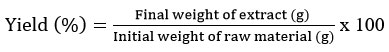Effect of Different Drying Methods on Phytochemicals and Antioxidant Potential of Cymbopogon citratus
1School of Biological Engineering and Life Sciences, Shobhit Institute of Engineering and Technology, Meerut, India.
2Research and Development Cell, Swami Vivekanand Subharti University, Subhartipuram, Meerut, India.
3Depatartment of Neutraceutics, Institute of Nuclear Medicine and Allied Sciences, Defence Research and Development Organization, Delhi, India.
Corresponding Author Email: amarprakashgarg@yahoo.com
DOI : http://dx.doi.org/10.13005/bbra/3466
ABSTRACT:Phytochemicals are naturally occurring bioactive chemical compounds produced by plants and play a significant role in preventing chronic diseases like heart diseases, cancer and neurodegenerative disorders. The aim of the current study was to investigate the effects of different drying methods [sun-dried (SD), shade-dried (ShD), oven-dried (OD) and microwave-dried (MD)] on phytochemicals and antioxidant properties of leaves of Cymbopogon citratus. Following the drying process an aqueous extract of the leaves of C. citratus was prepared. Qualitative analysis of extract showed the presence of alkaloids, flavonoids, phenolics, Phytosteroids, tannins, carbohydrates, glycosides, terpenoids, proteins and amino acids. The range of total protein content was 122.43 ± 4.15 to 314.91 ± 5.11 µg BSAE / mg dry extract, carbohydrate content 147.75 ± 8.01 to 313.17 ± 3.00 µg GE / mg dry extract, phenol content 107.01 ± 7.30 to 155.94 ± 4.94 µg GAE / mg dry extract and flavonoid content 176.02 ± 5.85 to 340.84 ± 6.00 µg QE / mg dry weight in aqueous extract (SD, ShD, OD and MD) of C. citratus. Moreover, MD leaves extract showed the strong potential against DPPH free radicals with IC50 = 222.81 ± 7.57 µg/ml, the highest reducing capacity as evaluated by the ferric reducing antioxidant assay and the highest nitric oxide scavenging activity with IC50 = 141.98 ± 6.65 µg/ml. Overall, microwave drying (MD) was found better than sun drying, shade drying and oven drying methods for leaves of C. citratus based on higher availability of bioactive compounds and preserved.
KEYWORDS:Antioxidants; Cymbopogon citratus; Drying methods; Free radicals; Oxidation stress; Pphytochemicals
Introduction
Free radicals are molecules or atoms contain unpaired electrons, making them highly reactive and unstable in chemical interactions with other molecules. They originate by three elements viz oxygen, giving rise to reactive oxygen species (ROS), nitrogen, giving rise to reactive nitrogen specie (RNS) sand sulfur, giving rise to reactive sulfur species (RSS).1 First time free radicals were identified in the 1950s in biological system.2 Free radicals are generated from two sources:- Endogenous and Exogenous sources (Fig 1).3 They are the cause of oxidative stress which is defined as the imbalance between the ROS/RNS and cellular oxidant defenses.4 It leads to cell death (apoptosis), oxidative stress associated with many diseases including cancer diabetes, neurodegenerative diseases, hypertension, aging, and cardiovascular diseases etc.5-6 Plants are rich sources of natural antioxidants such as polyphenols, flavonoids, anthrocyanin, tannins and, can be used in many forms such as fresh and raw or dry plant materials, in the form of tea, and extract form with different solvents7-8 which play important role in neutralizing free radicals or reduce oxidative stress and prevent different human diseases.9-11
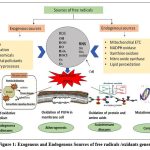 |
Figure 1: Exogenous and Endogenous Sources of free radicals /oxidants generation.
|
Cymboposgon citratus
Lemon grass (also known as citronella) is a common name of Cymbopogon citratus, a member of Poaceae family. 140 species (apprx.) of Cymbopogon are widely spread, grow across the semi-temperate and tropical regions of Asian, American and African continents. Lemon grass is also known as aromatic grass due to its volatile oils.12 Leaves of lemon grass are used as sedatives for the central nervous system.13 Several bioactive compounds of Lemon grass have medicinal value with various ethnopharmacological applications.14 According to WHO, more than two-thirds of the population depend on herbal medicine that an important part of healthcare industries in developing countries.15 Scientific name of lemon grass Cymbopogon derived from the Greek words “Kymbe” (boat) and “pogon” (beard), which refers to the flower spike arrangement.16
Several studies have shown the presence of carbohydrates, fats, proteins, minerals, fibres and other bioactive components in lemon grass that play a vital role in human health. These can be classified under several categories including alkaloids, flavonoids, tannins, terpenoids, phenols and saponins. Furthermore, phlobotannins, anthraquinones, steroids and cardiac glycosides were found in the extract of lemon grass.17-18 Lemon grass has been used in the treatment of cough, flu, fever, leprosy, malaria, digestive disorders and many other diseases.19-20 Infusion of lemon grass are also consumed as beverage for treatment of the inflammatory diseases due to its therapeutic properties.21 The aqueous extract of lemon grass has a potential to reduce the level of blood sugar and cholesterol.22 Lemon grass has also been utilized by people of the subcontinent to cure rheumatic pain, nausea, headaches and cold. 23 Herbals teas with lemon grass have been commonly used as a pleasant tonic, beverage and to cure fever hacking and cold.24
 |
Figure 2: Flavonoids compounds are abundant in leaves of C. citratus. |
The aim of current study was to find out the effect of different drying methods [(sun-dry (SD), shade dry (ShD), oven-dry (OD) and microwave-dry (MD)] on the qualitative and quantitative changes with high-quality bioactive molecules or materials that can be used for various applications.
Materials and Methods
Chemicals
All analytical-grade chemicals were used in this study including Benedict’s reagent, Folin-Ciocalteu reagent, benzene (C6 H6), ammonia (NH3), lead acetate Pb (CH3COO)2, Iodine solution (I2), ninhydrin (C9H6O4), nitric acid (HNO3), chloroform (CHCl3), tannic acid (C76H56O46), ferric chloride (FeCl3), potassium iodide, (KI), α-naphthol (C10H8O), sodium chloride (NaCl), sodium nitroprusside (SNP), sodium carbonate (Na2CO3), sodium hydroxide (NaOH), gallic acid (C7H6O5), quercetin (C15H10O7), ferric sulphate (FeSO4), potassium ferricyanide K4[FE(CN)6], trichloroacetic acid (TCA), and L-ascorbic acid were obtained from Mumbai, Sisco Research Laboratories Pvt, Ltd, India. Merck Life Science, Pvt, Ltd, Mumbai, India, provided the following: 1,1-diphenyl-2-picrykhydrazyl radical (DPPH), acetic anhydride (C4H6O3), aluminium chloride (AlCl3), sodium bicarbonate (Na2CO3), absolute ethanol, methanol and hydrochloric acid (HCl) were used.
Collection of leaves of C. citratus
In May 2022, young and soft leaves of C. citratus were obtained from the garden of Shobhit Deemed-to-be University, Modipuram, Meerut, were cut in to small pieces and washed with tap water followed by distilled water to remove dust particles present on the surface of leaves.
Drying process of leaves
Samples were divided into four equal parts and dried using different drying methods
Sun drying (SD): 250 g leaves were spread on a tray and dried in direct sun light. Weight of the samples were recorded after every one hour and continuously dried till constant weight.
Shade drying (ShD): 250 g leaves were spread on blotting paper and dried in shade at room temperature (25-27oC) till constant weight.
Oven drying (OD): 250 g leaves dried in hot air oven (UFB 500, Memmert, Germany) at 50 ± 5o C for 12 hours and weighed after every one hour, dried till constant weight.25
Microwave drying (MD): 250 g leaves were placed on glass plate in microwave (Sharp R-248E; 750W) for 4 minutes and weight was recorded after every 30 second till constant weight.26
Thereafter, samples were coarsely powdered using mortar and pestle followed by in electric grinder to obtain fine powder, stored in airtight container at room temperature.
