In silico design of Antimalarial drug from Catharanthus roseus (G. Don)alkaloid molecules against AMA1 protein
Department of Zoology, Sangam University, Bhilwara, Rajasthan, India
Corresponding Author E-mail:chetanasuvalka0408@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3470
ABSTRACT:The emergence and spread of drug resistance in Plasmodium falciparum, the parasite causing the most severe form of malaria is a major threat to malaria control and elimination program around the globe. With Plasmodium falciparum having evolved widespread resistance against a number of previously widely used drugs, currently artemisinine and its derivatives are the cornerstones of first line treatment of uncomplicated malaria. Growing incidences failure reflects artemisinine resistance. Pharmacology approach in this study is being used against AMA1 protein which can be used as major target for drug due to its participation in erythrocytic infection stage. This protein is conserved in all plasmodium species. The AMA1-PLAF8 structure is modelled through homology modelling method and virtual screening against Catharanthus roseus fraction is carries out using Maetrso. Secologanin,Vindoline,tabersonine,Vincristine,Serpentine, Ajmalicine,Catharanthine and catechol were taken for screening. Secaloganine showed the best binding results with lowest binding energy (-7.815 Kcal/mol) and shortest bond length i.e. 1.80881 Ǻ. This virtual screening investigation suggests that Secaloganin can be repurposed for malaria control and prevention.
KEYWORDS:Apical Membrane Antigen 1 (AMA 1); Artemesinine resistance; Malaria; Molecular docking; XP Gscore
Introduction
Malaria keeps on causing unsuitably significant degrees of illness and demise, as archived in progressive versions of the World Malaria report.1 As indicated by the most recent report, there were an expected 627000 lives lost in malaria 2020.2 Malaria is preventable and treatable and the worldwide need is to lessen the weight of infection and demise while holding the drawn out vision of Malaria destruction.
The only malaria vaccine, RTS,S/AS01, initiates incomplete adequacy through enlistment of antibodies against the sequence (Asn-Ala-Asn-Pro) of the circumsporozoite protein (CSP). However, there are many antigenic determinants of Plasmodium against which either medications or antibodies are formed. 3 The different standards for antigen choice as well as medication definition are summed up in table.1.
Table 1: Plasmodium exposed surface antigens are:
| S.No. | Protein ID | Protein name | Protein function |
| 1. | IPR010901 | Merozoite Surface 1, C-terminal | C-terminal region of merozoite surface protein 1 which helps in invasion of Red Blood Cells.4 |
| 2. | IPR010423 | Ookinete Surface antigen, EGF domain | Pvs25 and Pvs28 antigenic proteins present on surface of Plasmodium.5 |
| 3. | IPR032761 | Plasmodium falciparum erythrocyte membrane protein1,N terminal | Protein domain present on Plasmodium falciparum infected erythrocytes to adhere host endothelial receptors.6 |
| 4. | IPR003067 | Plasmodium Circumsporozoite Protein | Surface antigen on surface of sprozoite of Plasmodium which is passes from mosquito vector to mammalian host.7 |
| 5. | PF12319 | Tryptophan-Threonine-rich plasmodium antigen C terminal | C terminal of the surface antigen present on Plasmodium.8 |
| 6. | PF07462 | Merozoite surface protein 1 (MSP1) C-terminus | Represent C terminal region of merozoite surface protein 1 which induces merozoite infection to Red Blood Cells.9 |
| 7. | IPR029210 | Plasmodium falciparum erythrocyte membrane protein-1,N terminal segment | Protein domain present on Plasmodium falciparum infected erythrocytes to adhere host endothelial receptors.10 |
| 8. | IPR006373 | Variant surface antigen Rifin | RIFIN Expressed on free merozoites as well as on infected erythrocytes contributing to antigenic variation capacity of the parasite.11 |
| 9. | IPR010784 | Merozoite surface protein type | Merozoite invasion to erythrocytes involves multiple interaction with Merozoite Surface Protein.4 |
| 10. | PF15445 | Acidic terminal segments, variant surface antigen of PfEMP1 | These proteins help in evading host immune system by sticking to endothelial cells. |
| 11. | PF06247 | Pvs28 EGF domain | Group of okinete surface protein and induces immune responses in body |
| 12. | IPR006499 | Reticulocyte binding protein | These are reticulocyte binding protein which express Duffy antigen |
