Optimization and Evaluation of Dapagliflozin and Metformin HCl Bilayered Tablets Compared with Marketed Product
1Department of Pharmacology, Tagore College of Pharmacy, Rathinamangalam, Chennai, Tamilnadu, India
2Plainsboro, New Jersey, USA
3Department of Pharmacology, School of Pharmacy, Sathyabama Institute of Science and Technology, Chennai
Corresponding Author E-mail: kpcpharma81@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3462
ABSTRACT:The objective of this study was to formulate, optimize, and evaluate bilayered floating tablets containing dapagliflozin and metformin hydrochloride for sustained drug release and improved glycemic control compared with a marketed innovator product. Combination therapy of dapagliflozin, a sodium-glucose co-transporter-2 inhibitor, and metformin, a biguanide, provides effective management of type 2 diabetes mellitus but often requires multiple daily doses. To enhance therapeutic efficiency and patient compliance, a bilayered dosage form combining immediate and sustained release was developed. The formulation consisted of an immediate-release (IR) layer of dapagliflozin for rapid onset of action and a sustained-release (SR) layer of metformin HCl to maintain prolonged plasma concentration. Various batches (B7–B21) were prepared using different gas-generating agents and release-retarding polymers such as Hypromellose (HPMC K100M) and Carbopol 934P. Optimization was carried out to assess the effects of excipient concentration on key parameters including hardness, friability, floating lag time, total floating duration, and in vitro drug release. Among all formulations, Batch B17 showed superior performance with hardness of 5.4 kg/cm², friability of 0.19%, floating lag time of 60 seconds, and total floating duration of 12 hours. The in vitro release profile demonstrated 99.83% dapagliflozin release within 30 minutes and sustained metformin HCl release up to 12 hours (99.31%), closely comparable to the marketed product (XIDUGO XR). The combination of HPMC K100M and Carbopol 934P provided excellent matrix stability and prolonged gastric retention. In conclusion, the optimized bilayered floating tablet (B17) effectively combines immediate and sustained release profiles, reducing dosing frequency and improving patient adherence in diabetes management. Further in vivo, preclinical, and clinical studies are required to establish bioequivalence and confirm therapeutic performance.
KEYWORDS:Carbapol; Dapagliflozin; Formulation study; Metformin; Release Retarding agents
Introduction
A disturbance in glucose homeostasis, which raises blood glucose levels, is a hallmark of diabetes mellitus (DM), a metabolic disease.1 Type 2 diabetes is the most frequent of the two main forms, accounting for approximately 90% of all cases of diabetes.2Type 1 diabetes has been associated to a number of lifestyle variables, genetic predisposition, viral infections, and pollutants, In contrast, type 2 diabetes has proved associated with family history,3 ethnicity, age, and being overweight or obese. Currently, 9.3% of people worldwide suffer from diabetes; by 2030, that number is predicted to increase to 10.2%, and by 2045, to 10.9%.4 Due to people who have the condition, around 50% of diabetes cases globally go undetected.
Drug delivery is referred as tactics, formulations, techniques, and systems for providing a medicinal substance to the body when it’s essential to produce the expected therapeutic outcome. The most common drug administration route with the best patient compliance is oral administration.5 Specific benefits of oral delivery include Higher compliance is attained with non-invasive administration that is convenient for the patient. However, issues like limited solubility and permeability, irregular absorption, breakdown by gastrointestinal enzymes or flora, and first pass metabolism are frequently present.6 In this context, numerous ground-breaking technologies have been created to produce innovative dosage forms that improve medication delivery.7 In certain situations, extending the gastrointestinal retention of a delivery route is better in order to optimize the pharmacological components’ therapeutic efficiency.8
Floating Drug Delivery System
Due to their low density, these are meant to float on the contents of the stomach after delivery until the system breaks or until the device absorbs fluid to an extent when it loses its buoyancy and is able to exit the stomach more readily with a wave of motility that causes gastric emptying.9 The bilayered tablet is designed such that one layer distributes the medication instantly, causing it to start working quickly, and the other layer is made to float in the stomach (GI-fluid).10
Materials and Methods
In this study we have prepared different trail batches of bilayered tablets of Metformin and Dapagliflozin with different concentrations of gas generating agents and rate retarding agents in order to evaluate it to the commercial product to optimize and assess the floating duration and percentage of drug release
Table 1: Preliminary trial batches for Use of gas-generating agents- SR Layer
| LAYER- SR | Batch No. | |||
| Ingredients (mg) | B7 | B8 | B9 | B10 |
| Metformin HCl | 500.0 | 500.0 | 500.0 | 500.0 |
| Hypromellose K 100M | 160.0 | 160.0 | 160.0 | 160.0 |
| Sodium bi carbonate | 60.0 | – | – | – |
| Sodium carbonate | – | 60.0 | – | – |
| Potassium carbonate | – | – | 60.0 | – |
| Calcium carbonate | – | – | – | 60.0 |
| Microcrystalline cellulose pH 102 | 64.0 | 64.0 | 64.0 | 64.0 |
| Magnesium stearate | 8.0 | 8.0 | 8.0 | 8.0 |
| Talc | 8.0 | 8.0 | 8.0 | 8.0 |
| Total wt of SR layer (mg) | 800.0 | 800.0 | 800.0 | 800.0 |
Table 2: Preliminary trial batches for Use of gas-generating agents- IR Layer
| LAYER- IR | Batch No. | |||
| Ingredients (mg) | B7 | B8 | B9 | B10 |
| Dapagliflozin | 2.5 | 2.5 | 2.5 | 2.5 |
| Lactose | 20.0 | 20.0 | 20.0 | 20.0 |
| Microcrystalline cellulose pH 102 | 115.5 | 115.5 | 115.5 | 115.5 |
| Kyron T-314 | 6.0 | 6.0 | 6.0 | 6.0 |
| Talcum IP | 2.0 | 2.0 | 2.0 | 2.0 |
| Magnesium stearate IP | 2.0 | 2.0 | 2.0 | 2.0 |
| Iron oxide red | 2.0 | 2.0 | 2.0 | 2.0 |
| Total of IR layer (mg) | 150.0 | 150.0 | 150.0 | 150.0 |
| Total wt of bi-layered tablet (mg) | 950.0 | 950.0 | 950.0 | 950.0 |
Optimization of sodium bicarbonate concentration
A range of concentrations, including 5%, 7.5%, and 10% of the long-term release layer, have been employed to maximize the sodium bicarbonate concentration. Formulation of batches B7 (7.5%), B11 (5.0%) and B12 (10.0%) is mentioned in Table 2.