Preparation of extract (s) and percentage of extract recovery
50 g powder from each dried sample was mixed with boiled distilled water in separate beakers, placed on magnetic stirrer hot plate by continuous swirling for 30 minutes at 150 ± 2oC and 650 rpm. After that each extract was filtered using house hold tea strainer and finally with filter paper. All extracts were concentrated using rotavapor evaporator (BUCHI R-300, BUCHI Labortechnik, Flawil, Swetzerland) and thereafter lyophilized (Lyophilizer (-55oC, Sub-Zero Lab Instruments, Chennai, India)27 with minor modification, and stored in airtight glass container at 4oC for further analysis. Percentage of yield of samples was calculated using following formula:
Ash content analysis
Two g of sample was heated in muffle furnace for 6 hours at 550 ± 5oC. Ash was obtained free from carbon and cooled in a desiccators and weighed 28. Percentage was calculated using following formula:
Qualitative photochemical analysis
Qualitative photochemical analysis for Alkaloids, flavonoids, phenolics, steroids, tannins, carbohydrate, proteins and amino acids, terpenoids, glycolsides and saponin was made 29-30.
Tests for Alkaloids
Wagner’s reagent test
Crude extract was mixed with 5 mL of HCL (1%) and gently stirred and filtered, 1 mL of filtrate was added to Wagner’s reagent resulting in reddish-orange precipitate that confirmed the presence of alkaloids.
Tannic acid test
Two drops of Hager’s reagent were mixed with 1 mL of above filtrate and the formation of light-yellow precipitate indicated the presence of alkaloids.
Tests for Flavonoids
Alkaline reagent test
In 1 mL of extract added few fragments of magnesium ribbon and then conc. Hydrochloric acid was added drop wise, observation of a light pink colour confirmed flavonoids.
Shinoda test
1 mL of 2% aqueous NaOH (w/v) was mixed with 1 mL of extract, immediate observation of intense yellow colour which turned to colourless after addition of few drops of dilute HCl indicated the presence of flavonoids.
Test for Phenolic compounds
Ferric chloride test
Two or three drops of 5% ferric chloride solution (w/v) were gently mixed with 1 mL of extract. A dark green colour was observed that confirmed phenolic.
Iodine test
One or two drops of diluted 2% iodine solution (w/v) were mixed with 2 mL of extract, formation of transient red colour confirmed the presence of phenolic compounds.
Test for Phytosterols
Salkowski test
1 mL of extract was treated with 1 mL of chloroform and a few drops of concentrated sulfuric acid were added carefully, test tube was vigorously shaken and allowed to stand for 2 minutes. Observance of reddish colour in the lower layer of chloroform confirmed the presence of phytosterols.
Liebermann-Burchard test
2 mg of extract was dissolved in 2 mL of acetic anhydride, 1 mL of this solution was treated with two drops of concentrated sulfuric acid carefully. Observance of brown ring at the interphase and turned the upper layer a dark green colour indicated the presence of phytosterols.
Test for Carbohydrates
Molisch’s test
1 mL of extract was mixed with 1 mL of Molisch’s reagent (a solution of α-naphthol in ethanol) then added one mL of concentrated sulfuric acid carefully. Formation of a purple ring formation at the interface between two layers confirmed the presence of carbohydrates.
Benedict’s reagent test
2 mL of extract treated with Benedict’ reagent and boiled, precipitate of reddish-brown (brick red) colour settles down at the bottom of the test tube indicated the presence of carbohydrates.
Test for Glycosides
Killer Kilani test
1 mL extract was treated with glacial acetic acid and few drops of ferric chloride (5%) and two drops of concentrated sulfuric acid. Reddish- brown colour was observed between two layers, confirmed glycoside was present.
Modified Borntrager’s test
1 mL of extract was mixed with 5 % solution of ferric chloride and boiled for 1 minute in hot water bath, cooled at room temperature and treated with an equal volume of benzene and ammonia solution. Blood-red colour appeared in the ammonia layer indicated the presence of glycosides.
Test for Tannins
Ferric chloride test
1 mL of extract mixed with 1-2 drops of ferric chloride solution (5 %), appearance of brownish-green colour confirmed the presence of tannins.
Lead acetate test
1 mL of extract was treated with 2 drops of lead acetate solution (10 %), detection of creamy precipitate indicated the presence of tannins.
Test for Proteins and amino acids
Ninhydrin test
2 mL of extract was treated with 5 % ninhydrin solution which prepared in acetone and kept in hot water bath for 5 minutes, the development of violet colour was indication of proteins and amino acids.
Xanthoproteic test
1 mL of extract was mixed with 2-3 drops of conc. Nitric acid. A yellow colour confirmed the presence of proteins.
Test for saponins
Foam test
2 mL extract was taken in a test tube, mixed with 2 mL of distilled water, vortexed for 2 minutes. Formation of approximately 2 cm. thick layer in the test tube which was stable for 10-15 minutes, confirmed the presence of saponins.
Olive oil test
2 mL of extract was mixed with three drops of olive oil and vortexed. Formation of soluble emulsion was indication of presence of saponins.
Test for Terpenoids
2 mL of extract was treated with 2 mL of chloroform (CHCl3) and then added conc. sulfuric acid along the side of test tube carefully. A reddish-brown layer observed in the inner phase of this solution, confirmed the presence of terpenoids.
Quantitative Biochemical Analysis
The protein content
The protein content was determined using Lowry’s method 31 with few modifications. About 60 µl of extract was mixed with 40 µl of distilled water and 100 µl of freshly prepared Lowry’s reagent was added, allowed to stand at room temperature for 10 minutes. Thereafter, 50 µl of Follin-Ciocalteu reagent (1:10) was added and further incubated for 30 minutes at room temperature. The absorbance of test extract was measured at 650 nm against blank. The protein content of extract was expressed as µg of BSA (Bovine Serum Albumin) equivalent per mg of dried extract.
The Carbohydrates content
The carbohydrates content was measured using Anthrone reagent method 32. 100 mg of extract was boiled with 5.0 mL of 2.5 N HCl in water bath and cooled at room temperature, then centrifuged at 3000 rpm for 5 minutes, 0.005 µl supernatant was taken to which distilled water was added make up the volume to 1 mL and suspended solid was discard. 2 mL of Anthrone reagent (Anthrone reagent was dissolved in ice cold 95 % sulfuric acid in 2:1 ratio) was added in this mixture and allowed to heat on a water bath at 100°C for 5-10 minutes and cooled at room temperature. The absorbance was measured at 630 nm. Different concentration of Glucose was used as standard calibration curve. The carbohydrate content of extract was expressed in µg of Glucose equivalent (GE) per mg of dried extract.
Quantitative Phytochemical Analysis
Total Polyphenolic content (TPC)
The total polyphenol content (TPC) was determined using Folin-Ciocalteu method described by Konicc et al., 2010 with minor modification.33 In brief, the extract (1mg/mL) was treated with Folin-Ciocalteu reagent (diluted with distilled water 1: 10) and 20 % sodium bicarbonate in 1:1:1 ratio, mixed thoroughly kept at room temperature for 20 minutes for development of colour. Absorbance was measured at 760 nm. Gallic acid was used as a standard calibration curve. Total polyphenolic content (TPC) was expressed as µg of gallic acid equivalent (GAE) per mg of dried extract.
Total Flavonoid content (TFC)
The total flavonoid content (TFC) of extract was estimated.34 The extract and AlCl3 solution (2 %) were mixed together in 1:1 ratio and incubated for 1 hour at 20°C for development of yellow colour. Absorbance was measured at 420 nm. Quercetin was used as standard calibration curve. Total flavonoid content (TFC) of the extract was expressed as µg of quercetin equivalent (QE) per mg of dried weight.
Antioxidant Analysis
Scavenging activity against DPPH (2, 2- diphenyl-1-hydrazyl) free radical
The free radical scavenging potential of aqueous extract of C. citratus was determined using DPPH assay 35 with minor modification. Prepared a DPPH solution in 95 % methanol (0.004 %) and stored in dark. Different concentrations of extract were mixed with DPPH solution in 1:1 ratio and incubated for 30 minutes at room temperature in dark for development of colour. The absorbance was measured at 517 nm. Ascorbic acid was used for standard calibration curve. Percentage of DPPH inhibition was calculated using the following formula:
Here, A0 is the absorbance of the control, and A1 is the absorbance of extracts / standard.
IC50 value (half maximal inhibitory concentration) was calculated using linear regression analysis to quantify the potency of an antioxidant.
FRAP (Ferric-Reducing / Antioxidant Power Assay)
The ferric reducing power of the aqueous extract was determined using the method of Shang et al., 2018 with some modification.36 Briefly, 100 µl extract / standard at different concentrations were mixed with 250 µl of sodium phosphate buffer (pH 6.6) and 250 µl potassium ferricyanide (1%). The reaction mixture was incubated at 50 ᵒC for 20 minutes, then 250µl trichloroacetic acid (10 %) was mixed with this mixture and centrifuged at 4000 rpm for 10 minutes. 50 µl supernatant was withdrawn from the mixture and was mixed with 50 µl of deionized water and 50 µl of ferric chloride (0.1%) solution and further incubated at room temperature for 20 minutes. The absorbance was measured at 700 nm. Ascorbic acid was used as reference.
