Manuscript accepted on : 08 December 2017
Published online on: --
Mohammed J. Haider1, Duha S. Ahmed2 , M. R. Mohammad2 and Adawiya J. Haider2
, M. R. Mohammad2 and Adawiya J. Haider2
1Electrical Engineering Department, University of Technology, Baghdad, Iraq.
2Applied Science Department, University of Technology, Baghdad, Iraq.
Corresponding Author E-mail: duhasaadi2015@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/2599
ABSTRACT: In the present work, a new and economic method of functionalized Multi walled Carbon Nanotubes (MWCNTs) with olive oil has been improved as compared to the traditional methods, that composed of catalysts such as fatty acids (FFA), glycerol and triacylglycerols of oxidation power. This method is initiated by ultrasoniction the mixture which leads to functionalize the surface during the process of interaction with oil olive for a short time (30 min). Then, the results of this work were examined by using Scanning Electron Microscopy (SEM) and High Resolution Transmission Electron Microscopy (HRTEM) to study the morphology of the surface treated with olive oil, X-ray diffraction (XRD) was employed to confirm the functionalized method and using Fourier transformed infrared spectroscopy (FTIR) that shows the formation of functional group on to MWCNTs surface such as C=O and COOH. The final part includes a study antibacterial activity of Multi walled Carbon nanotubes (MWCNTs) against gram negative (E. coli and Salmonella typhi) and gram positive (S. aureus) bacteria using direct cell counting. Besides, evaluating the antibacterial assays of treated MWCNTs after incubated for various time to elucidate the interaction between functionalized MWCNTs and cell membrane to determine the study the adsorption of bacteria on the surface of (MWCNTs), which are used in environmental applications, such as sensing of contaminated water and using as a filter.
KEYWORDS: Antibacterial Activity; Fatty Acids (FFA); Functionalized Multi Walled Carbon Nanotubes (Mwcnts); Traditional MethodsOil Olive;
Download this article as:| Copy the following to cite this article: Haider M. J, Ahmed D. S, Mohammad M. R, Haider A. J. Modification of Functionalized Multi Walled Carbon Nanotubes by Olive oil as Economic Method for Bacterial Capture and Prevention. Biosci Biotech Res Asia 2017;14(4). |
| Copy the following to cite this URL: Haider M. J, Ahmed D. S, Mohammad M. R, Haider A. J. Modification of Functionalized Multi Walled Carbon Nanotubes by Olive oil as Economic Method for Bacterial Capture and Prevention. Biosci Biotech Res Asia 2017;14(4). Available from: https://www.biotech-asia.org/?p=28636 |
Introduction
Carbon nanotubes (CNTs), have great importance in the field of nanotechnology because of its extraordinary physical, electronic properties, mechanical properties and high surface area and since its discovery in 1991 by Japanese scientist Sumio Ijima (Ijima 1991).1 Carbon nanotubes are a promising part of growth due to possible applications in the fields of biomedical, water treatment, and alternative energy (Agui et al.2008; Balasubramanian et al.2006; Mita et al.2007).2-4 The idea of using CNTs such as (Single-Walled or Multi-Walled) can be applied three important areas: First, CNTs (SWCNTs or MWCNTs) used as a good media for adsorption microorganisms (biothreat- pathogens) in water treatment system.Second, CNTs (SWCNTs or MWCNTs) can be used as an effective filter for pathogen removal in water treatment. So that the purpose of using CNTs in the detection of bacteria due to the unique functional, physical and structural properties of CNTs by using functionalizing method to increase ability of MWCNTs to adsorb bacteria and virus elements from wastewater samples.5,6 In addition, using nanotube shape in removal bacteria because of a small volume to large surface area which known as