Table 3: Optimization of sodium bicarbonate concentration- SR layer
| LAYER-SR | Batch No. | ||
| Ingredients | B7 | B11 | B12 |
| Metformin HCl | 500.0 | 500.0 | 500.0 |
| Hypromellose K 100M | 160.0 | 160.0 | 160.0 |
| Sodium bi carbonate | 60.0 | 40.0 | 80 |
| Microcrystalline cellulose pH 102 | 64.0 | 64.0 | 64.0 |
| Magnesium stearate | 8.0 | 8.0 | 8.0 |
| Talc | 8.0 | 8.0 | 8.0 |
| Total wt of SR layer (mg) | 800.0 | 800.0 | 800.0 |
Table 4: Optimization of sodium bicarbonate concentration- IR layer
| LAYER-IR | Batch No. | ||
| Ingredients | B7 | B11 | B12 |
| Dapagliflozin | 2.5 | 2.5 | 2.5 |
| Lactose | 20.0 | 20.0 | 20.0 |
| Microcrystalline cellulose pH 102 | 115.5 | 115.5 | 115.5 |
| Kyron T-314 | 6.0 | 6.0 | 6.0 |
| Talcum IP | 2.0 | 2.0 | 2.0 |
| Magnesium stearate IP | 2.0 | 2.0 | 2.0 |
| Iron oxide red | 2.0 | 2.0 | 2.0 |
| Total of IR layer (mg) | 150.0 | 150.0 | 150.0 |
| Total wt of bilayered tablet (mg) | 950.0 | 950.0 | 950.0 |
Preliminary trial batches to study the Effect of release retarding agent (stearic acid)
Using a chloroform solution, stearic acid—a hydrophobic agent—was combined with the medication. Chloroform is used to first create a drug complex with stearic acid. Other chemicals are then added, well mixed, and compressed. Stearic acid was utilized to maintain the dissolution of metformin hydrochloride at different concentrations (1, 2, and 3%).
Table 5: Preliminary trial batches to study the Effect of release retarding agent (Stearic acid)- SR layer
| LAYER- SR | Batch. No | ||
| Ingredients | B13 | B14 | B15 |
| Metformin HCl | 500.0 | 500.0 | 500.0 |
| Hypromellose K 100M | 160.0 | 160.0 | 160.0 |
| Sodium bi carbonate | 60.0 | 40.0 | 80 |
| Stearic acid | 8.0 | 16.0 | 24.0 |
| Microcrystalline cellulose pH 102 | 56.0 | 68.0 | 20.0 |
| Magnesium stearate | 8.0 | 8.0 | 8.0 |
| Talc | 8.0 | 8.0 | 8.0 |
| Total wt of SR layer (mg) | 800.0 | 800.0 | 800.0 |
Table 6: Preliminary trial batches to study the Effect of release retarding agent (Stearic acid)- IR layer
| LAYER-IR | Batch No | ||
| Ingredients | B13 | B14 | B15 |
| Dapagliflozin | 2.5 | 2.5 | 2.5 |
| Lactose | 20.0 | 20.0 | 20.0 |
| Microcrystalline cellulose pH 102 | 115.5 | 115.5 | 115.5 |
| Kyron T-314 | 6.0 | 6.0 | 6.0 |
| Talcum IP | 2.0 | 2.0 | 2.0 |
| Magnesium stearate IP | 2.0 | 2.0 | 2.0 |
| Iron oxide red | 2.0 | 2.0 | 2.0 |
| Total of IR layer (mg) | 150.0 | 150.0 | 150.0 |
| Total wt of bi-layered tablet (mg) | 950.0 | 950.0 | 950.0 |
Optimization of carbopol 934 P concentration
Release delaying agents have been included to the formulation in order to improve the delay in the drug release and to counteract the burst effect. The literature indicates that improved release rates are obtained when Carbopol 934P and Hypromellose are combined. Therefore, batches B16–B21 were created to examine how carbopol affected drug retardation.
