Manuscript accepted on : 15 July 2017
Published online on: --
Plagiarism Check: Yes
Mohammad Javad Hosseini and Reza Sadripour
Molecular Biology Research Center, BaqiyatallahUniversity of Medical Sciences, Tehran, Iran.
DOI : http://dx.doi.org/10.13005/bbra/2547
ABSTRACT: Today, many studies doneregarding the antibiotic resistance showthose patients in intensive care units (ICUs) are more likely to develop drug resistant pathogens.Pathogens are the major cause of nosocomial infections in intensive care units. Knowledge of antibiotic resistance of pathogens in the hospital is very important for treatment. The recent study aims at identifying drug resistant pathogens and antibiotic resistance pattern in the intensive care units of a tertiary care hospital in Tehran. In other words, the main purpose of the recent study is understanding the degree to which the resistant bacteria, which cause nosocomial infections is spreading in the ICUs of a tertiary care hospital and to determine the antibiotic- resistance pattern of these bacteria in the ICUs of that hospital. The recent research is a retrospective and cross-sectional study that has been conducted on the files of all patients who were hospitalized in ICUs, internal surgery, and neurosurgery sections of Baqiyatallah Hospital in Tehran in a five-year period (March 2009 to February 2015) and who were diagnosed with nosocomial infection caused by resistant bacteria. According to the results of this research, most of the separated bacteria were: Acinetobacterbaumannii 39.45%, Pseudomonas aeruginosa 20.69% and the most common location for infection was respiratory tract (67.11%). Also the most common pathogen was Acinetobacterbaumannii which is resistant to most antibiotics.
KEYWORDS: Antibiotic resistance; ICU; nosocomial infection
Download this article as:| Copy the following to cite this article: Hosseini M. J, Sadripour R. Antibiotic Resistance Pattern of Bacteria Isolated from Nosocomial Infection in Internal Surgery and Neurosurgery Intensive Care Unit (NICU) at a Tertiary Care Hospital in Tehran, Iran. Biosci Biotech Res Asia 2017;14(3). |
| Copy the following to cite this URL: Hosseini M. J, Sadripour R. Antibiotic Resistance Pattern of Bacteria Isolated from Nosocomial Infection in Internal Surgery and Neurosurgery Intensive Care Unit (NICU) at a Tertiary Care Hospital in Tehran, Iran. Biosci Biotech Res Asia 2017;14(3). Available from: https://www.biotech-asia.org/?p=26864 |
Introduction
Alexander Fleming’s discovery of the first antibiotic substance generated a huge revolution in the health and treatment systems worldwide. The antibiotic drugs have been considered the most important factors fighting pathogenic bacteria ever since. Nevertheless, some bacteria have developed a type of resistance against antibiotics nowadays due to the incorrect use of antibiotics. As a result, they are able to live and even reproduce when exposed to antibiotics.Bacteria which are very tolerant to antibiotics are known as (MROS). Methicillin- resistant Staphylococcusaureus (MRSA) and vancomycin- resistant EnterococcusSpp. (VRE) are amongst the most important bacteria which are resistant to treatment. These bacteria are considered one of the most crucial factors causing nosocomial infections. Infections occur 48 to 72 hours after the patient has been admitted to the hospital provided the patient does not have obvious infection symptoms at the time of admission and the disease is not in its incubation period.1 The response to therapy of these infections has changed and antibiotic resistance incidence has increased at an alarming pace over the past 10 years. About 50%–60% of hospital-acquired infections are caused by resistant strains.2 A number of factors related to infection with resistant microorganisms have been reported, including previous use of antibiotics,3-6 corticosteroid therapy, mechanical ventilation, length of hospital stay5-8 and use of invasive devices such as catheters.9,10 Many researches have indicated that ICU sections are the center of these infections and the number of the patients suffering from nosocomial infection is in fact much greater in ICUs than in other wards.11,12 The level of the resistant bacteria has also increased in the ICUs due to repeated use of antibiotics. Nosocomial infections caused by resistant bacteria affect the increase of mortality and also the increase of hospital expenses.13 Therefore preventing these bacteria from appearing and spreading is of vital importance for controlling nosocomial infections. Proper supervision, which includes selecting the proper antibacterial, the dose, and also the treatment duration, can prevent or delay the appearance of the antibiotic- resistant bacteria.14
Method
