Manuscript accepted on : 06 November 2016
Published online on: --
Plagiarism Check: Yes
The Potential Yield of Some Superior Breeding Lines of Upland Rice of Southeast Sulawesi Indonesia
Gusti R. Sadimantara, Muhidin*, Sahta Ginting and Ni Wayan Sri Suliartini
Department of Agrotechnology, Faculty of Agriculture, Universitas Halu Oleo Kendari 93232 Southeast Sulawesi Indonesia.
Corresponding Author E-mail: muhidinunhalu@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/2341
ABSTRACT: Demand for rice as a source of food in Indonesia continues to increase in line with the growth of population, while the capacity to produce paddy rice increasingly limited. One of the efforts is to develop upland rice that tolerant to drought and high production potential the hybridization. The study was conducted at the experimental field of the Faculty of Agriculture Halu Oleo University. Production characters were observed and assessments are tillers number, a number of productive tillers, panicle length, number of filled grain/panicle and weight 1,000 seed grains. The results showed that there is eight promising progeny of upland rice that are tolerant to drought and high potential yield
KEYWORDS: Upland rice; Progeny; Hybridization; Drought; Mineral stress
Download this article as:| Copy the following to cite this article: Sadimantara G. R, Muhidin M, Ginting S, Suliartini N. W. S. The Potential Yield of Some Superior Breeding Lines of Upland Rice of Southeast Sulawesi Indonesia. Biosci Biotech Res Asia 2016;13(4). |
| Copy the following to cite this URL: Sadimantara G. R, Muhidin M, Ginting S, Suliartini N. W. S. The Potential Yield of Some Superior Breeding Lines of Upland Rice of Southeast Sulawesi Indonesia. Biosci Biotech Res Asia 2016;13(4). Available from: https://www.biotech-asia.org/?p=17006 |
Introduction
Rice has become dominant and important staple food in Indonesia 1. The rice demand continues to increase in line with population growth, while the production rice capacity to increase is very limited. An effort to increase rice production continuously is facing severe challenges, due to the high rate of population growth at one side and at the other side; there is a reduction in the productive wetland area. So that role of upland rice to support rice production and strengthen food security is needed more increasing.
Indonesia has more than 50 million hectares of dryland but that are suitable for development food crops only reached 35 million hectares 2, including marginal dry land for developing upland rice. As a result, the potential upland rice production that can be achieved is still low. The low productivity of upland rice is due to the severity constraints on upland rice cultivation, as compared with the constraints on rice production on the wetland. These constraints partly because upland rice grown on the acid soils, which have low nutrients, especially N, P, K, Ca, Mg and Mo 3-5. Soil acidity has become a limiting factor for growth and increase rice production on dry land. In the acid soil, the toxicity of aluminum is a major factor constraining crop performance 3,6-9, and low availability of phosphor will be a problem in upland rice production 10-12. Water holding capacity on the dry land also very low and soil easily eroded. Aluminum (Al) affect primarily on the root system, causing inhibition the elongation root. As a result, the fertilizer effectiveness and water absorption will reduce 13. The high Al also can inhibit the transport of nutrients from the roots to the top of the plant 13-14. Increased concentrations of Al also reduce the absorption of micronutrients 15 in rice plants 16. Al toxicity occurred since the beginning of Al induces instability of cellular membranes 17-19 and toxic the metabolic activity 20.
One of the efforts to increase the upland rice production in acid soil is to create the tolerant cultivars through crossing 21. Upland rice has a great diversity against Al and the tolerant cultivar only showed a small decrease in growth 22. Rice plants have the genetic variability tolerant to Al toxicity and have the ability to use the different nutrient potassium 23.
Each type of plant and each different growth phases has a different water needs. Water has an important role in the transportation and translocation of nutrients 24, opening and closing of stomata 25. According to a response of plants to drought stress is differentiated to tolerant and sensitive 26. Tolerant plants are able to accumulate large amounts of dissolved compounds, whereas sensitive crops not able to accumulate the dissolved compounds. Genetic improvement through plant breeding is an effort to overcome some constraint on the upland rice production. The development of upland rice lines that tolerant to abiotic and biotic stress is a cheaper and easier approach to using at the farm level.
Material and Methods
Preparation of sample
Research was conducted in Agriculture Farm Department of Agrotechnology Faculty of Agriculture Universitas Halu Oleo Kendari. Soil analysis conducted in the soil laboratory Faculty of Agriculture Universitas Halu Oleo.
Field arrangement
The research was arranged in randomized completely block design consist of eight progeny i.e., V1 (GS12-2), V2 (GS44-2), V3 (GS16-2), V4 (GS11-2), V5 (GS44-1), V6 (GS16-1), V7 (GS12-1) and V8 (GS11-1). Production characters were observed and assessments (1) number of tillers at harvest, (2) number of productive tillers, (3) panicle length (cm), (4) number of filled grain /panicle and weight of 1000 grains of seeds.