Table 7: Optimization of carbopol 934 P concentration- SR layer
| LAYER-SR | Batch No. | |||||
| Ingredients | B16 | B17 | B18 | B19 | B20 | B21 |
| Metformin HCl | 500.0 | 500.0 | 500.0 | 500.0 | 500.0 | 500.0 |
| Hypreomellose K 100M | 80.0 | 80.0 | – | – | – | – |
| Hypromellose K15M | – | – | 80.0 | 80.0 | – | – |
| Hypromellose K4M | – | – | – | – | 80.0 | 80.0 |
| Carbopol 934P | 80.0 | 96.0 | 80.0 | 96.0 | 80.0 | 96.0 |
| Sodium bicarbonate | 80.0 | 80.0 | 80.0 | 80.0 | 80.0 | 80.0 |
| Microcrystalline cellulose pH 102 | 44.0 | 28.0 | 44.0 | 28.0 | 44.0 | 28.0 |
| Magnesium stearate | 8.0 | 8.0 | 8.0 | 8.0 | 8.0 | 8.0 |
| Talc | 8.0 | 8.0 | 8.0 | 8.0 | 8.0 | 8.0 |
| Total wt of SR layer (mg) | 800.0 | 800.0 | 800.0 | 800.0 | 800.0 | 800.0 |
Table 8: Optimization of carbopol 934 P concentration-IR layer
| LAYER-SR | Batch No. | |||||
| Ingredients | B16 | B17 | B18 | B19 | B20 | B21 |
| Dapagliflozin | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 |
| Lactose | 20.0 | 20.0 | 20.0 | 20.0 | 20.0 | 20.0 |
| Microcrystalline cellulose pH 102 | 115.5 | 115.5 | 115.5 | 115.5 | 115.5 | 115.5 |
| Kyron T-314 | 6.0 | 6.0 | 6.0 | 6.0 | 6.0 | 6.0 |
| Talcum IP | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 |
| Magnesium stearate IP | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 |
| Iron oxide red | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 |
| Total of IR layer (mg) | 150.0 | 150.0 | 150.0 | 150.0 | 150.0 | 150.0 |
| Total wt of bi-layered tablet (mg) | 950.0 | 950.0 | 950.0 | 950.0 | 950.0 | 950.0 |
Results
Evaluation of Trail Batches for Use of Gas-Generating Agents
Table 9: Powder blend properties of trail batches for Use of gas-generating agents
| Dapagliflozinwith metformin HCl | Batch no | Bulk density (gm/cm3) | Tapped density (gm/cm3) | Angle of Repose (θ) | Carr’s Index (%) | Hausner’s ratio |
| B7 | 0.46±0.04 | 0.51±0.01 | 26.33±1.4 | 19.6±1.4 | 1.14±0.04 | |
| B8 | 0.41±0.03 | 0.56±0.0 | 25.92±1.2 | 20.1±1.3 | 1.18±0.02 | |
| B9 | 0.43±0.01 | 0.58±0.01 | 25.64±1.6 | 18.8±1.4 | 1.16±0.02 | |
| B10 | 0.44±0.02 | 0.53±0.02 | 26.19±1.3 | 19.4±1.2 | 1.18±0.03 |
Table 10: Physical parameters of trail batches for Use of gas-generating agents
|
Dapagliflozin with Metformin HCl |
Batch No | Weight variation (mg) | Thickness(mm) | Friability (%w/w) | Hardness (kg/cm2) |
| B7 | 949±0.55 | 6.9± 0.02 | 0.61±0.12 | 5.4±0.18 | |
| B8 | 952±0.49 | 6.7 ± 0.01 | 0.65±0.18 | 5.4±0.15 | |
| B9 | 950±0.66 | 6.6 ± 0.04 | 0.63±0.09 | 5.2±0.16 | |
| B10 | 951±0.63 | 6.7 ± 0.02 | 0.68±0.18 | 5.3±0.19 |
Table 11: Floating behavior of trail batches for Use of gas-generating agents
|
Dapagliflozin with Metformin HCl |
Batch No | Floating lag time (sec) | Total floating time (hrs) |
| B7 | 205 | 07 | |
| B8 | 234 | 05.5 | |
| B9 | 246 | 04.5 | |
| B10 | 259 | 05 |
Table 12: Physical parameters of tablets of trail batches B7, B11, B12
| Dapagliflozin
with Metformin HCl |
Batch No | Weight variation (mg) | Thickness(mm) | Friability (%w/w) | Hardness (kg/cm2) |
| B7 | 949±0.55 | 6.9± 0.02 | 0.61±0.12 | 5.4±0.18 | |
| B11 | 950±0.63 | 6.5± 0.01 | 0.54±0.15 | 5.3±0.13 | |
| B12 | 951±0.57 | 6.7 ± 0.04 | 0.59±0.18 | 5.3±0.11 |
Table 13: Floating behavior of tablets of trail batches B7, B11, B12
| Dapagliflozin
with Metformin HCl |
Batch No | Floating lag time (sec) | Total floating time (hrs) |
| B7 | 205 | 07 | |
| B11 | 125 | 10.5 | |
| B12 | 60 | 12 |
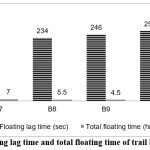 |
Figure 1: Floating lag time and total floating time of trail batches B7-B10
|
Floating lag time and total floating time of trial batches B7–B10. Sodium bicarbonate showed the shortest lag time and longest buoyancy. Batch B7 demonstrated optimum floating behavior among all formulations Shown in fig 1.
Table 14: % Drug content of Dapagliflozin and Metformin HCL of batches B7-B10
| Batch No | % Drug content | |
| Dapagliflozin | Metformin HCl | |
| B7 | 99.39 ±0.27 | 98.98 ±0.22 |
| B8 | 99.19±0.19 | 99.16±0.16 |
| B9 | 98.97±0.18 | 98.92±0.24 |
| B10 | 99.33±0.22 | 99.17±0.17 |
Evaluation of Batches for Optimization of Sodium Bicarbonate Concentration
Table 15: Powder blend properties of trail batches B7, B11, B12
| Batch no | Bulk density (gm/cm3) | Tapped density (gm/cm3) | Angle of Repose (θ) | Carr’s Index(%) | Hausner’s ratio | |
| Dapagliflozinwith Metformin HCl | B7 | 0.46±0.04 | 0.51±0.01 | 26.33±1.4 | 19.6±1.4 | 1.14±0.04 |
| B11 | 0.48±0.03 | 0.59±0.0 | 26.13±1.5 | 18.6±1.3 | 1.19±0.03 | |
| B12 | 0.45±0.02 | 0.52±0.01 | 26.28±1.3 | 21.3±1.4 | 1.17±0.04 |
Mean ± SD, n=3
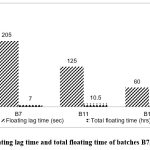 |
Figure 2: Floating lag time and total floating time of batches B7, B11 and B12
|
Floating lag time and total floating time of batches B7, B11 and B12. An increase in sodium bicarbonate concentration reduced lag time effectively. Batch B12 exhibited maximum floating duration of 12 hours in fig 2.