The present research is a retrospective, cross- sectional study which has been conducted on the files of all the patients who were hospitalized in internal, surgery, and neurosurgery ICUs of a tertiary care hospital in Tehran in a 5- year period (from Farvardin of 1388 to Esfand of 1392) (March 2009 to February 2015) and who were diagnosed with nosocomial infection caused by resistant bacteria. Bacterial cultures of the resistant- to- treatment bacteria which were separated from different samples of the patients such as their blood, urine, wound, etc., were identified through visiting the molecular diagnosis and clinical laboratory of Baqiyatallah (peace be upon him) Hospital in the present research. The files of the patients were examined based on these data. Clinical specimens included blood, urine, pus, and discharge from endotracheal tubes and post surgical wound swabs were collected and cultured on Eosin Methylen Blue (EMB), Blood agar, chocolate agar, thioglycollate and Trypticase Soy broth (TSB) media and incubated at 37°C for 24 – 48 h. Thioglycollate cultures and TSB bottles were reincubated for at least 7 days and subcultured on EMB and blood agar or chocolate agar plates, as necessary. The pathogenic isolates were identified by Gram staining, biochemical reactions and diagnostic tests included catalase, tubecoagulase and Manitol Salt agar in order to identify Staphylococcus aureus from Coagulase Negative Staphylococci (CoNS). Reaction of the isolates in TSI, SIM, Urea and Simmon’s citrate medium and oxidase tests were used for identification of gram negative bacteria. Antibiogram pattern of microorganisms was determined by Kirby Bauer method on Mueller Hinton agar medium (Baily and Scott, 1990). Results were recorded according to the standards provided by National Committee for Clinical Laboratory Standards (NCCLS, 2003).15-18
Ampicillin, penicillin, vancomycin, tetracycline, ciprofloxacin, norfloxacin, nitrofurantoin, cefotaxime, erythromycin, clindamycin, cefepime, chloramphenicol, co-trimoxazole, gentamicin, amikacin, and meropenem were examined on positive- gram bacteria and piperacillin-tazobactam, ceftazidime, cefepime, ceftriaxone, imipenem, amikacin, tetracycline, ciprofloxacin, ampicillin, colistin, co-trimoxazole, norfloxacin, nitrofurantoin, chloramphenicol, cefoxitin, meropenem, nalidixic acid and tobramycin were examined on negative- gram bacteria. All the demographical information including age, gender, hospitalization season, hospitalization duration, background diseases, mortality cases, the cause of the diseases, etc. were extracted from the patients’ files and recorded. The collected data were eventually analyzed with SPSS software and the obtained results were registered.
The medical ethics issues and the patients’ rights were also observed based on the international treaties and the suggestions of the Medical Ethics District Committee.
Results
According to the results, 37.3 percent of the 397 cultured samples were male patients who applied to this center and 62.7 percent were female. Regarding age distribution, 58.1 percent of the patients were over 60 years old and the mean age of all the patients was 57±28 years old. Considering the fact that the present research is a retrospective study, 34.2 percent of death cases were observed. Most infection cases occurred in summer, 29.4, and the minimum number of cases was reported in the spring. The mean hospitalization duration was also 34 ±28 days and 25.2 percent of the patients had higher than normal temperatures. It is also worth mentioning that the results of the microbial culture of all the under- study individuals prove that these patients were affected by nosocomial infections caused by resistant bacteria. For Acinetobacterbaumannii& Pseudomonas aeruginosa, lower resistance rates were observed for amikacin (91.2&75.7). Also, Acinetobacterbaumannii shows 100% resistance to most antibiotics. Pathogens frequency of multi drug resistance (MDR) was 18.3%.
Infection Distribution
These infections are mainly distributed in 4 locations: the respiratory system 67.11%, the urinary system 6%, and wound infection after surgery 13.9%, blood infection 5.4%, and the other 7.5 percent.
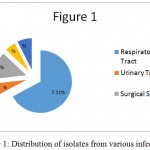 |
Figure 1: Distribution of isolates from various infections
|
The respiratory system has the largest infection percentage while 47.33% of cases of death were directly affected by respiratory disorders. This was 8% in brain patients, 6.66% for kidney disorders, 4.66% for cancer, 4.66% for digestive diseases, and 22.66% for other diseases. The results also indicated that 38.4 percent of the patients were hospitalized due to respiratory disorders and 32.6 percent of them had a respiratory disorder background.