| 13. | PF03805 | Cytoadherence linked asexual protein | Induces binding of Pf to host endothelial cells |
| 14. | PF15448 | N terminal segments of Plasmodium falciparum erythrocyte membrane protein | This is a transmembrane protein that shares domains with molecules expressed on infected erythrocytes |
| 15. | PF07133 | Merozoite surface protein (SPAM) | It is a platform for binding to human erythrocytes by Plasmodium falciparum |
| 16. | IPR003298 | Apical membrane antigen 1 | Expressed on invasive merozoite which are causative agent of malaria |
| 17. | IPR026894 | DNAJ containing protein, X domain | This domain is found in Plasmodium falciparum ring infected erythrocyte surface antigen and is shown to bind to spectrin and stabilize tetramer.12 |
| 18. | IPR024056 | Apical membrane antigen 1 (AMA-1) domain superfamily | AMA-1 appears to be transported to the merozoite surface close to the time of schizont rupture.13 |
| 19. | IPR005553 | Cytoadherence Linked Asexual Protein (CLAP) | CLAP is found to be associated in binding of P.falcipaprum infected erythrocytes to host endothelial cells. |
| 20. | IPR010884 | 6-Cysteine (6-Cys) domain | 6-Cys domain is found in Plasmodium proteins that are expressed in all stages of parasite life in both vertebrate and mosquito hosts.14 |
| 21. | PF02009 | RIFIN (Repeated Interspersed families) | These are expressed on the surface of infected erythrocytes.15 |
| 22. | PS50095 | PLAT (Polycystin-1, lipoxygenase and alpha toxin) domain profile | It is involved in protein-protein, protein-lipid interaction. In P.falciparum it is expressed on surface of gametocyte and interacts with mammalian triglyceride lipase.16 |
| 23. | PS50092 | Thrombospondin type-1 (TSP1) repeat profile | It is present in circumsporozoite protein of Plasmodium.17 |
| 24. | PF15447 | N terminal segments of PfEMP1 | It is variable part of the variant surface antigen family Plasmodium falciparum and adhere to host endothelial receptors.18 |
| 25. | IPR043226 | Neural cytotoxicity triggering receptor 3 | It stimulates NK cells cytotoxicity. |
| 26. | IPR009484 | Protein of unknown function DUF1103 | This family function is unknown. |
| 27. | IPR014886 | La protein, xRRM domain | La protein motifs are generally involved in binding to UUU-3’OH sequence of various RNA Polymerase III where xRRM domain induces conformational changes.19 |
| 28. | IPR034204 | Subtilisin SUB1-like catalytic domain | It activates the merozoite surface protein allows it to bind to host erythrocyte membrane prior to egress.20 |
| 29. | PS50234 | VWFA domain profile | Von Willebrand Factor (VWF) is a large multimeric protein required for normal hemostasis.21 |
| 30. | IPR036383 | Thrombospondin type-1(TSP1) repeat superfamily | These are multimeric glycoprotein that function at cell surfaces and in extracellular matrix.22 |
The current situation is confronting many difficulties in vector control and parasite disposal. One of the most significant challenges is drug obstruction in Plasmodium. While obstruction is regularly evaluated by drug adequacy concentrates on that uncover applicant point transformations, whose pervasiveness are checked. The genuine method of activity of obstruction is related with changes which are frequently less clear. Notwithstanding, a significant comprehension of the sub-atomic instrument hidden medication opposition can prompt the improvement of new synthetic mixtures that can re-establish drug productivity. Witnessing the need for antimalarial drug, there is need of virtual screening of some potential drug candidates to push them up for further trials.
Catharanthus roseus is an important medicinal plant of family Apocynaceae with 70 different types of alkaloids, steroids and chemotherapeutic agents which are effective as anticancerous, antimalarial and antimicrobial activity. Vinblastine and Vincristine are two important alkaloids that are being used widely to treat diabetes.23 Catharanthus roseus (L.) G. Don contains terpenes and alkaloids which exhibits great pharmacological activities. 24 It has wide range of medicinal properties such as antioxidant, 25-27 anticancer, 28 antidiabetic, 29 antimicrobial,30 antiulcer,23 hypertensive,24,31 wound healing, 31 hypolipidemic 27 and memory enhancement. 28,29 With virtual screening the objective is to explore antimalarial effect of various alkaloid fractions of the plant.