Nitric oxide radical scavenging activity
The nitric oxide scavenging ability of the aqueous extract was determined using the method of Green et al., 1996 with slight modification.37 0.5 µl of extract/standard at different concentrations was mixed with 1 mL of 10 mM sodium nitroprusside (prepared in 0.025 M phosphate buffer pH 7.4). The mixture was incubated for 150 minutes at room temperature in dark. After that, 0.1 mL mixture was withdrawn from incubated mixture and mixed with 0.1 mL of Griess reagent (1 % sulphanilamide reagent and 0.1 % napthylenediamine dichloride, prepared in 5 % phosphoric acid) and allowed to stand for 30 minutes at room temperature for development of pink colour. Ascorbic acid was used as reference. The absorbance was measured at 540 nm. The percentage of nitric oxide inhibition was calculated using following formula:
Here, A0 is the absorbance of the control, and A1 is the absorbance of extracts / standard. IC50 value was calculated using linear regression analysis to quantify the potency of antioxidant.
Functional group analysis
Functional groups were analysed using ATR-FTIR (Attenuated Total Reflection Fourier-Transform Infrared spectroscopy) (PerkinElmer Frontier) in a scanning range of 450 – 4000 cm-1, scanning 16 times at a spectra resolution of 4 cm-1.
Statistical analysis
In this study, all treatments and analysis were performed in triplicate for each sample and all results are presented as mean ± standard deviation. Graph Prism Pad software, version 5 was used to analyse statistical data and P < 0.05 was considered statistically significant. One-way ANOVA along Tukey’s test with 95% confidence interval was conducted to show the statistical significance difference between means.
Results
Physical nature and Yield percentage
Table 1 showed colour, form, solubility (colour) and turbidity of the extract and maximum yield percentage obtained from MD extract (17.64 ± 0.26 %) and minimum from ShD extract (15.02 ± 0.07 %) by maceration method while SD and OD extracts revealed 17.22 ± 0.24 % and 15.02 ± 0.07 % of yield respectively.
Ash content (%)
MD extract revealed 8.00 ± 0.00 % ash content which was less compared to SD, ShD and OD extract, 7.00 ± 0.03 %, 5.5 ± 0.03 % and 7.00 ± 0.05 % respectively (Table 1). No significant difference observed.
Table 1: Physical and physiochemical analysis
| Sample | Aqueous extract of leaves of C. citratus | ||||
| Drying Process | SD(sun-dried) | ShD(shade-dried) | OD(oven-dried) | MD (microwave-dried) | P value |
| Appearance (colour) | Light brown | Dark brown | Light brown | Dark brown | – |
| Appearance (form) | Powder | Powder | Powder | Powder | – |
| Solubility (colour) | Light brown | Light brown | Light brown | Light brown | – |
| Solubility (turbidity) 20 mg/ml, H2O | Hazy | Hazy | Hazy | Slightly Hazy | – |
| % Yield (w/w) | 17.22 ± 0.24 | 15.02 ± 0.07* | 16.46 ± 0.45 | 17.64 ± 0.26$& | < 0.0001 |
| Ash content (%) | 7.00 ± 0.03 | 5.5 ± 0.03 | 7.00 ± 0.05 | 8.00 ± 0.00 | 0.0728 |
Physical and physiochemical properties of aqueous extract of C. citratus under different drying methods
Qualitative Phytochemicals analysis
The phytochemical analysis of aqueous extract of C. citratus (SD, ShD, OD and MD) tested are summarized in Table 2 that shows the presence of various phytochemical like alkaloids, flavonoids, phenolic compounds, phytosterols, carbohydrates, glycosides, tannins, proteins and amino acids and terpenoids in all extract however saponins are not presence.
Table 2: Qualitative phytochemicals analysis
| Sample | Aqueous extract of leaves of C. citrates | |||
| Phytochemicals | SD (Sundried) | ShD(Shade dried) | OD(oven dried) | MD(microwave dried) |
| Alkaloids | ||||
| Wagner’ test | ++ | ++ | +++ | +++ |
| Tannic acid test | ++ | ++ | ++ | +++ |
| Flavonoids | ||||
| Alkaline test | ++ | + | ++ | ++ |
| Lead acetate test | ++ | + | ++ | +++ |
| Phenolics | ||||
| Ferric chloride test | ++ | ++ | +++ | +++ |
| Iodine test | ++ | ++ | +++ | +++ |
| Steroids | ||||
| Salkowski test | ++ | + | ++ | +++ |
| Liebermann-Burchard reaction | ++ | + | ++ | +++ |
| Carbohydrates | ||||
| Molisch test | ++ | ++ | ++ | +++ |
| Benedict’ test | ++ | ++ | ++ | +++ |
| Glycosides | ||||
| Keller-Killani test | ++ | ++ | +++ | +++ |
| Modified Borntrager’s test | + | ++ | +++ | +++ |
| Tannins | ||||
| Ferric chloride test | ++ | ++ | +++ | +++ |
| Lead acetate test | ++ | ++ | +++ | +++ |
| Protein and amino acids | ||||
| Ninhydrin test | + | ++ | ++ | +++ |
| Xanthoproteic test | + | ++ | ++ | +++ |
| Saponin | ||||
| Foam test | – | – | – | – |
| Olive test | – | – | – | – |
| Terpenoids | ||||
| Salkowski’s test | ++ | + | ++ | +++ |
Qualitative phytochemical screening of leaves extracts of C. citratus under different drying methods, indicating presence or absence of different plants metabolite with (-) indicates absence, (+) indicates positively (++) indicates strong positively in test while (+++) indicates very strong positive in test.
Quantitative Biochemical Analysis
The Protein content
Present study revealed that, protein content was significantly affected by the different drying methods. Protein content of different extracts (SD, ShD, OD and MD) was analysed between 122.43 ± 4.15 to 314.91 ± 5.11 µg BSAE / mg dry extract (Table 3) wherein MD extract showed higher protein content 314.91 ± 5.11 µg BSAE / mg dry extract and ShD lowest 122.43 ± 4.15 µg BSAE / mg dry extract. SD and OD extracts contained 202.06 ± 8.08 µg BSAE / mg and 231.91 ±3.51 µg BSAE / mg dry extract respectively.
The carbohydrate content
The carbohydrate content was found 313.17 ± 3.00 µg GE / mg dry in MD extract which was higher as compared to all (Table 3). While ShD extract showed least amount of carbohydrate content (147.75 ± 8.01 µg GE / mg dry extract) and SD and OD contained 222.67 ± 2.49 µg GE / mg dry extract and 243. 01 ± 7.18 µg GE / mg dry extract respectively.
Table 3: Quantitative Biochemical and Phytochemical Analysis
| Sample | Aqueous extract of leaves of C. citrates | ||||||
| Drying treatment | SD(sun dried) | ShD(shade dried) | OD(oven dried) | MD(microwave dried) | P value | ||
| The protein content (µg BSAE / mg dry wt.) | 202.06 ± 8.08 | 122.43± 4.15* | 231.74 ±3.51# | 314.91 ± 5.11@$& | < 0.0001 | ||
| The carbohydrate content (µg GE / mg dry wt.) | 222.67 ± 2.49 | 147.75 ± 8.01* | 243.01 ± 7.18# | 313.17 ± 3.00@$& | < 0.0001 | ||
| Total phenolic content (µg GAE / mg dry wt.) | 117.50 ± 2.96 | 107.01 ± 7.30* | 141.22 ± 5.98# | 155.94 ± 4.94@$& | < 0.0001 | ||
| Total flavonoid content (µg QE / mg dry wt.) | 207.52 ± 5.79 | 176.02 ± 5.85 | 278.85 ± 4.53**# | 340.84 ± 6.00@$& | < 0.0001 | ||
The results are expressed as mean and standard deviation. Key: BSAE (Bovine Serum Albumin Equivalent), GE (G lucose Equivalent), GAE (Gallic Acid Equivalent) and QE (Quercetin Equivalent). Significant changes were obtained using one Way ANOVA Post Tukey test. Inter-group comparisons were represented as *p< 0.05 SD vs ShD; **P<0.05 SD vs OD; @p<0.05 SD vs MD; #P<0.05 ShD vs OD; $P<0.05 ShD vs MD; &P<0.05 OD vs MD.