The Separated Bacteria
76.99 percent of the 517 pathogenic bacteria which caused nosocomial infection and were examined in this study were negative-gram bacteria and 23.01 percent were positive-gram bacteria. Most of the separated bacteria were: Acinetobacter baumannii 39.45%, Pseudomonasaeruginosa 20.69%, Staphylococcusaureus 12.95%, klebsiellapneumoniae 7.35%, E.coli 4.83%, Enterococcus spp., 4.25, Enterobacter spp., 3.86, Streptococcus spp. 3.48%, Staphylococcusepidermidis 2.12%, and 0.96% other bacteria. Table 1 shows the number of the separated bacteria based on the infected location
Table 1: Microorganisms reported from internal, surgery and neurosurgery ICUaccording to the site of infection
| Microorganism | Respiratory Tract N (%) | Surgical Site N (%) | Urinary Tract N (% | Blood stream N (%) | Others Sites N (%) | Total N (%) |
| Acinetobacter baumonnii | 153 (44.1) | 20 (27.8) | 7 (22.6) | 7 (25) | 17 (43.6) | 204 (39.5) |
| Pseudomonas aeruginosa | 79 (22.8) | 15 (20.8) | 5 (16.1) | 2 (7.1) | 6 (15.4) | 107 (20.7) |
| Staphylococcus aureus | 47 (13.5) | 12 (16.7) | 1 (3.2) | 3 (10.1) | 4 (10.3) | 67 (13) |
| Klebsiella Pneumoniae | 20 (5.8) | 6 (8.3) | 7 (22.6) | 4 (14.3) | 1 (2.6) | 38 (7.4) |
| Entrobacter Spp. | 10 (2.9) | 5 (6.9) | 2 (6.5) | 1 (3.6) | 2 (5.1) | 20 (3.9) |
| Entrococcus Spp. | 13 (3.7) | 3 (4.2) | 3 (9.7) | 1 (3.6) | 2 (5.1) | 22 (4.3) |
| Escherichia Coli | 13 (3.7) | 5 (6.9) | 2 (6.5) | 1 (3.6) | 4 (10.3) | 25 (4.8) |
| StaphylococcusEpidermis | 1 (0.3) | 0 (0) | 1 (3.2) | 9 (32.1) | 0 (0) | 11 (2.1) |
| Streptococcus Spp. | 9 (2.6) | 5 (6.9) | 3 (9.7) | 0 (0) | 1 (2.6) | 18 (3.5) |
| Proteus | 2 (0.6) | 0 (0) | 0 (0) | 0 (0) | 2 (5.1) | 4 (0.8) |
| Staphylococcus Saprophyticus | 0 (0) | 1 (1.4) | 0 (0) | 0 (0) | 0 (0) | 1 (0.2) |
| Total | 347 (100) | 72 (100) | 31 (100) | 28 (100) | 39 (100) | 517 (100) |
Actinobacterbaumannii(44.1%) and Pseudomonasaeruginosa (22.8%) had the largest percentage in respiratory infections. 22.6 percent of the bacteria separated from urinary infections belonged to Klebsiellapneumoniae (22.6) and this percentage also belonged to Acinetobacter baumannii. Also, in 32.3 percent of the cases the patient was simultaneously fighting Candidaalbicans. In blood samples, the largest percentage was related to Staphylococcusepidermidis (32.1%) and Acinetobacter baumannii (25%). Acinetobacter baumannii (20.8%) and Pseudomonasaeruginosa (20.8) also comprised the largest percentage of the intended positive cultures in surgical wounds. The positive- gram bacteria were mostly seen in the respiratory system (58.8%) and in the post surgical wounds (17.6%) in general. However, the negative-gram bacteria were mostly present in the respiratory system (69.6%).