Table 16: % Drug content of Dapagliflozin and Metformin HCl of batches B7, B11and B12
| Batch No | % Drug content | |
| Dapagliflozin | Metformin HCl | |
| B7 | 98.76 ±0.16 | 99.21 ±0.18 |
| B11 | 99.13±0.14 | 98.56±0.12 |
| B12 | 98.94±0.12 | 98.93±0.19 |
| Mean ± SD, n=3 | ||
Table 17: In-vitro dissolution profile data of formulations B7, B11, B12- Dapagliflozin
| Cumulative % Drug release (Dapagliflozin) | B7 | B11 | B12 |
| 5 min | 60.12±0.27 | 58.86±0.33 | 60.01±0.28 |
| 10 min | 92.31±0.36 | 89.96±0.13 | 91.18±0.16 |
| 15 min | 96.22±0.19 | 94.41±0.14 | 96.63±0.42 |
| 20 min | 98.85±0.21 | 96.63±0.16 | 97.44±0.10 |
| 30 min | 99.41±0.18 | 99.81±0.11 | 99.93±0.27 |
Table 18: In-vitro dissolution profile data of formulations B7, B11, B12- Metformin HCl
| Cumulative % Drug release (Metformin Hcl) | B7 | B11 | B12 |
| 0.5 hr | 20.69±0.10 | 21.21±0.21 | 22.19±0.22 |
| 1 hr | 29.13±0.28 | 28.56±0.11 | 33.67±0.17 |
| 2 hr | 36.72±0.14 | 33.18±0.17 | 37.25±0.15 |
| 3 hr | 42.78±0.28 | 41.37±0.13 | 41.27±0.21 |
| 4 hr | 48.89±0.21 | 46.86±0.11 | 46.63±0.10 |
| 6 hr | 64.56±0.15 | 58.19±0.14 | 53.76±0.18 |
| 8 hr | 89.61.28±0.22 | 65.97±0.21 | 67.49±0.14 |
| 10 hr | – | 91.25±0.16 | 80.24±0.15 |
| 12 hr | – | – | 95.62±0.11 |
| Mean ± SD, n=3 | |||
 |
Figure 3:(a,b): In- vitro Dissolution profile comparison for release of Dapagliflozin IR layer and Metformin HCl SR layer Bilayer tablets using sodium carbonate
|
In Fig. 3 (a), (b) In-vitro dissolution profile for Dapagliflozin (IR) and Metformin HCl (SR) using sodium carbonate. Rapid Dapagliflozin release was achieved within 30 minutes. Metformin HCl sustained its release for up to 12 hours.
Evaluation of Trail Batches to Study the Effect of Release Retarding Agent (Stearic Acid)
Table 19: Powder blend properties of trail batches B13- B15
| Batch no | Bulk density (gm/cm3) | Tapped density (gm/cm3) | Angle of Repose (θ) | Carr’s Index(%) | Hausner’s ratio |
| B13 | 0.44±0.02 | 0.56±0.04 | 25.59±1.6 | 18.8±1.3 | 1.14±0.02 |
| B14 | 0.46±0.02 | 0.52±0.03 | 26.35±1.3 | 19.6±1.4 | 1.16±0.03 |
| B15 | 0.45±0.03 | 0.51±0.01 | 26.33±1.4 | 21.5±1.9 | 1.15±0.01 |
| Mean ± SD, n=3 | |||||
Table 20: Physical parameters of trail batches B13- B15
| Batch No | Weight variation (mg) | Thickness(mm) | Friability(%w/w) | Hardness (kg/cm2) |
| B13 | 952±0.61 | 6.7± 0.03 | 0.58±0.12 | 5.4±0.18 |
| B14 | 949±0.55 | 6.4± 0.04 | 0.61±0.16 | 5.3±0.17 |
| B15 | 951±0.49 | 6.6 ± 0.03 | 0.59±0.15 | 5.5±0.18 |
| Mean ± SD, n=3 | ||||
Table 21: Floating behavior of trail batches B13- B15
| Batch No | Floating lag time (sec) | Total floating time (hrs.) |
| B13 | 204 | 07 |
| B14 | 228 | 06.5 |
| B15 | 240 | 06 |
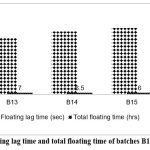 |
Figure 4: Floating lag time and total floating time of batches B13, B14 and B15
|
In Fig. 4 Floating lag time and total floating time of batches B13–B15. Formulations with stearic acid showed slightly increased lag time. B13 exhibited the most desirable floating characteristics.
Table 22: % Drug content of Dapagliflozin and Metformin HCL of batches B13-B15
| Batch No | % Drug content | |
| Dapagliflozin | Metformin HCl | |
| B13 | 99.34 ±0.16 | 99.13 ±0.14 |
| B14 | 99.19±0.11 | 98.94±0.16 |
| B15 | 99.65±0.17 | 98.99±0.20 |
| Mean ± SD, n=3 | ||
Table 23: In-vitro dissolution profile data of formulations B13-B15- Dapagliflozin
| Cumulative % Drug release (Dapagliflozin) | B13 | B14 | B15 |
| 5 min | 59.96±0.23 | 60.12±0.23 | 60.26±0.19 |
| 10 min | 91.99±0.31 | 89.66±0.15 | 90.38±0.17 |
| 15 min | 95.43±0.25 | 94.49±0.16 | 96.07±0.21 |
| 20 min | 98.59±0.27 | 96.18±0.11 | 97.86±0.16 |
| 30 min | 99.63±0.22 | 99.27±0.13 | 99.34±0.23 |
Table 24: In-vitro dissolution profile data of formulations B13-B15- Metformin HCl
| Cumulative % Drug release (Metformin Hcl) | B13 | B14 | B15 |
| 0.5 hr | 20.11±0.10 | 21.26±0.21 | 20.01±0.12 |
| 1 hr | 28.59±0.28 | 28.55±0.10 | 27.49±0.18 |
| 2 hr | 35.58±0.14 | 33.08±0.19 | 32.29±0.16 |
| 3 hr | 46.92±0.28 | 42.27±0.16 | 41.95±0.12 |
| 4 hr | 55.64±0.21 | 57.39±0.13 | 56.29±0.19 |
| 6 hr | 68.93±0.15 | 76.39±0.17 | 79.28±0.14 |
| 8 hr | 89.93±0.22 | – | – |
| 10 hr | – | – | – |
| 12 hr | – | – | – |
| Mean ± SD, n=3 | |||
 |
Figure 5: (a,b). In- vitro Dissolution profile comparison for release of Dapagliflozin IR layer and Metformin HCl SR layer from Bilayer tablets using stearic acid.