high aspect ratio improving bacteria adsorption on functionalizing MWCNTs surface as compared to the activated carbon (AC).In addition to the excellent features of MWCNTs mentioned before, it is characterized by a strong van der Waals bond making it high aggregation material, poor dispersion and bundling in MWCNTs, which is considered as one of the significant problems when using as a adsorbed media in water treatment and poor adhesion of metal ions or bacteria on the surface of the MWCNTs.7-12 Since, the procedure of reducing aggregation raw-MWCNTs represented by functionaliztion of raw-MWCNTs using conventional method, (like mixture of concentrated acid solution such as H2SO4/HNO3 and HCL) to improve their wettability and improve dispersion [13-16]. For the purpose of developing a new and economic method to functionalize MWCNTs, organic solvent, such as (oil olive) is used to improve functionaliztion the surface of the MWCNTs compared to the traditional methods, which results in destruction of material structure.This method involve treated Raw-MWCNTs in Seed oil like (oil olive) as an economic method to increase their wetability and dispersion because of the olive oil composition contain a certain amount of glycerol and triacylglycerols that composed of a mixture of fatty acids (FFA) shown in Fig (1). The chemical formula of oil olive represented by: CH3 (CH2)nCOOH where (n) is typically an even number between 12 and 22 which represented oxidizing agent leads to oxidized the surface and generate functional groups ( like OH, and COOH) on the surface of MWCNTs. The carbon chains of all fatty acids have a carboxyl group (COOH) at one end.17 Also, Olive oil is analyzed for minor compounds, including the phenolics and the sterols. These compounds give olive oil its unique flavour and contribute greatly to the nutritional benefits and they play a major role as antioxidants.18 Since, the Seed solvent showing hydrophilic behaviors, and they lead to functionalize (MWCNTs) without destroying the structures of (MWCNTs) by using ultrasonic microwave technology when they are treated with organic solutions (oil olive) because raw–MWCNTs cannot be used in any biological applications, since raw–MWCNTs cannot be used as adsorbing of bacteria or viruses and other biological materials on the surface of MWCNT , as a result the introduction of functional groups represented by (COOH, -OH) on the surface of (MWCNTs) which facilitates the possibility of dispersion of MWCNTs after functionaliztion in polarity or non polarity solutions and proving the compatibility between the (MWCNTs) and host materials (such as metal oxide, bacteria, viruses ……. etc.). This method is one of modern methods because till nowadays there is no more information about using olive oil as solvent in functionalization of (MWCNT) through our research of outsourcing magazines or foreign research and according to research that are talking about functionalization method by using concentrated or dilute acids or catalyst factors.19 Functionalization MWCNT known by its ability to penetrate wall of bacteria because of its nanostructure shape and flexibility which facilitates their capture and kills bacteria proving their antimicrobial activity on the pathogens trapped on them.20 In this work, we developed functionalization method of MWCNTs using Seed oil like (oil olive) as an economic method to increase their dispersion and increase bacterial inactivation. Besides, the antibacterial assays were studied on bacterial organisms especially those found in diverse environments such as like Escherichia coli, Salmonella typhi and Staphylococcus aureus which their presence in water system is an evidence of contamination. Also, study the penetration or interaction of CNTs with cell bacterial walls during the adsorption process by testing their toxicity.