Antibiotic- Resistance Pattern
Acinetobacterbaumannii was the most resistant negative- gram bacteria against the tested antibiotics, in such a manner that it was only to some extent sensitive to imipenem and amikacin (4.9 and 8.8%). It was resistant to almost all the tested antibiotics. Pseudomonasaeruginosa was 100 percent resistant to nalidixic acid, ceftazidime, and chloramphenicol and was least resistant to amikacin (75.7). Klebsiellapneumoniae was most resistant to cephalosporins, third generation (ceftriaxone, 100%, cefoxitin, 100%, and ceftazidime, 97.4%). Nitrofurantoin, tobramycin, ampicillin, and piperacillin also displayed complete resistance to this bacterium. Klebsiella pneumoniae was least resistant to imipenem (47.4%) and amikacin (65.8%). Except for amikacin against which Enterobacter Spp. showed an 80- percent resistance, it was 90 percent resistant to all other antibiotics and was very much resistant to most antibiotics. E.coli was 100 percent resistant to nalidixic acid, cefepime, tobramycin, and ceftazidime and was least resistant to amikacin (40%) and imipenem (48%). All the positive- gram bacteria were 100% resistant to penicillin, norfloxacin, gentamicin, erythromycin, and cefepime. In other cases, Staphylococcusaureus was 100% resistant to nitrofurantoin and least resistant to vancomycin (6%). Vancomycin (68.2%) and nitrofurantoin (86.4%) were least resistant to Enterococcus Spp. and the rest of the antibiotics showed a resistance over 90 percent. Tables(2) and (3) show the antibiotic resistance model of the separated pathogens. It is worth mentioning that the multi- drug resistance (MDR) was 18.3 percent in this study.
Table 2: Antibiotic resistance of Gram-positive microorganisms isolated from infections in ICUs)N = 119)
| organisms | S. Aureus | Entrococcus Spp | S. Epidermis | streptococcus Spp | S.Saprophyticus | Total |
| V | 6% | 68.20% | 9.10% | 50% | 0% | 24.40% |
| FM | 100% | 86.40% | 90.90% | 88.90% | 100% | 95% |
| SXT | 67.20% | 100% | 100% | 100% | 100% | 81.50% |
| AMK | 92.50% | 90.90% | 90.90% | 83.30% | 100% | 90.80% |
| TE | 89.60% | 100% | 100% | 88.90% | 0% | 91.60% |
| MRP | 98.50% | 100% | 100% | 94.40% | 100% | 98.30% |
| AM | 98.50% | 90.90% | 100% | 100% | 100% | 97.50% |
| C | 94.00% | 90.90% | 63.60% | 100% | 100% | 91.60% |
| CTX | 98.50% | 100% | 100% | 100% | 100% | 99.20% |
| CC | 98.50% | 100% | 90.90% | 94.40% | 100% | 97.50% |
| GM | 100% | 100% | 100% | 100% | 100% | 100% |
| NOR | 100% | 100% | 100% | 100% | 100% | 100% |
| P | 100% | 95.50% | 100% | 100% | 100% | 99.20% |
| CIP | 98.50% | 100% | 90.90% | 94.40% | 100% | 97.50% |
| CPM | 100% | 100% | 100% | 94.40% | 100% | 99.20% |
| E | 100% | 100% | 90.90% | 100% | 100% | 99.20% |
| CN | 98.50% | 100% | 90.90% | 100% | 100% | 98.30% |
S. Aureus- Staphylococcus aureus, S. Epidermis- Staphylococcus Epidermis,S.Saprophyticus– Staphylococcus Saprophyticus, V – vancomycin, FM – nitrofurantoin, SXT – co-trimoxazole, AMK – amikacin, TE – tetracycline, MRP – meropenem, AM – ampicillin, C – chloramphenicol, CTX – cefotaxime, CC – clindamycin, GM – gentamicin, NOR – norfloxacin, P – penicillin, CIP – ciprofloxacin, CPM – cefepime, E – erythromycin, CN – cephalexin
Table 3: Antibiotic resistance of Gram-negative microorganisms isolated from infections in ICUs(N=398)
| Organisms | A. baumonnii | P. aeruginosa | K. Pneumonia | Entrobacter Spp | E. Coli | Proteus | Total |
| FM | 100.00% | 99.10% | 100.00% | 100.00% | 92.00% | 100.00% | 99.20% |
| SXT | 100.00% | 98.10% | 73.70% | 100.00% | 88.00% | 100.00% | 96.20% |
| AMK | 91.20% | 75.70% | 65.80% | 80.00% | 40.00% | 50.00% | 80.40% |