|
Evaluation of Trail Batches to Study the Effect Carbopol Concentration
Table 25: Powder blend properties of trail batches B16- B21
| Batch no | Bulk density (gm/cm3) | Tapped density (gm/cm3) | Angle of Repose (θ) | Carr’s Index(%) | Hausner’s ratio |
| B16 | 0.43±0.02 | 0.54±0.04 | 26.18±1.6 | 18.8±1.3 | 1.18±0.01 |
| B17 | 0.42±0.02 | 0.51±0.03 | 26.56±1.3 | 19.6±1.4 | 1.12±0.02 |
| B18 | 0.44±0.03 | 0.51±0.01 | 25.98±1.4 | 21.5±1.9 | 1.14±0.04 |
| B19 | 0.43±0.04 | 0.56±0.04 | 26.25±1.6 | 19.3±1.3 | 1.13±0.03 |
| B20 | 0.45±0.01 | 0.68±0.03 | 26.22±1.3 | 19.9±1.4 | 1.17±0.04 |
| B21 | 0.46±0.03 | 0.54±0.01 | 25.49±1.4 | 21.2±1.9 | 1.19±0.02 |
| Mean ± SD, n=3 | |||||
Table 26: Physical parameters of trail batches B16- B21
| Batch No | Weight variation (mg) | Thickness(mm) | Friability (%w/w) | Hardness (kg/cm2) |
| B16 | 948±0.42 | 6.7± 0.03 | 0.58±0.12 | 5.4±0.18 |
| B17 | 949±0.37 | 6.4± 0.04 | 0.61±0.16 | 5.3±0.17 |
| B18 | 950±0.26 | 6.6 ± 0.03 | 0.59±0.15 | 5.2±0.18 |
| B19 | 952±0.24 | 6.7± 0.03 | 0.72±0.12 | 5.4±0.10 |
| B20 | 950±0.37 | 6.4± 0.04 | 0.68±0.15 | 5.3±0.13 |
| B21 | 951±0.49 | 6.6 ± 0.03 | 0.59±0.13 | 5.2±0.11 |
| Mean ± SD, n=3 | ||||
Table 27: Floating behavior of trail batches B16- B21
| Batch No | Floating lag time (sec) | Total floating time (hrs) |
| B16 | 255 | 10 |
| B17 | 221 | 13.5 |
| B18 | 268 | 8 |
| B19 | 270 | 11 |
| B20 | 267 | 9 |
| B21 | 301 | 10 |
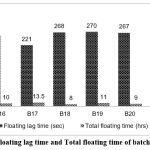 |
Figure 6: Floating lag time and Total floating time of batches B16-B21
|
In Fig. 6 Floating lag time and total floating time of batches B16–B21. Carbopol 934P combinations enhanced tablet buoyancy and duration. Batch B17 maintained floating for 13.5 hours with minimal lag time.
Table 28: In-vitro dissolution profile data of formulations B16-B21-Dapagliflozin
| Cumulative %drug release (Dapagliflozin) | ||||||
| B16 | B17 | B18 | B19 | B20 | B21 | |
| 5 min | 60.01±0.19 | 60.12±0.23 | 59.72±0.55 | 59.84±0.18 | 60.03±0.12 | 60.26±0.12 |
| 10 min | 91.13±0.17 | 89.66±0.19 | 91.29±0.43 | 91.00±0.15 | 90.07±0.24 | 90.22±0.13 |
| 15 min | 95.02±0.18 | 94.98±0.13 | 96.69±0.22 | 95.28±0.16 | 95.16±0.26 | 96.19±0.21 |
| 20 min | 97.18±0.19 | 96.07±0.17 | 97.76±0.86 | 98.19±0.22 | 98.22±0.21 | 97.88±0.18 |
| 30 min | 99.34±0.21 | 99.60±0.14 | 99.93±0.29 | 99.29±0.2 | 99.61±0.23 | 99.21±0.19 |
Table 29: In-vitro dissolution profile data of formulations B16-B21- Metformin HCl
| Cumulative %drug release (Metformin HCl) | ||||||
| B16 | B17 | B18 | B19 | B20 | B21 | |
| 0.5 hr | 16.38±0.21 | 20.08±0.21 | 19.94±0.22 | 18.65±0.15 | 19.69±0.15 | 17.96±0.11 |
| 1 hr | 25.16±0.28 | 28.55±0.10 | 25.59±0.19 | 23.76±0.18 | 26.43±0.23 | 24.80±0.18 |
| 2 hr | 29.65±0.10 | 33.08±0.19 | 34.11±0.21 | 32.76±0.20 | 34.83±0.11 | 36.49 ±0.13 |
| 3 hr | 43.61±0.15 | 46.63±0.16 | 43.35±0.18 | 40.02±0.18 | 41.76±0.18 | 45.12±0.16 |
| 4 hr | 54.27±0.11 | 57.59±0.13 | 49.95±0.12 | 47.23±0.13 | 48.12±0.21 | 57.26±0.10 |
| 6 hr | 66.33±0.16 | 73.34±0.17 | 67.58±0.16 | 61.72±0.21 | 68.72±0.16 | 69.62±0.19 |
| 8 hr | 72.29±0.24 | 82.59±0.22 | 85.56±0.19 | 83.62±0.19 | 82.70±0.14 | 81.56±0.15 |
| 10 hr | 82.97±0.22 | 90.07±0.97 | 96.79±0.20 | 87.73±0.15 | 96.34±0.21 | 85.59±0.11 |
| 12 hr | 97.15±0.20 | 99.49±0.14 | – | 94.26±0.22 | – | 92.12±0.15 |
| Mean ± SD, n=3 | ||||||
 |
Figure 7: (a,b). In-vitro Dissolution profile comparison for release of Dapagliflozin IR layer and Metformin SR layer
|
In Fig. 7 (a), (b) In-vitro dissolution profile for Dapagliflozin (IR) and Metformin HCl (SR) using Carbopol 934P. Dapagliflozin released rapidly within 30 minutes. Metformin HCl in B17 showed controlled and sustained release up to 12 hours.