Quantitative Phytochemical Analysis
Total Polyphenol content
In this study, range of total polyphenol content lying in between 107.01 ± 7.30 µg GAE /mg dry extract to 155.94 ± 4.94 µg GAE /mg dry extract was found. ShD extract revealed 107.01 ± 7.30 µg GAE / mg dry extract which was less than SD extract (117.50 ± 2.96 µg GAE /mg dry extract), OD extract (141.22 ± 5.98 µg GAE /mg dry extract and MD extract (155.94 ± 4.94 µg GAE /mg dry extract) (Table 3). The results of TPC were found in increasing order ShD < SD < OD < MD.
Total Flavonoid content
Total flavonoid content of MD extract was found 340.84 ± 6.00 µg QE / mg dry extract which was significantly higher than SD extract (207.52 ± 5.78 µg QE / mg dry extract), ShD extract (176.02 ± 5.85 µg QE / mg dry extract and OD extract (278.85 ± 4.53 µg QE / mg dry weight (Table 3). Whereas ShD extract exhibited lowest total flavonoid content as compared to all extracts clearly showing the effect of drying method on biochemical analysis.
Antioxidant Analysis
Scavenging activity against DPPH (2, 2-Diphenyl-1-picrylhydrazyl) free radical
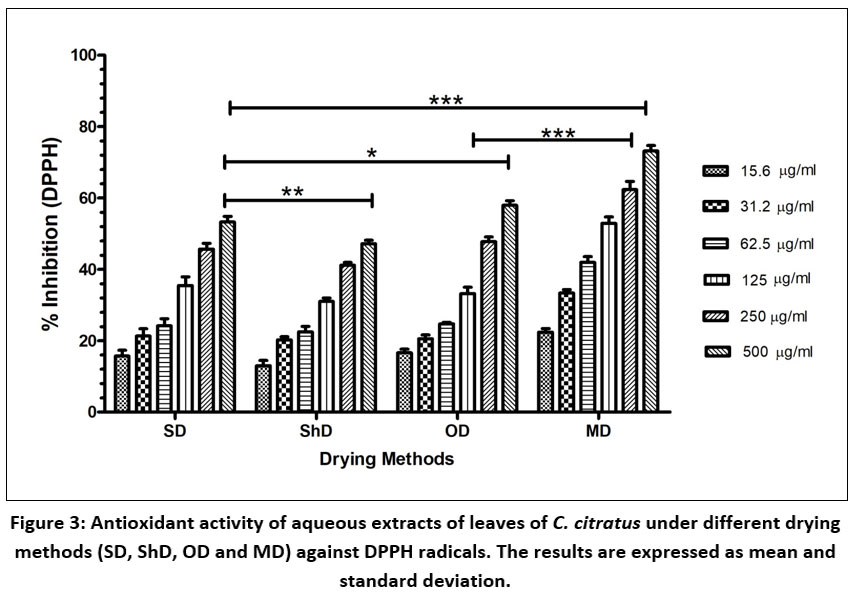
DPPH radical scavenging assay was conducted using all four extracts (SD, ShD, OD and MD) of C. citratus at different concentration (15.6, 31.2, 62.5, 125, 250 and 500 µg/ml) (Fig. 3). The dose dependent activity of extracts (SD, ShD, OD and MD) was observed with an increase in concentration (Fig3). SD extract showed 15.71 ± 2.89 % inhibition of free radical at 15.60 µg /ml concentration which increased till 53.26 ± 2.78 % at 500 µg/ml concentration that was near to 125 µg/ml concentration of MD extract. In case of ShD extract, highest free radical scavenging activity 47.21 ±1.68 % was noted at 500 µg/ml followed by 41.15 ± 1.40 % at 250 µg/ml, 31.06 ±1.58 % at 125 µg/ml, 22.51 ± 2.63 % at 62.50 µg/ml, 20.26 ± 1.51 % at 31.20 µg/ml and the minimum 13.01 ± 2.57 % free radical scavenging activity at 15.60 µg/ml. In OD extract, 20.56 ± 1.80 % scavenging activity was observed at 31.20 µg/ml which increased to 57.94 ± 2.20 % at 500 µg/ml. Free radical scavenging activity of MD was observed significantly highest (73.19 ±2.67 %) as compared to SD, ShD and OD extract at 500 µg/ml concentration. Amongst all four extracts, MD extract showed best free radical scavenging potential against DPPH free radical as compare to corresponding concentration of SD, ShD and OD extracts.
Moreover, MD extract showed least IC50 (190.99 ± 9.13 µg/ml) value which indicated strong free radical scavenging potential and ShD extract showed high IC50 value which indicated weak free radical scavenging potential. IC50 value of SD extract and OD extract showed 401.22 ± 5.60 µg/ml and 361.75 ± 4.40 µg/ml respectively (Table 4).
Reducing Power
The principle of reducing assay is based on electron donating capacity. The reducing power of extract/ standard solution were evaluated from their ability to reduce Ferric (Fe3+) to Ferrous ion (F2+) by the change of colour (yellowish to various shades of green colour). The intensity of green colour is directly proportional to the concentration of antioxidant present in the extract or standard. The result revealed that MD extract possessed strong ability to reduce the Ferric to Ferrous ion at all concentration as compared to all concentration of SD, ShD and OD extracts (Fig. 4). Wherein, ShD extract exhibited significantly weak reducing potential than SD, OD and MD extract. Moreover, the result represented in increasing order ShD < SD < OD < MD extract.
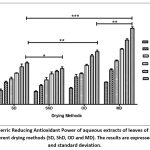 |
Figure 4: Ferric Reducing Antioxidant Power of aqueous extracts of leaves of C. citratus under different drying methods (SD, ShD, OD and MD). The results are expressed as mean and standard deviation. |
Nitric Oxide (NO) Scavenging Activity
Sodium nitroprusside produced nitric oxide in the extract at different concentration that reacts with oxygen to form nitrite ions, which can be detected using Griess reagent at 540 nm. By scavenging the nitic oxide, the extracts reduce the formation of nitrite ions at various concentrations. Fig. 5 shows the capacity of extract to scavenge nitric oxide at various concentrations. SD extract exhibited 5.97 ± 1.77 % scavenging potential at 7.8 µg/ml which was increased to 51.00 ± 1.65 % at 250 µg/ml while 4.25 ± 0.65 % scavenging potential at 7.8 µg/ml was observed for ShD extract which was increased to 43.36 ± 2.77 at 250 µg/ml. ShD extract showed weak scavenging potential at all tested concentrations in comparison to all extracts. In case of OD extract, 6.65 ± 0.73 % scavenging potential was observed at 7.8 µg/ml which was increased to 57.05 ± 1.39 % at 250 µg/ml. While MD extract showed the highest scavenging potential of 11.58 ± 2.64 %, 19.76 ± 3.56 %, 28.40 ± 2.00 %, 39.09 ± 3.89 %, 54.35 ±4.45 % and 68.28 ± 2.32 5 at 7.8 µg/ml, 15.6 µg/ml, 31.2 µg/ml, 62.5 µg/ml, 125 µg/ml and 250 µg/ml concentrations respectively. Additionally, MD extract showed lower IC50 value 141.98 ± 6.65 µg/ml than SD extract (222.81 ± 7.57 µg/ml), ShD extract (271.12 ± 5.01 µg/ml) and OD extract (189.68 ± 4.31 µg/ml) indicated higher scavenging potential of nitric oxide (Table 4). While ShD extract lower scavenging potential of nitric oxide due to higher IC50 value (271.12 ± 5.09 µg/ml).
Table 4: The IC50 value analysis (µg/ml)
| Sample | DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging assay | NO (nitric oxide)scavenging assay |
| AA (L-ascorbic acid) | 145.33 ± 3.69 | 54.25 ± 6.99 |
| SD (sun dried) | 401.22 ± 5.60 | 222.81 ± 7.57 |
| ShD (shade dried) | 484.15 ± 7.23 | 271.12 ± 6. 01 |
| OD (oven dried) | 361.75 ± 6.40# | 189.68 ± 4.31# |
| MD (microwave dried) | 190.99 ± 9.13@$& | 141.98 ± 6.65@$ |
| P value | < 0.0001 | < 0.0001 |
The results are expressed as mean and standard deviation. Significant changes were obtained using one Way ANOVA Post Tukey test. Inter-group comparisons were represented as *p<0.05 SD vs ShD; **P<0.05 SD vs OD; @p<0.05 SD vs MD; #P<0.05 ShD vs OD; $P<0.05 ShD vs MD; &P<0.05 OD vs MD.