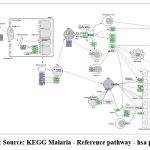 |
Figure 1: Source: KEGG Malaria – Reference pathway – hsa pathogen |
Table 2. Antimalarial drugs are classified on basis of its target:
| S.No. | Mode of action | Target Site | Type of drug | |
| 1. | Depending on stage of plasmodium it affects | Tissue Schizocidal | Primary Tissue forms | a. Sulfodoxine + Pyrimethininb. Proguanil
c. Atrovaquine d. Primaquine |
| Latent Tissue Forms(Hyphozoites) | a. Primaquineb. Tafenoquine | |||
| Blood Schizontocidal | Rapid action | a. Chloroquineb. Artemesinin derivative
c. Quinine d. Mefloquine e. Atoquine f. Amodiaquine g. Lumefantrine |
||
| Slow action | a. Proguanilb. Pyrimethamine + Sulfadoxine
c. Clirdomycin |
|||
| Gametocidal | a. Artemesininb. Primaquine
c. Chloroquine and Quinine |
|||
|
2 |
Depending on clinical indications | Causal prophylaxis | No drugs for sporozoite from, these drugs will inhibit pre-erythrocytic stage | |
| Suppressive prophylaxis | a. Chloroquineb. Mefloquine
c. Doxycycline |
|||
| Clinical cure | a. Chloroquineb. Artemesinin derivative
c. Quinine d. Mefloquine e. Atoquine f. Amodiaquine g. Lumefantrine d. Proguanil e. Pyrimethamine + Sulfadoxine h. Clirdomycin |
|||
| To prevent relpase | a. Primaquine | |||
| Transmission to mosquitoes | a. Primaquine |
Table 3: Antimalarial drugsdivided on basis of groups
| S.no. | Group | Type of drug | |
| 1. | Quinoline derivative | a. Chloroquine | |
| b. Quinine | |||
| c. Atroquine | |||
| d. Primaquine | |||
| e. Mefloquine | |||
| f. Tefloquine | |||
| 2. | Artemesinin | Artemesinin derivative | a. Dihydroartemesininb. Artemether
c. Artesunate d. artether |
| Synthetic | a. Arteolane | ||
| 3. | Aryl alcohol derivative | a. Lumefantrine | |
| 4. | Antifolates | a. Pyrimethamineb. Sulfodoxine
c. Doprone d. Proguanil |
|
| 5. | Antimicrobials | a. Tetracyclinb. Clindamycin | |
The potential drugs and their mechanism of action is summarized in table 2, however, the effect and targets of various drugs is summarized in table 3. There are no medications for sporozoite form; the causal prophylaxis drugs repress erythrocytic stage. The suppressive prophylaxis acting medications smother clinical infection beginning. Primaquine is the main medication which targets hyphozoites of P.vivax and P.ovale as well as gametocytes of Plasmodium.
Following treatments are used to treat uncomplicated P. falciparum malaria (2015) Treating children and adults with uncomplicated P. falciparum malaria (except pregnant women in their first trimester) with one of the following ACTs:
artemether + lumefantrine
artesunate + amodiaquine
artesunate + mefloquine
dihydroartemisinin + piperaquine
artesunate + sulfadoxine-pyrimethamine (SP).
The current situation is confronting many difficulties in vector control and parasite elimination. One of the recent problems in malaria elimination is Plasmodium mutants. The mutation in Kelch protein resulted in resistance. To overcome such obstructions drug designing against target protein is been done to overcome future problem of resistance. As changes in the Kelch13 (PfK13) quality were distinguished as the vital sub-atomic markers and stayed to be key indicators of ART resistance. As far as drug delivery is concerned the most potential and safe target stage is invasion of Red Blood Cells. Regarding above fact and seeing the overview of Plasmodium infection in figure.1. Merozoite surface antigen 1,8 Plasmodium falciparum erythrocyte membrane protein 1,13 RIFIN, 15 Apical Membrane Antigen 1 are the most effective targets. However, AMA1 protein is chosen in this work because of its transmembrane nature and many unrevealed characters.13
This paper covers in silico designated drug adequacy against AMA1 protein quality and its viability to be utilized as medication in future for Malaria treatment.
Methodology
The FASTA grouping of P. falciparum for AMA 1 was recovered from the UniProt data set (https://www.uniprot.org/). The comparing UniProt ID explicitly for the P. falciparum strain utilized for this study was AMA1_PLAF8. The Protein sequence and 3D structure of PfK13 was derived from Uniprot data set.
Protein Modelling
Protein modelled through Homology modelling method through Swiss Model. The maximum homology was 97% with searched templates, the modelled prepared by choosing best matching template and best model selected on basis of lowest RMSD value and DOPE score. The model structure was validated through Ramachandran plot.
Ligand Preparation
The small molecule library was built based on a wide range of experimental activity against P. falciparum. Molecular preparations such as the insertion of hydrogen bonds, 2D to 3D conversion, stereoisomers production, neutralisation of charged structures, or identification of most likely ionisation state at a user-defined pH, and the entire preparation were carried out using Maestro’s LigPrep application.32
Protein preparation
Because the protein was simulated, the YASARA Energy Minimisation Server was used to perform an energy minimisation (Krieger et al. 2009)33. This minimised protein was then used in the Protein Wizard tool for further processing. Using Maestro’s Glide application, the receptor grid with given grid coordinates of X = -20, Y = -10, and Z = 0 was produced with a box size of 36 × 36 × 36 Å. 33
Molecular Docking
The produced ligand library was then docked with the protein grid using Glide’s XP (extra precision) docking function, yielding an XP Glide score as a consequence. To rank docking poses and determine protein–ligand binding affinities, the XP Glide scoring function is utilised. Using a “funnel-type” method, the Glide algorithm searches for the ligand’s position, orientation, and conformation in the enzyme’s active site. 34 Maestro’s Pose viewer & XP-visualizer software was used to analyse the output files.