Metformin HCl SR layer Bilayered tablets using Carbopol 934P
Table 30: Effect of hardness on floating behavior of optimized formulation B17
| Hardness (kg/cm2) | Floating lag time (sec) | Total floating time (hrs) |
| 3 | 176 | 8.5 |
| 4 | 190 | 10 |
| 5 | 230 | 11.5 |
| 6 | 365 | 14 |
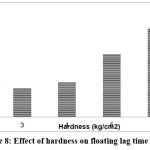 |
Figure 8: Effect of hardness on floating lag time of B17
|
Effect of hardness on floating lag time of B17. Higher compression increased lag time but extended total buoyancy. Optimal hardness was 5 kg/cm² for balanced floating and release shown in fig 8.
Comparison of evaluation parameters of optimized Dapagliflozin and Metformin HCl bilayered tablets with marketed product
Table 31: Post Compression Evaluation parameters for Optimized and reproducible batch 01 and Innovator (XIDUGO XR)
| Batch No | Weight variation (mg) | Hardness (kg/cm2 ) | Thickness (mm) | Friability (%) |
| B17 (01) | 952 ±2.13 | 5.4 ±0.08 | 6.4 ±0.18 | 0.19 |
| Innovator (XIDUGO XR) |
1050±0.06 | 6.3±0.03 | 7.4±0.08 | 0.11 |
Table 32: Cumulative % Drug release profile of optimized Dapagliflozin bilayered tablets and innovator product
| Time | Cumulative % drug release | |||
| Batch: B17 (01) | Innovator | |||
| Dapagliflozin | Metformin HCl | Dapagliflozin | Metformin HCl | |
| 0 | 0 | 0 | 0 | 0 |
| 5 min | 58.36±0.23 | – | 54.10±0.16 | – |
| 10 min | 86.33±0.11 | – | 82.31±0.21 | – |
| 15 min | 92.31±0.13 | – | 91.18±0.14 | – |
| 20 min | 96.19±0.14 | – | 95.64±0.12 | – |
| 30 min | 99.83±0.10 | – | 99.61±0.13 | – |
| 1 hr | – | 26.63±0.09 | – | 25.63±0.19 |
| 2 hr | – | 34.48±0.16 | – | 31.12±0.22 |
| 3 hr | – | 46.68±0.11 | – | 45.50±0.16 |
| 4 hr | – | 58.21±0.13 | – | 65.59±0.10 |
| 6 hr | – | 71.96±0.15 | – | 73.36±0.25 |
| 8 hr | – | 83.38±0.22 | – | 84.26±0.22 |
| 10 hr | – | 93.29±0.09 | – | 92.29±0.19 |
| 12 hr | – | 99.31±0.14 | – | 97.19±0.16 |
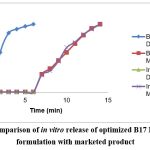 |
Figure 9: Comparison of in vitro release of optimized B17 Dapagliflozin formulation with marketed product
|
Comparison of in-vitro drug release of optimized B17 formulation with marketed product. Both formulations exhibited similar release kinetics for 12 hours. B17 showed 99.31% release, comparable to the innovator shown In fig 9.
Evaluation of Optimized Batch to Study Effect Of Hardness On Floating Behavior
Four batches of varying hardness were prepared and tested for floating lag time using the optimized batch B17.(Table 23)
In addition to reducing floating lag time, the lower hardness (3 kg/cm2, 4 kg/cm2) also reduced total floating duration. Both the floating lag time and the total floating time increase with increasing hardness. Both the desired floating lag time and the total floating lag time are displayed by the batch with a hardness of 5 kg/cm2. The floating lag time and overall floating time are increased for batches with a hardness of 6 kg/cm2. For B17, a hardness of 5 kg/cm2 was therefore ideal. When the tablets come into touch with the gastric contents, the hydrocolloid particles’ compacted surface cannot hydrate quickly, which greatly reduces the tablet’s resistance to floating. Results from the optimized B17’s innovative in vitro release comparison were displayed in 24. With a 99.31% release rate at 12 hours, dapagliflozin exhibits superior release as compared to innovator.
To determine the impact of variables such as the proportion and concentration of polymers on floating lag time and total floating time, a factorial design study must be conducted on the Dapagliflozin with Metformin HCl B17 formulation.
Discussion
Bilayer tablet technology represents a sophisticated pharmaceutical approach that allows several medications with different pharmacokinetic characteristics to be delivered simultaneously, improving therapeutic results and possibly increasing patient compliance.11 Bilayer tablets provide the ability to customize release profiles by separating incompatible drugs into distinct layers and managing their individual release mechanisms. This allows bilayer tablets to more effectively address complex treatment requirements than single-layer formulations.12-14
Several significant constraints in bilayer tablet research that have historically hampered pharmaceutical development are shown by the literature now under publication. Problems like layer separation during production, irregular drug release patterns, and difficulties preserving structural integrity under varied stress settings have all been repeatedly noted in earlier research.15-17 The consistent compression of several drug layers has shown to be quite challenging, according to research, which can result in unequal drug distribution and reduced therapeutic efficacy.18 Furthermore, the interface between layers frequently poses a challenging issue, with possible interactions between excipients and APIs that may impact the stability and release kinetics of the medication.19, 20 Numerous studies have documented mechanical issues, such as layer delamination during storage and transit, underscoring the complexity of creating reliable bilayer tablet formulations.21, 22 The development of a truly optimized bilayer tablet system that preserves consistent pharmacokinetic properties under a variety of physiological conditions has also proven difficult due to enduring concerns about potential variations in drug absorption and the variability in dissolution rates between different layers.23-25
By contrasting the physicochemical and pharmacological characteristics of bilayered tablets carrying dapagliflozin and metformin hydrochloride (HCl) with a commercially available marketed product, the current study sought to optimize and assess these tablets. Our investigation’s findings offer an assortment of important new perspectives on the design and functionality of this combination medication delivery system. To overcome the difficulties involved in mixing two medications with different physicochemical characteristics, we created a bilayered tablet formulation. The solubility, dissolution rates, and stability properties of metformin HCl, a biguanide antidiabetic drug, and dapagliflozin, an inhibitor of the sodium-glucose cotransporter-2 (SGLT2), differ. By using a hybrid approach, we were able to preserve each active pharmaceutical ingredient’s (API) unique therapeutic efficacy while optimizing their release patterns and stability.