Functional Group Analysis
The presence of multiple functional groups of the biochemical compounds in aqueous extract of C. citratus can be observed by ATR-FTIR spectroscopic analysis. Following the extract passed through the ATR-FTIR region, the functional groups were separated to their corresponding bonding position, the resulting peaks confirmed the presence of wide range of functional groups of bioactive compounds such as alcohol, carboxylic acid, vinyl / phenyl ester, cyclic alkane or amine and halo groups (Fig. 6). The IR spectra of SD extract revealed the presence of two bands at 3251.56 cm-1 and 2923.52 cm-1 that indicated the presence of alcohol and carboxylic acid (O-H) functional groups. Two potential bands were observed in the 1650- 1566 cm-1 range at 1557.12 and 1567.60 cm-1 indicated cyclic alkane or amine (C=O). One bending band observed at 1384.57 cm-1 in the range of 1390-1310 cm-1 represented phenol, confirmed O-H functional group and one stretching band at 1047.00 cm-1 in the range 1050-1040 cm-1 represented the anhydride (CO-O-CO) functional group. One potential band was detected in the range of 600-500 cm-1 at 532.40 cm-1, that showed the presence of C-Br or C-I, confirmed halo compounds. The IR spectra of ShD extract observed at 3241.95 cm-1 confirmed alcohol and carboxylic acid (O-H) functional group. One band identified at 1566.74 cm-1 in the range 1650-1566 cm-1 represented cyclic alkane or amine (C=O) as functional group, and one bending band was present at 1378.93 cm-1 in the range of 1390-1310 cm-1 as an indication of phenol (O-H) functional group. One potential band was detected at 1042.72 cm-1 represented anhydride (CO-O-CO) functional group. One band at 527.27 cm-1 in the range of 600-500 cm-1 indicated C-Br or C-I, confirmed halo functional group. In case of OD extract, only four bands were observed at 3253.64 cm-1 that confirmed alcohol or carboxylic acid (O-H) functional group, second bending band at 1573.95 cm-1 represented (N-H) cyclic alkane or primary amine, third bending band at 1384.66 cm-1 in the range 1390-1310 cm-1 confirmed phenols, O-H functional group and in the last band at 1042.23 cm-1 confirmed anhydride, CO-O-CO functional group was similar to ShD extract. The IR spectra of MD extract showed a band at 3259. 04 cm-1 indicated the presence of alcohol or carboxylic acid (O-H) functional group. One potential band was observed in the range of 2000-1650 cm-1 at 1998.64 cm-1 suggested the presence of vinyl/phenyl ester (C=O) functional group. One potential band was identified at 1567.11 cm-1 in the range 1650-1566 cm-1 that showed the presence of cyclic alkene or amine. One band was observed at 1379.50 cm-1 in the wavelength range of 1390-1310 cm-1 that confirmed phenol (O-H, bending) functional group. Two bands were observed at 1202.75 cm-1 and 1261.32 cm-1 in the range 1400-1000 cm-1 that indicated the presence of (C-O) alkyl aryl ether or vinyl ether functional group. One band was detected at 1047.90 cm-1 that confirmed the anhydride functional group last one band at 530.47 cm-1 that indicated (C-Br or C-I) halogen functional groups.
 |
Figure 6: FTIR spectrum analysis of aqueous extract of leaves of C. citratus under different drying methods- SD (sun dried), ShD (shade dried), OD (oven dried) and MD (microwave dried). |
Discussion
In the current study, the physical nature and yield % of extract expressed the efficiency of the different extraction conditions. Mostly essential oil extracts by leaves of C. citratus and the yield of essential oil depended on the optimum particle size that obtained the highest yield of essential oil.38 Analysis of ash content indicated that aqueous extract is rich source of minerals. Minerals of C. citratus are known to improve bone health and electrolytic balance, classifying it as neutraceutical.39 Present findings of qualitative phytochemicals for saponins were similar to those reported by Owusu and Owusu-Ansha.40-41 Phytochemicals including phenolics, flavonoids, saponins, alkaloids, tannins and steroids in extract of C. citratus have earlier been suggested with beneficial effects for health.42 C. citratus possess most abundant flavonoids were luteolin and apigenin derivatives such as 6-C-Hexosyl-8-C-pentosyl luteolin, 6-C-pentosyl-8-C-hexosyl apigenin, 6-C-Glucosyl luteolin, 7-O-Glucosyl luteolin, 6-C-Pentosyl luteolin, and X”-O-Rhamnosyl C-(6-deoxy-pento-hexos-ulosyl) C-(6-deoxy-pento-hexos-ulosyl).43 Most essential component of C. citratus are flavonoids that have pharmacological and therapeutic activities 44 and contribute significantly to neutralize free radicals which leads to oxidation stress.45 The presences of tannins in extracts are important in herbal medicine and are used to stop bleeding injuries, coagulate protein over the food tract lining and also react as antioxidant playing important role for scavenging of free radicals.41 Biochemical analysis demonstrated the different drying methods significantly affected the concentrations of proteins and carbohydrates. The quality of protein might be affected by variation of moisture content, salt ions and all-over drying process.46 OD and MD extracts might preserve proteins better due to shorter exposure to the environment and faster drying 47 whereas natural drying SD and ShD extracts showed lower protein content due to prolonged exposure to the environment resulting oxidation of protein content.48 Anti-nutritional factors reduced by OD and MD methods ensure better preservation of protein content.49-50 Carbohydrate is a natural source of energy,51 act as rapid energy source and improve its functional properties in food formulation. According to NHANES from 1999 to 2016, the estimated % of energy intake from total carbohydrate decreased from 52.5 % to 50 %, whereas consumption of plant-based proteins and high-quality carbohydrates increased.52 Present study supports that carbohydrates content obtained from OD and MD were significantly greater than naturally dried (SD and AD) methods.53 This may be credited to the different dried methods at appropriated drying temperatures of OD and MD, which likely increase the gelatinization of starch and thus enhancing digestibility of carbohydrates and resulting high carbohydrate content.54-55 In natural drying methods (SD and ShD), volatilization, oxidation and enzymatic degradation of carbohydrate may reduce the content due to long exposure to the open environment for drying.56
Total phenolics and flavonoids content are secondary metabolites in plants that act as antioxidant in human diet.57-58 The result of quantitative phytochemical analysis demonstrated the significant variation in concentration of total phenolic and flavonoid content (p < 0.05). Previous study has also shown that MD dried Mulbery leaves extract had rich sources of phenolics content.53 Furthermore, study demonstrated that Microwave drying improve stability of phenolics content and increased the interactions between the starch and polyphenolics in green tea noodles59 showing more preservation of polyphenol and minimizing damage of structure of bioactive molecules.60 Phenolic compounds in plant showed multiple pharmacological properties such as antimicrobial, antioxidants, anti-inflammatory, anti-infectious, cardio-protective, antiviral, anti-hyperglycemic, anti-allergic, mutagenic and anti-neuroprotective properties.61-63 Another study suggested that MD leaves extract of Mulbery obtained highest polyphenols including kaempferol, quercetin, caffeic acid p-hydroxycinnamic and sinapic acids etc.53 Further studies have confirmed that microwave dried sample retained more flavonoids than traditional thermal processing due to shorten duration of drying.64 Several scientific studies have reported antioxidant, anti-carcinogenic, anti-inflammatory, anti-rheumatic, antiprotozoal, antifungal, antimicrobial and cardio-protective activity of leaves of C. citratus.65-67 It has been employed to treat gastrointestinal infections, diabetes, anxiety or depression and also restrain the composition of platelets.68-69 Antioxidants assay revealed dose dependent activity of extracts in this study. DPPH is a synthetic and stable free radical which is commonly used for detect the activity of scavenging free radical of various natural compounds or extract of different parts of a plant. The antioxidants of leaves extract provide hydrogen atom to the DPPH free radical that neutralized to odd electron of nitrogen atom in DPPH solution.70 The present study suggested that MD leaves extract possess better free radical scavenging activity against DPPH at corresponding concentrations of OD, SD and ShD leaves extracts. The reducing power of antioxidants in the extract was often achieved by giving away hydrogen atom or break down the chains of free radicals.71 Hydroxyl group is linked to the benzene ring in the structure of polyphenols, the hydrogen atom which present in hydroxyl group is unstable and usually very good donor of hydrogen or electrons.72 Therefore, extract of C. citratus has a strong reducing ability that may be due to its high concentration of polyphenols. In nitric oxide scavenging assay, sodium nitroprusside produced nitric oxide in the extract at different concentration. By scavenging the nitic oxide, the extracts reduce the formation of nitrite ions at various concentrations. This study also confirmed that MD leaves extract have high promising activity against nitric oxide as compared to all corresponding concentration of SD, OD and ShD leaves extract. Previous investigation confirmed that different drying methods have affected the biological activity of sample before extraction.73 High antioxidant potential may be attributed to the high phenolic contents at high drying temperature74 and Phenolic compounds of the plants exhibited significant role play in antioxidant properties of extract in DPPH assay, and phenolic compounds inactivated of the metal ions such as ferric ions in reducing power assay.75 A comparative study showed that infusion of C. citratus presented better antioxidant activity than decoction of C. citratus.76 It is confirmed that MD extract showed strong antioxidant properties using DPPH free radical scavenging, FRAP, GSH, and CAT assay.53
ATR-FTIR is a modern technique used to detect functional groups, identify organic compounds without damaging them. The presence of multiple functional groups of the biochemical compounds in aqueous extract of C. citratus was observed by ATR-FTIR spectroscopic analysis. Following the extract passed through the ATR-FTIR region, the functional groups were separated to their corresponding bonding position, and the resulting peaks confirmed the presence of wide range of functional groups of bioactive compounds such as alcohol, carboxylic acid, vinyl / phenyl ester, cyclic alkane or amine and halo groups which were significantly different in different drying methods. There was no band present in the range of 2220-2260 cm-1 in all dried extracts indicated that the no cyanide groups was included in all extracts hence, the extracts contained no harmful substances.77 Several researchers confirmed that the analysis of phytochemical constituents of various plants by FTIR provided more rapid and accurate results.78-81 Our studies confirmed that MD leaves extracts contained better preserved qualitative and quantitative activities of useful biochemical that have various beneficial health effects.
