Exploring Nanoparticles: Types, Advantages, Challenges, and Applications in Drug Delivery and Technology
Department of Pharmaceutics, Govindrao Nikam College of Pharmacy, Sawarde, Maharashtra, India.
Corresponding Author Eimail:mamatakadamchavan@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/3297
ABSTRACT:Recently the use of particulate systems, such as nanoparticles, has physically altered and improved the pharmacokinetic and pharmacodynamic aspects of many different types of pharmaceutical compounds. They have been used in vivo to protect the drug entity in systemic circulation, restrict drug access to the desired locations, and deliver the drug at a steady and controlled rate to the site of action. Without a doubt, the size of medication delivery research is getting smaller and smaller. Nanotechnologies are therefore emerging as a field in medicine that promises to produce significant therapeutic benefits. One of the most challenging issues facing the pharmaceutical business is the development of effective, small delivery devices that can safely and accurately deliver a medication to a designated site of action. They are working to reformulate the blockbuster drugs that are presently on the market and add new indications in order to maintain valuable scientific findings and therapeutic advancements. Nano emulsions, liposomes, and lipid or polymeric nanoparticles are the fundamental elements of nano delivery systems.
KEYWORDS:Evaluation; Methods of Preparation; Nanoparticles; Synthesis; Types
Introduction
In recent years, nanotechnology has attracted a great deal of interest. Nanoparticles are the backbone of the nanotechnology industry. Particles composed of organic compounds, metals, metal oxides, or carbon that range in size from one nanometre to one hundred manometers are called nanoparticles. At the nanoscale, the particles’ physical, chemical, and biological characteristics are distinct from those of their larger-scale counterparts. This happens because of things like a higher surface area to volume ratio, better chemical stability or reactivity, more mechanical strength, and so on. Due to their unique characteristics, nanoparticles have found several practical uses. Nanoparticles come in a variety of forms, including hollow core, conical, spherical, cylindrical, tubular, flat, and wire. Additionally, its form may be asymmetrical. Nucleation products may have a smooth or uneven surface. They may also be found in crystalline and amorphous forms, either as single- or multi-crystal solids. Both loose and aggregated multi-crystal solids are possible1. These NPs’ differences in size and shape have a major impact on their physio-chemical characteristics. Because of their special physical and chemical characteristics, NPs have found remarkable success in a wide range of sectors, including imaging, chemical and biological sensing, energy-based research, medicine, and gas sensing. Due to its reputation as a key component of a sustainable and clean future, nanotechnology is drawing the attention of more researchers2. Modifying Given the particle’s form and polymer composition, it is possible to listen various controlled release aspects, allowing modest consistent doses for long durations of time. According to Liversidge and Cundy, a therapeutic particle’s accessibility comprised 77% more nanoparticles than in a formulation that used microspheres. The primary aim of this review was to focus on different types of nanoparticles using chemical, physiological, and biological means. Contrarily, biological therapies are straightforward, non-toxic, quick, and safe for the environment. In contrast, physical and chemical therapy are expensive damaging. It concludes with a list of applications and goes on to define the characteristics of nanoparticles3.
 |
Figure 1: Nanoparticles in NanometreClick here to view Figure |
Advantages4
Drug loading is crucial for maintaining drug activity, as pharmaceuticals can be added to systems without inducing any chemical reactions due to their high drug loading.
The effectiveness of a medicine can be improved by regulating or sustaining the release of the substance.
Smaller capillaries can be punctured by nanoparticles of small size, which permits medication to accumulate efficiently at specified regions.
Because liposomes and polymer nanoparticulate are typically biodegradable and never accumulate in the body, they may not pose a risk.
As the bioavailability of a medicine increases, its solubility also increases, allowing for more tailored drug administration.
Disadvantages
It is possible that the solvent system used in the preparation technique may have harmful effects.
Prescription leaks and unscheduled releases may be a significant issue.
The production expenses are significant and the efficiency of encapsulation is low.
Manipulating nanoparticles in both their wet and dry states is a significant challenge.
Limitations
| Nanomaterial | Limitations |
| 1.Silica-based nanomaterials(mesoporous silica and zeolites) | 1. Limited selectively and adsorption capacity2. poor recovery
3. Weak thermal, chemical, and mechanical stabilities 4. extensive cycling 5. Aggregation |
| 2. Carbonaceous nanomaterials | 1. Hydrophobicity2. Aggregation (negligible suspension into aqueous solution)
3. Low reactivity of carbon 4. High cost of carbon nanotubes 5. Possible health risk (direct toxicity) 6. poor recovery Of materials after treatment |
| 3. Nanoparticles (metals,
oxides, and quantum dots) |
1. Lack of chemical stability and mechanical strength(corrosion)
2. Propensity to aggregate 3. Lack Of for reactions in complex systems 4. Limited selectivity to organics and heavy metals 5. Limited activity under visible light (Ti02, ZnO) 6. Fast recombination of photogenerated charge carriers (semiconductor nanoparticles) |
Types of Nanoparticles
Semiconductor Nanoparticles5,6
Semiconductor materials are employed in a wide range of literary contexts due to their properties that lie between those of metals and nonmetals. Due to their large bandgaps, semiconductor nanoparticles (NPs) exhibit notable characteristics changes upon bandgap adjustment. When the zero-temperature coefficient is imposed at T = T0: the resistor ratio (R1/R3) and the bipolar emitter area ratio (m = ae1/ae2) can be adjusted properly (assuming R2 = R1). For this reason, they are crucial components for electrical devices, photocatalysis, and photopic. For instance, a range of semiconductor nanoparticles (NPs) have been discovered to be incredibly effective in water splitting applications because of their appropriate bandgap and bandage orientations.