Our bilayered formulation demonstrated similar, and in some cases better, release characteristics to the commercial product, according to the dissolution profile experiments. A more constant drug concentration in the bloodstream is guaranteed by the controlled release mechanism, which may increase patient compliance and lower the possibility of dose-related adverse effects.
Preliminary trail batches were performed to select good gas generating agent several gas generating agents were used. Sodium bicarbonate showed least floating lag time of 205sec for B7. Other gas-generating agents showed more floating lag time, which made their use undesirable. Batch B7 shows a satisfactory total floating time of 07 hours when sodium bicarbonate is used as the gas producing agent. (table.11) compared to all other batches. This may be due to the fact that Other gas-generating substances react slowly with hydrochloric acid, which slows down the generation of CO2 and increases the floating lag time. Thus, sodium bicarbonate is utilized as a gas generating agent for more research since it has an improved gas generating property than others and supplied the least floating lag time.
According to the data above, batch B7 and batch B11 had higher floating lag times because of lower gas producing agent concentrations. The lag time in batch B12 is floating within the specified bounds. Therefore, the floating lag time decreases as the sodium bicarbonate concentration rises. For additional research, the 100 mg of sodium bicarbonate was optimized. Gas is produced when the dose form comes into touch with the stomach’s acidic environment.
The formulation floats in the stomach’s acidic environment as the gas is trapped in its matrix of water-soluble polymers. The hydrophilic matrices cause the dosage form’s volume to expand, which lowers its density and causes it to begin floating.
In subsequent experiments, sodium bicarbonate at concentrations between 7.5 and 10% was employed to obtain the lowest floating lag time, as seen in Table 13. When the compressing force of tablets increases during compression, the porosity of the tablets decreases, allowing water to penetrate and changing the floating lag time. In these situations, it is preferable to use 10% sodium bicarbonate in order to reduce the floating time while the other ingredients are present.
When compared to batch B7, which had the same composition but no stearic acid, it demonstrates that the addition of stearic acid had no influence on the dissolution of metformin hydrochloride from the GFDDS (end point not reached). When stearic acid was added, the dissolution exhibited partial release. In order to achieve the appropriate medication release, it was determined that using carbopol in conjunction with hypromellose K100M was more beneficial than including stearic acid.(Table 18)
According to the results of the experimental trial, the profile of drug release of metformin hydrochloride in the recommended dosage unit changes when different amounts of carbopol are used, as compared to the dosage unit without carbopol, or Batch B7.
Under typical testing conditions, carbopol is insoluble in purified water and simulated stomach fluid. Cross-linking may be the cause of the fast release observed at the first time points in the trial in which carbopol was not used. The polymer expands and stays in the micro gelling network when the formulation enters into immediate proximity with the media or water. As a result, active material is delivered to the gut at a precise rate, which improves absorption. Compared to other batches and batches without Carbopol 934 P, the presence of Carbopol 934 P (Batch-B17) provided effective release delaying of metformin hydrochloride. Carbapol is used in formulation development to provide pills particular release and buoyancy. In order to keep the dosage form in the gastrointestinal tract, it also offers another feature called gastric retention. Therefore, the method of combining Hypromellose K100M with Carbopol 934P was employed.
Despite the encouraging findings of our investigation, a number of exceptions must be emphasized. Despite their reliability, the in vitro investigations fall short in simulating the intricate in vivo environment. To firmly prove our formulation’s therapeutic equivalency, more bioequivalence and clinical research are advised.
Conclusion
The floating time delay decreased as the HPMC ratio increased. K100M increased. HPMC K100M can maintain the matrix integrity and sustained the drug release up to 12hrs. B17(Dapagliflozin and Metformin HCl)is the best formulation containing HPMCK100M and Carbopol which showed higher similarity factor when compared with the marketed product and released maximum amount of drug 99.31%up to 12hrs.
Sodium bicarbonate, a gas-generating substance, was necessary for establishing in vitro buoyancy. A 10% formulation demonstrated a longer floating endurance (almost 12 hours) and a shorter floating time lag (60 seconds).
The conclusive results revealed that Dapagliflozin with Metformin HCl are best fit formulation among the other SGLT2 inhibitor when compared with marketed innovator product. As a combination therapy for diabetes, the improved bilayered floating tablet (B17) lowers dosage frequency and increases patient compliance. The utilization of bilayered floating tablets containing dapagliflozin and metformin HCl as an oral sustained release drug delivery technique demands preclinical and clinical research, as well as additional in-vivo studies to correlate in-vitro release tests.
Acknowledgement
We express our sincere gratitude to Tagore college of Pharmacy for providing support throughout our research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest-
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contribution
Sai Harini Sai Mohan: Conceptualization, Methodology
Deepika Srinivasan: Data collection, Writing- Original Draft
Karuna Priya Chitra: Conceptualization, Data collection, Analysis, Writing- Review & Editing
Srinath Nissankararao: Analysis, Methodology, Visualization, Supervision, Review & Editing
References
- Szablewski L,. Glucose Homeostasis – Mechanism and Defects. InTech. 2011. doi: 10.5772/22905
CrossRef - Galeone F., Fiore G., Arcangeli A., Mannucci E. Gliclazide and metformin combination in patients with type 2 diabetes. Preliminary data. Minerva Endocrinol. 1998; 23: 71-75.
- Farxiga [package insert]. Princeton, NJ: Bristol-Myers Squibb Company; 2014.