Conclusion
Various drying treatments significantly affected the composition of phytochemicals and antioxidant properties of leaves of C. citratus. The present study revealed that MD leaves extract possessed maximum concentration of important phytochemicals like proteins, carbohydrates, phenol and flavonoid contents as compared to SD, ShD and OD leaves extracts. These phytochemicals play significant role as antioxidants against free radicals. High antioxidant activity was observed in MD leaves extract due to high concentration of phytochemicals. Functional group analysis suggested that MD leaves extract are rich in bioactive phytoconstituents which can be used as antioxidant agents. It, is therefore, concluded that MD leaves extract preserved the phytochemicals better that possess important antioxidant properties for C. citratus and better than SD, ShD and OD drying methods. Further studies are suggested to find out the mechanism of action for developing a novel antioxidant medicine.
Acknowledgement
Authors acknowledge the help by research scholar Pritam Samal, Department of Neutraceutics, Institute of Nuclear Medicine and Allied Science, Defence Research and Development Organization, Timarpur, Delhi.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Beena Pal: Conducted the experimental work, analyzed data.
Amar Prakash Garg: Conceptualized the paper, finally corrected the paper, supervised the experiments
Damodar Gupta: Assisted in methodology design and development, preliminary analysis of data.
References
- Aslan, K., Kelle, K., Yilmaz, M.A., Erden Kopar, E. and Gulcin, I.,. Investigation of Cuckoo‐Pint’s (Arum maculatum) Phytochemistry, In Vitro Antioxidant Potential, Enzyme Inhibition, and Antimicrobial Activity. ChemistrySelect. 2024; 9(37).202403588.https://doi.org/10.1002/slct.202403588
CrossRef - Gulcin İ. Antioxidants: a comprehensive review. Archives of Toxicology. 2025;99(5):1893-1997. https://doi.org/10.1007/s00204-025-03997-2
CrossRef - Bagchi K, Puri S. Free radicals and antioxidants in health and disease. A review. EMHJ Eastern Mediterranean Health J. 1998;4:350-60.https://iris.who.int/handle/10665/118217
CrossRef - Waterman KC, Adami RC, Alsante KM, Hong J, Landis MS, Lombardo F, Roberts CJ. Stabilization of pharmaceuticals to oxidative degradation. Pharmaceutical development and technology. 2002;7(1):1-32. https://doi.org/10.1081/PDT-120002237
CrossRef - Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, Telser J. Free radicals and antioxidants in normal physiological functions and human disease. The international journal of biochemistry & cell biology. 2007;39(1):44-84. https://doi.org/10.1016/j.biocel.2006.07.001
CrossRef - Carocho M, Ferreira IC. A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food and chemical toxicology. 2013;51:15-25. https://doi.org/10.1016/j.fct.2012.09.021.
CrossRef - Škrovánková S, Mišurcová L, Machů L. Antioxidant activity and protecting health effects of common medicinal plants. Advances in food and nutrition research. 2012;67:75-139. https://doi.org/10.1016/b978-0-12-394598-3.00003-4.
CrossRef - Anderson KJ, Teuber SS, Gobeille A, Cremin P, Waterhouse AL, Steinberg FM. Walnut polyphenolics inhibit in vitro human plasma and LDL oxidation. The Journal of nutrition. 2001;131(11):2837-42. https://doi.org/10.1093/jn/131.11.2837.
CrossRef - Huyut Z, Beydemir Ş, Gülçin İ. Antioxidant and antiradical properties of selected flavonoids and phenolic compounds. Biochemistry research international. 2017;(1):7616791. https://doi.org/10.1155/2017/7616791.
CrossRef - Ziarno M, Kozłowska M, Ścibisz I, Kowalczyk M, Pawelec S, Stochmal A, Szleszyński B. The effect of selected herbal extracts on lactic acid bacteria activity. Applied Sciences. 2021;11(9):3898. https://doi.org/10.3390/app11093898.
CrossRef - Kozłowska M, Ścibisz I, Zaręba D, Ziarno M. Antioxidant properties and effect on lactic acid bacterial growth of spice extracts. CYTA-Journal of Food. 2015;13(4):573-7. https://www.tandfonline.com/ action/ showCitFormats?doi=10.1080/19476337.2015.1022228.
CrossRef - Adhikari S, Bandopadhyay T, Ghosh P. Assessment of genetic diversity of certain Indian elite clones of Cymbopogon species through RAPD analysis. Indian Journal of Biotechnology. 2013;12(1):109-14.
- Schaneberg BT, Khan IA. Comparison of extraction methods for marker compounds in the essential oil of lemon grass by GC. Journal of agricultural and food chemistry. 2002;50(6):1345-9. https://doi.org/10.1021/jf011078h.
CrossRef - Ravinder K, Pawan K, Gaurav S, Paramjot K, Gagan S, Appramdeep K. Pharmacognostical investigation of Cymbopogon citratus (DC) Stapf. Der Pharmacia Lettre. 2010;2(2):181-9.
- Andissa NO, Moussoungou A, Koloungous B, Abena A. Topical Anti-inflammatory effect of aqueous extract ointment of Ageratum conyzoïdes L. in wistarrat. Int J Phytopharm. 2015;5:37-41. https://doi.org/10.3390/molecules26102933.
CrossRef - Shah G, Shri R, Panchal V, Sharma N, Singh B, Mann A. Scientific basis for the therapeutic use of Cymbopogon citratus, stapf (Lemon grass). Journal of advanced pharmaceutical technology & research. 2011;2(1):3-8. https://doi.org/10.4103/2231-4040.79796.
CrossRef - Avoseh O, Oyedeji O, Rungqu P, Nkeh-Chungag B, Oyedeji A. Cymbopogon species; ethnopharmacology, phytochemistry and the pharmacological importance. Molecules. 2015;20(5):7438-53. https://doi.org/10.3390/molecules20057438.
CrossRef - Halabi MF, Sheikh BY. Anti‐Proliferative Effect and Phytochemical Analysis of Cymbopogon citratus Extract. BioMed research international. 2014;2014(1):906239. https://doi.org/10.1155/2014/906239.
CrossRef - Mirghani M, Liyana Y, Parveen J. Bioactivity analysis of lemongrass (Cymbopogan citratus) essential oil. International food research journal. 2012;19(2):569-75.
- Deepa Garg DG, Aditya Muley AM, Nishtha Khare NK, Thankamani Marar TM. Comparative analysis of phytochemical profile and antioxidant activity of some Indian culinary herbs. 2012.