| TE | 100.00% | 98.10% | 89.50% | 100.00% | 92.00% | 100.00% | 98.00% |
| MRP | 100.00% | 98.10% | 81.60% | 90.00% | 68.00% | 75.00% | 95.00% |
| IPM | 95.10% | 86.90% | 50.00% | 90.00% | 48.00% | 75.00% | 85.20% |
| TZP | 99.00% | 88.80% | 78.90% | 95.00% | 60.00% | 75.00% | 91.50% |
| AM | 100.00% | 99.10% | 100.00% | 100.00% | 92.00% | 100.00% | 99.20% |
| C | 99.50% | 100.00% | 84.20% | 95.00% | 96.00% | 100.00% | 97.70% |
| CTX | 100.00% | 98.10% | 100.00% | 100.00% | 96.00% | 100.00% | 99.20% |
| CAZ | 100.00% | 100.00% | 97.40% | 95.00% | 100.00% | 100.00% | 99.50% |
| NA | 100.00% | 100.00% | 94.70% | 100.00% | 100.00% | 100.00% | 99.50% |
| NOR | 99.50% | 99.10% | 92.10% | 100.00% | 92.00% | 100.00% | 98.20% |
| TM | 100.00% | 99.10% | 100.00% | 100.00% | 100.00% | 100.00% | 99.70% |
| CRO | 100.00% | 97.20% | 100.00% | 100.00% | 96.00% | 100.00% | 99.00% |
| CIP | 99.50% | 86.00% | 73.70% | 90.00% | 96.00% | 75.00% | 92.50% |
| CPM | 100.00% | 95.30% | 89.50% | 95.00% | 100.00% | 75.00% | 97.20% |
A. baumonnii– Acinetobacter baumonnii, P.aeroginosa– Pseudomonas aeroginosa, K. Pneumoniae- Klebsiella Pneumoniae, E. Coli– Escherichia Coli, FM – nitrofurantoin, SXT – co-trimoxazole, AMK – amikacin, TE – tetracycline, MRP – meropenem, IPM – imipenem, TZP – piperacillin-tazobactam, AM – ampicillin, C – chloramphenicol, CTX – cefotaxime, CAZ – ceftazidime, NA – nalidixic acid, NOR – norfloxacin, TM – tobramycin, CRO – ceftriaxone, CIP – ciprofloxacin, CPM – cefepime
Discussion
The creation of intensive care units and the progresses made in treating patients have helped heal the patients who were destined to die. On the other hand, prolonged hospitalization duration of these patients and using life-support machines, invasive monitoring, and different types of venous catheters cause nosocomial infections in these units.19 The factors which increase the danger of nosocomial infections in the intensive care units include: severity of the disease, physiological and psychological stress, age and other factors which lead to death, improper use of antibiotics, increase in the number of antibiotic-resistant organisms, stress-ulcer drug treatments, protein malnutrition, and the presence of the personnel who spread the infection among the patients.19-21 The increase in the number of antibiotic-resistant organisms is the most important factor among all the above- mentioned factors since most of the deaths caused by nosocomial infections are due to the antibiotic-resistance phenomenon. Therefore, the present study has been conducted with the aim of understanding the degree to which the resistant bacteria, which cause nosocomial infections is spreading in the ICUs of Baghiyatollah (peace be upon him) Hospital and to determine the antibiotic- resistance model of these bacteria in the ICU of that hospital.
From amongst the 397 samples that were cultured in our study, 67.11 percent were related to the respiratory system which was consistent with the results of Jamshidi et al.’s (2007) study (respiratory tract 54.2%).22 Also, the respiratory infection has been presented with a lesser percentage (31.5% and 38.8%) as the main locations in the researches conducted by Saeed et al. (2005) in Saudi Arabia23 and Bacli and Bayram (2006) in Turkey.24 Our results were, however, different from Sikka et al.’s (2012) research in India 25 in which the wound infection after surgery was presented as the main location which may be due to the difference between the type of hospital wards and the under- study population in their study and those of the present research.