Polymeric Nanoparticles7,8
These NPs are often based on organic materials, and they in the literature, these are called polymer nanoparticles (PNPs). Most of them have noncapsular or nanosphere-like shapes. At the outside edge of the spherical surface, the other molecules are adsorbed, whereas the Formers are matrix particles that usually have a solid total mass. In the latter case, the particle contains the complete solid mass. PNPs may be easily functionalized, which leads to wide range of applications in literature.
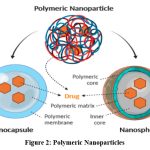 |
Figure 2: Polymeric NanoparticlesClick here to view Figure |
Carbon-Based Nanoparticles9,10
For fullerenes, carbon nanotubes (CNTs), and carbon-based NPs are the 2 principal types. Fullerenes include nanoparticles made of globular hollow cages, like allotropic forms of carbon. There has been a lot of commercial interest in them because of their electrical conductivity, high strength, structure, electron affinity, and versatility. These materials have carbon units that are organised in pentagonal and hexagonal shapes, with each carbon undergoing sp2 hybridization. consists of C60 and C70, two well-known fullerenes with diameters of 7.114 and 7.648nm, respectively. CNT have a 1-2 nm and are elongated, tubular structures (Ibrahim, 2013). We can predict whether they are semiconducting or metallic based on their diameter telicity. In terms of structure, these resemble sheets of graphite colliding head-to-head. Depending on the type of carbon nanotube, the rolled sheets are called either double-walled (DWNTs), multi-walled (MWNTs), or single-walled (SWNTs). whether have one, two, or many walls. They are commonly produced by depositing carbon precursors, particularly atomic carbons, onto metal particles using electric arc or laser vaporisation of graphite. Chemical vapour deposition, or CVD, has been used to create them more recently. Owing to materials with their own unique mechanical, chemical, and physical characteristics employed many contexts than just pure form.
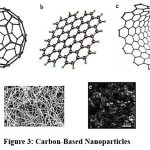 |
Figure 3: Carbon-Based NanoparticlesClick here to view Figure |
Metal Nanoparticles11,12
The only component of metal NPs is their precursor. These nanoparticles have distinct optoelectrical features because of their well-known localised surface plasmon resonance (LSPR) properties. LSPR is an optical phenomenon generated by a light wave trapped within conductive nanoparticles (NPs) smaller than the wavelength of light because of interactions between the incident light and surface electrons in a conduction band. The visible region of the electromagnetic sun spectrum has a large absorption band occupied by nanoparticles (NPs) of alkali noble metals, namely Cu, Ag, Au. The fabrication of metal nanoparticles that are regulated size and shape of the facet is essential modern advanced material. Utilizing metal nanoparticles (NPs) several scientific fields because of their superior optical characteristics. In order to enhance the electronic stream and generate high-quality findings, gold nanoparticle coating is widely utilized in SEM sampling.
Carbon Nanofibers13
Graphene sheets also make up carbon nanofibers (CNFs). This graphene arrangement shows layers as stacked plates, cups, or cones. CNFs are very electrically and thermally conductible and have outstanding mechanical qualities. They range in diameter from 10 nm to 500 nm. As a result, these CNFs are used in a wide range of industries, including photocatalysis, energy devices, sensors, medication delivery, and nanocomposites.
One Dimension Nanoparticles14,15
These might be thin sheets or artificial surfaces. Engineering, chemistry, and electronics have been their areas of expertise for a long time. A large number with very thin films or single layers ranging in thickness from 1-100 nm are used in solar cells and catalysis. Further examples include optical and magneto-optical devices, as well as fibre optic systems, information storage systems, and chemical and biological sensors.
Two-Dimension Nanoparticles16
Carbon nanotubes (CNTs) have size range of 1 nm to 100 nm and made up of hexagonally organised carbon atoms. CNTs come in two distinct forms: single-walled carbon nanotubes (SWCNTs) and multi-walled carbon nanotubes (MWCNTs). What distinguishes them from others is their tiny size and distinct mechanical, electrical, and physical properties. The ability of carbon nanotubes to absorb molecules is greater. Carbon nanotubes with extraordinary stability.
Synthesis of Nanomaterials
Vacuum Deposition and Vaporization17
The vacuum deposition method involves the vaporisation and subsequent deposition of elements, alloys, or compounds in an enclosed space without air. The vaporisation source is the only one that employs heat processes to evaporate materials. The procedure run in vacuum levels of 10 to 0.1 MPa and at pressures of less than 0.1 Pa (1 m Torr). The substrate might be 500°C or room temperature. The saturation or equilibrium vapour pressure of a material is the vapour pressure of that substance in balance with the solid or liquid surface. In the event that the rate of vaporisation is elevated, a respectable deposition rate can be achieved for vacuum deposition. In dense vapour clouds, multibody collisions may cause the nucleation of the vapour phase; To produce the necessary collision and cooling for nucleation, the atoms are circulated through a gas. These constituents called ultra fine particles or clusters due to their size range of 1 to 100 nm.
Nanolithography18,19
The study creating structures at when one dimension is at least one nanometre in size,1 and 100 nm, Nanolithography is the term for it. A variety of nano lithographic techniques are available, including optical, electron-beam, multiphoton, nanoimprint, and scanning probe lithography. Lithography is a broad term for the process of selectively removing material to print a required form or arrangement on a delicate substance to light. Producing a single nanoparticle to a cluster with the appropriate size and form is one of nanolithography’s primary benefits. The drawbacks include the need for pricey, sophisticated equipment and the related costs.