Protein FASTA sequence
QKY59679.1 AMA1MKSSNTKMQCIVKKLSLLAMPVVIAAILSLKIVPAGAAFVAFQTDPPSSRGNRRSSRGGRNQQAAGRQAQNEA EGTERAGGR SSSSKIIQQTPWTKYMIKYDIARCHGSGIYVDMGGYEAIGGKHYRMPIGKCPVMGKVINLASGADFLEPISADNPRYRGLGFPETVLK HTGALAGALTGTANNAINLSPVSAEDLRKWGYKGNPVTNCAEYANNIVPGSDTRTKYRYPFVYDGKDELCYVLYSPMQYNQGSRYC DADGSLEEGPSSLLCMKPYKSDLDAHLYYGSSRIDPKWDVNCPMSPIRDAIFGKWVSGACVALESAFEEFVNSAEECASILFENSATDID VDVDAEGYNEINELYSGLKNLQLKQIAFSLFAPMSKSAATAKLSKGVGKNWANYESNLGICRILSETPTCLIINAGSLAMTALGSPLESDA INFPCDIDTVGYVEPRTRNGENGESRFPVTTALSIKTLKCTKYVHSKYSESCGTYYYCSEEKSSYLSRLYQFLSNHSVKKAMAITAALLALIF AIYWVYRRLYTTKIRREHEDYDRLMSKYEYDDVSHAVSEPEQQLKTDAYIWGEAAARPSDITPVHLTKLN
 |
Figure 2: Model prepared through modeller
|
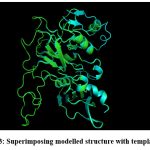 |
Figure 3: Superimposing modelled structure with template 6n87 |
Table 4: XP Gscore and drug targets
| S.no. | PDB | DRUG ID (PubChem) | XP GScore | |
| ID: | CHEMBL1235867 | |||
| Name: | SECOLOGANIN | |||
| Max Phase: | 0 Research | |||
| Molecular Formula: | C17H24O10 | |||
| Molecular Weight: | 388.37 | |||
| ChEMBL Synonyms: | Secologanin | |||
| Molecule Type: | Small molecule | |||
| 2. | Vindoline | ID: | CHEMBL526546 | -4.734 |
| Name: | VINDOLINE | |||
| Max Phase: | 0 Research | |||
| Molecular Formula: | C25H32N2O6 | |||
| Molecular Weight: | 456.54 | |||
| ChEMBL Synonyms: | Vindoline | |||
| Molecule Type: | Small molecule | |||
| 3. | Ajmalicine | ID: | CHEMBL1604074 | -6.230 |
| Name: | Ajmalicine | |||
| Max Phase: | 0 Research | |||
| Molecular Formula: | C21H24N2O3 | |||
| Molecular Weight: | 352.43 | |||
| Molecule Type: | Small molecule | |||
| 4. | Serpentine | ID: | CHEMBL3559488 | -6.286 |
| Name: | Serpentine | |||
| Max Phase: | 0 Research | |||
| Molecular Formula: | C21H21N2O3+ | |||
| Molecular Weight: | 349.41 | |||
| Molecule Type: | Small molecule | |||
| 5. | Catharanthine base | ID: | CHEMBL2163793 | -3.847 |
| Name: | CATHARANTHINE BASE | |||
| Max Phase: | 0 Research | |||
| Molecular Formula: | C21H24N2O2 | |||
| Molecular Weight: | 336.44 | |||
| ChEMBL Synonyms: | Catharanthine Base | |||
| Molecule Type: | Small molecule | |||
| 6. | Vincristine | ID: | CHEMBL90555 | -5.899 |
| Name: | VINCRISTINE | |||
| Max Phase: | 4 Approved | |||
| Molecular Formula: | C46H56N4O10 | |||
| Molecular Weight: | 824.97 | |||
| 7. | Tabersonine | ID: | CHEMBL2011511 | -4.990 |
| Name: | TABERSONINE | |||
| Max Phase: | 0 Research | |||
| Molecular Formula: | C21H24N2O2 | |||
| Molecular Weight: | 336.44 | |||
| ChEMBL Synonyms: | Tabersonine | |||
| Molecule Type: | Small molecule | |||
| 8. | Catechol | ID: | CHEMBL280998 | -3.622 |
| Name: | CATECHOL | |||
| Max Phase: | 0 Research | |||
| Molecular Formula: | C6H6O2 | |||
| Molecular Weight: | 110.11 | |||
| ChEMBL Synonyms: | Pyrocatechol | |||
| Molecule Type: | Small molecule | |||
| 9. | Artemesinin | ID: | CHEMBL567597 | -3.226 |
| Name: | ARTEMISININ | |||
| Max Phase: | 4 Approved | |||
| Molecular Formula: | C15H22O5 | |||
| Molecular Weight: | 282.34 | |||
| ChEMBL Synonyms: | (+)-Artemisinin ARTEMISININGNF-PF-5341 NSC-369397 | |||
| Trade Names: | Artemisin | |||
| Molecule Type: | Small molecule | |||
 |
Table 5: Structures of all compounds taken from PubChem |
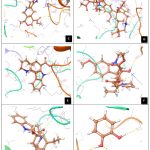 |
Figure 4: Showing docking pose of A. Secologanin, B. Serpentine, C. Ajmalicine, D. Vincristine, E. Tabersonine, F. Vindoline, G. Catharanthine, H. Catechol, I. Artemisinine |
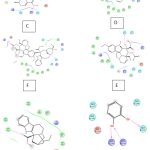 |
Figure 5: Showing protein ligand binding of A. Secologanin , B. Serpentine, C. Ajmalicine, D. Vincristine ,E. Tabersonine , F.Vindoline G. Catharanthine, H. Catechol ,I. Artemisinine |
Table 6: Details of the ligand protein interactions and participating amino acids
| S.No. | Name of Compound | Participating residues | Bond length(Ǻ) | Backbone/ Sidechain | Donar angle | Acceptor angle | Binding energy(Kcal/mol) |