- Hammer KA, Carson CF, Riley TV. Antimicrobial activity of essential oils and other plant extracts. Journal of applied microbiology. 1999;86(6):985-90. https://doi.org/10.1046/j.1365-2672.1999.00780.x.
CrossRef - Adeneye AA, Agbaje EO. Hypoglycemic and hypolipidemic effects of fresh leaf aqueous extract of Cymbopogon citratus Stapf. in rats. Journal of ethnopharmacology. 2007;112(3):440-4.https://doi.org/10.1016/j.jep.2007.03.034.
CrossRef - Poddar S, Sarkar T, Choudhury S, Chatterjee S, Ghosh P. Indian traditional medicinal plants: A concise review. International Journal of Botany Studies. 2020;5(5):174-90.
- Van Wyk B-E, Gorelik B. The history and ethnobotany of Cape herbal teas. South African Journal of Botany. 2017;110:18-38. https://doi.org/10.1016/j.sajb.2016.11.011.
CrossRef - Mabai P, Omolola A, Jideani AI. Effect of drying on quality and sensory attributes of lemongrass (Cymbopogon citratus) tea. 2018. http://dx.doi.org/10.5539/jfr.v7n2p68.
CrossRef - Ozkan IA, Akbudak B, Akbudak N. Microwave drying characteristics of spinach. Journal of food engineering. 2007;78(2):577-83. http://dx.doi.org/10.1016/j.jfoodeng.2005.10.026.
CrossRef - Trusheva B, Trunkova D, Bankova V. Different extraction methods of biologically active components from propolis: a preliminary study. Chemistry Central Journal. 2007;1(1):13. https://doi.org/10.1186/1752-153x-1-13
CrossRef - Idris-Hermann KT, Raoul TTD, Giscard D, Gabche AS. Preparation and characterization of activated carbons from bitter kola (Garcinia kola) nut shells by chemical activation method using H3PO4; KOH and ZnCl2. Chemical Science International Journal. 2018;23(4):1-15. https://doi.org/10.9734/CSJI/2018/43411.
CrossRef - Harborne A. Phytochemical methods a guide to modern techniques of plant analysis: springer science & business media; 1998. https://doi.org/10.1007/978-94-009-5570-7.
CrossRef - Brain KR, Turner TD. The practical evaluation of phytopharmaceuticals. (No Title). 1975.
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J biol Chem. 1951;193(1):265-75. https://doi.org/10.1016/S0021-9258(19)52451-6.
CrossRef - Hedge J, Hofreiter B, Whistler R. Carbohydrate chemistry. Academic Press, New York. 1962;17:371-80.
- Končić MZ, Kremer D, Gruz J, Strnad M, Biševac G, Kosalec I, et al. Antioxidant and antimicrobial properties of Moltkia petraea (Tratt.) Griseb. flower, leaf and stem infusions. Food and Chemical Toxicology. 2010;48(6):1537-42. https://doi.org/10.1016/j.fct.2010.03.021.
CrossRef - Quettier-Deleu C, Gressier B, Vasseur J, Dine T, Brunet C, Luyckx M, et al. Phenolic compounds and antioxidant activities of buckwheat (Fagopyrum esculentum Moench) hulls and flour. Journal of ethnopharmacology. 2000;72(1-2):35-42. https://doi.org/10.1016/S0378-8741(00)00196-3.
CrossRef - Ololade Z, Olawore N. Characterization of essential oil from the seed of Eucalyptus cloeziana and evaluation of its modes of medicinal potentials. Edorium Journal of Infectious Diseases. 2017;3(1):1-8. https://doi.org/10.5348/I03-2017-5-OA-4.
CrossRef - Shang H-M, Zhou H-Z, Yang J-Y, Li R, Song H, Wu H-X. In vitro and in vivo antioxidant activities of inulin. PloS one. 2018;13(2):e0192273. https://doi.org/10.1371/journal.pone.0192273.
CrossRef - Green MH, Lowe JE, Delaney CA, Green IC. [23] Comet assay to detect nitric oxide-dependent DNA damage in mammalian cells. Methods in enzymology. 1996;269:243-66. https://doi.org/10.1016/s0076-6879(96)69026-0.
CrossRef - Okpo SO, Edeh I. Parametric Study on the Extraction of Essential Oil from Lemongrass (Cymbopogon Citratus). International Journal of Food Science and Agriculture. 2022;6(3). http://dx.doi.org/10.26855/ijfsa.2022.09.007.
CrossRef - Yuniarti DW, Sulistiyati TD, Suprayitno HE. Pengaruh suhu pengeringan vakum terhadap kualitas serbuk albumin ikan gabus (Ophiocephalus striatus): Brawijaya University; 2013.
- Owusu DA, Afedzi AEK, Quansah L. Phytochemical and proximate content of Carapa procera bark and its antimicrobial potential against selected pathogens. Plos one. 2021;16(12):e0261755. https://doi.org/10.1371/journal.pone.0261755.
CrossRef - Owusu-Ansah P, Alhassan AR, Ayamgama AA, Adzaworlu EG, Afoakwah NA, Mahunu GK, et al. Phytochemical analysis, enumeration, isolation, and antimicrobial activity of lemongrass and moringa leaves extracts. Journal of Agriculture and Food Research. 2023;12:100579. http://dx.doi.org/10.1016/j.jafr.2023.100579.
CrossRef - Asaolu M, Oyeyemi O, Olanlokun J. Chemical compositions, phytochemical constituents and in vitro biological activity of various extracts of Cymbopogon citratus. Pakistan Journal of Nutrition. 2009;8(12):1920-2. https://doi.org/10.3923/pjn.2009.1920.1922.
CrossRef - Mechchate H, Es-Safi I, Haddad H, Bekkari H, Grafov A, Bousta D. Combination of Catechin, Epicatechin, and Rutin: Optimization of a novel complete antidiabetic formulation using a mixture design approach. The Journal of Nutritional Biochemistry. 2021;88:108520. https://doi.org/10.1016/j.jnutbio.2020.108520.
CrossRef - Zannou A, Konfo T, Gbaguidi A, Ahoussi-Dahouenon E. Antimicrobial activity of extracts from Cymbopogon citratus L. and of Mentha spicata L. against fungal and bacterial strains isolated from peuhl’s cheese (Waragashi) produced in Benin. Int J Adv Res. 2015;3(10):1684-95.
- Kiani HS, Ali A, Zahra S, Hassan ZU, Kubra KT, Azam M, et al. Phytochemical composition and pharmacological potential of lemongrass (Cymbopogon) and impact on gut microbiota. AppliedChem. 2022;2(4):229-46. https://doi.org/10.3390/appliedchem2040016.
CrossRef - Kinsella J, Srinivasan D. Nutritional, chemical, and physical criteria affecting the use and acceptability of proteins in foods. Criteria of Food Acceptance. 1981:296-332.
- Ramachandran RP, Nadimi M, Cenkowski S, Paliwal J. Advancement and innovations in drying of biopharmaceuticals, nutraceuticals, and functional foods. Food Eng Rev 2024;16(4):540–66 [Internet].2024 https://doi.org/10.1007/s12393-024-09381-7
CrossRef - Babu AK, Kumaresan G, Raj VA, Velraj R. Review of leaf drying: Mechanism and influencing parameters, drying methods, nutrient preservation, and mathematical models. Renewable and sustainable energy reviews. 2018;90:536-56. https://doi.org/10.1016/j.rser.2018.04.002
CrossRef - Huang, J. J., Zhang, M., Mujumdar, A. S., Semenov, G., & Luo, Z. J.. Technological advances in protein extraction, structure improvement and assembly, digestibility and bioavailability of plant-based foods. Critical Reviews in Food Science and Nutrition. 2023;64:11556–74 https://doi.org/10.1080/10408398.2023.2240892
CrossRef - Sa A. G. A, Y. M. F, & Cariofi B. A. M. Food precessing for the improvement of plant proteins digestibility. Critical review in food science and Nutrition. 2020;60:3367-86.
CrossRef - Ludwig DS, Hu FB, Tappy L, Brand-Miller J. Dietary carbohydrates: role of quality and quantity in chronic disease. Bmj. 2018;13;361. https://doi.org/10.1136/bmj.k2340
CrossRef - Shan Z, Rehm CD, Rogers G, Ruan M, Wang DD, Hu FB, et al. Trends in dietary carbohydrate, protein, and fat intake and diet quality among US adults, 1999-2016. Jama. 2019;322(12):1178-87. https://doi.org/10.1001/jama.2019.13771.