Gas Condensation20
Synthesising nanocrystalline metals and alloys was initially accomplished using gas condensation. Under this technique, a metallic or inorganic substance is evaporated in an environment of 1 to 50 m employing thermal evaporation techniques, including electron beam evaporation, in a pub devices or Joule heated refractory crucibles. Using this technique, ultra fine particles (100 nm) are formed by gas phase collision at high residual gas pressure. As evaporating atoms collide with leftover gas molecules, ultrafine particles are created. More than 3 mPa (10 torr) of pressure are needed in this gas. High energy electron beams, low energy electron beams, resistive heating, and inducting heating are examples of vaporisation sources. Atoms in the gas phase incorporate into one another to form clusters, which grow in the region of the source by homogeneous nucleation. The system consists of a compaction device, a liquid nitrogen-filled cluster collecting device, and an evaporation source equipped with an ultra-high vacuum (UHV) system, and a cold finger scraper assembly. The supersaturation zone around the Joule heating device is where atoms condense during heating. A scraper that takes the shape of a metallic plate removes the nanoparticles. From W, Ta, or Mo refractory metal crucibles, evaporation is to be carried out. The electron beam evaporation method is to be applied if the metals react with crucibles. A few of the method’s drawbacks include temperature ranges, inconsistent evaporation rates in an alloy, and incompatibility between the source and precursor. An atmosphere of inert gas, such as He, is formed as Fe evaporates. The Fe atoms that have evaporated lose their energy when they collide with other atoms and condense into tiny crystallite crystals, which eventually build up into a loose powder. Instead of using thermal evaporation, sputtering or laser evaporation can be employed.
Pyrolysis21
To produce nanoparticles on a big scale, the most often utilised technique in industry is pyrolysis. A precursor is burned by flame in this process. High pressure comes via a little hole where it burns is used to deliver precursor, which can either liquid or vapour, into the furnace. Air classification is then applied to the combustion or by-product gases to recover the nanoparticles. To attain elevated temperatures that enable straightforward evaporation, some furnaces utilise lasers, plasma in place of flames. Simple, effective, economical, continuous, and high-yield processes are the benefits of pyrolysis.
Sol-gel22,23
Particles dispersed colloidally in a liquid phase are known as sols. Gels are solid macromolecules that dissolve in liquids. Due to its simplicity in generating most nanoparticles and ease of usage, the bottom-up approach that is most frequently utilised is sol-gel. Wet-chemical means that first step in creating an integrated system of discrete particles is the use of a chemical solution. As precursors, metal oxides and chlorides typically needed for sol-gel process. Subsequently, precursor the host liquid is combined via sonication, shaking, or stirring form system consisting of solid liquid phase. After a phase separation, the nanoparticles are recovered using a variety of methods, such as sedimentation, filtering, and centrifugation; drying is then employed to eliminate any more moisture.
Spinning24
To spin produce nanoparticles, spinning disc reactor (SDR) is utilized. It features rotating disk within reactor or chamber that allows for temperature and other physical parameter adjustments. Nitrogen or other inert gases are frequently pumped into reactor to inhibit chemical reactions and eliminate oxygen. At varying speeds, the disk revolves as water and precursor are pushed in. The spinning, precipitating, collecting, and Atoms or molecules fuse together as the mixture dries. The liquid flow rate, properties of nanoparticles synthesized from SDR are influenced by many operational factors including disc rotation speed, liquid/precursor ratio, feed location, disc surface.
Thermal Decomposition25
Heat causes a compound’s chemical bonds to break, resulting in thermal breakdown, an endothermic chemical process. The decomposition temperature is the precise temperature at which an element undergoes chemical breakdown. The metal is broken down at particular temperatures, causing a chemical reaction that yields secondary compounds, and this process creates the nanoparticles.
Chemical Precipitation26,27
This method uses the arrested precipitation approach to regulate the size. The fundamental approach has been to synthesise and study the nanomaterial in situ, that is, inside the same liquid media, to prevent small crystallites from aggregating and from going through physical changes. By employing non-aqueous solvent at lower temperature for synthesis, double layer repulsion of crystallites regulated both Oswald ripening and thermal coagulation. The amalgamation required reaction between the component materials in an appropriate solvent. The purpose of the surfactant is to keep the particles apart. Centrifugation is used to separate resulting nanocrystal, which then cleaned hoover dried. In order to provide real quantum confinement, After the material had dried, it was subjected to ultraviolet light curing in the hopes of polymerizing the surfactant coating on the nano cluster’s surface.
Methods of Preparation of Nanoparticles
Emulsification – Diffusion28
It’s an altered variation of the solvent evaporation technique. This technique uses the water miscible solvent as the oil phase, with a tiny quantity of water immiscible organic solvent added. Small particles arise as a result of an interfacial turbulence that formed between the two phases by the spontaneous diffusion of solvents. Particle size can reduce concentration water miscible solvent rises. Drugs that are hydrophilic or hydrophobic can be treated with either the diffusion approach or solvent evaporation. When a medication is hydrophilic, it is necessary to create an internal aqueous phase-dissolved multiple w/o/w emulsion containing the drug. As such, technique has the benefit of not requiring the use of salts, which avoids the need for extensive purification stages even though it is a variant of the salting out operation. One limitation of this approach is that hydrophilic medicines in nanoparticles are not well entrapped by it. The effectiveness of water-soluble medications in nanospheres can be increased, nevertheless, by mixing medium chain glycerides with an aqueous solution.