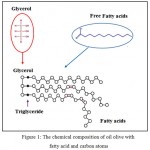 |
Figure 1: The chemical composition of oil olive with fatty acid and carbon atoms
|
Experimental Method
Functionalization of MWCNTs
In this step (raw-MWCNTs) are functionalized by treated with oil olive by using 0.1g of raw-MWCNTs with 100 mL of oil olive in flask of 500ml. In order to produce a stable suspension the flask was vortex for 10min and ultrasonic in water bath ultrasonic at temperature of 25°C for 30 min as shown in Fig (2a,b) that shows the mixture of MWCNTs and oil olive in glass tube before and after ultrasonic method. After that, the functionalized MWCNTs was collected via filtered method under vacuum in a Buchner filter with a 0.22 micro- membrane and then washed thoroughly 400ml of Chloroform ( ChCl3 ) to remove oil from each sample. The obtained film was easily peeled from the 0.22 membrane and dried at temperature 50oC for 24 h under vacuum in order to obtain the powder of MWCNTs containing functional groups as shown in Fig (3 a,b) and the schematic diagram of functionalized raw-MWCNTs with oil olive is shown in Fig (4 a,b) which represented the mechanism of treated and introduction of functional groups on the surface of (MWCNTs). These functional groups present in the treated carbon nanotubes by oil olive (F-MWCNTs) allow that their suspension remain stable by long periods
 |
Figure 2: Image of treated MWNTs by olive oil for 30 min a) MWNTs and b) functionalized MWNTs by olive oil solution after exposure to ultrasound waves for 30 minutes
|
 |
Figure 3: images of mixture collected F-MWNTs after filtration using vacuum-filtered through a 0.22 µm polycarbonate membrane, a) after drying treated MWNTs at T=60oC for 5 min ,b) shows MWCNTs after the filtration method with specifications pore size:0.2µm ,Diameter:47 mm
|
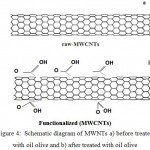 |
Figure 4: Schematic diagram of MWNTs a) before treated with oil olive and b) after treated with oil olive
|
Characterization
The surface morphology of treated-MWCNTs was performed using High Resolution Transmission Electron Microscopy (HRTEM, JEOL, JEM-2100 model) analysis. Studying the morphology of adsorption Gram negative and Gram positive bacteria on treated MWCNTs using SEM analysis.. X-Ray Diffraction (XRD, 6000-Shimadzu X-ray Diffractmeter) has been used to determine the crystal structure of raw and treated-MWCNTs. a dispersion test of raw and treated-MWCNTs by setting them in a polar solution (99%ethanol) with concentration of 5mg/ml and sonication 5min then monitored for one week to sediment in a polar solvent (ethanol) and compared it with the raw material before the treatment and recording notes about the deposition process of MWCNTs for each of the raw-MWCNTs before treatment and the sample treated with olive oil over a period of time. The antibacterial activity of the treated-MWCNTs with oil olive was performed towards bacterial strains of (E. coli and Salmonella typhi) as gram negative and (S. aureus) as gram positive bacteria which is provided by (Nanotechnology and advanced material research center in Iraq). The various types of bacterial culture were pre-culture on nutrient agar (N. agar) and incubated 24 h at 37°C. One milliliter of bacterial suspension was add to different concentrations (1,3,7) mg/ml of F-MWCNTs and cultivation at 370C overnight in shaker incubator at 160 rpm. Then, 100µ of bacterial suspension is used to inculcate on N. agar in Petri dishes and incubated at 37°C for 24 h. and counting the number of bacteria colonies survived after incubation and determined the percentage of survived bacteria.
Results and Discussion
Characterization Studies of Functionalized MWCNTs with oil Olive
Fig (5 a,b), indicate XRD patterns of raw and functionalized MWCNTs with oil olive (F-MWCNTs) samples.26 The results reveal significant diffraction peaks of raw-MWCNTs and F-MWCNTs that appeared at 2Ɵ of 26.1° as shown in Fig. (5 a,b) ,which indicates that the functionalization method using oil olive does not damage the structure of the MWCNTs. Besides, (002) reflection peaks was observed at the same 2Ɵ values in both raw and functionalized MWCNTs diffractions. According to these results the structure of MWCNTs after treated by oil olive is protected and did not make structural damages to the nanotubes as confirmed from XRD analysis.
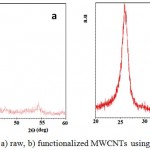 |
Figure 5: XRD patterns for a) raw, b) functionalized MWCNTs using oil olive treatment
|
In Fig (6 a,b) ,The FTIR spectrum indicate the presence of main absorption bands of raw-MWCNT (green line)and functionalized F-MWCNTs by oil olive (red line) in the range about 500 to 4000 cm-1, respectively. The FTIR results of raw and functionalized MWCNTs demonstrate absorption peak at 3433 cm-1 is due to O-H stretch indicating the presence of hydrogen bonded hydroxyl (-OH). Particularly, the peak sited around 2360 cm-1 is assigned as stretching vibration of strong H-bond. Besides, the absorption peaks C=C and C-C bonding is due to aromatic rings of the carbon skeleton and known as carbonyl group structure were found at 1631and 1377 cm 1, respectively.24,25 Also, observe typical band (C=O) at 1781 cm-1 that correspond to carboxyl groups (-COOH) produced on the functionalized MWNT surface. These absorption bands has been confirmed the introduction of functional groups (like carboxyl and hydroxyl groups) on F-MWCNTs after treated by oil olive without a severe damage in the structure of MWCNTs.