CrossRef - Zhao C, Li T, Zhang C, Li H, Wang Y, Li C, Wang Z, Zhao M, Shen M, Zhao W. Drying methods affect nutritional value, amino acids, bioactive compounds, and in vitro function of extract in mulberry leaves. Food Chemistry. 2025;30;481:144018. https://doi.org/10.1016/j.foodchem.2025.144018
CrossRef - Mann J, Cummings JH, Englyst HN, Key T, Liu S, Riccardi G, Summerbell C, Uauy R, Van Dam RM, Venn B, Vorster HH. FAO/WHO scientific update on carbohydrates in human nutrition: conclusions. European journal of clinical nutrition. 2007;61(1):S132-7. https://doi.org/10.1038/sj.ejcn.1602943
CrossRef - Nishida C, Nocito FM. FAO/WHO scientific update on carbohydrates in human nutrition: introduction. European journal of clinical nutrition. 2007;2:61. https://doi.org/10.1038/ sj.ejcn.1602935
CrossRef - Shirkole SS, Mujumdar AS, Raghavan GS. Drying of foods: principles, practices and new developments. InDrying technology in food processing 2023;13-29. Woodhead Publishing. https://doi.org/10.1016/B978-0-12-819895-7.00020-1
CrossRef - Arias A, Feijoo G, Moreira MT. Exploring the potential of antioxidants from fruits and vegetables and strategies for their recovery. Innov. Food Sci. Emerg. Technol. 2022;77(10.1016). https://doi.org/10.1016/j.ifset.2022.102974
CrossRef - Jacobo-Velázquez DA, Cisneros-Zevallos L. Bioactive phenolics and polyphenols: current advances and future trends. International Journal of Molecular Sciences. 2020;21(17):6142. https://doi.org/10.3390/ijms21176142.
CrossRef - Yu K, Huang X, He W, Wu D, Du C. Kinetics of polyphenol losses during cooking of dried green tea noodles as influenced by microwave treatment of dough. LWT. 2023;180:114675. https://doi.org/10.1016/j.lwt.2023.114675
CrossRef - Jiang Hao JH, Liu ZhiGang LZ, Wang ShaoJin WS. Microwave processing: effects and impacts on food components. Critical Reviews in Food Science. 2018;58(14):2476-89. https://doi.org/10.1080/10408398.2017.1319322
CrossRef - Kowalska J, Tyburski J, Matysiak K, Jakubowska M, Łukaszyk J, Krzymińska J. Cinnamon as a useful preventive substance for the care of human and plant health. Molecules. 2021;26(17):5299. https://doi.org/10.3390/molecules26175299.
CrossRef - Cardile V, Graziano ACE, Venditti A. Clinical evaluation of Moro (Citrus sinensis (L.) Osbeck) orange juice supplementation for the weight management. Natural product research. 2015;29(23):2256-60. https://doi.org/10.1080/14786419.2014.1000897.
CrossRef - Li W, Li Y, Bi J, Ji Q, Zhao X, Zheng Q, et al. Effect of hot air drying on the polyphenol profile of Hongjv (Citrus reticulata Blanco, CV. Hongjv) peel: A multivariate analysis. Journal of food biochemistry. 2020;44(5):e13174. https://doi.org/10.1111/jfbc.13174.
CrossRef - Guzik P, Szymkowiak A, Kulawik P, Zając M. Consumer attitudes towards food preservation methods. Foods. 2022;11(9):1349. https://doi.org/10.3390/foods11091349.
CrossRef - Ajayi E, Sadimenko A, Afolayan A. GC–MS evaluation of Cymbopogon citratus (DC) Stapf oil obtained using modified hydrodistillation and microwave extraction methods. Food chemistry. 2016;209:262-6. https://doi.org/10.1016/j.foodchem.2016.04.071.
CrossRef - Ekpenyong CE, Akpan E, Nyoh A. Ethnopharmacology, phytochemistry, and biological activities of Cymbopogon citratus (DC.) Stapf extracts. Chinese journal of natural medicines. 2015;13(5):321-37. https://doi.org/10.1016/s1875-5364(15)30023-6.
CrossRef - Chukwuocha UM, Fernández-Rivera O, Legorreta-Herrera M. Exploring the antimalarial potential of whole Cymbopogon citratus plant therapy. Journal of ethnopharmacology. 2016;193:517-23. https://doi.org/10.1016/j.jep.2016.09.056.
CrossRef - Karkala Manvitha KM, Bhushan Bidya BB. Review on pharmacological activity of Cymbopogon citratus. 2014.
- Costa G, Grangeia H, Figueirinha A, Figueiredo IV, Batista MT. Influence of harvest date and material quality on polyphenolic content and antioxidant activity of Cymbopogon citratus infusion. Industrial Crops and Products. 2016;83:738-45. http://dx.doi.org/10.1016/j.indcrop.2015.12.008.
CrossRef - Veerapur VP, Prabhakar KR, Parihar VK, Kandadi MR, Ramakrishana S, Mishra B, Satish Rao BS, Srinivasan KK, Priyadarsini KI, Unnikrishnan MK. Ficus racemosa Stem Bark Extract: A Potent Antioxidant and a Probable Natural Radioprotector. Evid Based Complement Alternat Med. 2009;6(3):317-24. https://doi.org/10.1093/ecam/nem119.
CrossRef - Xu P, Zheng Y, Zhu X, Li S, Zhou C. L-lysine and L-arginine inhibit the oxidation of lipids and proteins of emulsion sausage by chelating iron ion and scavenging radical. Asian-Australasian journal of animal sciences. 2017;31(6):905. https://doi.org/10.5713/ajas.17.0617.
CrossRef - Zhang L, Lin Y, Leng X, Huang M, Zhou G. Effect of sage (Salvia officinalis) on the oxidative stability of Chinese-style sausage during refrigerated storage. Meat Science. 2013;95(2):145-50. https://doi.org/10.1016/j.meatsci.2013.05.005
CrossRef - Gupta S, Cox S, Abu-Ghannam N. Effect of different drying temperatures on the moisture and phytochemical constituents of edible Irish brown seaweed. LWT-Food Science and Technology. 2011;44(5):1266-72. https://doi.org/10.1016/j.lwt.2010.12.022.
CrossRef - Lou S-N, Lai Y-C, Huang J-D, Ho C-T, Ferng L-HA, Chang Y-C. Drying effect on flavonoid composition and antioxidant activity of immature kumquat. Food chemistry. 2015;171:356-63.
CrossRef - Mohan S, Gupta D. Role of Nrf2-antioxidant in radioprotection by root extract of Inula racemosa. Internation Journal of Radiation Biology. [Research]. 2019 18.04.2019(April):1-13. https://doi.org/10.1080/09553002.2019.1607607.
CrossRef - Cheel J, Theoduloz C, Rodríguez J, Schmeda-Hirschmann G. Free radical scavengers and antioxidants from Lemongrass (Cymbopogon citratus (DC.) Stapf.). Journal of agricultural and food chemistry. 2005;53(7):2511-7. https://doi.org/10.1021/jf0479766.
CrossRef - Ragavendran P, Sophia D, Arul Raj C, Gopalakrishnan V. Functional group analysis of various extracts of Aerva lanata (L.,) by FTIR spectrum. Pharmacologyonline. 2011;1:358-64.
- Saxena M, Saxena J. Evalution of phytoconstituents of Acorus calamus by FTIR and UV-VIS spectroscopic analysis. International Journal of Biological & Pharmaceutical Research. 2012;3(3):498-501.
- Helal A, Tagliazucchi D, Verzelloni E, Conte A. Bioaccessibility of polyphenols and cinnamaldehyde in cinnamon beverages subjected to in vitro gastro-pancreatic digestion. Journal of Functional Foods. 2014;7:506-16. http://dx.doi.org/10.1016/j.jff.2014.01.005.
CrossRef - Gorgulu ST, Dogan M, Severcan F. The characterization and differentiation of higher plants by Fourier transform infrared spectroscopy. Applied Spectroscopy. 2007;61(3):300-8. https://doi.org/10.1366/000370207780220903.
CrossRef - Kumar JK, Prasad AD. Identification and comparison of biomolecules in medicinal plants of Tephrosia tinctoria and Atylosia albicans by using FTIR. Romanian Journal of Biophysics. 2011;21(1):63-71.
Accepted on: 17-11-2025
Second Review by: Dr. Makhabbah Jamilatun
Final Approval by: Dr. Eugene A. Silow