Solvent Displacement/ Nanoprecipitation29
Using this procedure, semi-polar water miscible solvent comprising a polymer, drug, and lipophilic surfactant solution is combined and then put into a stabiliser solution while being stirred. Nanoparticles are formed as a result of rapid solvent diffusion. Because hydrophilic medications do not interact well with polymers, the drug diffuses from the polymer in the organic phase into the surrounding aqueous environment, resulting in lower drug loading efficiency than hydrophobic pharmaceuticals. Barichello et al. used the nano precipitation technique to show that PLGA nanoparticles increased the bioavailability of peptides and proteins. Precipitation was developed Jaing et al. to create ibuprofen (IBU) nanoparticles stabilised by DEAE dextran (Dex). Through creation of core shell particles, medications that are poorly soluble in water may be efficiently distributed and have relatively excellent stability while being stored. IBU is precipitated in a highly saturated solution as part of the process, and Dex is then electrostatically deposited onto the precipitated IBU particles. Which medication, polymer, solvent, or non-solvent solution the nanoparticles would be placed in created drug effectively encapsulated is a challenge in this manufacturing procedure.
Emulsion/ Evaporation30
This method can produce particles that are a few nanometres to micrometres in size by varying the stirring rates and conditions, and it has shown to be quite effective in integrating lipophilic medications. A stabilizer-containing aqueous solution is used to emulsify an organic solution of polymer medication. In order to achieve a fine aqueous dispersion of nanoparticles, A high energy source is used to decrease the size of the droplets, and the organic phase is then allowed to evaporate under vacuum or reduced pressure before being freeze dried for storage.
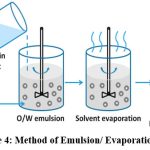 |
Figure 4: Method of Emulsion/ Evaporation methodClick here to view Figure |
Coacervation or Ionic Gelation Method31
These days, a lot of study is being done on nanoparticles made of hydrophilic polymers that are biodegradable, like sodium alginate, chitosan, and gelatin. A technique for creating hydrophilic chitosan nanoparticles by ionic gelation was presented by Calvo and associates. The process entails combining two aqueous phases: one containing the polymer chitosan and other poly anion sodium tri-polyphosphate. PEO-PPO is a di-block co-polymer. In this process, the positively charged chitosan amino group combines with the negatively charged tripolyphosphate to generate coacervates that are nanoscale in size. While ionic gelation includes a substance transitioning from a lipid to a gel under ionic contact circumstances at ambient temperature, coacervates are the outcome of electrostatic interaction between two aqueous phases.
Spray Drying32
The process of spray-drying has been frequently employed to produce particles of a micron size. Spray drying is the technique of turning solution droplet into dry particle in a single step by allowing the solvent to evaporate. Spray-drying of temperature-sensitive substances including proteins and enzymes has proven effective. Evidence has shown that it is possible to produce particles including various polymers medications, both soluble and insoluble in water, having to worry about the pharmaceuticals seeping into another phase. As a result, drug can be recovered almost entirely from the particles, and the properties of the particles, particularly their morphology, can be controlled by the spray-drying variables and solvent properties.
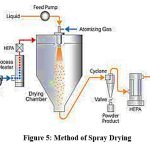 |
Figure 5: Method of Spray DryingClick here to view Figure |
Salting Out33
Medication and polymers soluble in polar solvent, such ethanol or acetone, can be processed with this method. Aqueous solution comprising stabiliser and salting out agent is stirred before a somewhat water miscible solvent; a polymer and medicine solution is introduced. The organic solvent diffuses into the aqueous phase of the o/w emulsion when a tiny amount of water is introduced for dilution, resulting in particles in the nano size range. Compared to nano precipitation approach, this procedure is different. When using the salting out procedure, it is completely miscible with the external aqueous phase, the organic phase, but its miscibility is hindered PVA saturation of the external aqueous phase.
Evaluation of Nanoparticles
Particle Size34
When characterising nanoparticles, one of the most fundamental and significant measurements is the particle. It establishes whether a particle is nano- or micro-scale based on its size, distribution, and composition. Electron microscopy is most typically used to measure the particle size and distribution. Scanning electron microscopy and transmission electron microscopy are used to measure particles and clusters, whereas laser diffraction methods are used to assess bulk samples in the solid phase. Photon correlation spectroscopy and centrifugation are used to quantify particles in a liquid phase. Because of its superior speed and accuracy compared to alternative approaches, a Scanning Mobility Particle Sizer (SMPS) is utilized for this purpose, because using PET scans of gaseous particles are challenging and disrespectful.
Structure and Cyclability35
It is possible to determine structure and crystallinity in a few ways. X-ray diffraction often used to determine both crystallinity and structure. The initial stage of x-ray imaging is produced when stream very energetic electrons collide with metal object. A filter blocks in the vicinity of the x-ray source. These low energy photons, or high energy beams, are the only ones that pass through the patient and onto a sheet of film to be photographed. Liquids, gases, and solids may all be visible to the x-ray. X-ray beams, wavelength, and intensity all influence the penetration’s strength, calibre, and location.
Zeta Potential36
It is usual practice to characterise a nanoparticle’s surface charge property using its Zeta potential. It is a reflection of the particle’s electrical potential, which is based on the
makeup as well as the medium in which it is distributed. Because the surface charge stops the particles from aggregating, it has been demonstrated that nanoparticles with zeta potentials exceeding (±) 30 mV are stable in suspension.
Drug Entrapment Efficiency37
By using ultracentrifugation for 30 minutes at 500C and 10,000 rpm, nanoparticles were extracted from aqueous medium. Following that, a decanter was used to distribute the supernatant solution into phosphate buffer saline pH 7.4. Fully eliminate drug molecules that were not ensnared, the process was therefore carried out again. The amount of drug contained in the nanoparticles was calculated by deducting the amount of drug utilized to make the nanoparticles from the amount of drug in the water-based medium.
