Cheng L, Xia F, Li Z, et al. Structure, function and drug discovery of GPCR signaling. Mol Biomed. 2023;4(1):46. doi:10.1186/s43556-023-00156-w
CrossRef - Structure, function and drug discovery of GPCR signaling | Molecular Biomedicine. Accessed December 6, 2025. https://link.springer.com/article/10.1186/s43556-023-00156-w
- Calcitonin Gene-Related Peptide (CGRP): A New Target for Migraine | Annual Reviews. Accessed December 6, 2025. https://www.annualreviews.org/content/journals/10.1146/annurev-pharmtox-010814-124701
- Bamalan OA, Moore MJ, Al Khalili Y. Physiology, Serotonin. In: StatPearls. StatPearls Publishing; 2025. Accessed December 6, 2025. http://www.ncbi.nlm.nih.gov/books/NBK545168/
- Spekker E, Tanaka M, Szabó Á, Vécsei L. Neurogenic Inflammation: The Participant in Migraine and Recent Advancements in Translational Research. Biomedicines. 2021;10(1):76. doi:10.3390/biomedicines10010076
CrossRef - Guirland C, Buck KB, Gibney JA, DiCicco-Bloom E, Zheng JQ. Direct cAMP Signaling through G-Protein-Coupled Receptors Mediates Growth Cone Attraction Induced by Pituitary Adenylate Cyclase-Activating Polypeptide. J Neurosci. 2003;23(6):2274-2283. doi:10.1523/JNEUROSCI.23-06-02274.2003
CrossRef - Exploration of purinergic receptors as potential anti-migraine targets using established pre-clinical migraine models – Kristian A Haanes, Alejandro Labastida-Ramírez, Frank W Blixt, Eloisa Rubio-Beltrán, Clemens M Dirven, Alexander HJ Danser, Lars Edvinsson, Antoinette MaassenVanDenBrink, 2019. Accessed December 6, 2025. https://journals.sagepub.com/doi/10.1177/0333102419851810
- Calcitonin Gene-Related Peptide: Physiology and Pathophysiology | Physiological Reviews | American Physiological Society. Accessed December 6, 2025. https://journals.physiology.org/doi/ full/10.1152/physrev.00034.2013
- Balcziak LK, Russo AF. Dural Immune Cells, CGRP, and Migraine. Front Neurol. 2022;13. doi:10.3389/fneur.2022.874193
CrossRef - Jakubowska B, Sowa-Kućma M. Gepants: targeting the CGRP pathway for migraine relief. Front Pharmacol. 2025;16. doi:10.3389/fphar.2025.1708226
CrossRef - Monoclonal antibodies for the prevention of migraine: Expert Opinion on Biological Therapy: Vol 19 , No 12 – Get Access. Accessed December 6, 2025. https://www.tandfonline.com/doi/full/ 10.1080/14712598.2019.1671350?utm_source=researchgate.net&utm_medium=article
- Versijpt J, Paemeleire K, Reuter U, MaassenVanDenBrink A. Calcitonin gene-related peptide-targeted therapy in migraine: current role and future perspectives. The Lancet. 2025;405(10483):1014-1026. doi:10.1016/S0140-6736(25)00109-6
CrossRef - PACAP and its role in primary headaches | The Journal of Headache and Pain. Accessed December 6, 2025. https://link.springer.com/article/10.1186/s10194-018-0852-4
- Juhasz G, Gecse K, Baksa D. Towards precision medicine in migraine: Recent therapeutic advances and potential biomarkers to understand heterogeneity and treatment response. Pharmacol Ther. 2023;250:108523. doi:10.1016/j.pharmthera.2023.108523
CrossRef - PACAP38 induces migraine-like attacks in patients with migraine without aura | Brain | Oxford Academic. Accessed December 6, 2025. https://academic.oup.com/brain/article-abstract/132/1/16/290515?redirectedFrom=fulltext
CrossRef - Therapeutic Potential of Phosphodiesterase Inhibitors against Neurodegeneration: The Perspective of the Medicinal Chemist | ACS Chemical Neuroscience. Accessed December 6, 2025. https://pubs.acs.org/doi/10.1021/acschemneuro.0c00244
- Pivotal effects of phosphodiesterase inhibitors on myocyte contractility and viability in normal and ischemic hearts | Acta Pharmacologica Sinica. Accessed December 6, 2025. https://www.nature.com/articles/aps20081
- Harvey RD, Clancy CE. Mechanisms of cAMP compartmentation in cardiac myocytes: experimental and computational approaches to understanding. J Physiol. 2021;599(20):4527-4544. doi:10.1113/JP280801
CrossRef - Phosphodiesterase 4-targeted treatments for autoimmune diseases | BMC Medicine. Accessed December 6, 2025. https://link.springer.com/article/10.1186/1741-7015-11-96
- PDE4-Mediated cAMP Signalling. Accessed December 6, 2025. https://www.mdpi.com/2308-3425/5/1/8
CrossRef - PDE4A – an overview | ScienceDirect Topics. Accessed December 6, 2025. https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/pde4a
- UCR1 and UCR2 Domains Unique to the cAMP-specific Phosphodiesterase Family Form a Discrete Module via Electrostatic Interactions* – Journal of Biological Chemistry. Accessed December 6, 2025. https://www.jbc.org/article/S0021-9258(19)89918-0/fulltext
- PDE4 inhibitors: current status – Spina – 2008 – British Journal of Pharmacology – Wiley Online Library. Accessed December 6, 2025. https://bpspubs.onlinelibrary.wiley.com/ doi/10.1038/ bjp.2008.307
- Richter W, Conti M. Dimerization of the Type 4 cAMP-specific Phosphodiesterases Is Mediated by the Upstream Conserved Regions (UCRs)*. J Biol Chem. 2002;277(43):40212-40221. doi:10.1074/jbc.M203585200
CrossRef - PDE5 inhibitors – pharmacology and clinical applications 20 years after sildenafil discovery – Andersson – 2018 – British Journal of Pharmacology – Wiley Online Library. Accessed December 6, 2025. https://bpspubs.onlinelibrary.wiley.com/doi/10.1111/bph.14205
- Stief CG, Ückert S, Becker AJ, et al. Effects of sildenafil on cAMP and cGMP levels in isolated human cavernous and cardiac tissue. Urology. 2000;55(1):146-150. doi:10.1016/S0090-4295(99)00371-4
CrossRef - An overview on pharmaceutical applications of phosphodiesterase enzyme 5 (PDE5) inhibitors | Molecular Diversity. Accessed December 6, 2025. https://link.springer.com/article/10.1007/s11030-024-11016-2
- Frontiers | Rethinking of phosphodiesterase 5 inhibition: the old, the new and the perspective in human health. Accessed December 6, 2025. https://www.frontiersin.org/journals/endocrinology/ articles/10.3389/fendo.2024.1461642/full
Accepted on: 25-12-2025
Second Review by: Dr. Shahin Vahora
Final Approval by: Dr. Wagih Ghannam