| 1. | Secaloganin | Chain A: LYS 149 | 1.80881 | Backbone | 162.125 | 159.578 | -7.815 |
| Chain B: LYS 149Chain B :GLN 147 | 1.77052.00271 | SidechainBackbone | 129.785178.615 | 150.832 | |||
| 2. | Serpentine | Chain A: LYS 149 | 1.82641 | Backbone | 171.464 | -6.286 | |
| Chain A: LYS 149 | 2.02224 | Backbone | 148.236 | ||||
| 3. | Ajmalicine | Chain A: PRO 218Chain A: LYS 149 | 1.84322.38055 | BackboneBackbone | 131.325109.751 | 162.126 | -6.230 |
| Chain B:LYS 149 | 2.12241 | Backbone | 145.232 | ||||
| 4. | Vincristine | Chain A: LYS 149 | 2.16061 | Sidechain | 168.287 | -5.899 | |
| Chain B: GLN 147 | 2.36326 | Backbone | 141.045 | 123.219 | |||
| 5. | Vindoline | Chain B : GLN147 | 2.03706 | Backbone | 142.347 | 173.627 | -4.734 |
| In chain B: LYS 149 | 1.911687 | Sidechain | 124.405 | ||||
| 6. | Tabersonine | -4.99 | |||||
| 7. | Catharanthine base | LEU 221 | 2.09897 | Backbone | 153.093 | -3.847 | |
| 8. | Catechol | Chain A :LYS 149 | 2.003264 | Backbone | 158.85 | 149.116 | -3.662 |
| Chain A :LYS 149 | 2.02838 | Sidechain | 119.466 | ||||
| Chain B :GLN 147 | 2.02045 | Backbone | 151.825 | 162.144 | |||
| 9. | Artemisinine | Chain A :LYS 149 | 2.0194 | backbone | 108.321 | -3.226 |
Results
Protein FASTA sequence downloaded from NCBI, BLASTp done with protein datasets. The maximum similarity was found to be 97% which suggests that homology modelling method can be used. Swiss Model software was used for searching best matched templates and best identical template was then used for preparing AMA1 model. 6n87 was found to be best match sequence and used as template for building model. Modeller was also used for building the model. Best model was selected among various models prepared on basis of DOPE score. The best model showed DOPE score -20753.71484 (Fig. 2). The modelled structured was validated by superimposing it with best match templates on basis of Root Mean Square Deviation (RMSD) which was found to be 1.65764 (Fig. 3). The structure was validated with Ramachandran plot, the plot statistics reveal that 82.6% of the residues of modelled structure were in favourable region and additional 15% were in allowed region. Only 0.7% of the residues were in disallowed region. More than 97% of the structure was in allowed regions which shows the modelled protein was good enough to carry out virtual screening. For virtual screening by using Glide’s XP (extra precision) docking function. The results were analysed by Maestro’s Pose viewer and XP- visualize software. YAARA energy minimization serevr was used to perform energy minimization. Catharanthus roseus alkaloid fraction molecules were used as ligands against the modelled protein. Secaloganin showed Hydrogen bonding with chain A and chain B with Lysine 149 and Glutamine 147 (Fig.5 A). The shortest length H- bond is formed with Lysine 149 of Chain A of bond length 1.80881 Å. Serpentine showed two H-bond with Lysine 149 with varied bond lengths (Fig.5 B). Ajmalicine showing H-bond with Porline 218 and Lysine 149 of chain A, while one more H-bond is formed between Lysine 149 of chain B (Fig.5 C). Vincristine is a known anticancerous compound showing H-bond with Lysine 215 and Glutamine 147 of chain A and B (Fig. 5 D) respectively. Tabersonin showing hydrophilic interactions with Leucine 187, Phenylalanine 196, proline 190 of chain B and proline 220, proline 218 and glutamine 148 of chain A (Fig. 5 E). Vindoline showing H-bond with Lysine 149 and Glutamine 147 of chain B (Fig. 5 F). Catharanthine showing H-bond with Leucine 221 (Fig 5 G). Catechol forming H-bond with Lysine 149 of chain a and Glutamine 147 of chain B (Fig. 5 H). Artemisinine which is known drug of malaria shows one H- bond with Lysine 149 of chain A (Fig. 5 I).