UV-Visible Absorption Spectroscopy38
To ascertain the optical characteristics of a solution, absorbance spectroscopy is employed. The amount of light that is gathered is measured after shining light on the sample solution. when each wavelength’s absorbance is measured and the wavelength is altered. The concentration of a solution may be ascertained from the absorbance by applying Beer-Lambert’s Law. When using ultraviolet-visible spectrophotometers for optical measurements, there are many absorbance peaks, including one at 410 nm.
X-Ray Diffraction Analysis39
Crystal structure and shape can be ascertained by the widely used method of X-ray diffraction. The intensity changes with the quantity of a component. In addition to giving details about the location of the atoms and the electron density inside the unit cell—this technique also provides information on the form and dimension of the translational symmetry It determines whether a particle is metallic by analysing its component cell from peak to valley. A Rota Flex Diffraction Metre associated with an X and = 1.5406 were utilised to compute XRD patterns using Cu K radiation.
Scanning Electron Microscopy40
Morphology analysis with direct visualisation is provided by scanning electron microscopy (SEM). In morphological and sizing analysis, electron microscopy-based methods have several benefits; yet, the size distribution and actual population average are not well-represented. Nanoparticle solution should be dried and then mounted on a sample holder for SEM analysis. A sputter coater should then be used to coat the sample with conductive metal, such gold. A concentrated fine beam of electrons is then employed to scan the material. From the secondary electrons released from the sample surface, one may determine the surface properties of the material. Since the electron beam might harm the polymer, the nanoparticles need to be able to endure vacuum. Findings from dynamic light scattering are similar to the mean size determined by SEM. These methods also need additional information on size distribution, which can be expensive and time-consuming.
Transmission electron microscope41
While operating a different premise than SEM, TEM frequently produces data of same kind. Because the sample must extremely thin for the electron transmission, TEM sample preparation difficult, time-consuming. Support grids or films are coated with the dispersion of nanoparticles. Negative staining materials, such phosphotungstic acid, or its derivatives are used to fix nanoparticles so they can tolerate the instrument vacuum and be easier to handle. An alternative technique involves immersing the sample in vitreous ice and then subjecting it to liquid nitrogen temperatures. By passing ultra-thin sample through an electron beam and interacting with it during Atomic Force Microscopy, one may determine the surface properties of the sample. Using an atomic-scale probe tip to physically scan materials at the sub-micron level, atomic force microscopy (AFM) provides extremely high resolution in particle size assessment. A topographical map of the sample is produced by instrument using forces acting on the sample surface and tip. Generally, depending on the characteristics of the sample, either contact or noncontact scanning is used. An atlas of the terrain is developed. in contact mode by tapping the probe on the sample’s surface, and in non-contact mode, the probe hovers above the conducting surface.
Stability of Nanoparticles42
The stability of the generated nanoparticles was investigated by keeping the formulation in 90 days at 4°C, 1°C, 30°C, and 2°C in the stability chamber. After that, samples were checked at different intervals—0, 1, 2, and 3 months—to check for physical changes or to find out how much medication was in them and how quickly it was released.
Applications of Nanoparticles
Cosmetics And Sunscreens43
Long-term stability while use is absent from traditional UV protection sunscreens. There are several benefits to using sunscreen that contains nanoparticles, including titanium dioxide. Certain sunscreens use titanium oxide and zinc oxide nanoparticles because of their ability to both absorb and reflect UV radiation while remaining transparent to visible light. A pigment in certain lipsticks is made of iron oxide nanoparticles.
Targeted Drug Delivery44
Accurately delivering the medication to the desired cells or tissue is a crucial component of drug delivery. medicine targeting systems have to have the ability to regulate how a medicine enters the body. The so-called “magic bullet,” which Paul Ehrlich first envisaged at the turn of the 20th century and in which the medication is precisely targeted to the specific side of action, is substantially different from the delivery technology of today. Delivering the medication at the appropriate time and place presents another problem made possible by nanotechnology.
In the coming years, nanotechnology is predicted to bring about a significant shift in manufacturing and have a profound effect on the life sciences, encompassing medication delivery, diagnostics, nutraceuticals, and biomaterials production.
Medicine45,46
Because nanoparticles can be used to deliver drugs more efficiently, nanotechnology has benefited medicine. The medication delivered to specific cells using nanoparticles. Through administration of the drug in prescribed dosage location, overall amount of medicine consumed and its adverse effects are greatly reduced. Side effects and expense are decreased using this strategy. Tissue engineering is the technique of using nanotechnology to reproduce damaged tissue and make it whole again. The tissue engineering method possesses potential to replace traditional treatments such as artificial implants and organ transplants. One example of this creation of bone scaffolds made of carbon nanotubes. Gold possesses long been used in medicine. Gold is employed in several Ayurvedic procedures, an Indian medicinal system. Using gold to improve memory is one such recommendation. As part of several medical procedures, a newborn gold’s mental health is improved.
Nanoparticles In Food47,48
Foods that have manmade nanoparticles added during growth, production, processing, or packaging are referred to as nanofoods. These foods may also involve nanotechnology tools, methods, or both. The development of nanofood is being done for several reasons. improved flavour and nutrition, lower manufacturing and customer expenses, and increase of food safety are some of them. Moreover, nanofood offers a number of advantages, such as extended shelf life, novel flavour combinations, and ingredients that support health. Food processing, bioavailability enhancement, agricultural uses, and other facets of the food chain are all being affected by the fast-emerging field of nanotechnology in food.