Results and Discussions
Number of tillers
Based on number tillers character appears that the progeny V2 (GS44-2) has the highest number of tillers. Followed by the progeny V5 (GS44-1) and V4 (GS11-2). While the progeny V3 (GS16-2) has the lowest number of tillers (Table 1).
Table 1: Different productive characteristic of uplane rice progeny
| Progeny | Number of tiller (tiller clump -1) | Number of grain (seed panicle -1) | Production (g clump-1) | Weight of 1,000
Grains (gram) |
| V1 (GS12-2) | 15.1 | 251.93 | 57.30 | 24.16 |
| V2 (GS44-2) | 25.2 | 178.67 | 61.61 | 28.62 |
| V3 (GS16-2) | 12.6 | 230.00 | 45.83 | 26.70 |
| V4 (GS11-2) | 20.1 | 210.13 | 48.47 | 26.49 |
| V5 (GS44-1) | 25.0 | 164.93 | 55.89 | 30.79 |
| V6 (GS16-1) | 14.5 | 199.40 | 41.32 | 27.15 |
| V7 (GS12-1) | 15.1 | 191.67 | 38.34 | 25.67 |
| V8 (GS11-1) | 17.4 | 170.27 | 35.92 | 25.14 |
Number of grain
Base on the number of grain on panicle appears that the progeny V1 (GS12-2) has the highest number of grain, followed by progeny V3 (GS16-2) and V4 (GS11-2). While the progeny V5 (GS44-1) has the lowest number of grain (Figure 1). Based on the character of number of grain, the promising progeny are V1 (GS12-2) has the highest number of grain, followed by progeny V3 (GS16-2) and V4 (GS11-2).
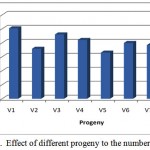 |
Figure 1: Effect of different progeny to the number of grain |
Potential Production
In the character of grain production appears that the progeny V2 (GS44-2) has the highest grain production, followed by progeny V1 (GS12-2) and V5 (GS44-1). While the progeny V8 (GS11-1) has the lowest grain production (Figure 2). Based on the grain production, the promising progeny is at V2 (GS44-2), V1 (GS12-2) and V5 (GS44-1).
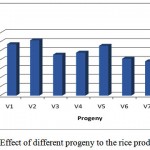 |
Figure 2: Effect of different progeny to the rice production |
Weight of 1000 Grain (WTG)
Based on the weight of thousand grain (WTG) appears that the progeny V5 (GS44-1) has the highest of WTG, followed by the progeny V6 (GS16-1) and V2 (GS44-2). While the progeny V1 (GS12-2) has the lowest WTG (Figure 3). Based on the WTG characters, the promising progeny is at V5 (GS44-1) has the highest of WTG, followed by the progeny V6 (GS16-1) and V2 (GS44-2).
Based on the productive character that includes the number of tillers, number of grains, grain production and grain weight; it appears that there is a significant differences character between progeny. Therefore, the development and plant breeding programmed must be adjusted to the main objective of the cross itself. When the crossing aims are to obtain progeny that have the highest grains per panicle, the most promising cultivars is the progeny V1 (GS12-2) that has reached 251.93 grains per panicle. If the purpose of crossbreeding is to obtain the plants with the highest production potential, the most promising progeny is crosses V2 (GS44-2) with 61.61 grams per panicle that equivalent to 12 ton ha-1. However, if the goal of crossbreeding is to choose the progeny that has the big size of rice grain, the progeny V5 (GS44-1) has the highest weight of grains.
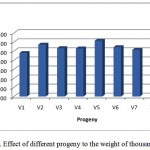 |
Figure 3: Effect of different progeny to the weight of thousand grains |
Conclusion
Based on the research concluded that there are eight promising progeny of upland rice that are tolerant to drought and has a high potential yield, included progeny V1 (GS12-2), V2 (GS44-2), V3 (GS16-2), V4 (GS11-2), V5 (GS44-1), V6 (GS16-1), V7 (GS12-1) and V8 (GS11-1).
Acknowledgements
The authors extend their gratitude to the Minister of Ministry of Research, Technology and Higher Education Republic of Indonesia for providing research grant under the scheme of MP3EI 2016.
References
- Panuju, D.R., Mizuno, K., B. H. Trisasongko. The dynamics of rice production in Indonesia 1961–2009. Journal of the Saudi Society of Agricultural Sciences., 2013 12 : 27-37
CrossRef - Agus, , Wiratno and Suwardi. Status of Indonesian Soil Resources. Asian Soil Partnership Consultation Workshop on Sustainable Management and Protection of Soil Resources 13-15 May 2015, Bangkok, Thailand. 31p
- Ponnamperuma, F.N. Role of Phosphorus in Global Food Production in Global Aspects Food Production. Tycooly publishing Ltd. London. 1986.