Acinetobacterbaumannii was the most common (51.3%) microorganism obtained in the present research among the negative-gram bacteria which was consistent with the research conducted by Krishnamurthy et al. (2006- 2010) on patients suffering from VAP in India.26 This matter was, however, different from the study of Sikka et al.25 who reportedE.coli 58% and the study of Saravanan and Raveendaran (2012) which reported E.coli 32.8%.27 Staphylococcusaureus was the most usual pathogen (56.3%) among the positive- gram bacteria which was similar to the results of the research of Jamshidi et al.22 (39.2%) and that of Krishnamurthy et al.26 (42.15%). The results, however, were different from the results of Wattal et al. (2008- 2011)28 which introduced Staphylococcus coagulase negative (20.3% of all the microorganisms) as the most common pathogen. It seems that the difference between the above- mentioned results is due to the difference in the type of the under- study disease and the fact that the latter research was conducted in many centers in comparison with the present study.
It became clear in the present study that among the positive-gram bacteria, Staphylococcusaureus (94%) and Staphylococcus coagulase negative (100 and 90.9%) are especially sensitive to vancomycin, which was consistent with the study of Randrianirina et al. (2008) (29) in Madagascar and that of Saeed et al. (2009) in Saudi Arabia23 where Staphylococcusaureus and Staphylococcus coagulase negative displayed 100 percent sensitivity. This antibiotic is in fact still effective in the under- study patients. Streptococcus and Enterococcus Spp. are, respectively, 50 and 31.8 percent sensitive to this antibiotic while these amounts were 100 and 96.7 percent in Randrianirina et al.’s (2008) 29 research. In their research, Saeed et al.23 stated that Enterococcus Spp. is 59.1 percent resistant to vancomycin. The degree of antibiotic resistance was 63.6 percent in chloramphenicol which is related to Enterococcus Spp, while other positive-gram bacteria also displayed a more than 90 percent resistance to chloramphenicol. Staphylococcusaureus was only sensitive to vancomycin (100%) and trimetoprima/ sulfametoxazol (61%) in Balci and Bayram’s 24 study but it was highly resistant to other bacteria. StreptococcusSpp. and Enterococcus Spp were highly resistant to a wide range of antibiotics in such a way that except for vancomycin and chloramphenicol, they were more than 80 percent resistant to other cases.
Acinetobacter baumannii was only slightly sensitive to imipenem (4.9%) and amikacin (8.8%) among the negative-gram bacteria. The resistance percentage of meropenem was 22.2 in Sikka et al.’s 25 study and Acinetobacterbaumannii was more than 77 percent resistant to other antibiotics. Acinetobacterbaumannii was least resistant against colistin (0%) and tigecycline (43%) in Saeed et al.’s23 study. Also, Pseudomonasaeroginosa was 24.3 percent sensitive to amikacin and other antibiotics were more than 85% resistant to this bacterium. The minimum resistance percentage was also related to amikacin in Kumari et al.’s (2002)30 study and more than 80 percent resistant to other antibiotics were observed.
But in the research of Wattal et al., 28 the minimum resistance was related to colistin (0%) and piperacillin-tazobactam (45%), and in the study of Saeed et al. 23 it was related to colistin (8%) and amikacin (15%) which was different from the results obtained from our study. Also, the minimum resistance was related to imipenem (0%) and piperacillin-tazobactam (50%) in Sikka et al.’s 25 study. The greatest sensitivity of Klebsiella pneumoniae was also related to imipenem (88%) and amikacin (84%) in the study conducted by Balci and Bayram.24 On the other hand, the maximum sensitivity was related to gentamycin (52.2%) and ciprofloxacin (52.2%) in the research carried out by Khalili et al. (2008) 31 on negative- gram bacteria with the nosocomial infection origin, these bacteria were relatively highly resistant to other cases in this study. Enterobacters spp. were only 20 percent sensitive to amikacin and they were more than 90 percent resistant to other cases. E.coli was also mostly sensitive to amikacin (60%), imipenem (52%), and tazocin (40%) which was similar to the results of Balci and Bayram’s (24) study, amikacin (81.6%) and imipenem (86.9%). It was, however, different from Sikka et al.’s 25 study (meropenem 95.8%).