Cardiovascular Diseases49
The medical industry has made extensive use of nanotechnology to provide innovative platforms for therapy and diagnosis that can replace conventional techniques. Cardiovascular nanomedicine (CVN) focused mostly on creative explanations for the shortcomings of existing CVD therapies. The original goal of CVN’s nano systems was to improve the bioavailability, stability, and safety of already-approved medications. Nanomaterials vary from ordinary materials in their physicochemical characteristics, as their high surface energy enhanced surface area to volume ratio, which affect biological activity and protein adhesion. Therefore, the regulated and targeted administration of different functional components for the management of lipid metabolism disorders and other illnesses associated to CVD is made possible by nanotechnology, which offers a safe and effective platform. Many researchers are actively working on developing nano-drug carrier systems for CVD detection and treatment. In order to combat atherosclerosis and other CVDs, several medicines can be encapsulated in nanoparticles. There are two uses for encapsulation in nanomedicine. First, it protects the environment in vivo or off-target characteristics from hazardous medications by verifying that the drug’s release is regulated by the material’s features and that its surface qualities allow for targeted delivery of the drug to the afflicted area. Additionally, the encapsulation technique protects pharmaceuticals from unwanted changes and deterioration. Polymeric nanoparticles, silica nanoparticles, carbon nanotubes, polymeric micelles, quantum dots, nanofibers, and nanocrystals are a few examples of nano formulations for controlled drug delivery. Surface functionalization is also the most effective way to target specific components of plaque or avoid immune system and clearance by the body. For this concept to work, a high surface area to volume ratio is essential. Targeting ligands are the primary moieties (antibodies, peptides, aptamers, or small molecules) attached to nanoparticles that are specific for components in plaque and PEGylation, which discusses stability and stealth in vivo.
Nanomedicine50
Nanoparticle-based tailored drug delivery technologies that increase the accuracy and potency of cancer therapies. creation of imaging agents at the nanoscale for quicker and more precise illness detection
Asthma51
Chronic airway inflammation, bronchial hyperresponsiveness, and reversible airway blockage are the hallmarks of asthma, an inflammatory lung disease. Prolonged exposure to allergens triggers an inflammatory reaction in the lungs, which leads to lung tissue destruction and constriction of the tiny airways. Previous research has demonstrated the anti-inflammatory properties of nanoparticles in a range of inflammatory conditions. By aiding in the drug’s transport to the intended tissue and enhancing its deposition in the lungs, nanoparticles boost the therapeutic impact. Several investigations, both in vivo and in vitro, documented the impact of nanoparticles on asthma.
Chronic obstructive pulmonary diseases52
The chronic inflammatory lung disease COPD obstructs airways. Breathing difficulties, coughing, and increased mucus flow are some of its symptoms. Long-term exposure to irritating or airborne particles—which are frequently brought on by cigarette smoke—causes this condition. In COPD, the airways were less flexible and narrower, which led to air trapping in the lungs. Because they are used in small amounts and may be precisely targeted to the sick tissue milieu, nanomaterial medicines have demonstrated therapeutic potential against a variety of respiratory illnesses with minimal adverse effects. Several in vitro and in vivo investigations have demonstrated the impact nanoparticles on COPD.
Conclusion
For a range of drugs, nanoparticles provide a viable drug delivery method. A revolutionary technology that has transformed several sectors is nanotechnology. Around the world, researchers are looking at newer applications for this technology. Nanoparticle technology is generally applicable and can be beneficial for drugs with limited solubility and bioavailability. Any medication may be transformed into drug nanoparticles, which improve a drug’s saturation solubility, rate of dissolution, and overall ability to stick to surfaces. Nanoparticulate drug delivery systems are becoming more regarded as an efficient means of administering biological drugs. Furthermore, nanoparticles offer targeted and regulated release, which further enhances their effectiveness as a therapeutic approach. For these reasons, nanoparticulate medicine delivery systems appear to be a promising and feasible commercial approach for the biopharmaceutical industry.
Acknowledgement
The authors are thankful to the management of Govindrao Nikam College of Pharmacy. Sawarde.
Conflict of Interest
The authors declare there is no conflict of interest
Funding Sources
The authors’ received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Authors’ Contribution
All authors have collected data and arranged as per article
References
- Arole VM, Munde S V. Fabrication of Nanomaterials By Top-Down and Bottom-Up Approaches-an Overview. JAASTMaterial Sci (Special Issue. 2014;1(2):2-89.
- Saba H. A Review on Nanoparticles : Their Synthesis and Types. Res J Recent Sci Res J Recent Sci Uttar Pradesh ( Lucknow Campus ). 2014;4(February):1-3.
- Dubchak S, Ogar A, Mietelski JW, Turnau K. Influence of silver and titanium nanoparticles on arbuscular mycorrhiza colonization and accumulation of radiocaesium in Helianthus annuus. Spanish J Agric Res. 2010;8(SPL ISS.). doi:10.5424/sjar/201008s1-1228
CrossRef - Sapra P, Tyagi P, Allen T. Ligand-Targeted Liposomes for Cancer Treatment. Curr Drug Deliv. 2005;2(4):369-381. doi:10.2174/156720105774370159
CrossRef - Kumar Sahu M. Semiconductor Nanoparticles Theory and Applications. Int J Appl Eng Res. 2019;14(2):491-494. http://www.ripublication.com
- Matyushkin LB, Ryzhov OA, Aleksandrova OA, Moshnikov VA. Synthesis of metal and semiconductor nanoparticles in a flow of immiscible liquids. Semiconductors. 2016;50(6):844-847. doi:10.1134/S1063782616060166
CrossRef - Aleksandra Zieli ´nska 1 2, , Filipa Carreiró 1, Ana M. Oliveira 1, Andreia Neves 1 BP 1, 3 DNV, et al. Polymeric Nanoparticles: Production, Characterization, Toxicology and Ecotoxicology. Molecules. 2020;25:3731.