- Conde, L.D., Z. Chen., H. Chen., H. Liao. Effects of phosphorus availability on plant growth and soil nutrient status in the rice/soybean rotation system on newly cultivated acidic soils. American Journal of Agriculture and Forestry. 2014; 2(6): 309-316
CrossRef - Sarker, A., A. Kashem., K.T. Osman. Influence of lime and phosphorus on growth performance and nutrient uptake by indian spinach (Basella alba L.) grown in soil. Open Journal of Soil Science., 2014; 4 : 98-102.
CrossRef - Poschenrieder, C., B. Gunsé., I. Corrales., J.Barceló. A glance into aluminum toxicity and resistance in plants. Sci Total Environ., 2008; 400 (1/3): 356-–368.
CrossRef - Zhou, S., Sauvé., T.W. Thannhauser. Proteome changes induced by aluminum stress in tomato roots. J Exp Bot., 2009. 57: 4201-4213.
- Alvim, M.N., F.T. Ramos., D.C. Oliveira., R.M.S. Isaias., M.G.C Franca. Aluminum localization and toxicity symptoms related to root growth inhibition in rice (Oryza sativa) seedlings. J. Bio Sci., 2012; 37(6) : 1079-1088.
CrossRef - Pandey, P., K. Srivastava., R.S. Dubey. Salicylic acid alleviates aluminum toxicity in rice seedlings better than magnesium and calcium by reducing aluminum uptake, suppressing oxidative damage and increasing antioxidative defense. Ecotoxicology., 2013; 22(4) : 656-670.
CrossRef - De Data. Major Research of Upland Rice. IRRI Los Banos Philipine. 1975; 225p
- Slaton, N.A., Wilson Jr, C.E., Norman, R.J., Ntamatungiro, S., Frizzell, D.L. Rice response to phosphorus fertilizer application rate and timing on alkaline soils in Arkansas, Agronomy Journal. 2002; 94 : 1393-1399.
CrossRef - Bhattacharyya, P., A.K. Nayak., M. Shahid., R. Tripathi., S. Mohanty., A. Kumar., R. Raja., B.B. Panda., B. Lal., P. Gautam., C.K. Swain, K.S. Roy, P. K. Dash. Effects of 42-year long-term fertilizer management on soil phosphorus availability, fractionation, adsorption – desorption isotherm and plant uptake in flooded tropical rice. The Crop Journal., 2015; 3: 387-395.
CrossRef - Foy, C. D. The physiology of plant adaptation to mineral stress. Iowa State J. Res., 1983; 57:355-392.
- Watanabe, T., M. Osaki., H. Yanoi., I.M. Rao. Internal mechanism of plant adaptation to aluminium toxicity and phosphorus starvation in three tropical forages. Journal of Plant Nutrition., 29: 1243-1255.
CrossRef - Abate, E., S. Hussien., M. Laing and F. Mengistu. Aluminium toxicity tolerance in cereals: Mechanisms, genetic control and breeding methods. J. Agric. Res., 2013; 8(9) : 711-722.
- Fageria, N.K., J.R.P. Carvalho. Influence of aluminum in nutrient solution on chemical composition in upland rice cultivar. Plant and Soil. 1982; 69 (1): 31-44.
CrossRef - Blum, A. Plant Breeding for Stress Enviroment. CRC Press Inc. Boca Ratoon. 1988. 223p.
- Matsumoto, H. Inhibition of proton transport activity of microsomal membrane vesicles of barley root by aluminium. Soil Sci. Plant. Nutr. 1988; 34(4): 499-506
CrossRef - Roy, B., S.B. Hadra. Effects of toxic levels of aluminium on seedling parameters of rice under hydroponic culture. Rice Science, 2014, 21(4): 217-223.
CrossRef - Delhaize, R., R. Ryan. Aluminium toxicity and tolerance in plant. Plant Physiol., 1995; 107:315-321.
CrossRef - Kamparath, E. Soilacidity in well drained soil of a tropic as a constraint to food production. IRRI and New York State College Agriculture and Life Science. Cornell University IRRI Los Banos Philipine. 1980.
- Garnessan, K., Sankarnarayanan, and T. Balakumar. Physiological basis of differential aluminium tolerance in rice genotypes. Commun. In Soil Sci. Plant Anal. 1993. 24:2179-2191.
CrossRef - Von Uexkull. Availibility and management of potassium in wetland rice soils. Proceeding of workshop held 26 March to 5 April 1984 under the join sponsorship of the IRRI, Soil Management Support Services Agnecy for Internastional Development and United States Depattemen of Agric. Bureau of Soils Philipine Minsstry of Agric. IRRI Los Banos. 1985.
- Hsiao, T.C. Plant respond on water stress. Rev. Plant Physiol. 1973. 24: 519-570.
CrossRef - Bahrun, A., C.R. Jensen,, Asch., V.O. Mogensen. Drought induced changes in xylem pH, ionic composition and ABA concentration act as early signals in field grown maize (Zea mays L.). Journal of Experimental Botany, 2002; 53:1-3.
CrossRef - Bohnert, H.J., R.G. Jensen. Strategies for engineering water stress tolerance in plants. , 1996; 14 : 89-97.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.