Conclusion
According to the examinations carried out in the present study, the male patients and also the group aged over 60 years of age are the most vulnerable groups among the patients hospitalized in the ICUs. It also became clear that the degree to which patients are affected by the nosocomial infections caused by resistant bacteria is greater in the summer in ICUs rather than in other seasons, which must be further studied. The present research indicates that Acinetobacterbaumannii is the most common pathogen which is resistant to treatment and is, in fact, very important when it comes to the spread of nosocomial infections. This important matter must therefore be noted when treating infections that are caused by it so that this process will decrease to the minimum. Moreover, according to the results, the respiratory system more than other organs is prone to nosocomial infections caused by resistant bacteria. Therefore, attention to methods which control the respiratory infections in the ICU may help prevent the spread of nosocomial infections. The studies indicated that vancomycin is the most effective antibiotic in positive- gram bacteria and amikacin and imipenem are the most effective antibiotics when it comes to negative-gram bacteria. It is therefore advisable to use these antibiotics for controlling and fighting these bacteria. On the other hand, taking into consideration the possibility that the resistant genes may transfer to other bacteria, also high resistance levels which were observed in the present study, the health authorities must pay more attention to planning and carefully supervising control over the nosocomial infections and utilizing accurate and effective treatment protocols for the purposes of eliminating the drug- resistant microorganisms. It is also essential to prescribe antibiotics, to which bacteria have multiple and high resistance, rationally and scientifically (32).
Acknowledgement
The authors are thankful for the advice, guidance and support of the Research Department of Clinical Research of Baqiyatallah hospital.
References
- Meric M., Willke A., Caglayan C., Toker K. Intensive care unit acquired infections: incidence, risk factors and associated mortality in a Turkish university hospital. Jpn J Infect Dis. 2005;58:297-302.
- Kollef M. H. et al. The impact of nosocomial infections on patient outcomes following cardiac surgery. Chest. 1997;112:666–675.
CrossRef - Ganguly P., Yunus M., Khan A., Malik A. A study of nosocomial infection in relation to different host factors in an Indian teaching hospital. J Res Soc Health. 1995;115(4):244-6.
CrossRef - Maa S. H., Lee H. L., Huang Y. C., Wu J. H., Tsou T. S., MacDonald K., Abraham I. Incidence density and relative risk of nosocomial infection in Taiwan’s only Children’s Hospital, 1999-2003. Infect ContHosp Ep. 2008;29 (8):767-70.
CrossRef - Chen M. L., Sun C. C., Chen W. H., Pan H. J., Yang L. S et al. Antimicrobial drug resistance in pathogens causing nosocomial infections at a university hospital in Taiwan, 1981-1999. Emerg Infect Dis. 2002;8:63-68.
CrossRef - Coia J. E., Duckworth G. J ., Edwards D. I., Farrington M., Humphreys F. H., et al. Guidelines for the control and prevention of Meticillin-Resistant Staphylococcus aureus (MRSA) in healthcare facilities. J Hosp Infect. 2006;63 (1):S1-44.
CrossRef - Collee J. G., Miles R. S., Watt B. Tests for identification of bacteria. In: Mackie and McCartney Practical Medical Microbiology, 14th edition, Collee J. G., Fraser A. G., Marmion B. P., Simmons A (Eds.), Churchill Livingstone: New York. 1996;166-7;131-49.
- Bauer A. W., Kirby W. M., Sherris J.C ., Turck M. Antibiotic susceptibility testing by a standardized single disk method. J ClinPathol. 1966;45(4):493-6.
CrossRef - Cavalcante S. S., Mota E., Silva L. R., Teixeira L. F., Cavalcante L. B. Risk factors for developing nosocomial infections among pediatric patients. Pediatr Infect Dis J. 2006;25(5):438-45.
CrossRef - Klevens R. M., Edwards J. R., Richards C. L., HoranT. C.,Gaynes R. P., Pollock D. A. Estimating Health care-associated infections and deaths in U.S. Hospitals, 2002. Public Health Rep. 2007;122(2):160-6.
CrossRef - Fridkin S. .K., Welbel S. F., Weinstein R. A. Magnitude and prevention of nosocomial infections in the intensive care unit. Infect Dis Clin North Am. 1997;11: 479-96.