CrossRef - Elmowafy M, Shalaby K, Elkomy MH, et al. Polymeric Nanoparticles for Delivery of Natural Bioactive Agents: Recent Advances and Challenges. Polymers (Basel). 2023;15(5):1-34. doi:10.3390/polym15051123
CrossRef - Holmannova D, Borsky P, Svadlakova T, Borska L, Fiala Z. Carbon Nanoparticles and Their Biomedical Applications. Appl Sci. 2022;12(15):1-21. doi:10.3390/app12157865
CrossRef - Zaytseva O, Neumann G. Carbon nanomaterials: Production, impact on plant development, agricultural and environmental applications. Chem Biol Technol Agric. 2016;3(1):1-26. doi:10.1186/s40538-016-0070-8
CrossRef - Mody V, Siwale R, Singh A, Mody H. Introduction to metallic nanoparticles. J Pharm Bioallied Sci. 2010;2(4):282. doi:10.4103/0975-7406.72127
CrossRef - Venkatesh N. Metallic Nanoparticle: A Review. Biomed J Sci Tech Res. 2018;4(2):3765-3775. doi:10.26717/bjstr.2018.04.0001011
CrossRef - Feng L, Xie N, Zhong J. Carbon nanofibers and their composites: A review of synthesizing, properties and applications. Materials (Basel). 2014;7(5):3919-3945. doi:10.3390/ma7053919
CrossRef - Zhao Y, Hong H, Gong Q, Ji L. 1D nanomaterials: Synthesis, properties, and applications. J Nanomater. 2013;2013:2013-2014. doi:10.1155/2013/101836
CrossRef - Garnett E, Mai L, Yang P. Introduction: 1D Nanomaterials/Nanowires. Chem Rev. 2019;119(15):8955-8957. doi:10.1021/acs.chemrev.9b00423
CrossRef - Benelmekki M. One-dimensional nanomaterials. Nanomaterials. Published online 2019:4-1-4-15. doi:10.1088/2053-2571/ab126dch4
CrossRef - Mattox DM. Introduction. Handb Phys Vap Depos Process. Published online 2010:1-24. doi:10.1016/b978-0-8155-2037-5.00001-0
CrossRef - Pimpin A, Srituravanich W. Reviews on micro- and nanolithography techniques and their applications. Eng J. 2012;16(1):37-55. doi:10.4186/ej.2012.16.1.37
CrossRef - Sebastian EM, Jain SK, Purohit R, Dhakad SK, Rana RS. Nanolithography and its current advancements. Mater Today Proc. 2019;26(xxxx):2351-2356. doi:10.1016/j.matpr.2020.02.505
CrossRef - Fan L, Harris BW, Jamaluddin A, et al. Understanding gas-condensate reservoirs. Oilf Rev. 2005;17(4):14-27.
- Demirbaş A, Arin G. An overview of biomass pyrolysis. Energy Sources. 2002;24(5):471-482. doi:10.1080/00908310252889979
CrossRef - Bokov D, Turki Jalil A, Chupradit S, et al. Nanomaterial by Sol-Gel Method: Synthesis and Application. Adv Mater Sci Eng. 2021;2021. doi:10.1155/2021/5102014
CrossRef - Chen YJ. Sol-Gel Methods Lisa klein. http://www.uio.no/studier/emner/matnat/ kjemi/KJM5100/h06/undervisningsmateriale/10KJM5100_2006_sol_gel_d.pdf
- Music O, Allwood JM, Kawai K. A review of the mechanics of metal spinning. J Mater Process Technol. 2010;210(1):3-23. doi:10.1016/j.jmatprotec.2009.08.021
CrossRef - Odularu AT. Metal Nanoparticles: Thermal Decomposition, Biomedicinal Applications to Cancer Treatment, and Future Perspectives. Bioinorg Chem Appl. 2018;2018. doi:10.1155/2018/9354708
CrossRef - Matthews JA. Chemical Precipitation. Encycl Environ Chang. 2014;3:141-142. doi:10.4135/9781446247501.n624
CrossRef - Nazari M, Ghasemi N, Maddah H, Motlagh MM. Synthesis and characterization of maghemite nanopowders by chemical precipitation method. J Nanostructure Chem. 2014;4(2):2-6. doi:10.1007/s40097-014-0099-9
CrossRef - Trotta M, Debernardi F, Caputo O. Preparation of solid lipid nanoparticles by a solvent emulsification-diffusion technique. Int J Pharm. 2003;257(1-2):153-160. doi:10.1016/S0378-5173(03)00135-2
CrossRef - Beck-Broichsitter M. Solvent impact on polymer nanoparticles prepared nanoprecipitation. Colloids Surfaces A Physicochem Eng Asp. 2021;625(March):126928. doi:10.1016/j.colsurfa.2021.126928
CrossRef - Staff RH, Schaeffel D, Turshatov A, et al. Particle formation in the emulsion-solvent evaporation process. Small. 2013;9(20):3514-3522. doi:10.1002/smll.201300372
CrossRef - Chatterjee S, Salaün F, Campagne C, Vaupre S, Beirão A, El-Achari A. Synthesis and characterization of chitosan droplet particles by ionic gelation and phase coacervation. Polym Bull. 2014;71(4):1001-1013. doi:10.1007/s00289-014-1107-4
CrossRef - Satoshi O, Izutsu K ichi, Lechuga-Ballesteros. Drying Technologies for Biotechnology And.; 2020.