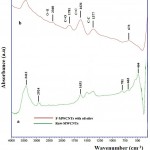 |
Figure 6: FTIR spectra of raw-MWCNTs a) and b) functionalized MWCNT with oil olive
|
Fig (7 a,b) display HRTEM (JEOL, JEM-2100 model) images of raw and functionalized MWCNTs. In Fig (7 b), it is noticed that there is no strong destruction occur in the structure of treated MWCNTs with oil olive as compared to raw-MWNTs before treatment in Fig (7 a). It is probably due to the mild condition of oil olive solvent as compared to the concentrated acids which caused sometimes severe structural damage to the nanotubes structure through tubes scission.21,22 HRTEM images of treated MWCNTs allowed reliable length measurements of only a few nanotubes, and all of them were between 1 and 3μm, similar to raw material.23Furthermore, aggregation property is reduced and the nanotube bundle are separated from each other which leads to enhanced of interaction between the bacteria and F-MWCNTs with increasing adsorption of cell by increasing MWCNTs surface area. Since, little agglomeration, debundle F-MWCNTs and high dispersion are resulted after treated with oil olive as compared to strong acids like H2SO4, HNO3 which leads to severe damage nanotubes structure.
 |
Figure 7: HRTEM images of the sample of Carbon nanotube a) before treatment b) after treatment with olive oil at magnification 200nm
|
Dispersion Test
The presence of functional groups on the CNT surface can be identified by dispersion test which is a common technique since it is fast, cheap and provides qualitative information through estimation the time required for MWCNTs to sediment in a polar solvent.
As shown in Fig (8 a,b), treated MWCNTs by oil olive is remained as stable suspension in case 2 after a tow hours and one week, respectively as compared to raw-MWCNTs in case 1, that shows MWCNTs solution before and after the treatment, respectively. These results refers to the presence of functional (COOH, -OH) groups on the surface of the carbon nanotubes which led to a reduction of van der Waals interactions among them which promotes their separation and dispersion in ethanol as compared with as-synthesized MWCNTs through its treatment with oil olive and change from the hydrophobic to hydrophilic material during the functionalization therefore it can be used in other applications.
 |
Figure 8: Photograph of MWCNTs in ethanol for: 1) raw -MWCNTs and 2) functionalized MWCNTs with oil olive: a) after two hours b) after one week
|
The Antimicrobial Activity of F-MWCNTs Against Bacteria
The results in Fig (9 a,b,c), indicate treated-MWNTs with oil olive have an obvious antimicrobial activity against gram positive and negative . Fig (10) shows the number of colonies survival against positive (S.aureus) and gram negative (E.coli, Salmonella typhi) using plate count method.27 As shown in Fig (9 a,b), limited number of survival cells present against E. coli and Salmonella while there is a large colonies of S.aureus at high concentrations of MWNTs after incubated for 24h as shon in Fig (9 b). The results in Fig (10), illustrate the effect of increasing concentrations of treated-MWNTs on the growth of bacteria with the role of longtime of shaker incubator (overnight) which leads to direct interaction of bacteria membrane with MWCNTs at high concentrations.
In Fig (11a,b,c), the SEM analysis images after treatment with cells represent the microbial adsorption or capturing efficiency of bacteria on functionalization F-MWCNTs by olive oil as shown in Fig (11 a,b) with E. coli, Salmonella typhi While S.aureus adsorb and wind with F-MWCNTs as shown in Fig (11 c) as a result of the existence functional groups on the surface of the carbon nanotube, suggest direct interaction between F-MWCNTs and cell membrane which cause cells death and thus enhance the antimicrobial activity against the E. coli, Salmonella typhi as shown and S.aureus .In addition, the results of SEM suggest the presence of the functional groups which increase surface area and thus, high molecular weight portion of bacteria is adsorbed relatively strongly.