Discussion
In this study alkaloid fraction of Catharanthus roseus were taken for docking against AMA protein. An extensive survey of literature related to Catharanthus roseus was conducted up to Janauary,2022. Catharanthus roseus has been of prime importance in the traditional medicine systems and has wide therapeutic applications for many centuries. During phytochemical investigation total of 344 compounds including monoterpene indole alkaloids, bisindole alkaloids, flavonoids, phenolic acids and volatile were found active against many diseases in Catharanthus roseus.35 AMA is a surface protein which is crucial for erythrocyte invasion of parasite, the AMA 1_PLAF8 structure is not revealed yet so protein modelling done through Swiss Model and structure validated by superimposing modelled protein with available templates. Catharanthus roseus is revealed for many therapeutic values but anti-malarial molecule is underinvestigation.36 Catharanthus roseus leaf and flower extract is proved to inhibit parasite 66% Megha et al, 2017. However, when pharmacology of Catharanthus roseus leaves and flowers was done, majority of alkaloids fractions were revealed. The major molecules present in Catharanthus roseus leaf and flower extract are Secologanin, Vindoline, tabersonine, Vincristine,Serpentine, Ajmalicine,Catharanthine 37 and a little fraction of catechol is been observed which is phenolflavanoid. Virtual screening is been done through Maestro and protein was simulated by YASARA energy minimization software. In virtual screening the best XP Gscore is shown by Secaloganine i.e. -7.815 Kcal/ mol while that of artemisinine is -3.226 Kcal/mol which is a known drug. Artemisinine are derived from extracts of weet wormwood and are well established for the treatment of malaria, including highly drug resistant dtrains. Its efficacy also extends to phyllogenetically unrelated parasitic infections such as schistosomiasis. In modelled protein Lysine 149 in chain A is participating in Hydrogen bond formation with secaloganin, Serpentine, Ajmalicine. Vincristine, Catechol and atremisinine. Chain Glutamine 147 is participating in hydrogen bond formation with Secaloganine, Vinscristine, Vindoline and catechol. Chain A proline 218 is participating in Hydrogen bond formation with Ajmalicine . The shortest Hydrogen bond if formed between Ajmalicine and proline 218 (1.8492 Ǻ), Chian A lysine 149 and Sepentine (1.82641 Ǻ) and Secaloganin and ChainA Lysine 149 (1.80881 Ǻ). Tabersonine is making Hydrophillic interactions with the protein’s Proline 190, Lutamine 147 pocket. Tabersonine is a monoterpenoid indole alkaloid with cytotoxic activity. It has a role as an antineoplastic agent and a metabolite.
Conclusion
The above data reveal that Secaloganine can be further taken explored as a drug molecule for antimalarial drug formulation, and chemoinformatics techniques can be used to future investigate the most stable form of the molecule in the future.
Acknowledgement
I would acknowledge the opportunity provided to me to learnt computational Biology techniques To Dr. Imran Siddiqui, Senior Scientist, Central Drug Research Institute, Lucknow, Uttar Pradesh, India.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
References
- Uhomoibhi P . Redoubling efforts to sustain seasonal malaria chemoprevention, Lancet Child Adolesc. Heal., 2022; 6( 3) :142–144. Doi- 10.1016/S2352-4642(22)00007-4
CrossRef - W. H. Organization,THE E-2020 ELIMINATING 2019 progress report, 2020.