- Sadeghi R, Jahani F. peg的盐析Salting-In and Salting-Out of Water-Soluble Polymers in Aqueous Salt Solutions Supporting Information. J Phys Chem B. 2012;116:5234-5241.
CrossRef - de Rooij GH. Methods of Soil Analysis. Part 4. Physical Methods. Vadose Zo J. 2004;3(2):722-723. doi:10.2136/vzj2004.0722
CrossRef - Szabó DV, Kilibarda G, Schlabach S, Trouillet V, Bruns M. Structural and chemical characterization of SnO 2-based nanoparticles as electrode material in Li-ion batteries. J Mater Sci. 2012;47(10):4383-4391. doi:10.1007/s10853-012-6292-7
CrossRef - Gupta V, Trivedi P. In Vitro and in Vivo Characterization of Pharmaceutical Topical Nanocarriers Containing Anticancer Drugs for Skin Cancer Treatment. Elsevier Inc.; 2018. doi:10.1016/B978-0-12-813687-4.00015-3
CrossRef - Ghadiri M, Fatemi S, Vatanara A, et al. Loading hydrophilic drug in solid lipid media as nanoparticles: Statistical modeling of entrapment efficiency and particle size. Int J Pharm. 2012;424(1-2):128-137. doi:10.1016/j.ijpharm.2011.12.037
CrossRef - Grand J, Auguié B, Le Ru EC. Combined Extinction and Absorption UV-Visible Spectroscopy as a Method for Revealing Shape Imperfections of Metallic Nanoparticles. Anal Chem. 2019;91(22):14639-14648. doi:10.1021/acs.analchem.9b03798
CrossRef - Dorofeev GA, Streletskii AN, Povstugar I V., Protasov A V., Elsukov EP. Determination of nanoparticle sizes by X-ray diffraction. Colloid J. 2012;74(6):675-685. doi:10.1134/S1061933X12060051
CrossRef - Mohan AC, Renjanadevi B. Preparation of Zinc Oxide Nanoparticles and its Characterization Using Scanning Electron Microscopy (SEM) and X-Ray Diffraction(XRD). Procedia Technol. 2016;24:761-766. doi:10.1016/j.protcy.2016.05.078
CrossRef - Filippov SK, Khusnutdinov R, Murmiliuk A, et al. Dynamic light scattering and transmission electron microscopy in drug delivery: a roadmap for correct characterization of nanoparticles and interpretation of results. Mater Horizons. 2023;10(12):5354-5370. doi:10.1039/d3mh00717k
CrossRef - Tso CP, Zhung CM, Shih YH, Tseng YM, Wu SC, Doong RA. Stability of metal oxide nanoparticles in aqueous solutions. Water Sci Technol. 2010;61(1):127-133. doi:10.2166/wst.2010.787
CrossRef - Fytianos G, Rahdar A, Kyzas GZ. Nanomaterials in cosmetics: Recent updates. Nanomaterials. 2020;10(5):1-16. doi:10.3390/nano10050979
CrossRef - Singh AP, Biswas A, Shukla A, Maiti P. Targeted therapy in chronic diseases using nanomaterial-based drug delivery vehicles. Signal Transduct Target Ther. 2019;4(1):1-21. doi:10.1038/s41392-019-0068-3
CrossRef - Haleem A, Javaid M, Singh RP, Rab S, Suman R. Applications of nanotechnology in medical field: a brief review. Glob Heal J. 2023;7(2):70-77. doi:10.1016/j.glohj.2023.02.008
CrossRef - Murthy SK. Nanoparticles in modern medicine: state of the art and future challenges. Int J Nanomedicine. 2007;2(2):129-141. http://www.ncbi.nlm.nih.gov/pubmed/ 17722542%0A http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC2673971
- Couto C, Almeida A. Metallic Nanoparticles in the Food Sector: A Mini-Review. Foods. 2022;11(3):10-12. doi:10.3390/foods11030402
CrossRef - Dash KK, Deka P, Bangar SP, Chaudhary V, Trif M, Rusu A. Applications of Inorganic Nanoparticles in Food Packaging: A Comprehensive Review. Polymers (Basel). 2022;14(3):1-17. doi:10.3390/polym14030521
CrossRef - Hu Q, Fang Z, Ge J, Li H. Nanotechnology for cardiovascular diseases. Innovation. 2022;3(2):100214. doi:10.1016/j.xinn.2022.100214
CrossRef - Chen G, Roy I, Yang C, Prasad PN. Nanochemistry and Nanomedicine for Nanoparticle-based Diagnostics and Therapy. Chem Rev. 2016;116(5):2826-2885. doi:10.1021/acs.chemrev.5b00148
CrossRef - Zuo X, Guo X, Gu Y, et al. Recent Advances in Nanomaterials for Asthma Treatment. Int J Mol Sci. 2022;23(22). doi:10.3390/ijms232214427
CrossRef - Xu Y, Liu H, Song L. Novel drug delivery systems targeting oxidative stress in chronic obstructive pulmonary disease: a review. J Nanobiotechnology. 2020;18(1):1-25. doi:10.1186/s12951-020-00703-5
CrossRef
Accepted on: 13-08-2024
Second Review by: Dr. Bhavana Gundavarapu
Final Approval by: Dr. Ali Mohamed Elshafei