CrossRef - M et al., Society for Healthcare Epidemiology of America and Infectious Diseases Society of America joint committee on the prevention of antimicrobial resistance: Guidelines for the prevention of antimicrobial resistance in hospitals. Infect Cont Hosp Ep. 1997;18:275–291.
- Marik P. The ICU therapeutics hand book. St louis, Mosby Co. 1996;240-253.
- Jamshidi M., Javadpour S., EghbalEftekhrai T., Moradi N., Jomehpour F. Antimicrobial resistance pattern among intensive care unit patients.Afr J Microbiol Res. 2009;3(10):590-594.
- Saeed N., Kambal A., El-Khizzi N. Antimicrobial-resistant bacteria in a general intensive care unit in Saudi Arabia. Saudi Med J. 2010;31(12):1341-49.
- Bayram A., Balci I. Patterns of antimicrobial resistance in a surgical intensive care unit of a university hospital in Turkey. BMC Infect Dis. 2006;155.
CrossRef - Sikka R., Mann J. K et al. Prevalence and Antibiotic Sensitivity Pattern of Bacteria Isolated from Nosocomial Infections in a Surgical Ward. Indian J ClinPract. 2012;22(10):519-25.
- Krishnamurthy V., Kumar G. S., Prashanth H. V., Prakash R., Kumar M. Ventilator associated pneumonia: bacterial isolates and its antibiotic resistance pattern.Int J Biol Med Res. 2013;4(2):3135-3138.
- Saravanan R., Raveendaran V. Antimicrobial resistance pattern in a tertiary care hospital:An observational study. Journal of Basic Clin Pharmacy. 2013;4(3): 56-63.
CrossRef - Wattal C., Raveendaran R., Goel N., Obreoi J. K., Rao K. B. Ecology of blood stream infection and antibiotic resistance in intensive care unit at a tertiary care hospital in North India. Braz J Infect Dis. 2014;18(3):245–251.
CrossRef - Randrianirina et al. Antimicrobial resistance in pathogens causing nosocomial infections in surgery and intensive care wards in Antananarivo, Madagascar. J Infect DevCtrie. 2010;4(2):074-082.
- Fagon J. Y et al. Nosocomial pneumonia in patients receiving continuous mechanical ventilation. Prospective analysis of 52 episodes with use of a protected specimen brush and quantitative culture techniques. Am Rev Resp.Dis. 1989;139:877–884.
CrossRef - Kumari V. S. N and Chandramuki A. Antimicrobial Resistance Pattern among Aerobic Gram-negative Bacilli of Lower Respiratory Tract Specimens of Intensive Care Unit Patients in a neurocentre. Indian J Chest Dis & Allied Sciences. 2007;49: 20-22
- Khalili H., Soltani R., Afshami S., Dashti-Khavidaki S., Alijani B. Antimicrobial resistance pattern of Gram-negative bacteria of nosocomial origin at a teaching hospital in the Islamic Republic of Iran. East Mediterranean Health. 2012;18(2).172-77.
CrossRef - Oh H. S., Chung H. W., Kim J. S., Cho S. I. National survey of the status of infection surveillance and control programs in acute care hospitals with more than 300 beds in the Republic of Korea. Am J Infect Control. 2006;34(4):223-33.
CrossRef - Husni R. N et al. Risk factors for an outbreak of multi-drug-resistant Acinetobacter nosocomial pneumonia among intubated patients. Chest. 1999;115:1378–138.
CrossRef - Rello J et al. Risk factors for infection by Pseudomonas aeruginosa in patients with ventilator-associated pneumonia. Intense Care Med. 1994;20:193–198.
CrossRef - Trouillet J. L et al. Ventilator-associated pneumonia caused by potentially drug-resistant bacteria. Care Med. 1998;157:531–539.
CrossRef - Ibelings M. M., Bruining H. A. Methicillin-resistant Staphylococcus aureus: acquisition and risk of death in patients in the intensive care unit. Eur J Surg. 1998;164:411–418.
CrossRef - Bonten M. J et al. External sources of vancomycin-resistant enterococci for intensive care units. 1998;26:2001–2004.
- Richards M. J et al. Nosocomial infections in medical intensive care units in the United States. National Nosocomial Infections Surveillance System Critical Care Medicine. 1999;27:887–892.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.