 |
Figure 9: photograph of a) E.coli, b) Salmonella typhi, and c) S. aureus colonies grown on Nutrient Agar plate as a function of F-MWNTs concentration (1, 3 , 7mg/ml)
|
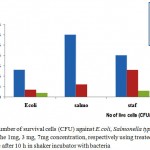 |
Figure 10: Number of survival cells (CFU) against E.coli, Salmonella typhi, and S. aureus in the 1mg, 3 mg, 7mg concentration, respectively using treated-MWCNTs with oil olive after 10 h in shaker incubator with bacteria
|
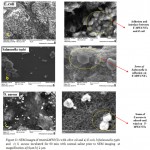 |
Figure 11: SEM images of treated-MWNTs with olive oil and a) E.coli, b)Salmonella typhi and c) S. aureus incubated for 60 min with normal saline prior to SEM imaging at magnification a)10μm b) 2 μm
|
Conclusion
In this study, the results reveals the possibility of introducing functional groups (COOH, OH) on the surface of multi walled carbon nanotubes (MWCNTs) by functionalization with (oil olive) as improved by FTIR and SEM analysis and dispersion test because oil olive contains certain amount of phenol and few rates of acids in, which makes it a good solvent for the functionalization process , providing a good quality and inexpensive method compared to traditional methods represented by using concentrated acid, which are often expensive as well as their dangerous to the user in the laboratories. Furthermore, such a treatment improved antibacterial agent of F-MWCNTs against different pathogens and enhancement its role as filter in capture and removal of microorganisms like (S.aureus) and (E.coli, Salmonella typhi) bacteria from an aqueous solution by using MWCNTs functionalized with olive oil.
Acknowledgements
Authors gratefully acknowledge nanotechnology and advanced material research centre, University of Technology, Baghdad, Iraq for conducting the SEM (VEGA Easy Probe), X-Ray diffractometer (XRD, 6000-Shimadzu),(FTIR,8400S, Shimadzu model) spectra, High Resolution Transmission Electron Microscope (HREM , JEOL, JEM-2100 ) and biotechnology lab in center for making different antibacterial activity tests and for their help and support.
References
- Ijima S. Helical microtubules of graphitic carbon. Nature. 1991;354:56-58.
CrossRef - Agui L, Sedeno P.Y, Pingarron J.M. Role of carbon nanotubes in electroanalytical chemistry. Anal. Chim. Acta. 2008;622:11–47.
CrossRef - Balasubramanian K, Burghard M. Biosensors based on carbon nanotubes. Anal bional chem. 2006;385:452-468.
CrossRef - Mita D.G, Attanasio A, Arduini F, Diano N,Grano V, Bencivenga U, Rossi S, Amine A, Moscone D. Enzymatic determination of BPA by means of tyrosinase immobilized on different carbon carriers. Biosensors & Bioelectronics. 2007;23:60-65.
CrossRef - Srivastava A, Srivastava O.N, Talapatra S, Vajtai R, Ajayan P.M. Carbon nanotube filters. Nat Mater. 2004;3(9):610–4.
CrossRef - Vecitis C.D, Schnoor M.H , Rahaman M.S , Schiffman J.D,Elimelech M. Electrochemical multiwalled carbon na otube filter for viral and bacterial removal and inactivation. Environ Sci Technol. 2011;45(8):3672–9.
CrossRef - Li C.C,Lin J.L,Huang S.J, Lee J.T,Chen C.H. A new and acid-exclusive method for dispersing carbon multi-walled nanotubes in aqueous suspensions. Colloids and Surfaces A: Physicochem Eng. Aspects. 2007;297:275-281.
CrossRef - Bal S, Samal S.S. Carbon nanotube reinforced polymer composites-A state of the art. Mater. Sci. 2007;30:379-386.
CrossRef - Vaisman L, Wagner H.D, MaromG. The role of surfactants in dispersion of carbon nanotubes. Advances in Colloid and Interface Science. 2006;128-130:37-46.
CrossRef - Adawiya J.H, Mohammed R. M, Duha S.A. Influence of functionalization MWCNTs using acid treatment on Gram negative and Gram positive bacteria. IJAP. 2014;10(3):29-33.