- Wilding J.P., Woo V., Soler N.G., and et al. Long-term efficacy of dapagliflozin in patients with type 2 diabetes mellitus receiving high doses of insulin: a randomized trial. Ann Intern Med. 2012; 156 (6):405-415.
CrossRef - Yunoos M., Gowri Sankar D. A Validated Stability Indicating High-Performance Liquid Chromatographic Method For Simultaneous Determination Of Metformin Hcl And Dapagliflozin In Bulk Drug And Tablet Dosage Form. Asian J Pharm Clin Res. 2015; 8(3), ISSN – 0974-2441.
- Jani B.R., Shah K.V., Kapupara P.P., Development And Validation Of Uv Spectroscopic Method For Simultaneous Estimation Of Dapagliflozin And Metformin Hydrochloride In Synthetic Mixture. Intern J Res Dev in Pharm Life Sci. 2015; 4(3): 1569-1576.
- List J. F., Woo V., Morales E., Tang W., Fiedorek F. T., Sodium-Glucose Cotransport Inhibition With Dapagliflozin in Type 2 Diabetes, Diabetes Care. 2009; 32 (4): 650-657.
CrossRef - Mante G. V., Gupta K. R and Hemke A. T. Estimation of Dapagliflozin from its tablet formulation by uv-spectrophotometry. Pharm methods. 2017; 8(2): 102-107.
CrossRef - Ferrannini E., Ramos S., Salsali A., Tang W., List J. Dapagliflozin monotherapy in type 2 diabetic patients with inadequate glycemic control by diet and exercise. Diabetes Care. 2010; 33: 2217–2224.
CrossRef - Sanghavi S., Polara M., Patel M., Shah N. Technologies in Bilayer Tablet Manufacturing: A Review of State of the Art. Res J Pharm Dosage Forms Technol. 2021;13(3):13.
- Akhtar M., Jamshaid M., Zaman M., Zeeshan Mirza A. Bilayer tablets: A developing novel drug delivery system. J. Drug Deliv. Sci. Technol. 2020; 60 (102079). Doi: 10.1016/j.jddst.2020.102079.
CrossRef - Nazneen S. S.,Shahidullah S. M. Bilayer Tablets in Drug Delivery: A Concept of Modified Release. Int. J. Pharm. Sci. Rev. Res. 84(11). DOI: 10.47583/ijpsrr.2024.v84i11.003.
CrossRef - Haran P. A., Pratapwar A. S., Thakre M. M., Haran A. R. Bilayer Tablet: An Dual Release Drug Delivery System. J. Emer Techn Innov Res. 2024; 11 (1).
- Walsh E., Maclean N., Turner A., Alsuleman M., Prasad E., Halbert G., Ter Horst J.H. Markl D. Manufacture of tablets with structurally-controlled drug release using rapid tooling injection moulding Int. J. Pharm., 2022; 624 (121956). Doi: 10.1016/j.ijpharm.2022.121956.
CrossRef - Crişan AG, et al. Development of a Bilayer Tablet by Fused Deposition Modelling 3D Printing for Fast + Sustained Release of Diclofenac Sodium. Pharmaceuticals (Basel). 2023;16(9):1321
CrossRef - Viswanathan G., Singh B. Real-world safety of Dapagliflozin + Metformin in patients with Type 2 Diabetes. Diabetes Res Clin Pract. 2024
- Sindhu S., Jyothi M., Richitha S., Hemanth G. Formulation and Evaluation of Bilayer Tablets of Sustained Release Microspheres of Anti Diabetic Drugs. Int J Pharm Res Sch (IJPRS). 2018;7(3):77–86. doi:10.31638/IJPRS.V7.I3.00056.
CrossRef - Huiyu Z, Cong Z., Guiting L., Rong C., Shaoyun G. The effect of layer thickness ratio on the drug release behavior of alternating layered composite prepared by layer-multiplying co-extrusion. Front Bioeng Biotech. 2023. 11. DOI=10.3389/fbioe.2023.1217938
CrossRef - Jackson K., Young D., Pant S. Drug–excipient interactions and their affect on absorption. Pharm Sci Tech Today. 2000; 3(10): 336-345. Doi: 10.1016/S1461-5347(00)00301-1
CrossRef - Miller B., Chavkin L. The Use of Polyethylene Glycol as a Binder in Tablet Compression. J Am Pharm Assoc (Scientific ed.). 1954; 43 (8): 486-488. Doi: 10.1002/jps.3030430813.
CrossRef - Sayantan C. Sticking and Picking in Pharmaceutical Tablet Compression: An IQ Consortium Review. J Pharm Sci. 107 (9): 2267 – 2282
CrossRef - Kottala N., Abebe A., Sprockel O. et al. Evaluation of the Performance Characteristics of Bilayer Tablets: Part II. Impact of Environmental Conditions on the Strength of Bilayer Tablets. AAPS PharmSciTech. 2012; 13: 1190–1196.
CrossRef - Li S. P., Karth M. G., Feld K. M., Di Paolo L. C., Pendharkar C. M., & Williams R. O. (1995). Evaluation of Bilayer Tablet Machines—A Case Study. Drug Dev Indus Pharm. 21(5), 571–590. Doi: 10.3109/03639049509048124.
CrossRef - Huiyu Z., Cong Z., Guiting L., Rong C., Shaoyun G. The effect of layer thickness ratio on the drug release behavior of alternating layered composite prepared by layer-multiplying co-extrusion. Front Bioeng Biotechnol. 2023;11. doi:10.3389/fbioe.2023.1217938.
CrossRef - Sindhu S., Jyothi M., Richitha S., & Hemanth, G. Formulation and Evaluation of Bilayer Tablets of Sustained Release Microspheres of Anti Diabetic Drugs. Int J Pharm Res Sch (IJPRS). 2018; 7(3): 77-86. Doi: 10.31638/IJPRS.V7.I3.00056.
CrossRef
Accepted on: 17-11-2025
Second Review by: Dr. Raghavendra Kumar Gunda
Final Approval by: Dr. Eugene A. Silow