- Naaz R, Anjum N, and Anjum G, is malaria an entirely preventable and treatable. 2020;8(1): 1248–1256. Doi – https://www.openacessjournal.primarydomain.in/abstract/104
- Senosiain G, Kana A, Singh I, Chourasia.S, Das B, Dodoo M , Theisen D, Peripheral merozoite surface proteins are targets of naturally acquired immunity against malaria in both India and Ghana. Infection and Immunity. 2020 ;88(4): 10-1128. Doi- https://doi.org/10.1128/iai.00778-19
CrossRef - Jalei A, Chaijaroenkul A, Bangchang K. Genetic Diversity of Plasmodium vivax Surface Ookinete Protein Pvs25 and Host Genes in Individuals Living along the Thai–Myanmar Border and Their Relationships with Parasite Density. Microbiology Research; 2024; 15(2): 693-707. Doi – https://doi.org/10.3390/microbiolres15020045
CrossRef - Avril M, Bernabeu M , Benjamin M , Brazier J , and Smith D.Interaction between Endothelial Protein C Receptor and Intercellular Adhesion Molecule 1 to Mediate Binding of Plasmodium falciparum : Infected Erythrocytes to Endothelial Cells.mBio. 2016; 7 (4): 00615-16. https://doi.org/10.1128/mbio.00615-16
CrossRef - Aly A , Vaughan A ,Kappe S, Malaria Parasite Development in the Mosquito and Infection of the Mammalian Host, Annu. Rev. Microbiol .2009;63( 1):195–221. Doi- https://doi.org/10.1146/annurev.micro.091208.073403
CrossRef - Fofana F, Wele M, Diabate O, Cisse C, Diawara A, Toure M, Djimde A. (2025). Advances in Malaria Vaccine Development Targeting Plasmodium falciparum Surface Proteins. Annual Research & Review in Biology, 40(9), 1-17. DOI: https://doi.org/10.9734/arrb/2025/v40i92300
CrossRef - Yan H, Feng J, Chen M. Structural modelling prediction of recombinant Plasmodium falciparum K13-F446I and K13-C580Y gene by AlphaFold method and heterologous expression in Spodoptera frugiperda 9 cells. Pathogens. 2022;11(11): 1271.Doi- https://doi.org/10.3390/pathogens11111271
CrossRef - Kardam R, Jangra A, Kumar D, Kumar V, & Sharma V. Promising Molecular Targets of Plasmodium Falciparum, the Deadliest Parasite and Their Synthetic Inhibitors: A Review. ChemistrySelect. 2025; 10(17): 202405253. Doi- https://doi.org/10.1002/slct.202405253
CrossRef - Mwakalinga S,Expression of a type B RIFIN in Plasmodium falciparum merozoites and gametes. 2012 ;11(1):1–12.Doi- http://www.malariajournal.com/content/11/1/429
CrossRef - Pei X,The ring-infected erythrocyte surface antigen ( RESA ) of Plasmodium falciparum stabilizes spectrin tetramers and suppresses further invasion. 2007 ; 110(3) : 1036–1042. Doi- https://doi.org/10.1182/blood-2007-02-076919
CrossRef - Sun S, Segev-Zarko L, Pintilie G, Kim C, Staggers Y, Schmid R, Boothroyd M, Cryogenic electron tomography reveals novel structures in the apical complex of Plasmodium falciparum.Mbio. 2024; 15(4): 02864-23. Doi- https://doi.org/10.1128/mbio.02864-23
CrossRef - Dijk V, “A Central Role for P48 / 45 in Malaria Parasite Male Gamete Fertility,” 2001 vol. 104, pp. 153–164. DOI: 10.1016/S0092-8674(01)00199-4
CrossRef - Goel S. RIFINs are adhesins implicated in severe Plasmodium falciparum malaria, Nat. Med. 2015; 21( 4): 314–317. Doi- https://www.nature.com/articles/nm.3812
CrossRef - Yu D, Yang S, Li Y, Hu W.Co-expression network with protein–protein interaction and transcription regulation in malaria parasite Plasmodium falciparum. Gene. 2013;518(1): 7-16.Doi- https://doi.org/10.1016/j.gene.2012.11.092
CrossRef - Pan H., Lu X, Ye D, Feng Y, Wan, J,Ye, J. The molecular mechanism of thrombospondin family members in cardiovascular diseases. Frontiers in Cardiovascular Medicine. 2024 ; 11(1): 1337586. Doi- https://doi.org/10.3389/fcvm.2024.1337586
CrossRef - Juillerat A,Lewit A, Guillotte M, Gangnard S, and Hessel A, “Structure of a Plasmodium falciparum PfEMP1 rosetting domain reveals a role for the N-terminal segment in heparin-mediated rosette inhibition,” 2011 vol. 108, no. 13, pp. 5243–5248. https://doi.org/10.1073/pnas.1018692108
CrossRef - Connor C , Collins K. A Novel RNA Binding Domain in Tetrahymena Telomerase p65 Initiates Hierarchical Assembly of Telomerase Holoenzyme. 2006; 26( 6) :2029–2036. Doi-https://doi.org/10.1128/MCB.26.6.2029-2036.2006
CrossRef - Das S. Processing of Plasmodium falciparum Merozoite Surface Protein MSP1 Activates a Spectrin-Binding Function Enabling Parasite Egress from RBCs Article Processing of Plasmodium falciparum Merozoite Surface Protein MSP1 Activates a Spectrin-Binding Function Enabling Parasite Egress from RBCs. Cell Host Microbe. 2015;18(4):433–444. Doi-https://doi.org/10.1016/j.chom.2015.09.007