- Adawiya J.H , Mohammed M. R, Emad A, Al-Mulla J, Duha S.A. Synthesis of silver nanoparticle decorated carbon nanotubes and its antimicrobial activity against growth of bacteria. Rend. Fis. Acc. Lincei. 2014.
- Adawiya J.H, Mohammed M.R, Duha S. A. Preparation and characterization of multi walled carbon naotubes /Ag nanoparticles hybrid materials. International Journal of Scientific & Engineering Research. 2014;5:3.
- Dobrzański L.A, Pawlyta M, Krztoń A, Liszka B, Labisz K. Synthesis and characterization of carbon nanotubes decorated with platinum nanoparticles. Journal of Achievements in Materials and Manufacturing Engineering. 2010;39(2):184-189.
- Goyanes S, Rubiolo G.R, Salazar A, Jimeno A, Corcuera M. A and Mondragon I. Carboxylation treatment of multiwalled carbon nanotubes monitored by infrared and ultraviolet spectroscopies and scanning probe microscopy. Diamond and Related Materials. 2007;16(2):412–417.
CrossRef - Wu F.Y and Cheng H.M. Structure and thermal expansion of multi-walled carbon nanotubes before and after high temperature treatment. J. Phys. D. Appl. Phys. 2005;38:4302-4307.
CrossRef - Datsyuk V, Kalyva M, Papageli K, Parthenio J, Tasis D. Chemical oxidation of multiwalled carbon nanotubes. Carbon. 2008;46(8):33–840.
CrossRef - Kiritsakis A.K. Olive Oil From the Tree to the Table Second Edition (2ed) . Trumbull Connecticut USA. Food and Nutrition Press Inc. 1998.
- Boskou D. OLIVE OIL Chemistry and Technology, AOCS Press. 2006.
- Duha S. A, Adawiya J.H, Mohammed R.M. Comparesion of Functionalization of multi walled Carbon nanotubes treated by oil olive and nitric acid and their characterization. Energy Procedia. 2013;36:1111–1118.
CrossRef - Upadhyayula V.K, Deng S, Mitchell M.C, Smith G.B. Application of carbon nanotube technology for removal of contaminants in drinking water: a review. Sci Total Environ. 2009;408(1):1–13.
CrossRef - Avile´F. S, Cauich-Rodr J.V,´Guez L. Moo-Tah. Evaluation of mild acid oxidation treatments for MWCNT functionalization. Carbon 2009;47:2970–2975.
CrossRef - Ren-Shen L, Wen-Hsin C. Polymer-grafted multi-walled carbon nanotubes through surface initiated ringopening polymerization and click reaction. arrn-Horng Lin.Polymer. 2011;52:2180-2188.
CrossRef - Ganguly S.A, Mohana L.R and Ramaprabhu S. Biological Sensors Using DnaFunctionalized Multiwalled Carbon Nanotubes. Alternative Energy Technology Laboratory, Department of Physics, Indian Institute of Technology Madras, Chennai – 600 036, India. 2009
- Faraj A. A, Laoui T, Al-Harthi M and Ali M.A. Modification and Functionalization of Multiwalled Carbon Nanotube (MWCNTs) Via Fischer Esterification. The Arabian Journal for Science and Engineering. 2010;35:1.
- Lee G.W, Kim J, Yoon J.S, Bae B.C, Shin I.S, Kim W.O and Ree M. Structural characterization of carboxylated multi-walled carbon nanotubes Secretariat. Thin Solid Films. 2008;516:5781-5784.
CrossRef - Wu F.Y and Cheng H.M. Structure and thermal expansion of multi-walled carbon nanotubes before and after high temperature treatment. J. Phys. D: Appl. Phys. 2005;38:4302-4307.
CrossRef - Shan Y, Chen K, Yu X Gao L. Preparation and characterization of biocompatible magnetic carbon nanotubes. Applied Surface Science. 2010;257:362–366.

This work is licensed under a Creative Commons Attribution 4.0 International License.