CrossRef - Yan H, Feng J, Chen M. Structural modelling prediction of recombinant Plasmodium falciparum K13-F446I and K13-C580Y gene by AlphaFold method and heterologous expression in Spodoptera frugiperda 9 cells. Pathogens.2022;11(11):1271. Doi-https://doi.org/10.3390/pathogens11111271
CrossRef - Adams J, Thrombospondins: Multifunctional Regulators of Cell Interactions. Annu. Rev. Cell Dev. Biol. 2001; 17( 1): 25–51, Doi- https://doi.org/10.1146/annurev.cellbio.17.1.25
CrossRef - Hira F, Islam A,Uddin M, Immunomodulatory and Pharmacological Properties of Catharanthus Roseus: A Comprehensive Review. Letters in Applied Nanobioscience. 2025;14(4):91-106.Doi- https://doi.org/10.33263/LIANBS142.091
- Anvikar A, In vitro assessment of drug resistance in Plasmodium falciparum in five States of India. Indian J. Med. Res. 2012;135(4): 494–499. https://journals.lww.com/ijmr/fulltext/2012/35040 /in_vitro_assessment_of_drug_resistance_in.8.aspx
- Chanda E, Integrated vector management : a critical strategy for combating vector-borne diseases in South Sudan. Malaria journal. 2013;12(1):369-378. Doi- http://www.malariajournal.com/content/ 12/1/369
CrossRef - Rajput M, Nair V, Chauhan A, Jawanjal H, Dange V, Evaluation of Antidiarrheal Activity of Aerial Parts of Vinca major in Experimental Animals. Middle East J Sci Res. 2011; 7(5): 784–788. https://www.researchgate.net/profile/Mithun-Rajput/publication/267228391_Evaluation_of_ Antidiarrheal_Activity_of_Aerial_Parts_of_Vinca_major_in_Experimental_Animals/links/546b61b80cf2397f7831bdd3/Evaluation-of-Antidiarrheal-Activity-of-Aerial-Parts-of-Vinca-major-in-Experimental-Animals.pdf
- Takthsingji S and hospital G.evaluation of hypolipidemic activity of leaf juice of catharanthus roseus ( linn .) G . Donn . In guinea PIGS. Acta Poloniae. 2011; 68( 6): 927–935. https://ptfarm.pl/pub/File/Acta_Poloniae/2011/6/927.pdf
- Mishra J, Verma N, “A brief study on Catharanthus Roseus : A review,” 2017 vol. 2, no. 2, pp. 20–23. https://www.researchgate.net/profile/Navneet-Verma/publication/319007421_A_brief_study_ on_Catharanthus_Roseus_A_review/links/598ad641aca272435857d010/A-brief-study-on-Catharanthus-Roseus-A-review.pdf
- Ramya S, Govindaraji V, Kannan K, Navaneetha, Jayakumararaj R. In Vitro Evaluation of Antibacterial Activity Using Crude Extracts of Catharanthus. Ethnobot. Leafl. 2008; 12 (1) : 1067–1072. Doi- https://opensiuc.lib.siu.edu/ebl/vol2008/iss1/140/
- Salman M., Qadeer F, Pharmacological Actions and Therapeutic Potential of Trigonella foenum-graecum L. Fenugreek: Biology and applications. 2021;981(16): 523-537. Singapore: Springer Singapore. https://link.springer.com/chapter/10.1007/978-981-16-1197-1_22
CrossRef - Nayak B, Anderson M, and Pereira P. Evaluation of wound-healing potential of Catharanthus roseus leaf extract in rats. Fitoterapia. 2007; 78(7) : 540–544. Doi- https://doi.org/10.1016/j.fitote. 2007.06.008
CrossRef - Friesner R, Extra Precision Glide: Docking and Scoring Incorporating a Model of Hydrophobic Enclosure for Protein−Ligand Complexes. J. Med. Chem. 2006; 49( 21): 6177–6196. Doi- https://pubs.acs.org/doi/abs/10.1021/jm051256o
CrossRef - Krieger E, Joo K, Lee J, Lee J, Raman S, Thompson J, Karplus K. Improving physical realism, stereochemistry, and side‐chain accuracy in homology modeling: four approaches that performed well in CASP8. Proteins: Structure, Function, and Bioinformatics, 2016; 77(S9): 114-122. Doi- https://doi.org/10.1080/07391102.2018.1444510
CrossRef - Kumar S, Singh B, Singh R. Catharanthus roseus (L.) G. Don: A review of its ethnobotany, phytochemistry, ethnopharmacology and toxicities. J. Ethnopharmacol. 2022; 284 (2) :1016-1026.Doi- https://doi.org/10.1016/j.jep.2021.114647
CrossRef - Ravikumar S, Inbaneson J, Suganthi P, In vitro antiplasmodial activity of ethanolic extracts of South Indian medicinal plants against Plasmodium falciparum, Asian Pacific J. Trop. Dis.2012: 2(3): 180–183. Doi- https://doi.org/10.1016/j.jep.2021.114647
CrossRef - Joshi S, Huo C, Budhathoki R, Gurung A, Bhattarai S, Sharma K, Parajuli N. HPLC-ESI-HRMS/MS-Based Metabolite Profiling and Bioactivity Assessment of Catharanthus roseus. Plants.2025; 14(15): 2395-2410. https://doi.org/10.3390/plants14152395
CrossRef
Accepted on: 01-12-2025
Second Review by: Dr. Bidyutparna Bhattacharya Kundu
Final Approval by: Dr. Ali Elshafei









